Abstract
Preweaning colibacillosis is a major cause of economic loss to the swine industry in Vietnam. The aim of this study was to examine the enteropathogenicity of representative enterotoxigenic Escherichia coli (ETEC) strains obtained during an earlier epidemiologic survey conducted in five provinces in North Vietnam. This included isolates belonging to serotype O8 that produced heat-stable and heat-labile enterotoxins but did not produce any of the recognized fimbriae (F4, F5, F6, F41, F18). In vitro hemagglutination (unique mannose-resistant hemagglutination activity with guinea pig, sheep, human, and chicken red blood cells at 37°C, but not at 18°C) and enterocyte brush border attachment assays suggested that the F-ETEC strains produced an unidentified colonization factor that promoted adherence to the intestinal epithelium. Colostrum-deprived 1-day-old piglets challenged with an F-strain (1-2 × 109 bacteria) developed acute watery diarrhea within 4 hours of inoculation and suffered up to 20% weight loss, with comparable severity to piglets challenged with conventional F4 and F5 strains. At necropsy, viable counts and histopathologic examination of intestinal sections demonstrated colonization of the duodenum, jejunum, and ileum by F4-positive strains. In comparison, the F- and F5-positive strains attached exclusively to the ileum. Transmission electron micrographs of negatively stained F- cells grown at 37°C demonstrated the presence of fimbriae. These results confirm the presence of a potentially new pathogenic ETEC fimbrial type in piggeries in Vietnam, with a unique hemagglutination property and attachment characteristics similar to ETEC bearing F5 fimbriae.
Keywords
Enteric colibacillosis caused by enterotoxigenic strains of Escherichia coli (ETEC) is one of the most important diseases of pre- and postweaning pigs worldwide. 1, 4, 20 ETEC associated with neonatal diarrhea often belong to a limited number of O serogroups (O8, O64, O101, O138, O141, O147, O149, O157) and produce one or more fimbriae (F4, F5, F6, F41, F18) in association with enterotoxins STa, STb, LT, or a combination thereof. 1, 13, 31 In addition, several studies have shown that ETEC, apparently lacking these fimbrial antigens and belonging to nonclassical ETEC serogroups (O8, O9, O20, O101), is increasingly isolated from piglets with diarrhea, particularly in herds where E. coli vaccines have been used. 7, 15, 21, 42, 46 However, the pathogenicity of many of these strains is still questionable. In addition to the five common specific fimbriae, several less recognized adhesins also have been identified, for example F42, 47 F165, 14 CS1451, 6 and AIDA-I. 36
Vietnam is a developing country with a rapidly expanding commercial pig industry, although the majority of pigs are still raised in small numbers in village-based industries. Results from a survey of preweaning diarrhea in five commercial intensive piggeries in North Vietnam showed that ETEC was responsible for 43% of cases of diarrhea in the first 4 days of life and 23.9% of remaining cases until weaning. These ETEC isolates belonged to only a limited number of serogroups and pathotypes, including O149 : K91 (F4/STa/STb/LT, 33.3%), O64 (F5/STa, 16.7%), O8 : G7 (F4/STa/STb, 11.1%), and O101 (F4/STa/STb, 3.2%). A relatively high frequency of ETEC isolates (22.2%) belonged to serogroup O8, produced enterotoxins (STa/STb/LT), but did not possess any of the recognized fimbriae (designated F– ETEC). 11 The objectives of this study were to examine the adherence capability and pathogenicity of the most common ETEC serotypes and pathotypes in Vietnam, including the F– strains. The F– strains were compared with representative conventional ETEC pathotypes in assays to measure in vitro attachment. These included determining mannose-resistant hemagglutination activity (MRHA) to different red blood cell (RBC) types and adhesion to isolated porcine epithelial cells. Second, the strains were tested for pathogenicity in colostrum-deprived 1-day-old piglets.
Materials and Methods
Bacterial strains
The designations and characteristics of the 10 ETEC strains isolated from field cases and the 5 ETEC reference strains used for in vitro and in vivo study are outlined in Table 1. The pathotypes of the F– strains (NVP608, NVP613, NVP1268, NVP1390, NVP1452) together with fimbriated strains (NVP1402, NVP1271, NVP2081, NVP1372) were confirmed by agglutination with F4, F5, F6, and F41 antisera and by polymerase chain reaction (PCR; with primers specific for F4, F5, F6, F41, F18, STa, STb, and LT). 11, 12 The reference E. coli strains FV847a (F4-positive), 1547 (F5-positive), 1500 (F6-positive), and 1520 (F41-positive) were used as positive controls, and strain C600 (E. coli K12) was used as a fimbria-negative control in the in vitro assays.
Designation and characteristics of ETEC strains used in this study.∗
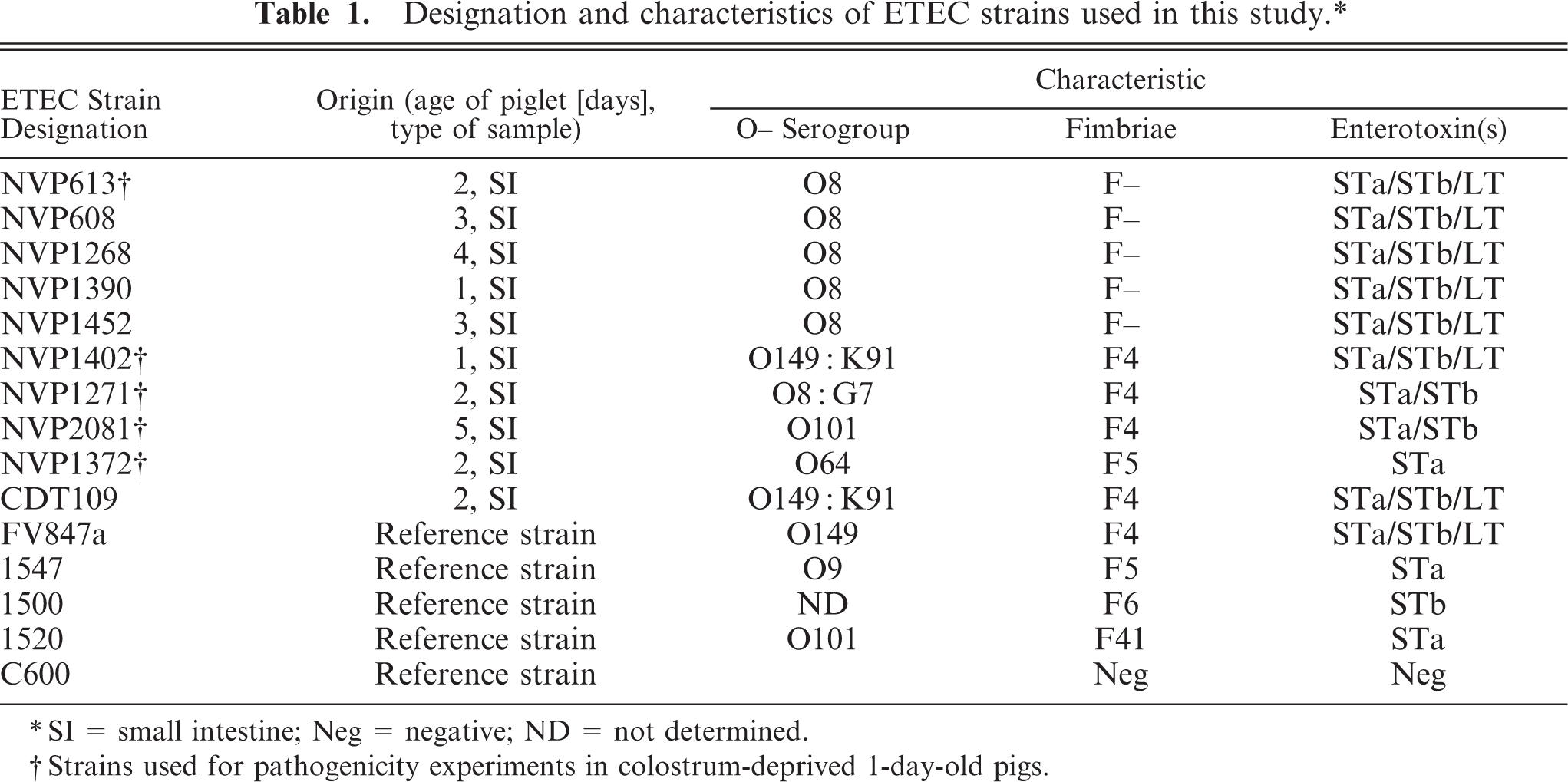
∗ SI = small intestine; Neg = negative; ND = not determined.
† Strains used for pathogenicity experiments in colostrum-deprived 1-day-old pigs.
Hemagglutination tests
The tests were performed in the presence or absence of
In vitro adhesion assay
Assays of isolated porcine intestinal epithelial cells were performed as previously described with some modifications.
40,
43,
45
Cultures of each ETEC strain were grown on agar as described in experiment 1 and suspended in Krebs–Henseleit buffer to a final concentration of approximately 109 cells/ml. Intestinal epithelial cells were obtained from newborn colostrum-deprived piglets within 5 hours of parturition as described.
40,
45
The cells were suspended in Krebs–Henseleit buffer (pH 7.4) and adjusted to a concentration of approximately 106 cells/ml. After incubation at 37°C for 30 min, the bacteria–epithelial cell suspension was washed three times in PBS, and a drop of the mixture was then placed on a microscope slide and stained with one drop of 0.1% carbol fuchsin. Bacteria adhering to individual epithelial cells were examined. The number of bacteria on 20 cells was counted, taking care to include only individual cells with intact brush borders. When more than 20 bacteria were present per individual cell, the number was also recorded as 20 because it was difficult to count the number exactly. The average number of adherent bacteria per epithelial cell was then calculated. The tests were performed in parallel in the presence of 1.5%
Experimental infection of conventionally reared, 1-day-old piglets
Preparation of cultures for challenge
Freeze-dried cultures were inoculated into tryptic soy broth, incubated at 37°C overnight, and transferred onto sheep blood agar (SBA) plates. On the following day, inocula were prepared for each E. coli strain by subculturing a single isolated colony into 20 ml of brain heart infusion (BHI) and incubating at 37°C for 6 hours with agitation (150 rpm) to obtain a desired cell count of approximately 109 bacteria/ml. Final viable counts (the average of two duplicates) were obtained as described. 27
Experimental animals and experimental design
Healthy newborn, colostrum-deprived Large White piglets were obtained from sows held at the Pinjarra Hills Piggery Unit at The University of Queensland. All sows were vaccinated during pregnancy with LEvac vaccine (Intervet, Australia) for prevention of leptospirosis and erysipelas, and porcine parvac vaccine (CSL, Australia) for prevention of porcine parvovirus. All experimental procedures were conducted with the approval of the Animal Ethics Committee of The University of Queensland, Australia (SVS/001/04/AUSAID). Animals showing severe illness or discomfort were immediately euthanatized and subjected to postmortem examination.
Sows were monitored during parturition to collect the piglets as soon as they were born to ensure that transfer of colostrum did not take place before ETEC dosing. Each piglet was marked for identification and transported in a warm (30°C) container to PC2 isolation animal pens and distributed into cardboard boxes in separate rooms. Ambient temperature was held at 34°C and straw was used as bedding. Time from birth to challenge was approximately 1–2 hours. Twenty-four colostrum-deprived piglets (from nine different litters) were divided into six groups of four. Each group was challenged with one of five ETEC strains (NVP613, NVP1402, NVP1271, NVP2081, NVP1372) administered by intragastric route (1 or 2 ml of bacterial suspension in 20 ml of 0.1% peptone water), except for group 6, which was given sterile peptone water only and served as sham-inoculated controls. 38, 39
Body weights for individual piglets were recorded before challenge and at the time of euthanasia. Following challenge, piglets were kept under continual observation for clinical signs. At 4 hours postchallenge (p.c.) each piglet received 20 ml of milk replacer (Pigiplus, Biostarch, Pty. Ltd., Australia) and then every 3 hours until 24 hours p.c. The piglets were individually examined for diarrhea at each feeding, and rectal temperature and fecal consistency were recorded. Severity of diarrhea was used to score feces on a scale of 0 = no diarrhea, 1 = mild diarrhea (viscous or creamy feces), 2 = moderate diarrhea (liquid mixed with fecal materials), and 3 = severe diarrhea (watery). Rectal swabs were also collected at an interval of 3 hours for bacteriologic culture. Piglets were immediately euthanatized (0.5 ml/kg body weight intravenous injection of sodium pentobarbitone, Lethabarb, Virbac, Australia) on showing severe clinical signs (dehydration, watery diarrhea, depressed state) or at 24 hours p.c. and necropsied. Rectal swabs were taken before inoculation and every 3 hours throughout the observation period for bacteriologic evaluation, except for the first sample collection at 4 hours p.c.
Postmortem procedure
The gross appearance of the carcass (including estimation of the degree of dehydration) and gastrointestinal tract were noted. Approximately 10 cm of duodenum (the segment immediately adjacent to the pylorus), jejunum (5 cm either side of the midpoint of the small intestine), and ileum (the last 15–30 cm proximal to the ileocecal valve) were tied off with sterile string and collected for bacteriologic investigation. Each segment was stored in sterile containers, and E. coli viable counts were performed on all segments. For histopathologic investigation, adjacent segments were obtained from each site at the same time and fixed in 10% buffered formalin.
Viable counts, isolation, and identification of ETEC
Rectal swabs and intestinal loops taken at necropsy were cultured aerobically at 37°C on SBA and Mac Conkey agar overnight. Identification of challenge strains was provisionally made on the basis of colony characteristics on SBA. This was confirmed by the slide agglutination test with the use of appropriate specific antisera raised in rabbits against each of the challenge strains on three to five representative colonies and by PCR assay for identification of specific virulence factors. The level of challenge strain shed in the feces was subjectively scored by estimating the degree of growth obtained from the inoculated rectal swabs on a scale of 0 = no inoculated strains detected, 1 = a few colonies of the inoculated strain were detected, 2 = 50% of colonies were the inoculated strain, and 3 = profuse growth of a pure or almost pure culture of the inoculated strain. The degree of association between the onset of diarrhea and isolation of the inoculated strain from rectal swabs was determined by an association index obtained by multiplying the values obtained for two assessment criteria: 1) fecal consistency graded from 0 (normal feces) to 3 (watery diarrhea) and 2) level of challenge strain recovered from culture plates scored from 0 (no isolates recovered) to 3 (heavy pure culture). Thus, an index of 9 indicated watery diarrhea associated with heavy pure growth of the inoculated strain, whereas an index of 0 indicated no signs of diarrhea, no inoculated strains detected in rectal swab samples, or both.
Viable counts for each segment of intestine were determined on SBA as described, 5 with some modifications. The contents of each segment were washed three times with 5 ml of PBS by syringe with an 18-gauge needle. The final volume of the contents was then adjusted to 30 ml with PBS (lumen contents). Each segment was then opened longitudinally, placed in a sterilized tissue grinder, and homogenized. Cold PBS (pH 7.2) was added, the sample was vortexed (900 rpm for 1 minute), and the total volume of the intestinal specimens was then adjusted to 30 ml (intestinal wall). The total viable E. coli count in a 10-cm segment was calculated by adding bacterial counts from the lumen contents and the intestinal wall together. A segment was defined as being colonized if it contained >108 (8 log) of the inoculated strain. 34 Confirmation of the challenge strains was determined by PCR and serology as described previously. 10
Histopathology
Formalin-fixed segments of duodenum, jejunum, and ileum were embedded in paraffin, and sections (∼5 µm thick) were cut, stained with hematoxylin and eosin (HE stain) and Gram's stain (Brown and Brenn method), and examined by light microscopy.
Transmission electron microscopy
One drop from the surface of a culture of strain NVP613 grown in nutrient broth at 37°C overnight without agitation was placed on a carbon and formvar–coated copper 200 mesh grid and allowed to absorb for 2 minutes. Excess liquid was then removed with filter paper, and one drop of 1% ammonium molybdate (pH 6.5) was added. After 30 seconds, the specimen was blotted dry with filter paper and observed under a JEOH 1010 transmission electron microscope operated at 80 kV. Images were captured with a Soft Imaging Megaview III digital camera.
Statistical analysis
Means of the number of adherent bacteria per epithelial cell were compared by Student's t-test (two-tailed P value) for the test strains and the positive and negative control strains. Mean weight changes and bacterial counts for each group of piglets were also compared for statistical significance by Student's t-test (two-tailed P value). Statistical significance was set at P < 0.05 (two-tailed) with GraphPad Instat version 3.01.
Results
Mannose-resistant hemagglutination activity of F– ETEC strains
The five F– strains were able to agglutinate guinea pig, sheep, human, and chicken RBCs, with the highest hemagglutination titer (256) observed with guinea pig RBCs (Table 2). Agglutination was not inhibited in the presence of 1.5%
Hemagglutination titers of ETEC strains grown at 37°C with different types of RBCs in the presence of
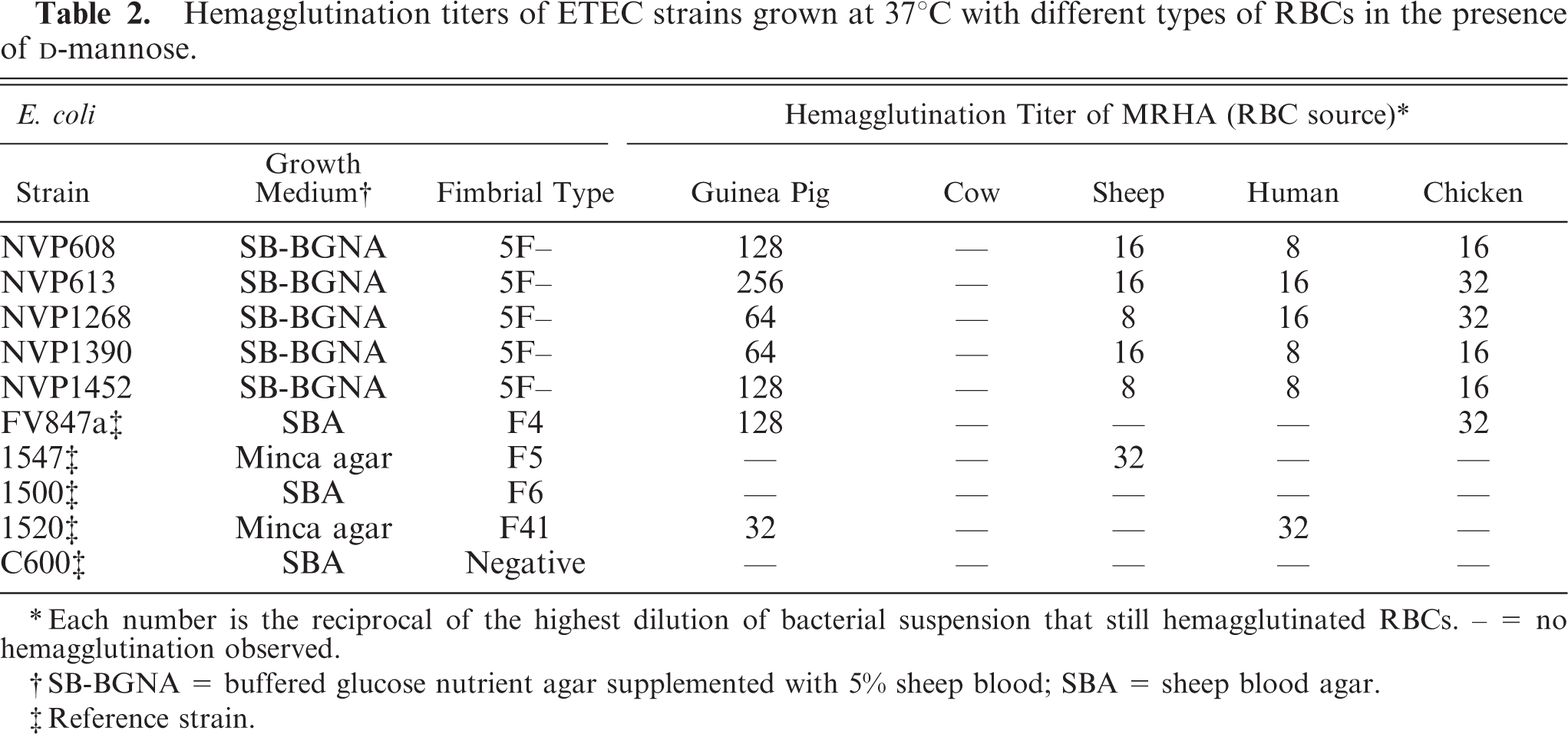
∗ Each number is the reciprocal of the highest dilution of bacterial suspension that still hemagglutinated RBCs. – = no hemagglutination observed.
† SB-BGNA = buffered glucose nutrient agar supplemented with 5% sheep blood; SBA = sheep blood agar.
‡ Reference strain.
In vitro adhesion of F– ETEC strains to isolated intestinal epithelial cells
Adherence to isolated intestinal epithelial cells from newborn piglets was demonstrated for all five F– strains grown at 37°C (Table 3). A large number of bacteria (on average, >18 bacteria/epithelial cell) adhered to and completely covered the brush borders, both in the presence and absence of
Mean(± SEM) number of adherent bacteria per porcine intestinal epithelial cell in the in vitro adhesion assays.
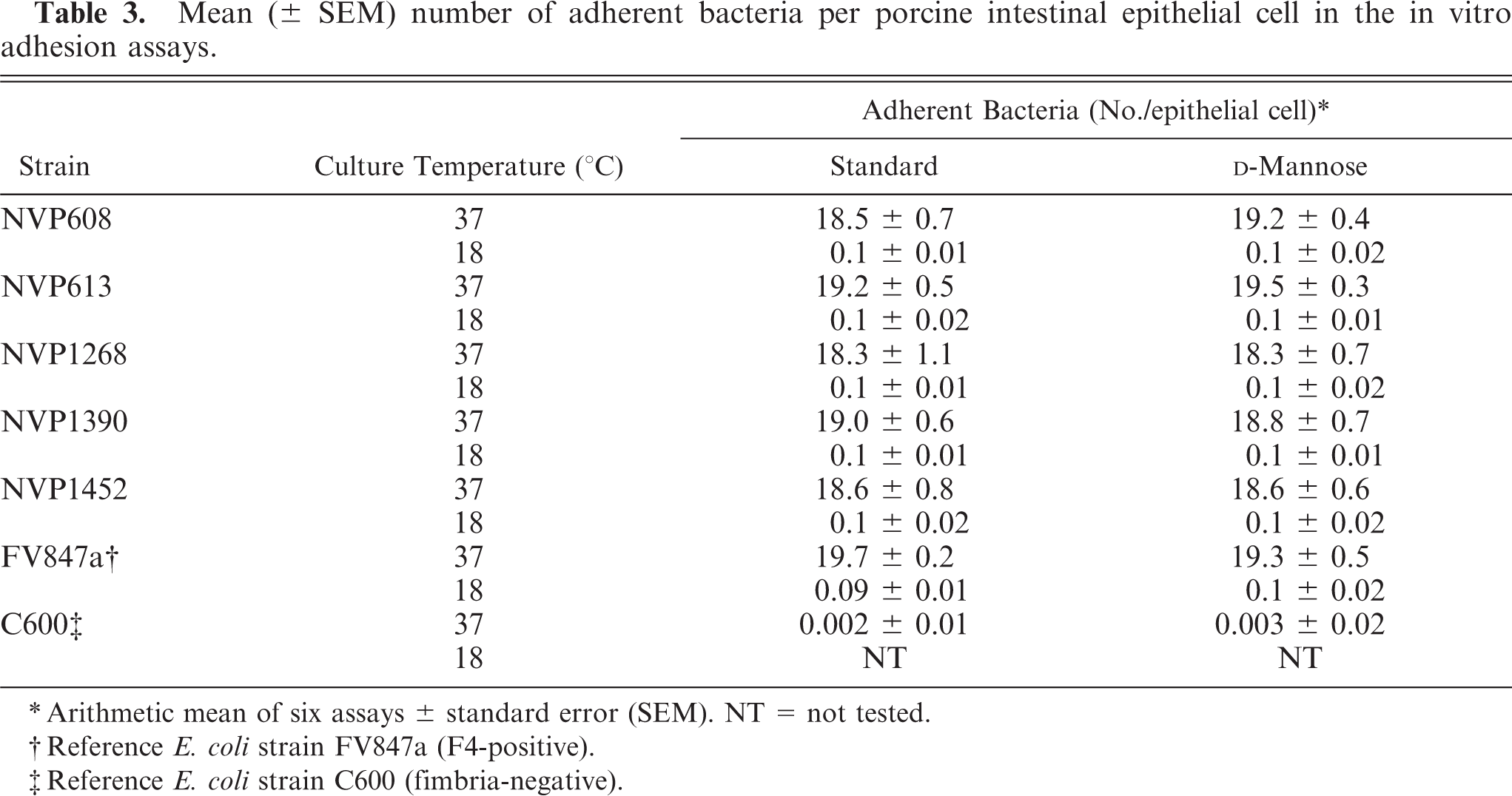
∗ Arithmetic mean of six assays ± standard error (SEM). NT = not tested.
† Reference E. coli strain FV847a (F4-positive).
‡ Reference E. coli strain C600 (fimbria-negative).
Pathogenicity of ETEC strains in newborn piglets
Watery diarrhea was observed in all piglets within 24 hours of intragastric administration of an ETEC strain. In contrast, none of the four piglets in the control group showed any illness, and they remained healthy until euthanasia at 24 hours p.c. Clinical signs of piglets produced by the five ETEC strains showed almost no differences, and the severity of disease produced was not influenced by the size of the dose given. The rectal temperatures of inoculated and control piglets did not show any significant differences during the observation period.
Altogether, 11 of the 20 challenge-exposed piglets became moribund between 11 and 24 hours p.c. The shortest observed period between exposure and euthanasia was 11 hours. By 24 hours p.c., all remaining challenge-exposed piglets were euthanatized and subjected to postmortem examination. All challenge-exposed piglets were confirmed to be excreting the inoculated strains by rectal swab culture. Within 24 hours of exposure, affected piglets suffered an average weight loss of approximately 20% of their initial body weight (Table 4). In contrast, piglets in the control group increased their body weight by 5.3%, a highly significant difference compared with the inoculated groups (P < 0.0001).
Weight change of piglets during the experiment.
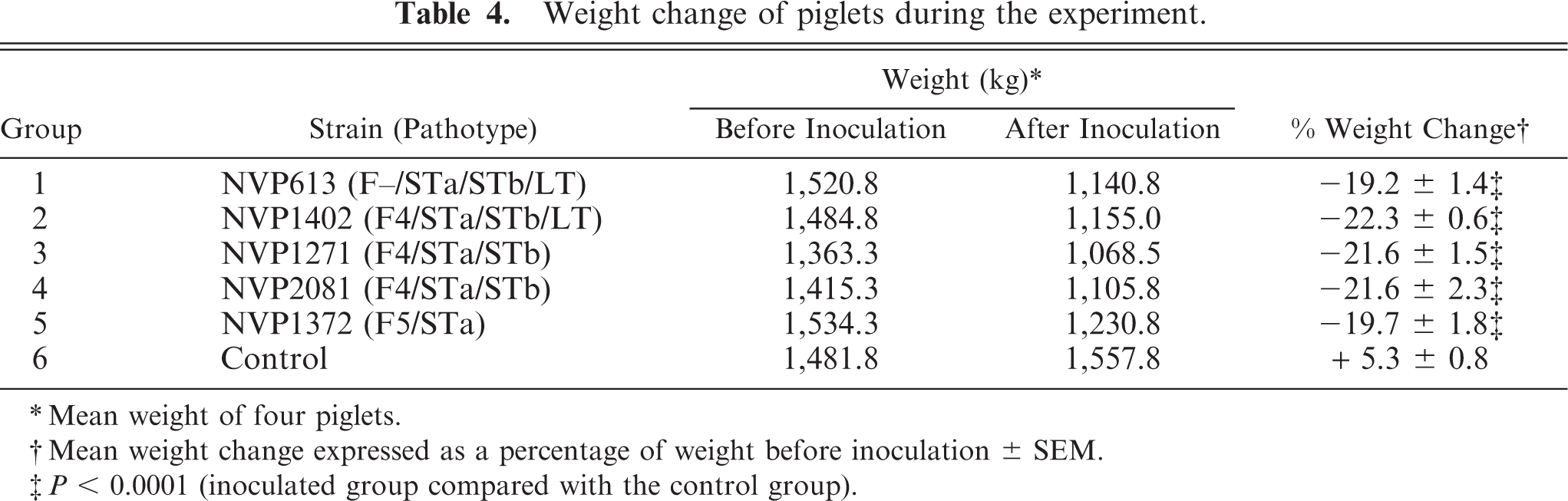
∗ Mean weight of four piglets.
† Mean weight change expressed as a percentage of weight before inoculation ± SEM.
‡ P < 0.0001 (inoculated group compared with the control group).
None of the rectal swabs taken just before inoculation yielded enterotoxigenic E. coli. However, as soon as diarrhea began at 4 hours p.c., the inoculated strain could be isolated from rectal swabs. Subsequently, the test organisms were recovered with increasing frequency. By 21 hours p.c., all surviving piglets from each challenge-exposed group scored the maximum index of 9.
Necropsy findings
The carcasses of piglets with diarrhea were dehydrated; the hair was raised and rough, and fluid feces were pasted around the perineum and tail. Internally, the stomach was distended with undigested milk curd, the small intestine was dilated from fluid accumulation, and the large intestine was distended with fluid and gas. Inflammation of the small intestine was not evident in the piglets. The colon of one piglet in group 2 showed slight hyperemia of the mucosal surface.
Bacteriologic findings
Numbers of viable bacteria from the lumen contents and the intestinal wall specimens for each segment of the small intestine are presented in Table 5. At the dilution at which individual colonies could be counted, only pure cultures of the infecting strain were recovered. Samples from control piglets yielded a variety of lactose-fermenting colonies, none of which were positive by slide agglutination or PCR assays for fimbriae or enterotoxins. The number of bacteria recovered from each segment of the inoculated groups was significantly greater when compared with the control group (P < 0.001). In piglets infected with F4-positive strains (NVP1402, NVP1271, NVP2081), the mean intestinal wall bacterial counts always exceeded those recovered from the lumen contents. At all levels of the small intestine, the differences were statistically significant (P < 0.05). A similar pattern of counts was observed in groups inoculated with the F– strain (NVP613) and the F5-positive strain (NVP1372). However, the difference in counts between the intestinal wall and lumen contents was only significant in the ileum (P < 0.01). Furthermore, the number of bacteria in the intestinal wall of the ileum was significantly higher than in the duodenum or jejunum (P < 0.05). When the number of bacteria from the lumen contents and the intestinal walls were added together to make the total count, it was difficult to find any segment in the infected piglets that contained < 6 log CFU/10-cm segment. When compared with the negative control, the three F4-positive strains (NVP1402, NVP1271, NVP2081) were consistently found to have significantly higher counts in all sections of the small intestine (total counts >9 log CFU/10-cm segment). For the F– and F5-positive strains (NVP613 and NVP1372, respectively), the counts were only significantly different from the controls in the ileum (mean count = 9–9.3 log CFU/10-cm segment), and not the duodenum and jejunum (mean count <7 log CFU/10-cm segment).
Number of viable E. coli recovered from the lumen contents or homogenized intestinal wall and the number of piglets showing histologic evidence of diffuse adherence of bacteria to the epithelial surface of the duodenum, jejunum, and ileum after challenge exposure with Vietnamese ETEC strains.
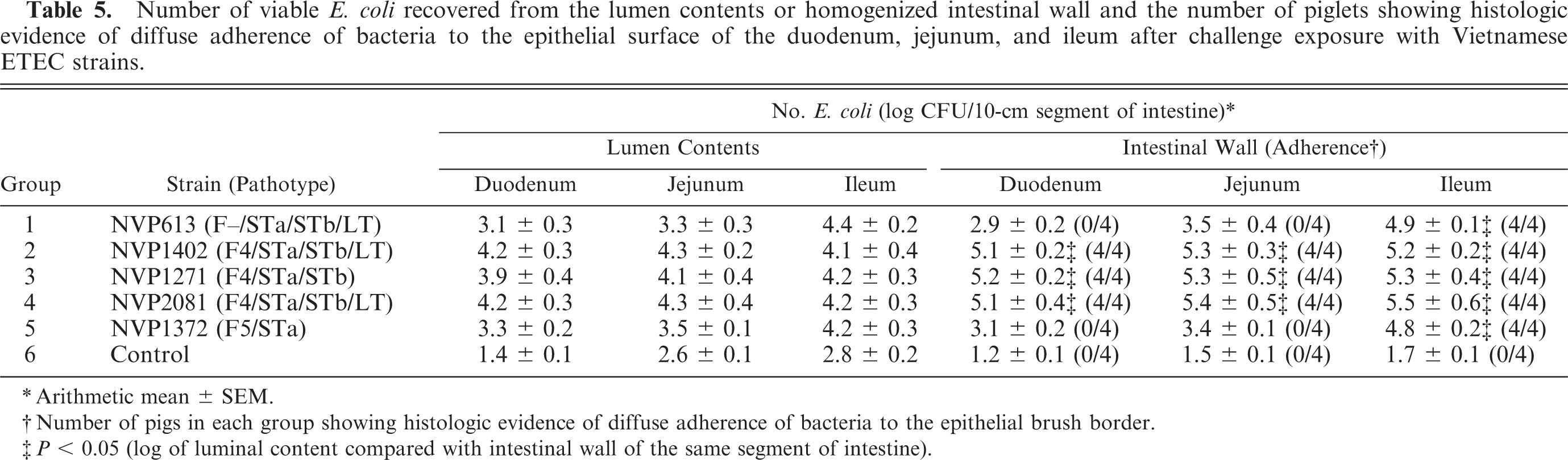
∗ Arithmetic mean ± SEM.
† Number of pigs in each group showing histologic evidence of diffuse adherence of bacteria to the epithelial brush border.
‡ P < 0.05 (log of luminal content compared with intestinal wall of the same segment of intestine).
Histopathologic findings
Most of the sections of duodenum, jejunum, and ileum showed few or no histologic changes. The epithelial cells lining the sides of the villi were columnar, and their brush borders were intact. No structural or morphologic damage to the villous epithelium, submucosa, muscularis externa, or serosa were observed. However, some piglets euthanatized because of severe clinical signs before 24 hours p.c. showed mild congestion and a mild infiltration of neutrophils in the jejunal or ileal lamina propria. Sections from the control piglets did not show any attachment of bacteria to the epithelium (Fig. 1). In contrast, in all segments from inoculated piglets in which the E. coli count was >9 log CFU/10-cm segment, diffuse layers of bacteria were found closely adhering to the epithelial surfaces on the sides or the tips of the villi. However, the epithelial cells underlying the adherent bacteria appeared normal (Fig. 2). Sections of duodenum and jejunum from piglets inoculated with the F– or F5 strains containing <7 log CFU/10-cm segment had only a few bacteria occasionally associated with the mucosa. However, both strains were shown to attach in large numbers to the surface of the ileum, consistent with the slightly higher mean counts of >9 log CFU/10-cm segment in these sections.
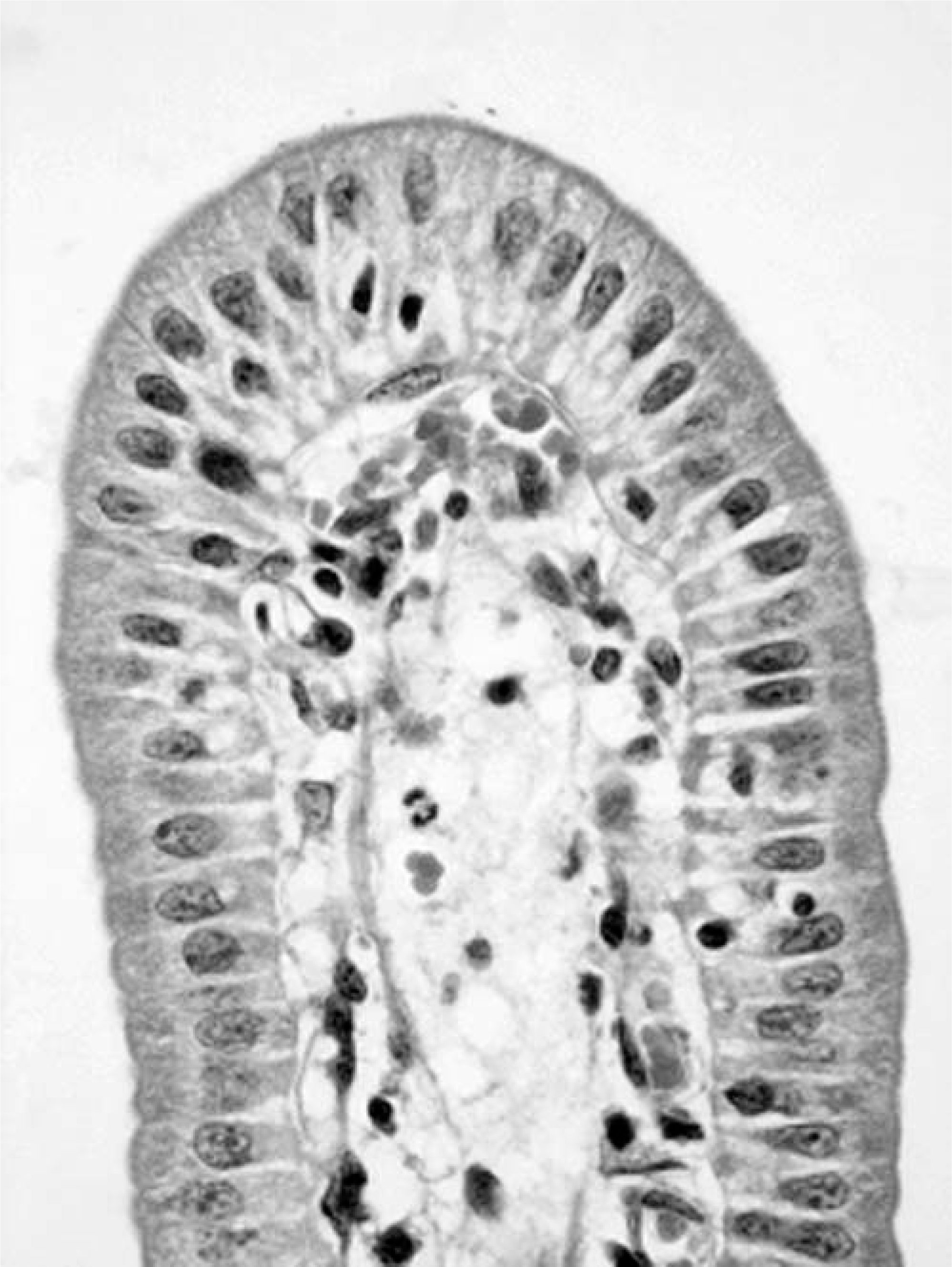
Ileum; control piglet. HE.
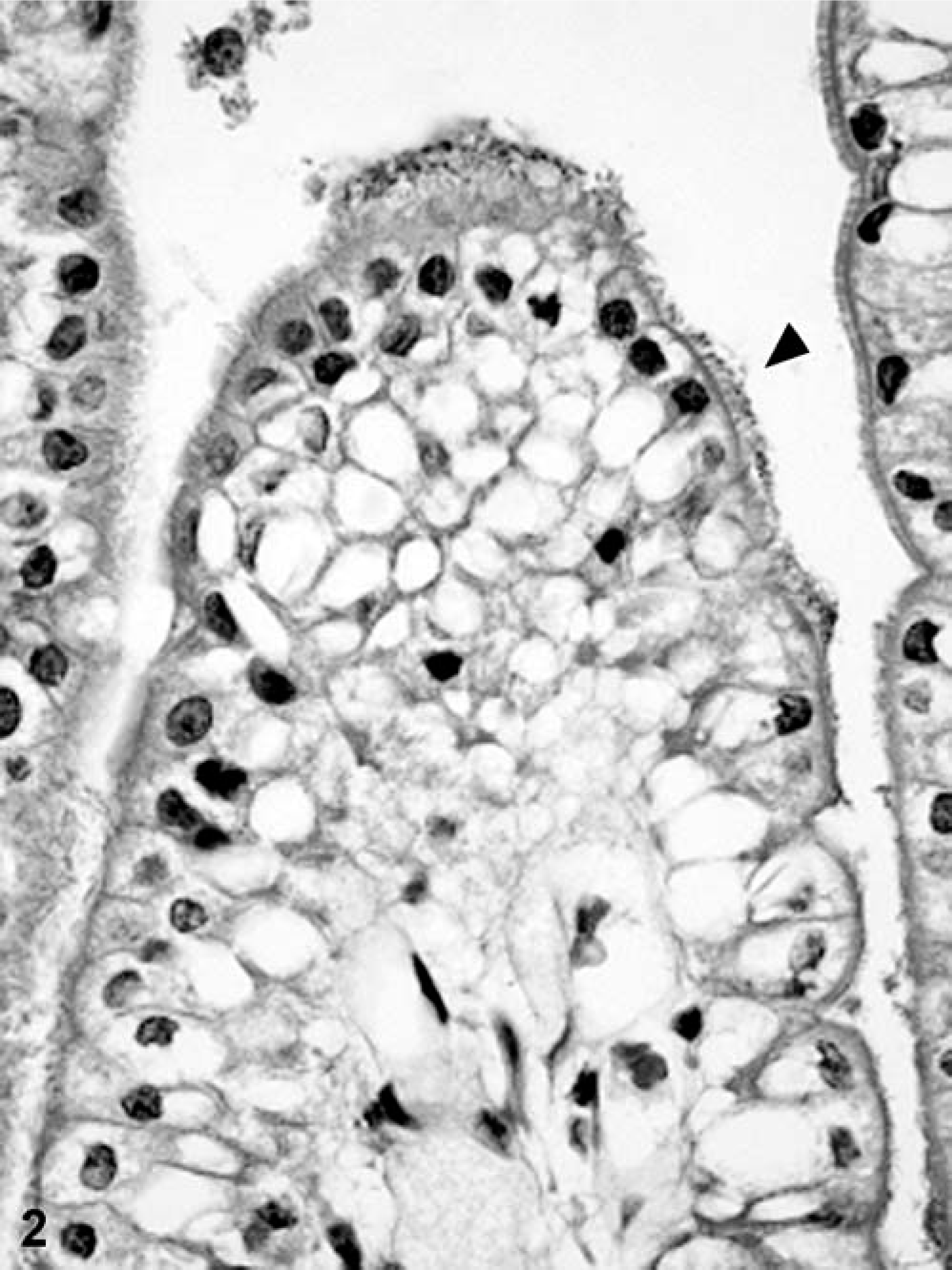
Ileum; piglet inoculated with enterotoxigenic Escherichia coli strain NVP613 (5F–/STa/STb/LT). Numerous bacteria (arrow) are attached to the surface of the enterocytes.
Transmission electron microscopy of negatively stained whole cells
Electron micrographs of negatively stained strain NVP163 bacteria cells showed that fimbriae were expressed following in vitro culture in BHI broth (Fig. 3).
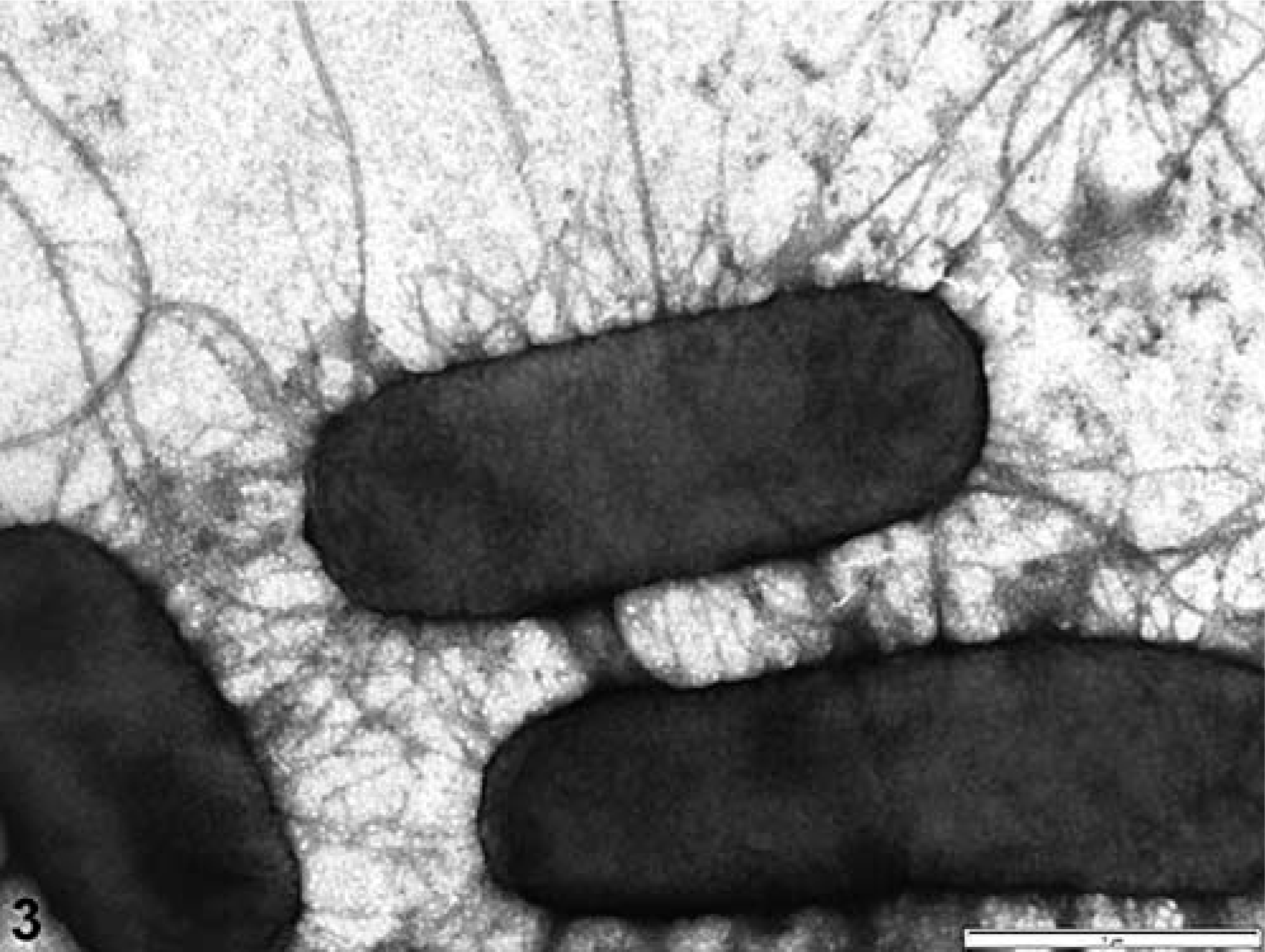
Transmission electron micrograph showing fimbriae on the surface of enterotoxigenic Escherichia coli strain NVP613 (5F–/STa/STb/LT) after growth in brain heart infusion broth. Bar = 1 µm.
Discussion
All five F– ETEC strains isolated from piglets with diarrhea in Vietnam (NVP613, NVP608, NVP1268, NVP1390, NVP1452) displayed a unique hemagglutination property. Besides showing MRHA with sheep, human, and chicken RBCs, a strong reaction was also obtained with guinea pig RBCs, but not with bovine RBCs. This characteristic hemagglutination pattern was observed only with cultures grown at 37°C and not with those grown at 18°C. In addition, the F– strains were observed to strongly attach to isolated porcine brush borders. This adhesive property was recorded only with cultures incubated at 37°C, and it was not inhibited by the presence of 1.5%
The results of in vivo pathogenicity testing supported the hypothesis that the atypical F– strains are pathogenic for piglets and carry a novel fimbrial type. The F– strains expressed STa, STb, and LT enterotoxins, and the one strain selected for pathogenicity testing caused hypersecretory diarrhea in all piglets with a similar severity to that observed for the other four tested strains that possessed recognized fimbriae. The clinical response and bacteriologic findings can be summarized as follows. 1) All test strains consistently produced profuse diarrhea in newborn, colostrum-deprived piglets, which was observed as early as 4 hours p.c. 2) Affected piglets consistently lost approximately 20% of their initial weight. 3) The occurrence of diarrhea and recovery of the test strain from rectal swabs were always closely related. 4) Postmortem examinations did not reveal any typical changes for most viscera, except for the alimentary tract, which was distended with fluid. Histopathologic changes in sections of the small intestines were minimal or absent. 5) Colonization of the duodenal, jejunal, and ileal surface (>8 log CFU/10-cm segment) was confirmed in all piglets inoculated with F4 fimbriated strains (NVP1402, NVP1271, and NVP2081), but was only demonstrated on the ileal surface of piglets inoculated with F5 (NVP1372) or the F– strain (NVP613).These results closely resembled descriptions of field outbreaks of neonatal colibacillosis 1, 4, 31 and are in agreement with other studies in which piglets were experimentally infected with strains obtained from naturally occurring cases of colibacillosis. 5, 19, 28– 30, 34, 38, 39, 44
Marked differences in clinical signs were not observed between groups of piglets inoculated with the different ETEC strains. However, in groups of piglets inoculated with the same test strain, diarrhea began in some piglets at different times. A 24-hour period of study has generally been sufficient to permit the development of diarrhea in inoculated pigs. 5, 7, 14, 28, 34, 38, 39, 41, 44 However, in most experiments conducted by other investigators, piglets were returned to their dams after inoculation. In this study, inoculated piglets were separated from their dams, kept in a PC2-level isolation room, and then euthanatized at 24 hours p.c. according to Australian quarantine rules for in vivo experiments with imported strains. Therefore, we were not able to investigate exactly how long diarrhea would have lasted in infected piglets or the period of excretion of the infecting challenge strains. Infected piglets have previously been reported to excrete the challenge strains for 6–7 days after inoculation. 38, 39
In several reports, E. coli strains lacking known fimbrial adhesins were isolated from neonatal pigs with diarrhea. 1, 4, 15, 21, 31, 42 However, only strains expressing the known fimbrial adhesins F4, F5, F6, and F41 have consistently been shown to be pathogenic to experimentally infected newborn piglets. 5, 28, 29, 34 Our results confirm that the Vietnamese F– strains do not possess any of the recognized fimbriae but might produce a novel adhesive factor that results in attachment of the strain to the ileum in a manner similar to F5-positive strains. However, an important difference is that the F– strains have been isolated from all ages of piglets in Vietnam from birth to weaning; 11 thus, the receptor mediating attachment might be similar to F4 fimbriae by not being age dependent. The novel adhesin also appears to be antigenically distinct from other well-recognized colonization factors because antisera prepared against these antigens (F4, F5, F6, F41) did not react with the F– bacteria. Furthermore, when the results were compared with other studies documenting the occurrence of strains that possess other less recognized fimbriae, some distinct differences were apparent. The Vietnamese F– E. coli strains all belonged to serogroup O8; produced STa, STb, and LT enterotoxins; and showed the highest MRHA titers with guinea pig RBCs. In contrast, F42-positive E. coli (one strain was O8 : H– and the other two were not serogrouped) produced only STa and possessed the strongest MRHA reaction with chicken RBCs. 47 Strains producing F165 were shown to belong to O115 and were enterotoxin negative. 14, 16 In addition, the Vietnamese F– strains differed from atypical F6-positive O8 : KX105 because these strains were all negative in the MRHA test. 7 Other less recognized E. coli adhesins, such as CS 1541 6 and AIDA-I, 36 were not assayed in this study.
A number of other factors might contribute to the ability of strain NVP613 and other F– strains to colonize the small intestine and induce diarrhea. First, the characteristics of attachment to the ileum were comparable to classical ETEC attachment by F4-, F5-, F6-, or F41-producing strains, which frequently have ileal counts >9 log CFU/10-cm segment. 28, 29, 33 It is conceivable that lipopolysaccharide O antigens could confer adherence, as suggested. 46 However, the bacteria were observed attached to both the tips and the sides of the villous epithelium in a fashion similar to the other fimbrial types. Second, the production of enterotoxins played an importance role in the enteropathogenicity of this strain, as demonstrated by the disease course in affected piglets. The close link between enterotoxin, particularly LT, and pathogenicity in the O8 strains is noteworthy because the production of LT enterotoxin is rarely observed in strains lacking F4 antigens. 13, 31 F4-negative, LT-positive O8 strains have been reported occasionally in other studies, but their pathogenicity in piglets has not yet been demonstrated. 7, 18, 21, 25, 26 LT enterotoxin might be an important virulence factor for F– O8 strains. In noninvasive ETEC strains, there is always a strong positive correlation between the production of enterotoxin and the presence of fimbrial adhesin (F2, F3, F4, F5, F6, F41). 17 Therefore, it is possible that the production of enterotoxins in these unusual strains is also genetically linked to the unknown colonization factor. The O8 : KX105 strains (STb/LT) carrying fimbrial antigen CS1541 were able to cause disease in newborn piglets in Canada, 6, 7 whereas in Australia, diarrhea was experimentally reproduced in pigs inoculated with an O8 : K85 (STa/F5) strain, which was well recognized as pathogenic for calves and lambs but apparently rarely found in pigs. 44 One strain belonging to O8 : H– was proven to produce F42 fimbriae and was associated with porcine neonatal diarrhea in Brazil. 47
Because the production of enterotoxins and most fimbriae (F4, F5, F6) is plasmid encoded, 32, 35 it is not surprising that new serotypes of enteropathogenic E. coli with different combinations of enterotoxin and fimbriae can appear from time to time in different geographic areas. Several studies have discussed that, under the high selection pressure of widespread use of ETEC vaccines containing fimbriated strains, novel fimbriae that have not yet been characterized might become more prevalent. 42, 46 This is one of the possible explanations for the appearance of these O8 ETEC strains carrying a probable new adhesin among the population of ETEC-causing diarrhea in piglets in Vietnam. The high frequency of O8 F– ETEC strains associated with diarrhea throughout the preweaning period in commercial piggeries in North Vietnam, together with the ability of one of the representative strains to reproduce diarrhea in experimentally challenged newborn piglets, confirms that these strains are extremely important in the etiology and control of preweaning colibacillosis in Vietnam. The appearance of these strains could be related to the mixing of imported breeds with local Vietnamese breeds of pig, which is a common practice with the increase in intensive pork production in recent years. These strains certainly appear to be novel compared with fimbria-negative strains isolated in other studies; therefore, they should be the subject of further investigation. Furthermore, they should be included in multivalent killed vaccines for the control of neonatal colibacillosis in Vietnam.
Footnotes
Acknowledgements
This work was funded by a small grant from the Australian Centre for International Agricultural Research (ACIAR), and T. N. Do was the recipient of an ACIAR John Allright Fellowship. We gratefully acknowledge Rick Webb, Center for Microscopy and Microanalysis, The University of Queensland, for assistance with electron microscopy.
