Abstract
The objective of our study was to compare the serum concentrations of procalcitonin (PCT), neopterin, tumor necrosis factor alpha (TNF-α), prostaglandin E2, malondialdehyde, interleukin 8, and gamma interferon (IFN-γ) in neonatal calves with septicemic colibacillosis (n = 15) with healthy controls (n = 15). Septicemic colibacillosis was diagnosed based on clinical signs and the isolation of Escherichia coli from heparinized blood in clinically suspected cases. PCT, neopterin, TNF-α ( p < 0.05), and IFN-γ concentrations in calves with septicemic colibacillosis were significantly higher than in the healthy controls ( p < 0.01). Serum PCT concentrations were ~4 times higher in the calves with septicemic colibacillosis than in the healthy calves, suggesting that PCT could be a useful marker of septicemic colibacillosis in neonatal calves.
Septicemic colibacillosis caused by pathogenic serotypes of Escherichia coli is a common disease of neonatal calves, and it generally occurs as peracute septicemia, resulting in sudden death. Affected calves 2–5 days of age have fever (40.5–41°C), anorexia, weak or absent suckle reflex, conjunctival hyperemia, weakness, tachycardia, decreasing body temperature in 1–2 days, and hyperpnea. The fatality rate is 100%.2,11,20 As one of the most important causes of calf mortality, the rapid diagnosis of septicemic colibacillosis in calves is of vital importance.
Procalcitonin (PCT) is the prohormone of calcitonin and, in humans, it is thought to be an effective marker used in early diagnosis of bacterial infections and to assist in the differentiation of bacterial from viral infections. In systemic infections usually caused by bacterial infections, baseline PCT serum levels are reported to increase 10–100-fold.3,5,6,9,10,22,24 Studies have shown that PCT is a useful early diagnostic marker of sepsis in neonatal human infants.1,21
Serum neopterin is an indicator of cell-mediated immunity reflecting the activation of macrophages and/or monocytes. 22 Gamma interferon (IFN-γ) is effective in the stimulation of neopterin production by macrophages.13,23 Cytokines are important pro-inflammatory mediators in the early phases of septicemia. Cytokines, PCT, and neopterin are used in human medicine for the diagnosis of infections in neonates and adults.5,6,10,12,16,17,23
In veterinary medicine, relatively few parameters are useful in the diagnosis of septicemia and in observing the response to treatment (e.g., C-reactive protein, 6 lactate, 18 haptoglobin, 25 and pro-inflammatory cytokines).17,19 These markers have a short half-life in plasma, poor specificity and poor response to bacterial infections, and limited stability in plasma. PCT has a long serum half-life (20–30 hr), is stable in blood specimens even at room temperature, unlike cytokines, and offers high sensitivity, no special conditions for storage, and a useful response to bacterial infections. 6 The objective of our study was to evaluate the diagnostic value of determining the serum levels of PCT, neopterin, tumor necrosis factor alpha (TNF-α), prostaglandin E2 (PGE2), malondialdehyde (MDA), interleukin 8 (IL-8), and IFN-γ in neonatal calves diagnosed with septicemic colibacillosis.
Our study was approved by the Animal Research Ethics Committee of Cumhuriyet University (Sivas, Turkey). Neonatal calves from dairy farms near Sivas, Turkey, which were admitted to the Internal Medicine Service of our animal hospital, were used in this study. Fifteen neonatal calves 2–10 days of age with septicemic colibacillosis and 15 clinically healthy age-matched control calves were included in this study. The diagnosis of E. coli septicemia was based on clinical findings and isolation of E. coli from heparinized blood. Calves suspected to be septicemic had variable body temperature (35.5–40°C), anorexia, weak or absent suckle reflex, conjunctival hyperemia, weakness, inability to stand, tachycardia, and hyperpnea on clinical examination. Control calves were determined to be healthy based on clinical findings and serum biochemical and hematological parameters within reference intervals.
Blood for biochemical analysis was taken from each calf by jugular venipuncture, allowed to clot, and then centrifuged at 4,000 rpm for 10 min. Sera were stored at −20°C until analyzed. Heparinized blood samples were taken to the laboratory immediately for isolation of E. coli and for hematological analysis with an automated hematology analyzer. a Blood samples were cultured on 5% blood agar, b MacConkey agar, b and in a 1:5 ratio in tryptic soy broth (TSB). c Cultures were incubated at 36°C with 5% CO2 for 1 week. Samples enriched in TSB c were passed to 5–7% blood agar, b eosin methylene blue (EMB) agar, b and MacConkey agar. b Identification of E. coli was performed according to colony morphology and Gram staining. 7 Colonies were identified as E. coli if they produced a distinctive metallic green sheen on EMB agar b plates, formed pink colonies on MacConkey agar b plates, were gram-negative, bacilli shaped, oxidase (–), catalase (+), motility (+/), glucose (+), lactose (+), O-nitrophenyl-β-galactoside (+), indole (+), methyl red (+), Voges Proskauer (–), citrate (–), H2S (–), and urease (–).
Serum concentrations of glucose, creatinine, total bilirubin, cholesterol, total protein, and albumin were determined using an automated biochemistry analyzer. d Serum, PCT, e IL-8, f and IFN-γ g concentrations were determined using a sandwich enzyme immunoassay, and serum neopterin, h TNF-α, i MDA, j and PGE2 k concentrations by a competitive inhibition enzyme immunoassay with an automated enzyme-linked immunosorbent assay reader. l
Systematic autopsies of the 4 calves that died during the study were performed, and samples of liver, kidney, heart, lung, intestines, and mesenteric lymph nodes were taken for histologic examination.8,14 Tissues were fixed in 10% buffered formalin. Paraffin blocks were prepared, sections were cut at 5–6 µm, and the sections were then stained with hematoxylin and eosin for histologic examination by light microscopy. 15
For the comparison of the studied parameters, Student t-test was used for parametric variables and chi-square test for nonparametric variables. Homogeneity of variances and intergroup comparisons were made through a software program. m
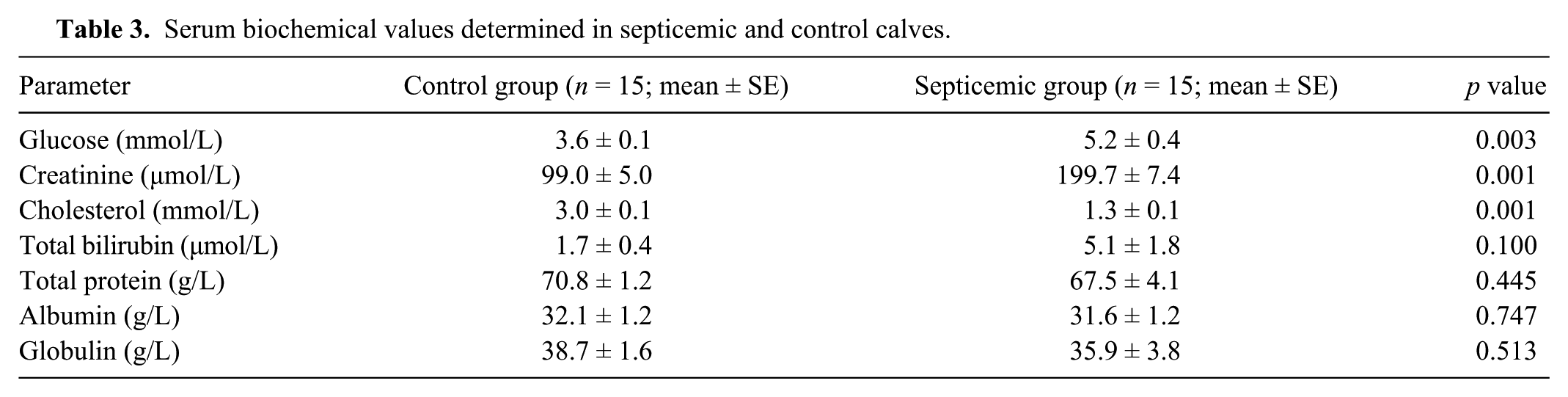
In the calves with septicemic colibacillosis, heart and respiratory rates were significantly higher compared to the healthy controls ( p < 0.01); however, body temperatures of the study groups were comparable ( p > 0.05; Table 1). The packed cell volume, leukocyte, and erythrocyte concentrations were significantly higher in the calves with septicemic colibacillosis compared with the healthy controls ( p < 0.01; Table 2). In the calves with septicemic colibacillosis, blood glucose and creatinine values were significantly higher compared with the healthy controls ( p < 0.01); however, the cholesterol value was significantly lower ( p < 0.01; Table 3).
Clinical examination findings in septicemic and control calves.

Hematological values determined in septicemic and control calves.
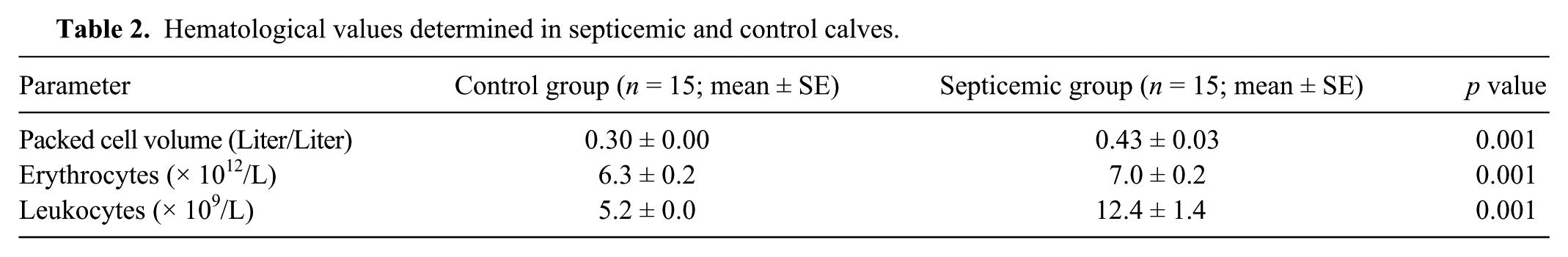
Serum biochemical values determined in septicemic and control calves.
PCT, neopterin, TNF-α ( p < 0.05), and IFN-γ concentrations were significantly higher in the calves with septicemic colibacillosis than in the healthy controls ( p < 0.01; Table 4). No statistical differences were found in PGE2, IL-8, and MDA levels between the study groups.
Serum procalcitonin, neopterin, tissue necrosis factor alpha (TNF-α), gamma interferon (IFN-γ), prostaglandin E2 (PGE2), malondialdehyde (MDA), and interleukin 8 (IL-8) values determined in septicemic and control calves.

Undetectable.
Autopsy findings in the 4 fatal cases were consistent with septicemic colibacillosis. There were petechial and ecchymotic hemorrhages on pleural and serosal surfaces. Alveolar septal thickening resulted from increased numbers of pulmonary macrophages and neutrophils. There was pulmonary congestion and serofibrinous exudation into alveoli. In the small intestines, lumens contained liquid content; mucosal tissues were congested and edematous; mucosal surfaces were covered with mucoid exudate. The liver was congested, and there was activation of Kupffer cells in the liver. There was petechial endocardial hemorrhage, kidneys were congested and swollen, and mesenteric lymph nodes were congested and edematous.
Procalcitonin appears to be a useful marker in neonatal calves with septicemic colibacillosis. In the current study, PCT concentrations were significantly higher in neonatal calves with septicemic colibacillosis compared with controls, and there was a positive correlation between PCT and the pro-inflammatory cytokines.
We found significant increases in TNF-α and IFN-γ concentrations in calves with septicemic colibacillosis, although the TNF-α concentration was lower than that found by others. 4 The increased level of IFN-γ was correlated with a high neopterin level, which may offer protection against some bacterial infections. 12
Of the studied biomarkers, only serum PCT, neopterin, TNF-α, and IFN-γ increased significantly in neonatal calves with septicemic colibacillosis. PCT in particular has merit as a diagnostic biomarker in neonatal calves with septicemic colibacillosis because it may increase more than 4 times, it has a long serum half-life, and is stable when held at room temperature.
Footnotes
Authors’ note
This study was presented at The World Congress on Controversies, Debates and Consensus in Veterinary Medicine (COVET), October 23–25, 2014, Prague, Czech Republic.
Authors’ contributions
All authors contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
BC2800 Vet, Mindray Medical International Ltd., Shenzhen, People’s Republic of China.
b.
Accumix, Ashby, MA.
c.
Acumedia, Lansing, MI.
d.
BS 200, Mindray Medical International Ltd., Shenzhen, People’s Republic of China.
e.
Procalcitonin ELISA kit (catalog number: CSB-E15004B), Wuhan Huamei Biotech Co. Ltd., Wuhan, Hubei Province, People’s Republic of China.
f.
IL-8 ELISA kit (catalog number: CSB-E13052B), Wuhan Huamei Biotech Co. Ltd., Wuhan, Hubei Province, People’s Republic of China.
g.
IFN-γ ELISA kit (catalog number: CSB-E09812b), Wuhan Huamei Biotech Co. Ltd., Wuhan, Hubei Province, People’s Republic of China.
h.
Neopterin ELISA kit (catalog number: CSB-EQ027403BO), Wuhan Huamei Biotech Co. Ltd., Wuhan, Hubei Province, People’s Republic of China.
i.
TNF-α ELISA kit (catalog number: CSB-E12020B), Wuhan Huamei Biotech Co. Ltd., Wuhan, Hubei Province, People’s Republic of China.
j.
MDA ELISA kit (catalog number: CSB-E14000B), Wuhan Huamei Biotech Co. Ltd., Wuhan, Hubei Province, People’s Republic of China.
k.
PGE2 ELISA kit (catalog number: CSB-E14237B), Wuhan Huamei Biotech Co. Ltd., Wuhan, Hubei Province, People’s Republic of China.
l.
Thermo Multiskan, Thermo Fischer Scientific Oy, Vantaa, Finland.
m.
SPSS 14.0 for Windows, SPSS Inc., Chicago, IL.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Scientific Research Projects Commission of Cumhuriyet University under project V-013.
