Abstract
A 23-year-old captive-bred red-tailed guenon (Cercopithecus ascanius) with a brief history of inappetence, lethargy, and seizures was submitted for necropsy. On postmortem examination, multiple cryptococcomas were identified in brain and heart. Cryptococcus neoformans organisms were also identified microscopically in kidney, eye, and pancreas. Fungal yeast formed rare pseudohyphae. The histologic diagnosis of cryptococcosis was confirmed by a positive test for C. neoformans antigen in a serum sample. Immunohistochemical staining confirmed that macrophages were the principal inflammatory cell in brain lesions and often contained phagocytosed yeast. As disseminate cryptococcosis is often associated with immune suppression, serology and immunohistochemical staining for simian immunodeficiency virus were performed but showed no evidence of SIV infection.
The dimorphic fungal pathogen Cryptococcus neoformans frequently invades the central nervous system with a propensity for the meninges in many mammalian species, including dogs, cats, horses, and primates. In contrast, systemic cryptococcosis is rare, primarily found in the context of immunosuppression or advanced neoplasia. 7 In a study of humans infected with the human immunodeficiency virus (HIV) that have progressed to acquired immunodeficiency syndrome (AIDS), C. neoformans infection was identified in 4.2% of patients and affected an average of 3.8 organs per patient, most commonly including meninges followed by brain, lung, lymph nodes, liver, spleen, kidney, adrenal gland, heart, and skin. 2, 6
A 23-year-old female captive-born, red-tailed guenon (Cercopithecus ascanius), a monkey species indigenous to West Africa, developed progressive lethargy, decreasing appetite, seizures, and finally blindness over a 5-day period. The animal was euthanized due to poor prognosis. On postmortem examination, gross lesions were identified in the brain and heart. The right caudal parietal and occipital lobes contained 1-cm diameter gelatinous cystic areas replacing cortical gray matter and extending into subjacent white matter, consistent with a diagnosis of multiple central nervous system (CNS) cryptococcomas. Overlying meninges appeared grossly normal (Fig. 1). In the heart, the interventricular septum contained a focal gelatinous nodule measuring 1 cm in diameter resembling the brain lesions in character (Fig. 2).
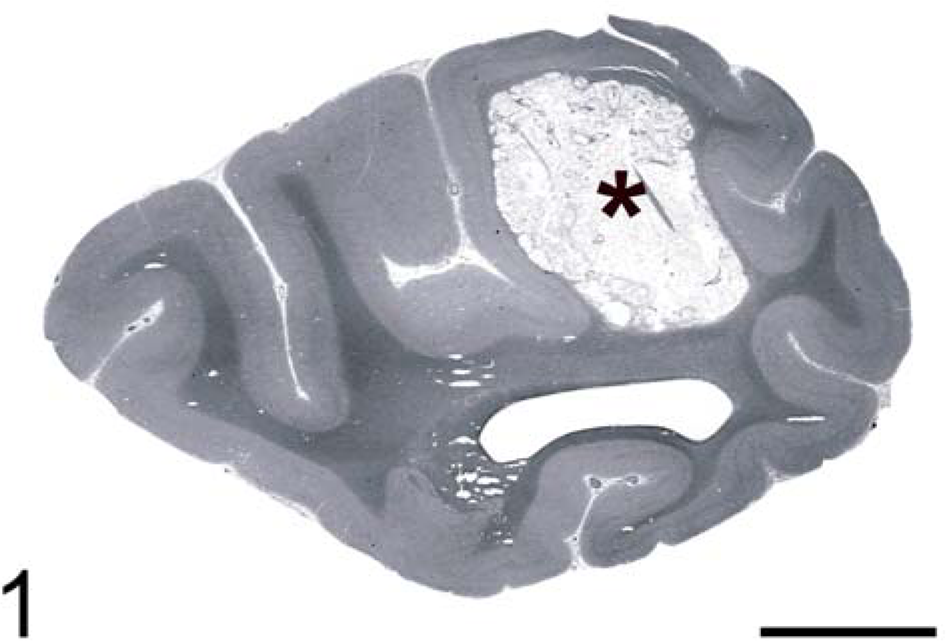
Brain, occipital cortex. Red-tailed guenon. Grossly visible cryptococcoma (∗). HE. Bar = 5 mm.
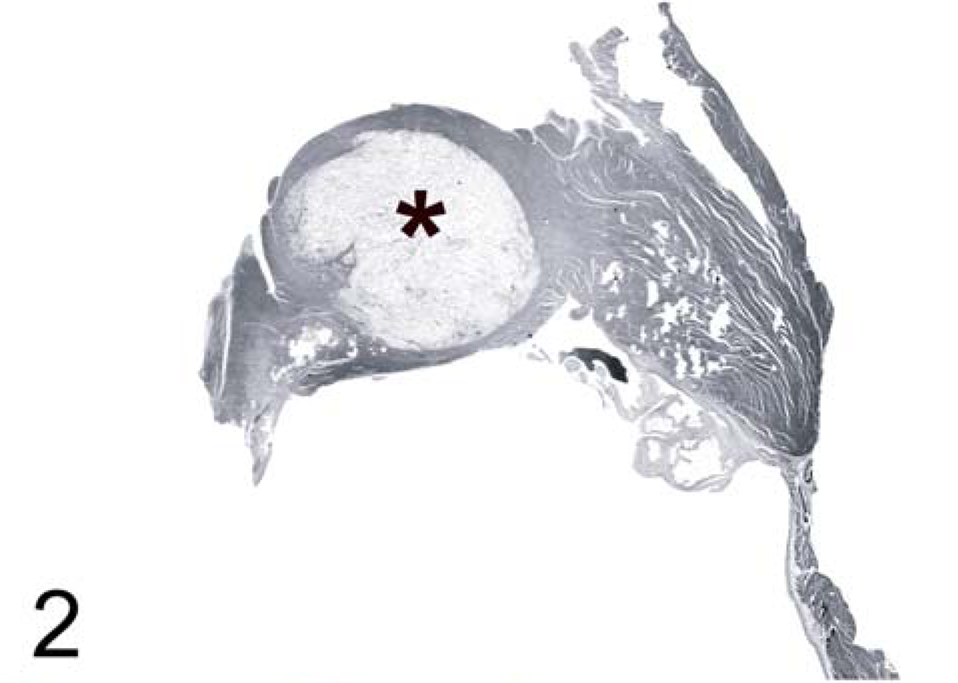
Heart, interventricular septum. Red-tailed guenon. Cryptococcoma in interventricular septum adjacent to tricuspid valve (∗). HE.
In the brain, microscopic examination revealed abundant intracellular and extracellular yeast forms accompanied by scattered inflammatory infiltrates composed predominantly of macrophages and occasional lymphocytes in a loose gelatinous matrix replacing CNS parenchyma (Fig. 3). The meninges also contained scattered yeast forms accompanied by occasional lymphocytes and plasma cells. Yeast forms were round to lemon shaped and varied in size from 7–20 × 10–30 um. Yeast contained homogenous amphophilic cytoplasm with a 5-µm-diameter eosinophilic nucleus. Yeast walls were thin and surrounded by a clear pericellular zone indicative of a thick capsule, consistent with C. neoformans. 6 Mucicarmine red staining confirmed the presence of a thick capsule around the yeast (Fig. 4). Gomori methenamine silver (GMS) staining highlighted narrow-based budding (Fig. 5). The presence of extracellular yeast forms in a gelatinous matrix accompanied by a modest inflammatory infiltrate is consistent with C. neoformans infection. 12 In this case, there also were rare instances of cryptococcal organisms forming pseudohyphal structures (Fig. 4). While pseudohyphae are not commonly seen, they have previously been reported in cytology specimens from cases that have been positive for C. neoformans by culture. 5
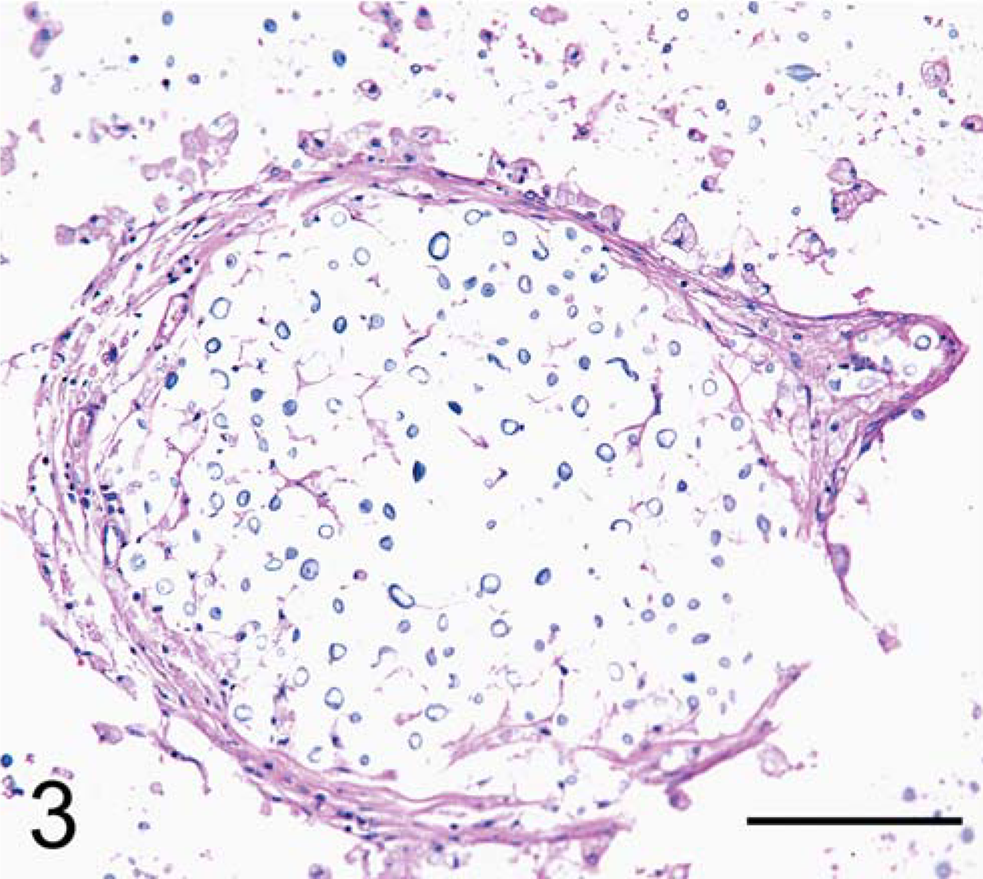
Brain. Red-tailed guenon. Numerous C. neoformans organisms within gelatinous matrix surrounded by macrophages. HE. Bar = 100 µm.
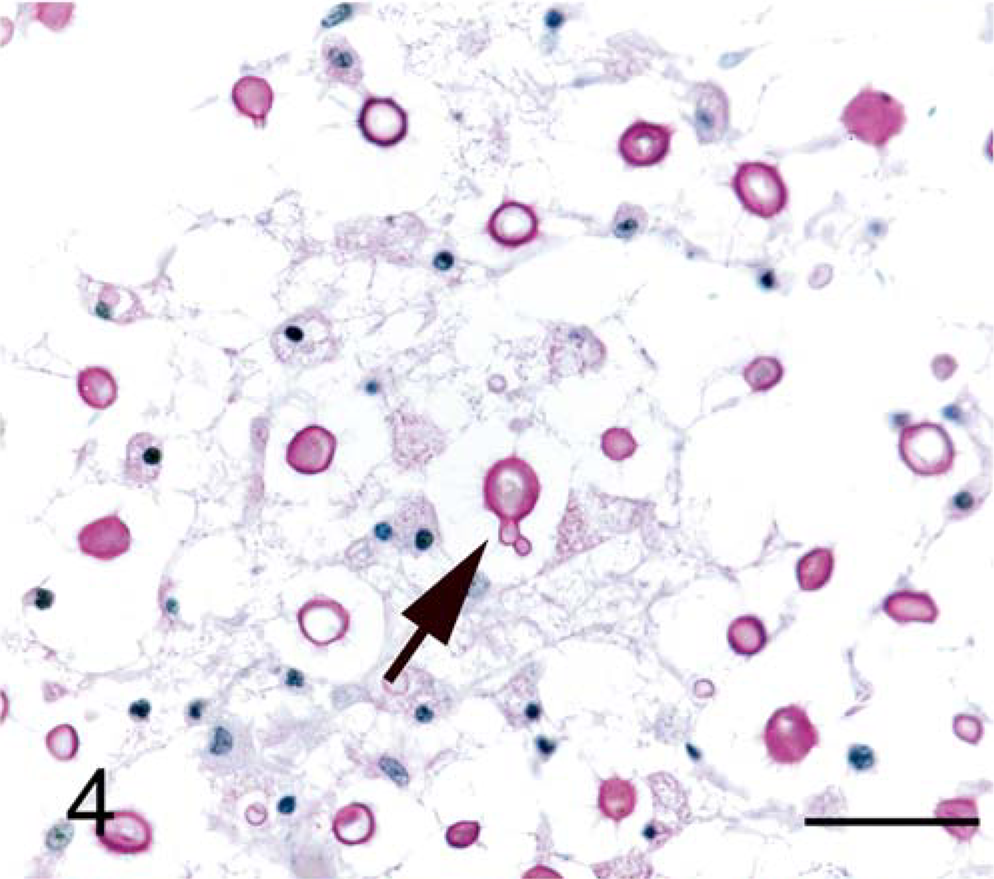
Brain. Red-tailed guenon. Positive staining of polysaccharide capsule with mucicarmine. Rare pseudohyphae are present (arrow). Mucicarmine red stain. Bar = 30 µm.
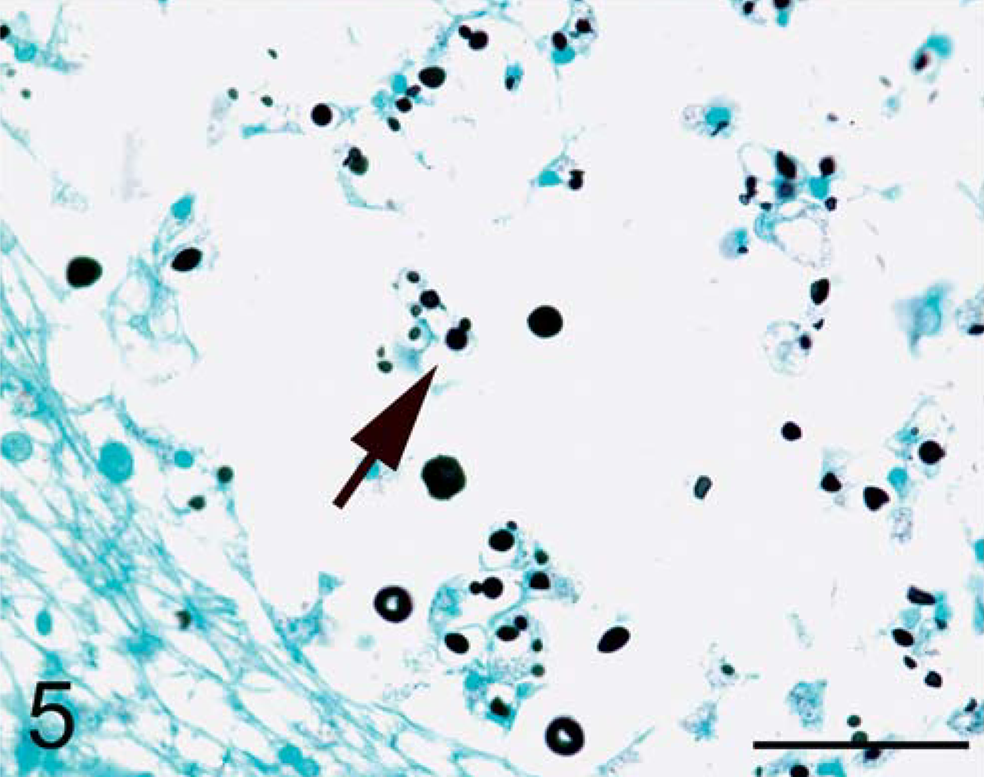
Brain. Red-tailed guenon. Narrow-based budding of C. neoformans visible with GMS (arrow). Gomori methenamine silver stain. Bar = 50 µm.
Immunohistochemical staining confirmed that macrophages were the principle inflammatory cell in brain lesions and often contained phagocytosed yeast (Fig. 6). Primary antibody against CD68 was used for detection of macrophage infiltration (KP-1, diluted 1 : 2,000; DAKO, Carpinteria, CA). Formalin-fixed, paraffin-embedded brain tissue sections were deparaffinized, rehydrated, and then postfixed for 20 minutes. After rinsing in water, tissues were heated in a microwave in sodium citrate buffer (0.01 M, pH 6.0) for 8 minutes to retrieve antigen. Endogenous peroxidase was quenched with 3% H2O2 for 10 minutes and then sections were blocked with buffered casein for 10 minutes. Primary antibody was applied to tissue sections for 60 minutes at room temperature, the tissues were washed in buffer, and then secondary biotinylated multilink antibody (Biogenex, San Ramon, CA) was added for 20 minutes. After washing, streptavidin-horseradish peroxidase was applied for 20 minutes, followed by diaminobenzidene tetrahydrochloride in buffer containing H2O2 for 10 minutes. After hematoxylin counterstain, sections were then washed, dehydrated, and coverslipped.
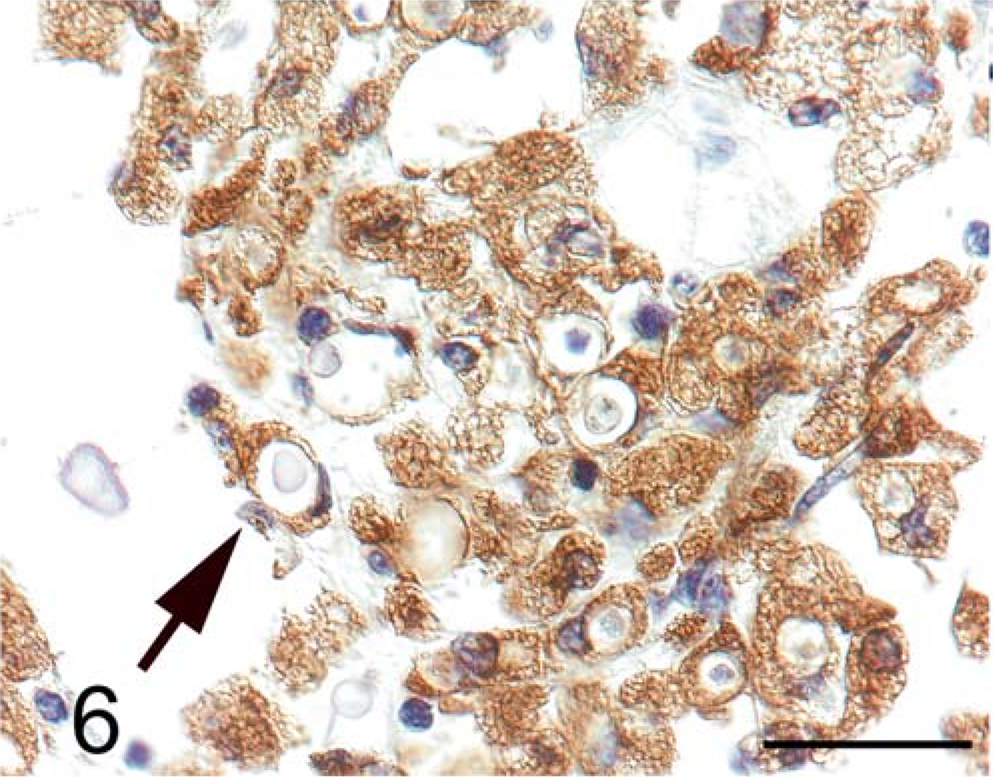
Brain. Red-tailed guenon. C. neoformans organisms are present within macrophages staining positive for CD68 (arrow). Immunohistochemistry for CD68. Bar = 30 µm.
Numerous extracellular yeast forms also were present within the interventricular septum of the heart (Fig. 2). Abundant fungal organisms separated and replaced myofibers, spanning the entire septum from right to left ventricle, with few intact myofibers remaining on either side of the nodule. The organisms were accompanied by scattered macrophages and lymphocytes as well as mild hemorrhage. Myocyte degeneration was focally extensive within the interventricular septum. Fungal organisms extended from the interventricular septum into the base of the tricuspid valve.
Although gross lesions were only identified in the brain and heart, fungal organisms were also found in the pancreas, kidney, and the sclera of the eye by microscopic examination. Clusters of yeast were present in the exocrine pancreas with no associated inflammatory response. The cortex of the right kidney contained multifocal clusters of yeast located within clear spaces that were surrounded by macrophages and lymphocytes enclosed by a dense fibrovascular stroma. Within the eye, a focal accumulation of yeast was identified in the sclera with associated lymphoplasmacytic inflammation. While cyst-like lesions induced by C. neoformans, often called cryptococcomas, are most common in the brain, they also can be found in other organs. 6 In previous reports of cryptococcosis in nonhuman primates, the brain, meninges, eye, thyroid gland, salivary gland, lung, heart, pericardium, liver, spleen, pancreas, small intestine, adrenals, kidney, blood vessels, subcutis, and spinal cord have all been reported to be affected. 4, 10– 12
The histologic diagnosis of cryptococcosis was confirmed by a positive test for C. neoformans antigen in a serum sample from this guenon (Meridian, Cincinnati, OH). In contrast, four clinically normal cage mates of this animal, including two other red-tailed guenons (Cercopithecus ascanius) and two black and white colobus monkeys (Colobus guereza), were negative for C. neoformans antigen in serum. Given that simian immunodeficiency virus (SIV) infection has been reported in guenons, we also tested this animal for SIV. Although SIV induces profound immunodeficiency in many Asian-origin macaque species, rare cases of immunodeficiency have also been reported in SIV-infected African nonhuman primates. 8 In this case, we did not detect SIV infection either by enzyme-linked immunosorbent assay for SIV antigen in plasma (Coulter, Hialeah, FL) or by performing immunohistochemistry for SIV on spleen and mesenteric lymph node sections using the anti-SIV antibody KK41 (diluted 1 : 400; AIDS Reagent Program, Bethesda, MD, per the protocol described previously for CD68 immunostaining). Whether this animal was immunocompromised as a consequence of advanced age is unknown; however, the presence of numerous macrophages capable of yeast phagocytosis in the brain suggests the capability of mounting innate immune responses. In addition to Cryptococcus-associated lesions, multifocal pancreatic islet amyloidosis was present. As diabetes mellitus has been reported to be a risk factor for development of cryptococcosis in humans, we examined blood glucose levels reported in this animal's medical history but did not find evidence of hyperglycemia. Other predisposing factors in non-AIDS human patients include steroid therapy, sarcoidosis, and lymphoma, but up to 50% of non-AIDS patients with cryptococcosis have no clear predisposing factor. 6
Disseminated cryptococcosis is rarely reported in animals and to date has been reported in less than 10 nonhuman primate species. 1, 3, 4, 9– 12 In this case, the presence of multiple extensive cryptococcomas within the brain and heart is unusual given that overt immunosuppression was not apparent. Although the animal was leukopenic 5 months prior to death, the leukogram was within normal limits 1 week prior to euthanasia. Whether immune system deficiencies were present due to the advanced age of this animal, being well past the life expectancy for this species, remains unknown. 10 Although neural involvement is most common, a significant proportion (up to 24%) of immunosuppressed individuals with systemic cryptococcosis present with extraneural involvement, similar to the lesion distribution in this case. 2 The pathology findings in this case are consistent with the observed clinical signs as cerebral cryptococcosis often manifests as fever, headache, neck stiffness, altered mental status, vomiting, asthenia, anorexia, cough, cranial nerve palsies, dyspnea, and seizures. 2
Footnotes
Acknowledgements
We are grateful to Ms. Patricia Wilcox for her histotechnology expertise, Ms. Suzanne Queen for immunohistochemistry, Dr. William Meri for antigen testing, Dr. M. Chris Zink for manuscript review, and Dr. Cory Brayton for photographic assistance. KLH was supported by NIH RR07002.
