Abstract
The present study describes immunophenotypic characteristics of inflammatory infiltrate in the skin and lung of lambs naturally infected with sheeppox virus (SPV). Three lambs revealed typical cutaneous and pulmonary lesions of sheeppox. Histologically, cutaneous and pulmonary lesions consisted of hyperplastic and/or degenerative changes in the epithelium with mononuclear cells, neutrophils, and typical sheeppox cells (SPCs), which had a vacuolated nucleus and marginated chromatin with occasional granular intracytoplasmic inclusions. The inflammatory infiltrate in pox lesions in both skin and lung was characterized by the presence of MHC II+ dendritic cells, CD4+, CD8+, γδ+ T cells, IgM+ cells, and CD21+ cells. Loss of expression of MHC I and MHC II antigens was observed in the affected areas of skin and lung. SPCs, stained with anti-SPV antibody, were also positive for CD14 and CD172A, antigens expressed on monocytes and macrophages. CD14 and CD172A negative SPCs were considered to be SPV infected degenerated epithelial cells or fibroblasts.
Sheeppox (SP) is a contagious disease of sheep caused by sheeppox virus (SPV), a member of the genus Capripox in the family Poxviridae. It exists currently in Africa, Asia, and the Middle East, whereas the disease is exotic to America, Australia, and New Zealand. The disease occurs in all ages of sheep, but is most severe in lambs, with mortality reaching 80–100%. 23 In SP, the most prominent pathological change is the accumulation of sheeppox cells (SPCs), Cellules claveleuses of Borrel, with inflammatory cells in the skin, respiratory tract, and other sites. SPCs have a distinctly vacuolated nucleus and marginated chromatin and occasionally show granular intracytoplasmic inclusions. Electron microscopic studies revealed that SPCs infected with virus are monocytes, macrophages, and fibroblasts, but not endothelial cells. 2, 18, 20, 23 SPCs have been regarded as sites of replication of SPV. 18, 20 Furthermore, immunohistochemical examination has shown positive labeling of poxvirus antigen only in SPCs, and in virus-infected epithelial cells resembling SPCs in the various tissues, such as the epidermis and bronchi. 9 The aim of the present study was to characterize the immune response to sheeppox virus infection in the skin and lung of lambs naturally infected with SPV and to determine the antigenic phenotype of SPCs.
Three native White-Karaman lambs, between 1 and 2 months of age, were obtained from outbreaks of SP seen in eastern Turkey. These flocks had not been vaccinated against SPV. The lambs were euthanized at the terminal stage of the illness. Necropsy was performed on the lambs. Tissue specimens were collected from skin (axillary region), lung, and then fixed in buffered formalin. Tissues were embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin (HE). Additional tissues were fresh frozen or fixed in zinc salt fixative (ZSF). 7 ZSF-fixed tissues were sectioned as described and deparaffinized for immunohistochemistry. Cryostat sections of fresh frozen tissues were cut at 5 µm and mounted onto organosilane (3-aminopropyl) triethoxysilane-coated glass slides and immediately fixed for 8 minutes in ice-cold absolute ethanol.
Monoclonal antibodies (mAb) used in this study were obtained from Washington State University, Monoclonal Antibody Center, Pullman, WA. The specificities included anti-ovine major histocompatibility complex (MHC) Class I (H58A, MHCl I) expressed on most nucleated cells, with exception of neural cells; anti-ovine MHC Class II (TH14B, MHCl II) expressed on B cells, activated T cells, and dendritic cells (DC); anti-ovine CD1 (TH97A) expressed on thymocytes and a subset of DCs; anti-ovine CD5 (ST1) expressed on all T cells and a subset of B cells; anti-ovine CD2 (MUC2A) expressed on αβ T cells; anti-ovine CD4 (GC1A) expressed on T-helper cells; anti-ovine CD8 (CACT80C) expressed on T-cytotoxic cells; anti-ovine WC1-N2 (BAQ4A) expressed on a major subset of γδ T cells present in ruminants; anti-B cells (BAQ44A and GB26A); anti-ovine CD21 (GB25A) expressed on mature B cells and follicular DCs; anti-ovine sIgM (PIG45A2) expressed on B cells; anti-ovine CD14 (CAM36A) expressed on monocytes and macrophages; anti-ovine CD172A (DH59B) expressed on granulocytes, monocytes, macrophages, and a subset of DCs. The polyclonal rabbit hyperimmune serum used to detect SPV antigen was developed in two adult rabbits against a commercial attenuated sheeppox vaccine (Poxvac™, Vetal A.S., Lab., Turkey) as previously described. 9
Both frozen and deparaffinized sections were prepared using a commercial streptavidin-biotin peroxidase system (DAKO, Carpinteria, CA). The frozen sections were immediately air dried, using a hair dryer (cold air) to avoid autolysis of cells, and then kept at −20°C. All sections were preincubated in 10% normal goat serum for 30 minutes at room temperature (RT) to block nonspecific binding of second-step antibody. Sections were reacted with primary antibodies overnight at 4°C and rinsed with phosphate-buffered saline (PBS), pH 7.4, at RT. The sections were reacted with biotin-conjugated second-step antibody (DAKO) for 10 minutes at room temperature and then rinsed in PBS at RT. For inactivation of endogenous peroxidase, sections were incubated in 0.3% H2O2/methanol for 60 minutes. The sections were then rinsed with PBS at RT and reacted with streptavidin-biotin-peroxidase complex (SABC) (DAKO) for 10 minutes at RT. After another washing with PBS, sections were incubated with 3-amino-9-ethylcarbazole (AEC) (DAKO) or diaminobenzidine (DAB) (DAKO) for 15 minutes and then counterstained with Mayer's hematoxylin. Double immunostaining was used to characterize the phenotypic features of SPV-infected SPCs. Sections were incubated with 10% normal goat serum for 30 minutes. The double staining included two stages. In the first stage, tissue sections were incubated with the polyclonal rabbit anti-SPV antibody for 3 hours at RT. After washing for 5 minutes in PBS, an alkaline phosphatase (AP)-labeled secondary rabbit-anti-mouse antibody (DAKO) was added for 1 hour at 37°C. After washing for 5 minutes in PBS, fast red was added and allowed to develop for 20 minutes. Sections were washed in PBS. In the second stage, sections were again incubated with 10% normal goat serum for 30 minutes, followed by subsequent incubation with mAb specific for CD14 and CD172A. The tissue sections were incubated overnight at 4°C. The sections were washed for 5 minutes in PBS and then incubated with SABC (DAKO) for 10 minutes at RT. After washing, the tissues were incubated for 20 minutes in DAB. Mayer's hematoxylin was used for counterstaining. Sections were mounted in aqueous mounting medium (Shandon, Pittsburgh, PA).
Negative controls included tissues incubated with an unrelated mAb or normal rabbit serum and second-step antibodies. Labeling of normal uninfected tissues from areas adjacent to the lesions, known to express the relevant tissue antigens, served as positive controls in place of normal tissue from uninfected animals.
The lambs presented with clinical signs of fever, seromucous nasal discharge, dyspnea, and typical discrete circumscribed lesions of pox. All lambs examined had skin lesions consisting of irregular, raised, and top-flattened papules. The lesions were predictably more severe and numerous in the areas of skin where the wool or hair was shortest, such as eyelids, cheeks, nostrils, ears, axillae, abdomen, thigh, udder, and ventral surface of the tail. The typical vesicular or pustular stages for poxvirus infections were not seen in any of the lambs. The subcutaneous tissues were very edematous. The pulmonary lesions consisted of subpleural whitish, multiple nodules, 2–3 cm in diameter on the dorso-caudal lobes of the lung. There was slight congestion around these lesions.
Histologic examination of the epidermis with HE staining revealed localized acanthosis with para and/or orthokeratotic hyperkeratosis, with occasional rete ridges. There were varying degrees of degenerative changes, ranging from hydropic-reticular keratinocytes to microvesiculation. Intracytoplasmic eosinophilic inclusion bodies were also present in most of the degenerated keratinocytes. The edematous underlying dermis and subcutis contained many aggregates of mononuclear cells and neutrophils, and loosely or scattered typical SPCs (Cellules claveleuses of Borrel) around the fragmented collagen bundles and blood vessels. SPCs had vacuolated nuclei with margination of nucleoli and chromatin. Their cytoplasm had a basophilic appearance. Intracytoplasmic eosinophilic inclusion bodies were seen in most of these cells. Many hair follicles and sebaceous glands also showed changes similar to those observed in the epidermis.
In the lungs, epithelial hyperplasia of the alveoli, bronchi, and bronchioli was detected. Many alveoli contained eosinophilic material and cubic-columnar epithelial cells, many neutrophils, macrophages, and SPCs. Mononuclear cells, SPCs, a few neutrophils, and karyorrhectic cellular debris were also detected in the peribronchial, peribronchiolar, and perivascular sheaths. The cytoplasm of degenerated bronchiolar epithelial cells and many SPCs had eosinophilic inclusion bodies. Mild hyperplasia and degeneration of the epithelium of trachea were observed. Examination of the submucosa revealed limited infiltration of lymphocytes and macrophages.
Some vessels in the skin and lung, particularly the venules and arterioles, showed vasculitis or perivasculitis accompanied with varying degrees of extensive necrosis in their walls that extended into the connective tissues. Usually, macrophages, lymphocytes, plasmacytes, and neutrophils were seen within and around these vessels. Several SPCs were observed in the walls of some affected arterioles. The degenerate epidermal cells and bronchial epithelium with typical pox lesions exhibited the same morphological features as SPCs, especially with the distinctive vacuolation of the nucleus.
In the skin, minimal epithelial changes, including mild acanthosis without degeneration of the epithelium, were considered to be early stages of lesion development, whereas epithelial degeneration with rete ridge formation and inclusion bodies (considered to be hallmarks of poxvirus-induced epithelial changes) were considered to be at the late stages of lesion development. Such distinguishing stages of lesion formation could not be made in the lung.
Optimal immunostaining was obtained by incubation with primary antibodies overnight at 4°C on both the frozen and ZSF-processed sections.
In the epidermis in early lesions, many keratinocytes had positive staining for MHC I. In the late lesions, MHC I immunostaining of the degenerated and hyperplastic epidermis was weak or absent. The labeling was localized to the keratinocyte membranes (Fig. 1). The hair follicles revealed little or no peripheral immunostaining, whereas nerves, capillary, endothelium, sweat, and sebaceous glands had strong immunostaining. Large populations of MHC I+ cells with lymphocyte or DC morphology were present in the perivascular and perifollicular spaces scattered throughout the dermal and subcutaneous tissues. Many MHC I+ SPCs were also found among the inflammatory infiltrates, especially in the dermis and occasionally in the subcutis. In the lung, intense labeling with MHC I was seen on normal intact bronchial epithelium and on round-spindle or DCs in the interalveolar tissues. Immunostaining was absent on many SPCs and interstitial tissues in the center of typical pox lesions, excluding some scattered individual round cells and bronchial epithelium that exhibited complete or occasional discontinuous labeling. The Bronchus-associated Lymphoid Tissue (BALT), inflammatory cells, and tissues lining the pox lesion exhibited strong immunostaining for MHC I. Table 1 shows the number of SPCs labeled with antibodies in skin and lung tissue sections.
The number of SPCs immunostained by antibodies in skin and lung-tissue sections of lambs infected with sheeppox virus.∗
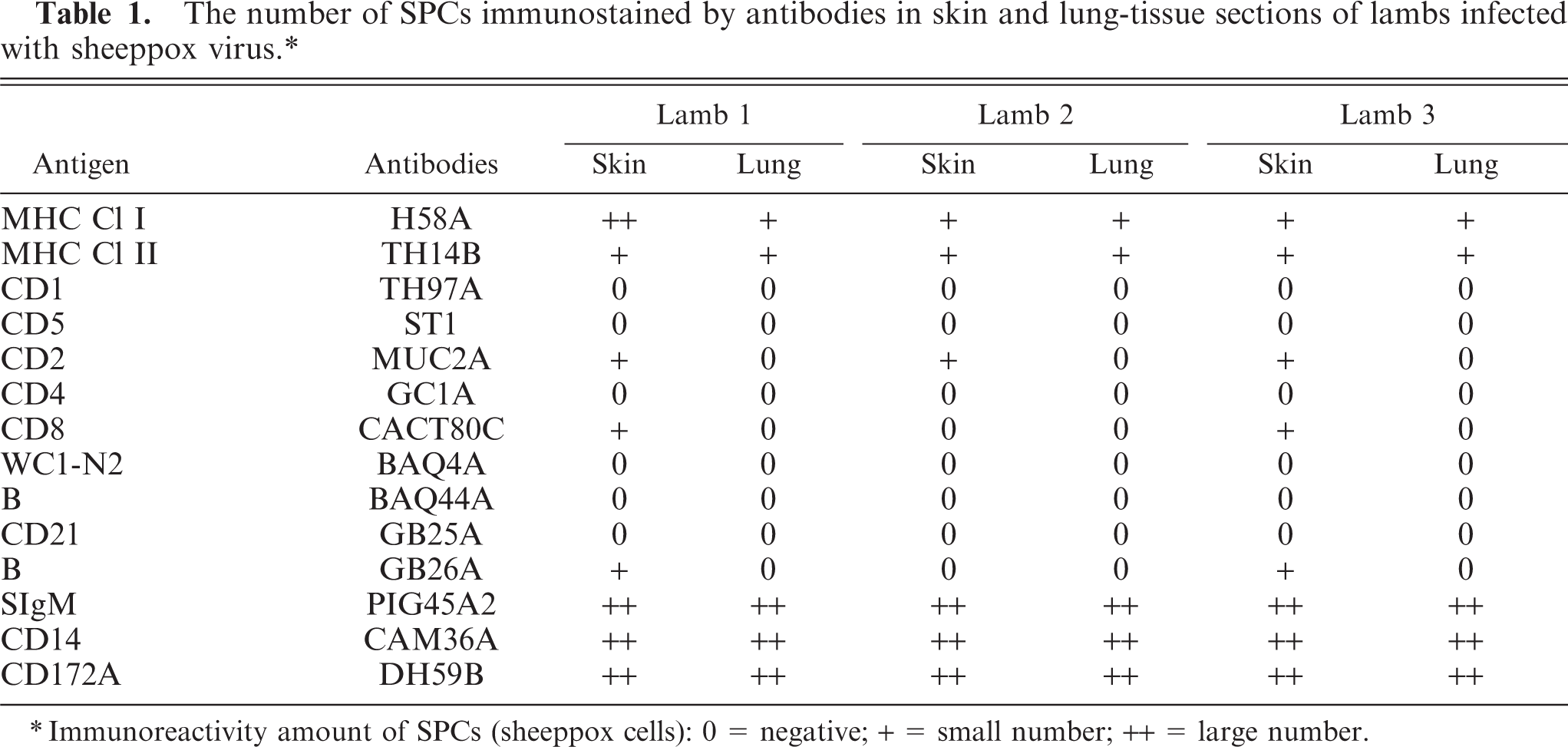
∗Immunoreactivity amount of SPCs (sheeppox cells): 0 = negative; + = small number; ++ = large number.
Skin; lamb No. 2. The loss of MHC I expression in the affected epidermis. Note dense MHC I expression in the stratum corneum. Arrows indicate epidermal-dermal junction. Immunostaining for MHC I. Frozen; SABC/AEC, hematoxylin counterstain. Bar = 100 µm.
MHC II+ cells had DC morphology and were prominent among the keratinocytes in early lesions, mostly at the transition of the basal to the suprabasal layer. The dense accumulation of MHC II+ cells with mostly dendritic and occasional lymphocyte morphology were found in the superficial dermis associated with vasculitis and neighboring adnexal elements at the dermoepidermal junction (Fig. 2). An increase of MHC II+ cells was also noted in the deep dermis and subcutis. Few dermal macrophages resembling SPCs were positive for MHC II antigen, with staining being more distinctive in the ZSF-processed tissues. The frequency of MHC II+ cells with DC morphology in severely affected epidermis was significantly decreased compared with the unaffected or minimally hyperplastic epidermis. In the lung, MHC II expression was found in cells with dendritic and mononuclear cell morphology throughout the intact tissues, the peripheral tissues surrounding the pox lesion, BALT, and occasionally within the normal or mildly hyperplastic bronchiolar epithelium. There was minimal or no expression of MHC II in the center of pox lesions. The majority of lymphocytes present in the periphery of pox lesions exhibited high expression of MHC II antigen, whereas few SPCs were positive for MHC II (Fig. 3).
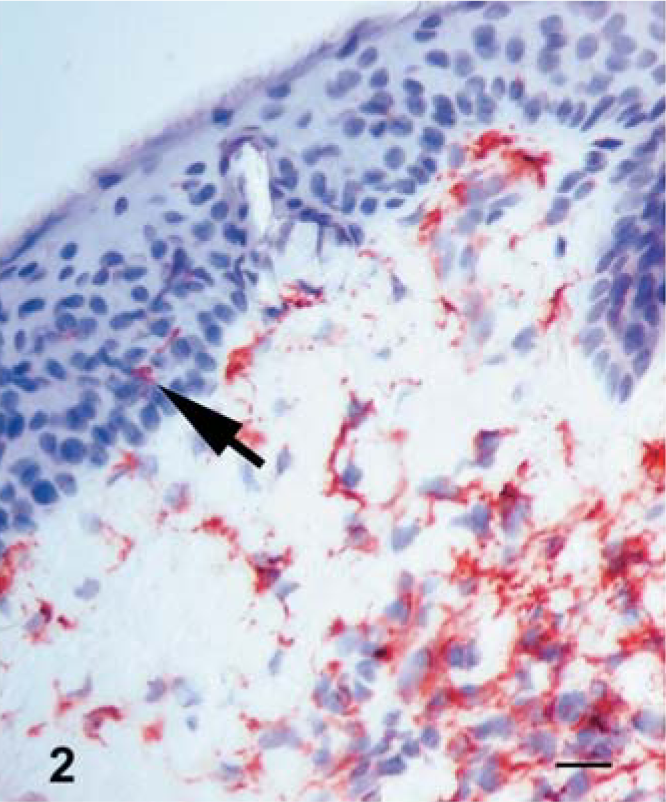
Skin; lamb No. 2. Dermal MHC II expressing dendritic cells. Hyperplastic epidermis included few MHC II+ dendritic cells (arrow). Note dendritic cells at the dermoepidermal junction. Frozen; SABC/AEC, hematoxylin counterstain. Bar = 100 µm.
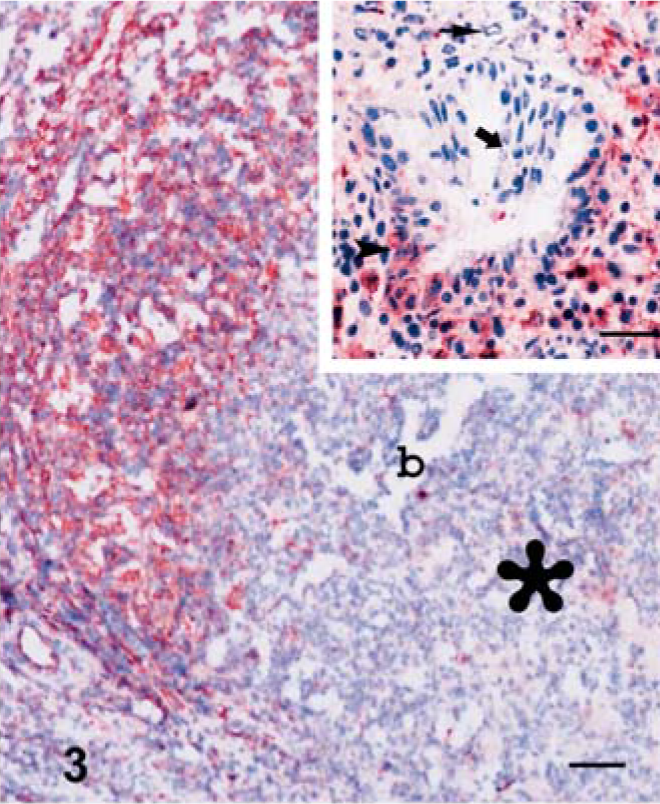
Lung, lamb No. 2. Note dense immunostaining at the periphery of pox lesion (∗), whereas weak immunostaining at its center for MHC II (b = bronchiole). ZSF; SABC/DAB, hematoxylin counterstain. Bar = 150 µm. Inset: MHC II negative SPC (thin arrow) and also positive (arrowhead) and negative (thick arrow) affected bronchial cells. ZSF; SABC/AEC, hematoxylin counterstain. Bar = 100 µm.
Some CD1-immunostained cells, which had dendritic or mononuclear morphology, were observed in the epidermis and dermoepidermal junction of early lesions. Cells in advanced pox lesions of the epidermis exhibited weak or no immunostaining for CD1. A few CD1+ cells with lymphocyte morphology were scattered throughout dermal and subcutaneous tissues. In the lung, CD1 labeling was detected in the lymphoid cells, especially in BALT, and occasionally in the interstitium of intact tissues or pox lesions. The DCs, which were positive for MHC II, were negative for CD1. The CD1+ cells that infiltrated lung tissues had a round-cell morphology. These cells were more numerous in the lung than in the skin. SPCs were negative for CD1.
In the epidermis, CD5+ cells were not found in early or late pox lesions. CD5+ cells were diffusely or focally concentrated throughout the dermis and subcutaneous tissues. In the lung, CD5+ cells were scattered in the interstitial tissues and within pox lesions, whereas they were locally concentrated in the BALT. SPCs in the lung and skin were CD5 negative. In the skin including late lesions, a small number of CD2+ cells were detected in the hyperplastic and degenerate epidermis. Immunostaining quality of the CD2+ cells revealed characteristics similar to that obtained with CD5. However, few SPCs were positive for CD2 antibody in the skin. In the lung, CD2+ cells were located at the periphery of pox lesions and distributed in the BALT and throughout the lung tissues. In contrast with the skin, no CD2+ SPCs were observed in the lung tissues.
A small number of both CD4+ and CD8+ cells were detected in the areas of epidermal changes in late pox lesions. Early lesions in the epidermis contained both T cell subsets. Numerous CD4+ and CD8+ positive cells with a similar distribution pattern were observed in the dermal and subcutaneous tissues. The frequency of both cell subsets was highest around blood vessels and the adnexa, especially in the MHC I- and MHC II-positive areas. Few cells with SPCs morphology were CD8+. None of these cells were positive for CD4. The frequency of CD4+ and CD8+ positive cells in the lung was high and similar to their frequency in the skin. Many CD4+ and CD8+ cells were present in BALT and interstitial tissue.
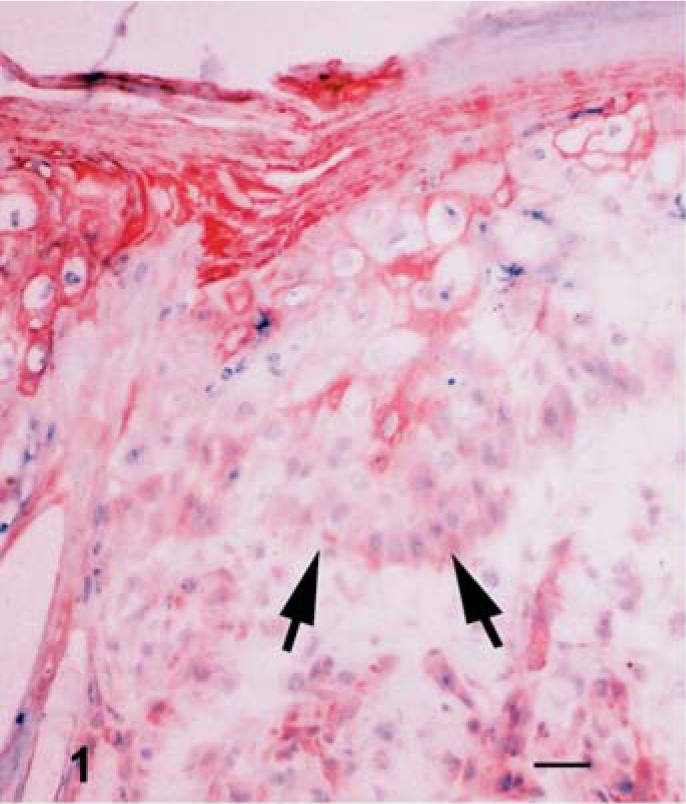
Many γδ+ T cells (WC1-N2+) were observed in small and large clusters in areas associated with dermal vessels, adnexa, axon and subcutaneous blood vessels, and fat tissues. In early lesions, a small number of γδ+ T cells were observed in the same cutaneous area containing other T cell subsets. The degenerate epithelium contained occasional γδ+ T cells, DC, and lymphocytes. A few scattered γδ+ T cells were present in the basal layers of the epidermis (Fig. 4). These cells were predominantly located in the superficial dermis, especially in dense areas of MHC I+, MHC II+, CD4+, CD8+ cells, but they were also present in the deep dermis and subcutis. In the lung, they were concentrated in the BALT and the periphery of pox lesions. Cells positive for WC1-N2 (γδ T cell subset) were scattered in areas that contained large numbers of other lymphocyte subsets in BALT and interstitial tissue. Some γδ+ T cells were present in and just beneath the hyperplastic bronchial epithelium. No positive immunostaining by WC1-N2 was observed on SPCs in the skin and the lung.
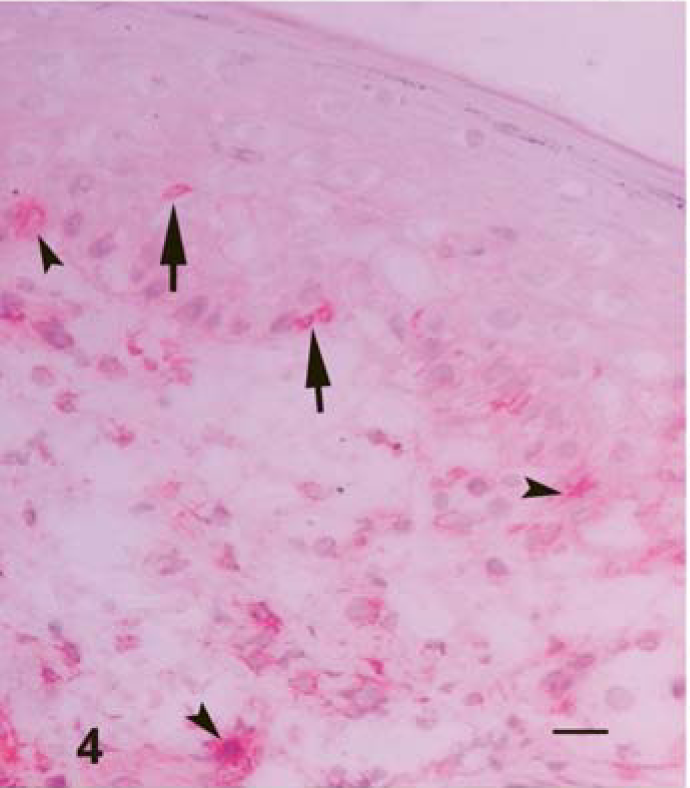
Skin; lamb No. 3. γδT cells (arrows), some of which have dendritic morphology (arrowheads), infiltrating into the affected epidermis and in the dermis. Frozen; SABC/DAB, hematoxylin counterstain. Bar = 100 µm.
In the skin, the best results for immunostaining B cells were obtained with the mAb GB26A as compared with staining with GB25A and BAQ44A. GB26A+ and BAQ44A+ and GB26A+ cells with lymphocyte morphology were present in small numbers scattered around the dermal and subcutaneous blood vessels, adnexal elements, and among the infiltrating cells underlying the lesion, whereas GB25A(CD21) +cells with dendritic or lymphocytic morphology were located in the superficial dermis or degenerated epidermis. In the lung, B cells labeled by mAbs BAQ44A and GB26A were located generally in the peribronchialar and perivascular cuffs. A few positive cells were present in the hyperplastic epithelium and interstitium. Numerous CD21+cells, in contrast with those in the skin, had dendritic or round and small cell morphology. These cells were dispersed throughout the lung tissue and at the periphery of pox lesions. A few CD21+ small and round cells were detected in the center of pox lesions (Fig. 5). In the lung, SPCs positive for every B cell marker were not detected.
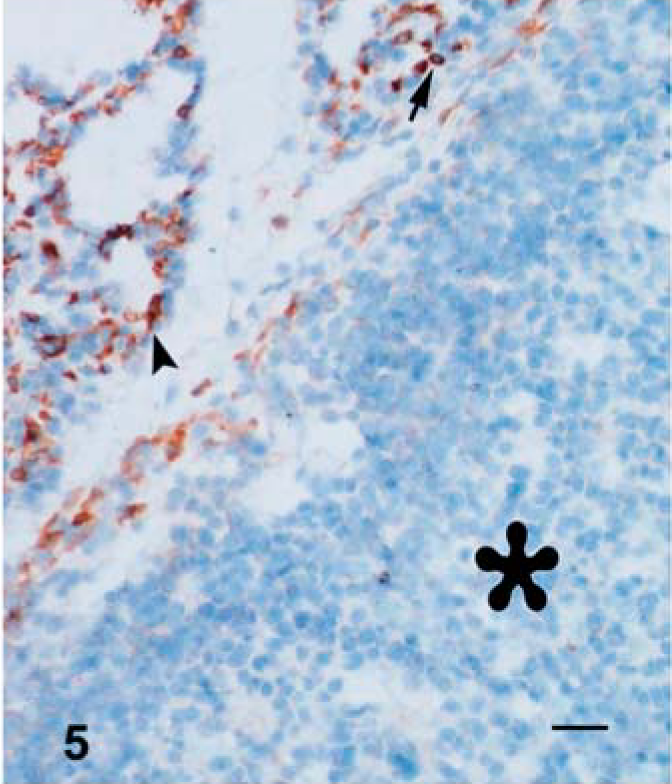
Lung; lamb No. 1. The lack of CD21 expression in the pox lesion (∗) and CD21+ lymphocytes (arrow) and dendritic cells (arrowhead). ZSF; SABC/DAB, hematoxylin counterstain. Bar = 100 µm.
In both the skin and lung, IgM staining gave good results in ZSF-processed tissues. B and plasma cells as well as SPCs and other stromal elements labeled for IgM in both early and advanced cutaneous lesions were very numerous (Fig. 6). A strong IgM immunostaining was detected in the ballooning degenerate epidermis and adnexal elements having typical pox lesion characteristics; conversely, staining in early lesions was weak. Vascular structures with vasculitis had intimal (endothelial)- and medial-positive labeling for sIgM. In the lung, strong labeling of sIgM was expressed on the cells with SPCs morphology within the pox lesions, as well as by groups of small, round cells scattered inside or outside of pox lesions. Amorphous eosinophilic material in the typical pox lesions seen in HE-stained lung sections reacted with IgM. Many SPCs exhibited perinuclear intense staining for sIgM (Fig. 6).
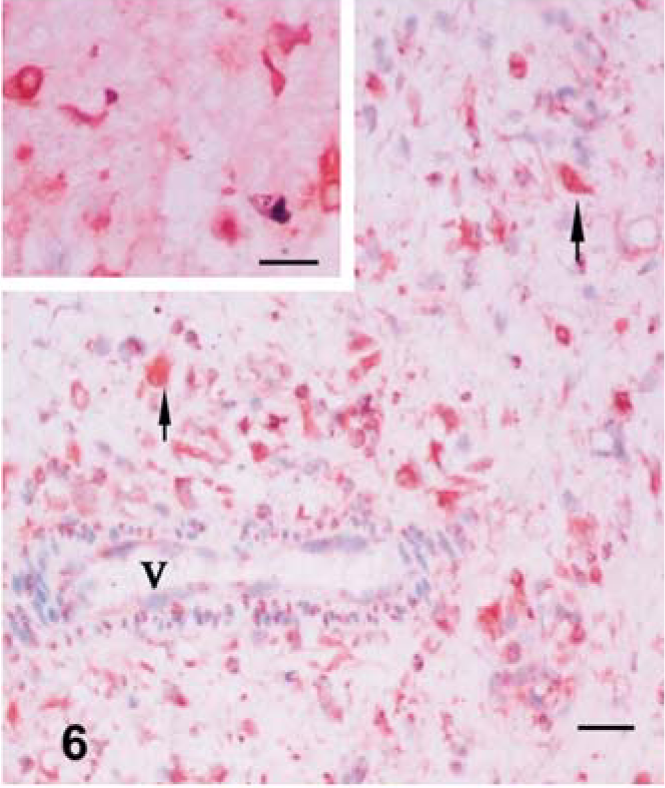
Skin; lamb No. 2. Dense immunostaining for SIgM of cellular and other elements with vasculitis (V = vessel) in the dermis. Arrows show SIgM+ SPCs. ZSF; SABC/DAB, hematoxylin counterstain. Bar = 150 µm. Note the perinuclear staining in the nucleus of SPCs (inset). ZSF; SABC/DAB, hematoxylin counterstain. Bar = 75 µm.
CD14 labeling was observed on large cells with pale nuclei and abundant cytoplasm throughout the dermal and subcutaneous tissues. Numerous CD14+ and smaller CD14_ SPCs were dispersed in perivascular areas and within the clusters of inflammatory infiltrates. The epidermal lesions did not contain CD14+ cells. In the lung, a small population of grouped and scattered large cells with clear nuclei and SPCs were CD14+ (Fig. 7). These cells were detected in alveolar spaces and interstitial tissues. No CD14+ cells were observed in the areas infiltrated with lymphocytes in the skin and BALT of lung.
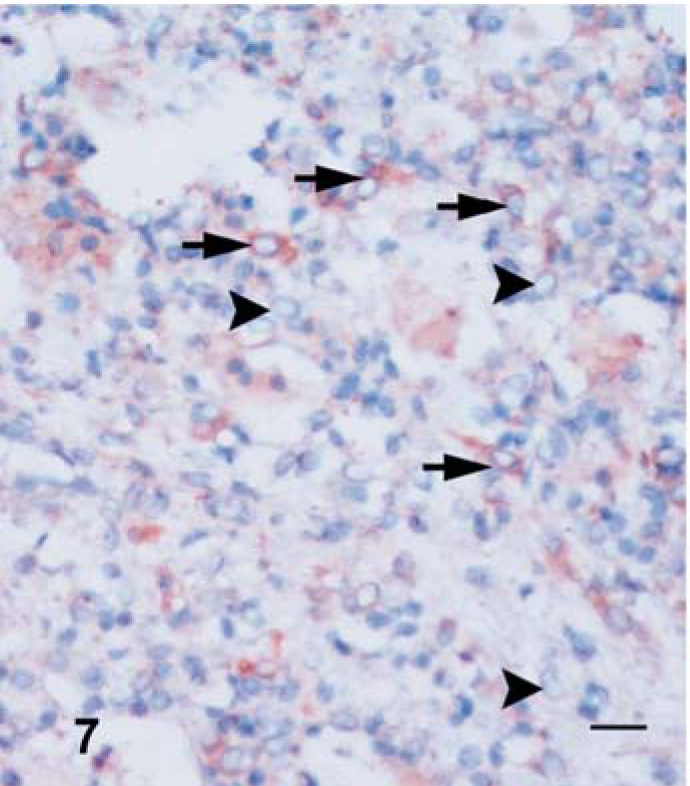
Lung; lamb No. 1. CD14 immunostaining of cells in pox lesion. Note positive (arrows) and negative (arrowheads) labeling of SPCs for CD14. ZSF; SABC/AEC, hematoxylin counterstain. Bar = 100 µm.
Of all antibodies used in the study, CD172A labeling with ZSF provided the best results in both histological detail and antigen preservation. In the early lesions, small numbers of CD172A+ cells with DC morphology were detected in the epidermis. In the early and advanced lesions, many DCs, SPCs, and granulocytes were observed at the dermo-epidermal junction. In the dermis and subcutis, MHC II+ areas also included dense immunostaining for CD172A on DCs, macrophages resembling SPCs, and numerous granulocytes, especially around the hair follicles and blood vessels. In the lung, CD172A was expressed on DCs, SPCs, and granulocytes in and between the alveoli in pox lesions. There was no labeling of cells with SPC morphology in the interalveolar septa or alveolar spaces (Fig. 8).
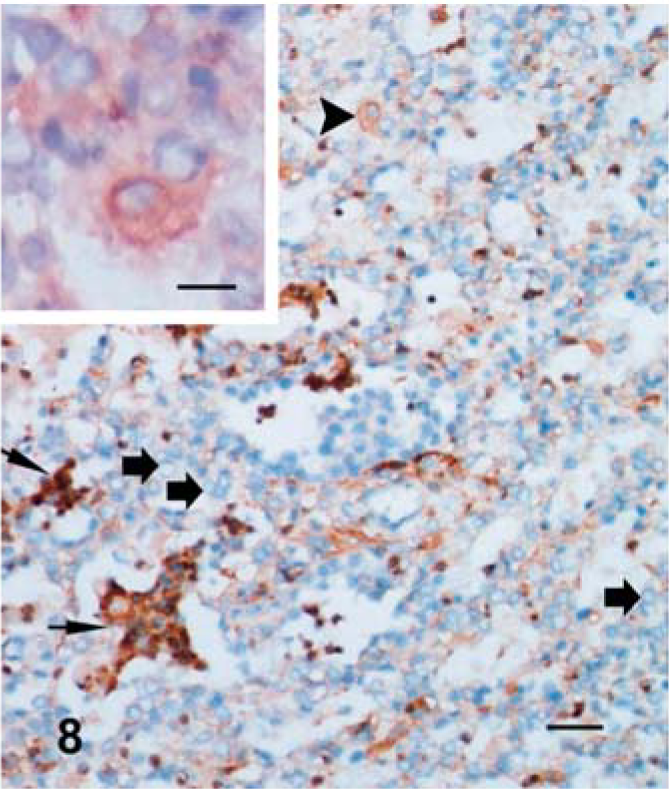
Lung; lamb No. 2. CD172A+ polymorphonuclear cells (thin arrows) in the alveolar spaces and SPC (arrowhead) and also negative SPCs (thick arrows). ZSF; SABC/DAB, hematoxylin counterstain. Bar = 150 µm. Inset: Higher magnification of SCPs indicated by arrowhead. ZSF; SABC/DAB, hematoxylin counterstain. Bar = 20 µm.
Immunostaining for SPV showed that viral antigen was localized in the cytoplasm of SPCs in the skin and lung sections, but sometimes only in the nucleus without evidence of cytoplasmic staining. Intracytoplasmic inclusion bodies could not be detected immunohistochemically (Fig. 9a). Moreover, double immunostaining for both cell markers and viral antigens could not easily differentiate SPCs, whereas their distinct vacuolated nucleus and marginated chromatin were more discernable features for detecting types of SPV-infected cells (Fig. 9b, c). SPCs that did not stain with macrophage and monocytes markers, especially with CD14 and CD172A, were considered to be fibroblasts in the skin and pneumocytes in the lung.
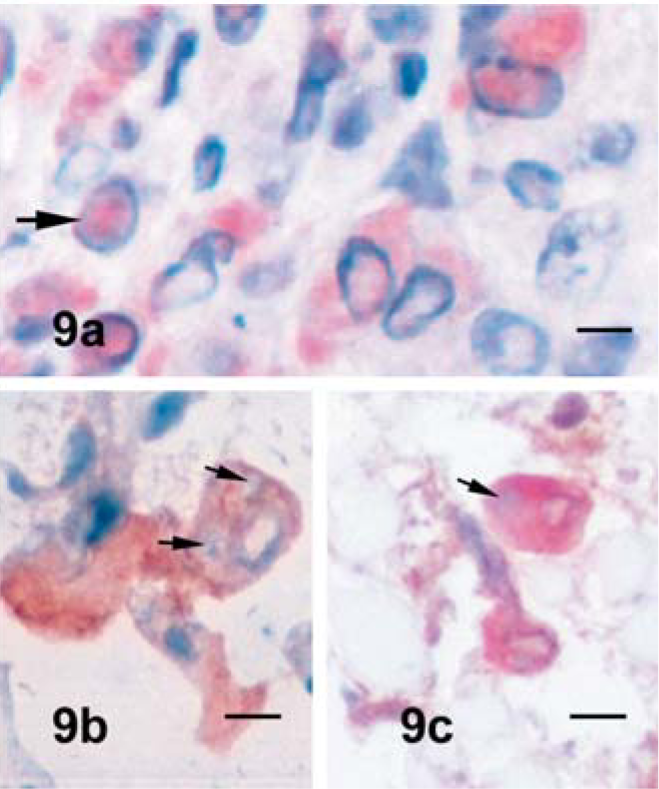
Skin; lamb No. 2. SPV+ SPCs in the dermis ( Fig. 9a ). Note the nuclear staining without cytoplasmic evidence of staining (arrow). ZSF; SABC/AEC, hematoxylin counterstain. Bar = 10 µm. Fig. 9b . Lung; lamb No. 2. CD14+SPC in the lung with typical nuclear morphology at higher magnification. Arrows indicate intracytoplasmic inclusions, ZSF; SABC/AEC, hematoxylin counterstain. Bar = 10 µm. Fig. 9c . Dermis; lamb No. 2. Double immunostaining for CD14 and SPV with purple-brown color in the SPCs in the dermis (arrow = intracytoplasmic inclusion). ZSF; AP/red substrate and SABC/DAB, hematoxylin counterstain. Bar = 20 µm.
SP is generally a systemic disease and it does not occur again in animals that recover from infection. They remain immune for the rest of their lives. 23 Neutralizing antibodies, cytotoxic T-cells, and interferon produced by infected macrophage play a role in recovery from poxvirus infection and development of protection against reinfection. 19 The present study shows the response to SPV may be complex and involve CD4+, CD8+ and γδ+ T cells, IgM+ B cells, and CD21+ monocytes/macrophages, and, to a lesser extent, B cells that express other B cell antigens. Moreover, in the skin and lung, dense accumulations of CD172A+ neutrophils as well as macrophages and monocytes determined were one of the striking findings. T cells appear to be the dominant cell type involved in clearance of SPV infection. However, the functional interaction of the various cell types remain to be determined. 22
In our study, MHC II and CD1 markers provided a clear way to identify ovine DCs in the epidermis. In the lung, expression of MHC II, CD21, and CD172A distinguished DCs. Cells that expressed CD1 had B cell or macrophage morphology. In contrast with the CD1 immunostaining, CD21 immunostaining with mAb GB25A yielded good results for distinguishing DCs as well as B cells in lung sections. The lack of CD1 expression in the DCs of the lung and the lack of CD21 expression in the skin are consistent with observations showing there is a difference in patterns of expression of CD1 on different DC subsets 6, 21 and in variations of expression of CD21 on subsets of DCs in the distinct tissues. Moreover, SPCs could not be distinguished in the skin and lung sections based on expression of CD1 and CD21. There was a decrease in the number of Langerhans cells and DCs positive for MHC II, CD1, and CD172A markers in the epidermis infected with SPV. The decreased MHCI+, MHC II+, and CD1+ cell numbers in the epidermis, associated with pox lesions, suggests that they may leave the epidermis and migrate to regional lymph nodes or that expression of MHC II is down-regulated following infection of these cells with pox virus. It is known that different families of viruses, including herpes, retro, and pox can obstruct the class II expression, so they avoid detection by CD4+ T cells. 11, 13 This study demonstrated that SPV also caused a down-regulation of MHC class I expression in both the skin and lung tissues, potentially affecting the generation CD8+ T cell responses to infection 11 as well as interference with the expression of MHC I.
In ruminants, WC1+ γδ T cells are the predominant subset in the peripheral blood, whereas WC1− γδ T cells are more abundant in spleen, with a similar abundance in the other secondary lymphoid organs and the mammary gland, intestine, skin, and uterus. The function of both populations remains to be elucidated. 5 γδ T cells have been purported to play a role in the first line of defense as effector cells and potentially play a role in the regulation of immune responses restricted by the MHC mediated through αβ T cells. 12 In contrast with other species, Artiodactyla, including the suborder of Ruminantia, contain an extra population of γδ T cells characterized by the expression of a unique molecule WC1 (workshop cluster 1). 1 WC1 is present on the surface of γδ T cells, which are CD4−CD8−, but which express a low level of CD5. WC1+ cells are the most prominent T cell subset in the skin of sheep at all ages and they are the predominant lymphocyte subpopulation at the skin surfaces in wooled sites. 17 However, they represent a small proportion of γδ T cells in other epithelial sites. 15 In the present study, the infected lambs were approximately between 1 and 2 months of age and the skin samples were collected from sites with minimal wool. Moreover, the numbers of γδ T cells in the skin and lung were large. Because sheeppox lesions have a predilection for the sparsely wooled areas of the skin, as well as being a systemic disease, 23 it is difficult to assess the immunological significance of γδ+ T cells in SPV infection and why sheeppox lesions have a predilection in sites not covered by wool. In normal bovine lung, WC+ γδ T cells are sparsely scattered in the interstitium of alveoli and the lamina propria of bronchioles. 1 We found WC+ γδ T cells concentrated in BALT and the periphery of pox lesion as well as in or just beneath the hyperplastic bronchial epithelium. The presence of WC+ γδ T cells in the skin and the lung suggest that they may play a role in the first line of defense at sites of infection as hypothesized by other investigators. In the present study, we detected few WC1+ DCs in the epidermis, dermoepidermal junction, and dermis, but not in lung sections. None of the T19+ cells, which are WC1+ γδ T cells, resembled the DCs in normal and acanthotic ovine skin, as observed by Gorrell et al., 8 whereas the γδ+ DCs that are found in the skin of many rodent species were first described by Bergstresser et al., 3 who described these cells as Thy-1+ dendritic T cells, and in UV-B-irradiated sheep skin described by Dandie et al. 4 If the cells described in the present study are Thy-1+ DCs, they may be acting as effector cells targeting SPV-infected keratinocytes or acting as antigen-presenting cells carrying viral antigens from the skin to the lymph node for presentation to suppressor T cells, as suggested by Dandie et al. 4 Moreover, immunohistochemical studies indicate that macrophage lineage cells and some nonlymphoid cells express a receptor for WC1. WC1 might serve as an accessory molecule involved in cell signaling and cell proliferation. 1 Thus, there may be a close association of WC1-expressing cells with SPCs in positive or negative signaling.
We detected an extensive leakage of IgM in the tissues as well as the expression on many SPCs and cells with round morphology, probably B cells. Vasculitis observed in sections infected with SPV from previous 9 and the present study was similar to an Arthus reaction secondary to deposition of antibody-antigen complexes, characterized by the presence of IgM immunostaining in blood vessel walls and activation of complement with an acute influx of many CD172A+ neutrophils around the pox lesions.
The mAbs used in this study revealed that many SPCs were CD14+ and CD172A+ monocytes and macrophages. The SP viral antigen was seen in the cytoplasm and nucleus. Sometimes staining was only seen in the nucleus of some SPCs. However, nuclear staining of some SPCs observed in the present study might be due to an artifact after tissue preparation as reported by other workers. 9, 19 CD14 labeling by mAb CAM36A was observed on the large cells with pale nuclei and abundant cytoplasm. Many of these cells had SPC morphology. They were dispersed throughout the dermal, subcutaneous, and lung tissues, similar to observations reported by Gupta et al. 10 However, CAM36A did not react with granulocytes. CD14 expression on SPCs, detected with CAM36A, also demonstrated that these cells were macrophages or monocytes. Some dermal-subcutaneous and many pulmonary SPCs were negative for these antibodies. These were considered to be virus-infected fibroblasts or pneumocytes. Efforts to use double immunostaining to phenotype infected cells yielded less than optimal results because the chromogens used in the study were too close in color to determine whether cells were labeled with one or both antibodies. In addition to CD14+ and CD172A+ SPCs, we also detected CD2 and CD8 antigens and B cell antigen with mAb GB26A on some SPCs. These were in low frequency in the skin of some lambs. The expression of CD2 may differ markedly between species, 16 and expression of the CD2 molecule has been described on all αβ T cells and on subsets of sheep macrophages at low levels. 14 Although the presence of CD8 and B cell antigens on sheep monocytes or macrophages, which are infected with SPV in this study, has not been documented, sheep cells of the common myeloid lineage may also express these antigens. Further studies are needed to verify this observation.
In conclusion, the inflammatory infiltrate in pox lesions in both skin and lung was characterized by the presence of MHC II+ DCs, CD4, CD8, γδ+ T cells, IgM+ cells, and CD21+ cells and, to a lesser extent, B cells expressing the other mAb-defined B cell antigens, suggesting cell-mediated immune response to SPV. The loss of expression of MHC I and MHC II antigens was observed in the affected areas of skin and lung. SPCs, stained with anti-SPV antibody, were monocytes and macrophages, whereas CD14_ and CD172A-negative SPCs were considered to be SPV-infected epithelial cells or fibroblasts.
Footnotes
Acknowledgements
We are particularly grateful to the Washington State University, Monoclonal Antibody Center for the donation of the mAbs for the study.
