Abstract
Adenoviral infections were diagnosed in three neonatal lambs that died spontaneously, and no other etiologic agents were identified. Clinical signs were anorexia, weakness, abdominal distention, and sudden death. Microscopic lesions consisted of multifocal necrotizing hepatitis, multifocal subacute interstitial nephritis, and loss of enterocytes from intestinal villi. Adenovirus inclusions were identified by light microscopy in the kidneys only. Adenoviral antigen, however, was identified in the liver, kidney, and intestine of the lambs by immunohistochemical techniques. An ovine adenovirus serotype 7, not previously isolated from sheep in the United States, was characterized from these lambs.
Six adenovirus serotypes have been isolated from sheep and characterized. 2 A seventh ovine adenovirus (OAdV) has been characterized and proposed as a new serotype and the prototype for the new genus Atadenovirus in the family Adenoviridae. 2 These prototype adenoviruses were isolated from nasal secretions and feces of clinically normal sheep and from sheep with clinical pneumonia and enteritis. Adenoviral infections are apparently common in sheep; the prevalence of antibodies to several serotypes of adenovirus is high. 8,9 Experimental adenoviral infections in lambs has produced clinical signs and lesions that can exacerbate Mannheimia (Pasteurella) haemolytica pneumonia. 4,6
An adenovirus isolated from lambs in Kansas that died spontaneously was serologically and genetically similar to the OAdV serotype 7 (OAdV-7) isolate, which has not previously been isolated from sheep in the United States. Here, we describe the signs, lesions, and characteristics of the adenovirus associated with this case.
Materials and Methods
Case history
During the spring of 2000, five of approximately 45 lambs in a 130-ewe flock died. Adenovirus infections were identified in three of these five lambs. The adenovirus-infected lambs were 9, 13, and 25 days of age at onset of clinical signs. One lamb was found dead with no previous clinical signs observed, and the other two lambs had clinical signs of anorexia, weakness, and abdominal distention that progressed to recumbency and death within 48 hours. Gross postmortem findings were limited to fluid distention of the gastrointestinal tract.
Histopathology
Tissues from the lambs were fixed in 10% buffered formalin, processed, and stained by routine methods. Tissues examined included lung, liver, kidney, spleen, small intestine, and brain.
Virus isolation
Low-passage ovine fetal turbinate cells were grown in Eagle's minimal essential medium (EMEM) supplemented with 10% heat inactivated bovine fetal serum (BFS) and gentamicin sulfate (50 µg/ml). Cultures were incubated at 37 C in a humidified atmosphere containing 5% CO2.
Pooled tissues (approximately 0.5 g each of lung, liver, and kidney) from the three lambs were ground in EMEM supplemented with 2% BFS, gentamicin sulfate (50 µg/ml), and amphotericin B (10 µg/ml) with a TenBroeck tissue grinder and clarified by low-speed centrifugation. Supernatant was used to inoculate tissue culture flasks containing subconfluent cells. The inoculum (0.5 ml) was allowed to adsorb for 2–4 hours and then was replaced with fresh medium. Flasks were examined daily for virus-induced cytopathic effect (CPE) and were subpassaged after 7–10 days if no CPE was observed. Cultures with CPE were subjected to a freeze–thaw cycle, and the supernatant was inoculated into new flasks of subconfluent cells. The isolated virus was biologically cloned by inoculating six wells of a 24-well tissue culture plate containing subconfluent cells with 0.1 ml of serial 10-fold dilutions of the virus. The plates were examined with an inverted microscope to identify wells with a single plaque, the plate was subjected to two freeze–thaw cycles, and the identified wells were used to make dilutions to repeat the process through three successive plaque purifications before stock virus was produced. Stock virus was harvested after two freeze-thaw cycles and stored at −80 C.
Virus characterization
For negative contrast and transmission electron microscopy, infected cultures were harvested by trypsinization when CPE involved >75% of the cells. The cells were pelleted by low speed centrifugation (1,000 × g), resuspended in either distilled water for negative-contrast electron microscopy or 2.5% glutaraldehyde for transmission electron microscopy, and prepared using standard procedures.
Serum virus neutralization tests for virus identification were performed in microtitration plates as previously described. 8 Rabbit antiserum prepared from biological cloned stock pools of the prototype OAdV and two caprine adenovirus (GAdV) serotypes were used in neutralization tests. Ovine fetal turbinate cells were used for replication of the OAdV and GAdV. Serial twofold dilutions of reference antisera were made starting at 1:16. The reference and test viruses were diluted to provide 100 TCID50 per well. Controls included back titration of the reference and test viruses and a fetal calf serum negative control. Serum antibody titers were expressed as the reciprocal of the highest dilution of serum preventing CPE in 50% of the wells after 7 days of incubation.
DNA from the adenovirus isolated from one of the lambs in this study was extracted from the infected cell monolayers and digested with restriction enzymes BamHI, HindIII, and PstI as previously described. 10 The hexon protein region of the adenovirus was sequenced using the ClustalW module of the Mac Vector program (Oxford Molecular, Hunt Valley, MD) and was compared with published adenovirus hexon sequences. 10
Immunohistochemistry
Tissues were fixed in 10% buffered formalin and fixed for 24–48 hours, embedded in paraffin, and sectioned onto Vectabond treated slides. Tissues were deparaffinized in three changes of xylene and rehydrated through graded alcohols to distilled water. Tissues were treated for 15 minutes with pepsin, quenched with 3% hydrogen peroxide for 5 minutes, and then treated with protein-binding agent (Shandon Lipshaw, Pittsburgh, PA) for 10 minutes. Primary antibody (polyclonal bovine anti-bovine adenovirus serotype 5 (BAdV-5), American BioResearch, Sevierville, TN), diluted to 1:2,000 was applied overnight at 4 C and then warmed to room temperature for 1 hour. Secondary antibody (biotin-labeled goat anti-bovine antibody, Kirkegaard-Perry, Gaithersburg, MD) diluted to 1:100 was applied for 15 minutes at room temperature. The enzyme (Vector ABC-HRPO, Vector Laboratories, Burlingame, CA) diluted to 1:200 was incubated for 15 minutes at room temperature, and chromogen (Vector DAB, Vector Laboratories) was applied for 2–6 minutes at room temperature. Slides were counterstained with hematoxylin.
Other testing
Intestinal contents were examined for protozoal organisms by standard flotation techniques and for viral particles by routine direct electron microscopy.
Results
Pathology
Microscopic examination of tissues revealed interstitial nephritis in all three lambs, which was characterized by multifocal infiltrates of neutrophils, lymphocytes, and macrophages throughout the interstitium of the kidney and most prominent in the cortex. Basophilic to chromophobic intranuclear inclusions of various sizes were present in endothelial cells of the renal cortex and to a lesser extent in the medulla (Fig. 1). Inclusions were most prevalent in the immediate subcapsular region of the kidney. Two lambs had multifocal random hepatic necrosis characterized by small foci with necrotic hepatocytes, infiltrates of neutrophils and macrophages, and fibrin exudation (Fig. 2). Viral inclusions were not identified in liver sections. Two lambs had villus atrophy in the small intestine. Affected villi were denuded and infiltrated by neutrophils. Occasional crypts were filled with sloughed cells and neutrophils. Viral inclusions were not identified in the intestinal sections.
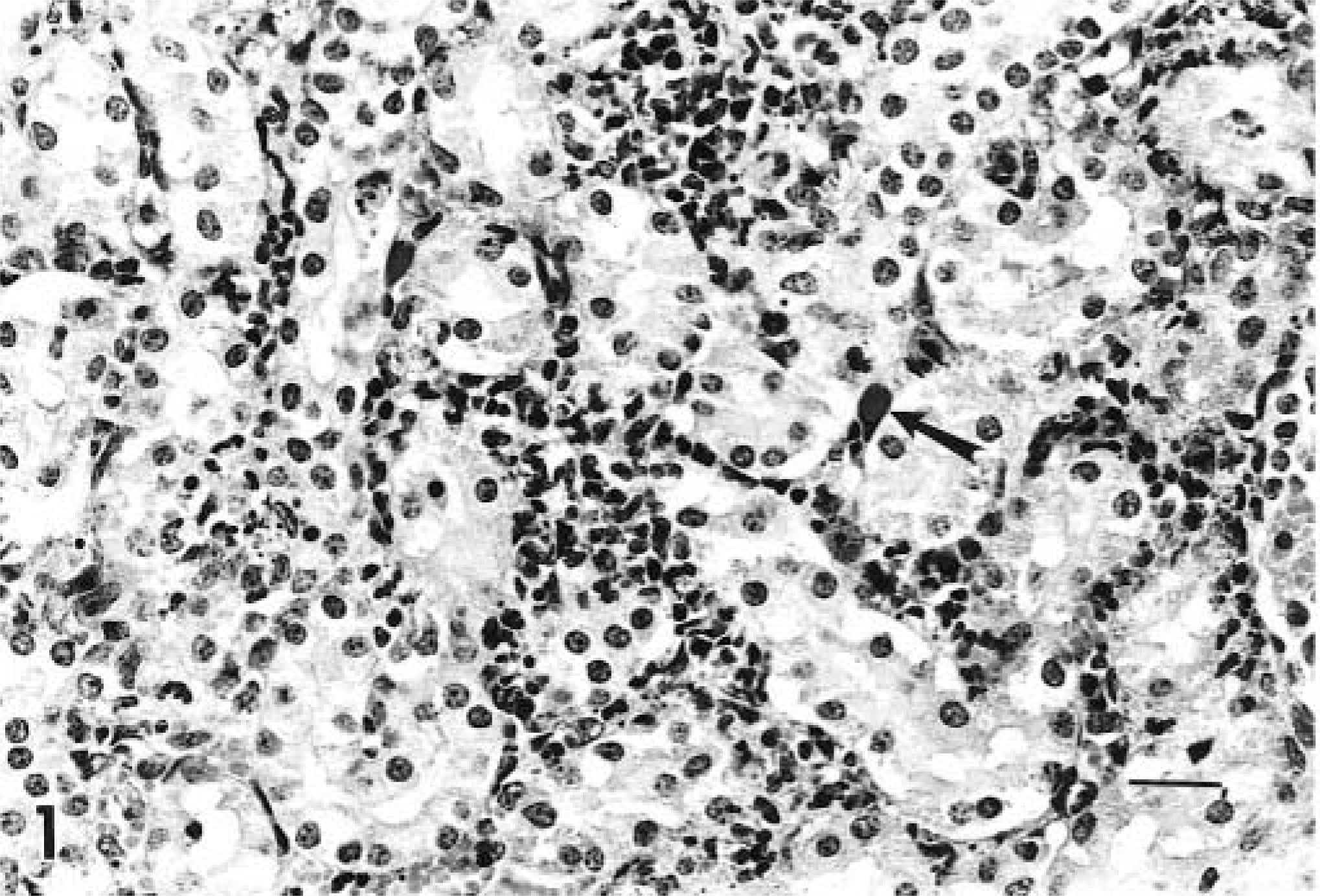
Kidney; lamb infected with adenovirus. Interstitial nephritis with intranuclear adenoviral inclusions within endothelial cells (arrow). HE. Bar = 67 µm.
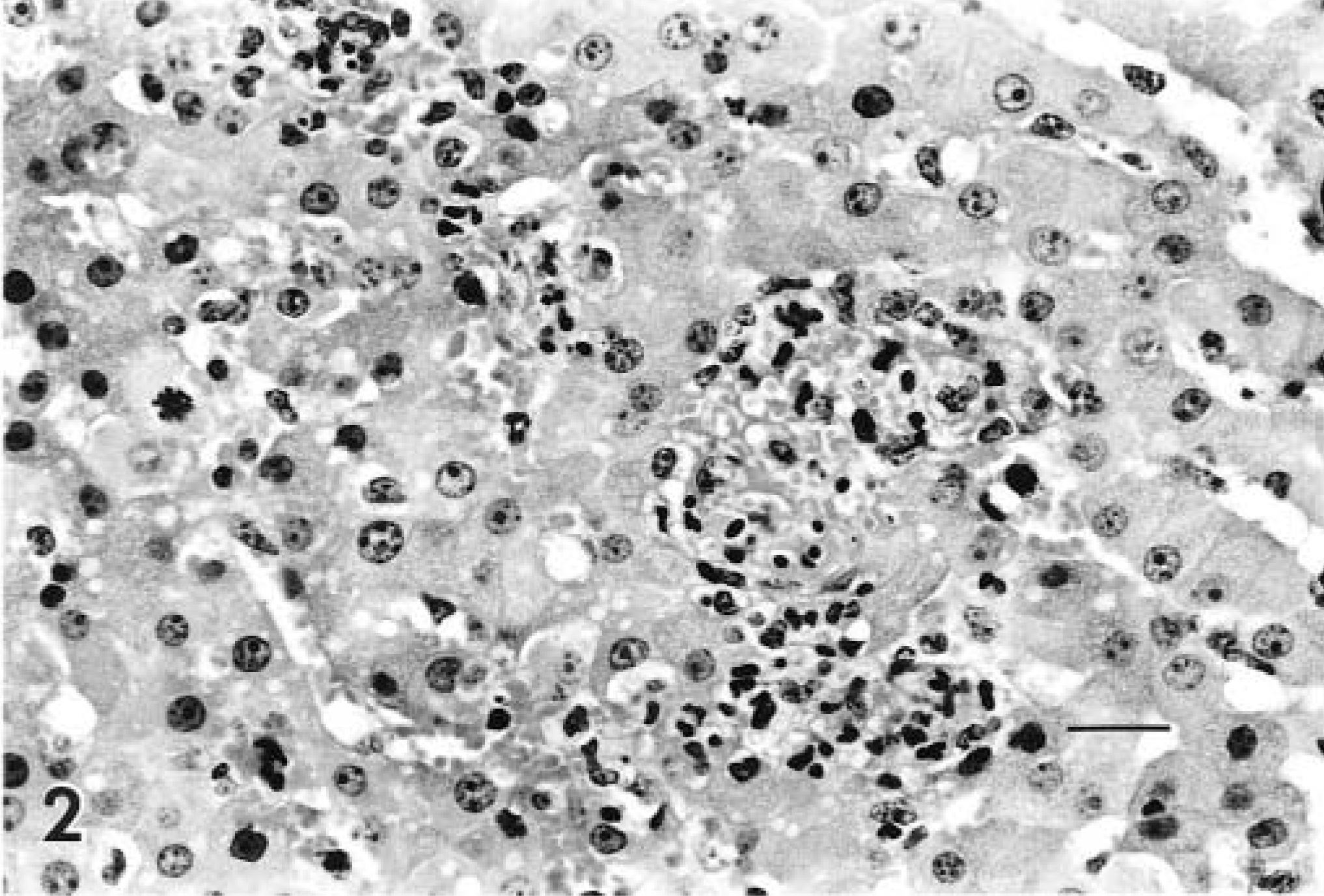
Liver; lamb infected with adenovirus. Inflammatory infiltrates within a focus of hepatocellular necrosis and loss. HE. Bar = 67 µm.
Virus isolation and characterization
Virus (ES 2560 and ES 3048) was isolated from the 9- and 25-day-old lambs. The CPE occurred as scattered refractile cells throughout the monolayer that became generalized over the entire monolayer, with detachment of rounded cells late in virus replication. Electron microscopic examination of cell lysate revealed hexagonal virions with equiangular triangular faces, each triangle containing six subunits (capsomeres) per side. The diameter of the virions was between 75 and 80 nm. Crystalline arrays of virus were seen infrequently in ultrathin sections of virus-infected cells. Antiserum to OAdV-7 and GAdV serotype 1 neutralized the isolated viruses. Neutralization with the other OAdV and GAdV serotypes was not observed. Restriction endonuclease cleavage patterns generated by the adenovirus isolate (ES 3048) were identical to those produced by OAdV-7, and the hexon amino acid sequence was virtually identical to that of OAdV-7, except at ES 3048 alignment sites 356 and 757, where there was a conserved substitution, and at alignment site 515, where there was a nonconserved substitution (Fig. 3).

Amino acid sequence alignment of adenovirus partial hexon protein. Hexon proteins of OadV-7 (OAV 287; GenBank No. AAA84979) and ES 3048 (GenBank No. AF372835) have been aligned (position 1 = amino acid 3 of OAdV-7 hexon gene). Identical amino acids are indicated by shadowed boxes. Conserved amino acids within the boxes are indicated by light background.
Immunohistochemistry
Adenoviral antigen was detected in kidneys (3/3 lambs), liver (2/3 lambs), small intestine (2/3 lambs), spleen (2/3 lambs), and lung (1/3 lambs). In the kidney, endothelial cells were positive for adenoviral antigen (Fig. 4), and in the liver, endothelial cells and hepatocytes contained antigen (Fig. 5). In the intestine, crypt epithelial cells and endothelial cells contained adenovirus antigen. Viral antigen was located within nuclei of some cells and sometimes within the cytoplasm and nuclei of cells (Fig. 5).
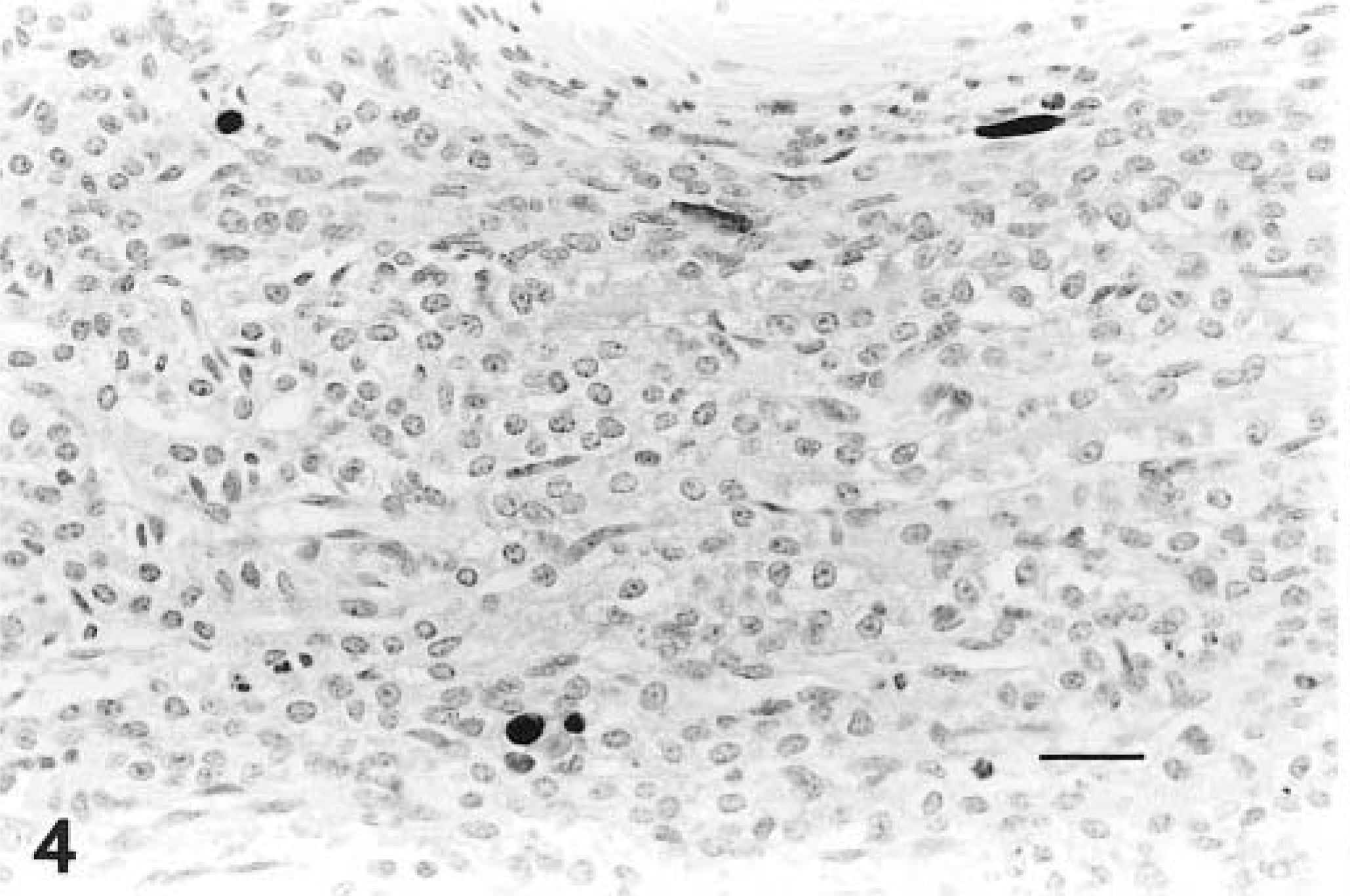
Kidney; lamb infected with adenovirus. Immunohistochemical stain with antibody to BAdV-5, revealing antigen positive endothelial cells within the interstitium. Avidin–biotin–peroxidase complex method, Gill's No. 1 hematoxylin counterstain. Bar = 67 µm.
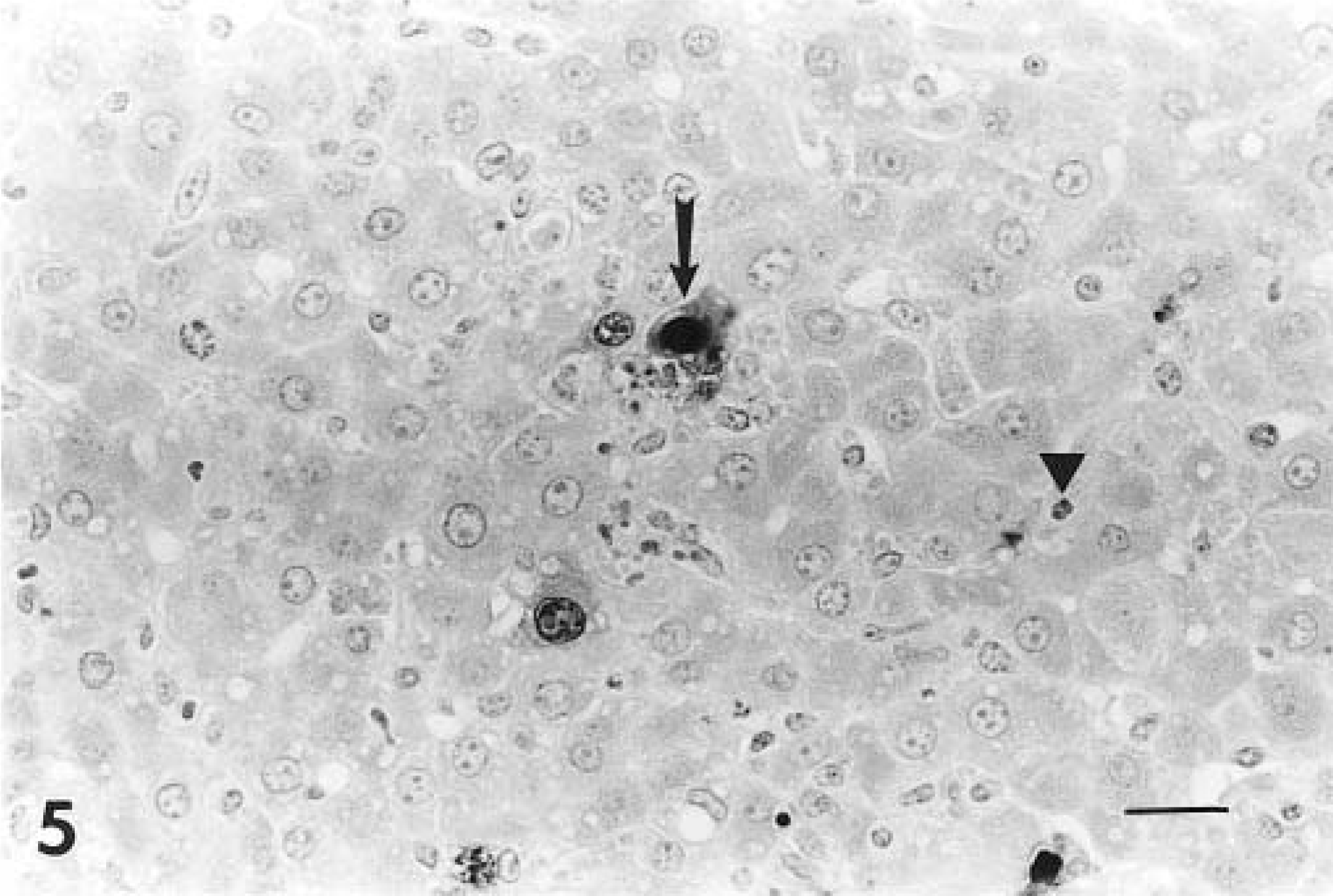
Fig. 5 Liver; lamb infected with adenovirus. Immunohistochemical stain with antibody to BAdV-5, revealing a hepatocyte containing intranuclear and intracytoplasmic antigen (arrow) and antigen-positive endothelial cell (arrowhead). Avidin–biotin–peroxidase complex method, Gill's No. 1 hematoxylin counterstain. Bar = 67 µm.
Additional laboratory testing
No bacterial pathogens were isolated from lung, liver, kidney, and intestine of the lambs by routine aerobic cultures or salmonellae enrichment techniques on intestinal contents. No viral particles were identified by direct electron microscopic examination of small intestinal contents, and no protozoal organisms were identified by flotation techniques from the contents of the small intestine.
Discussion
Virus isolates (ES 2560 and ES 3048) from the lambs in this study had the cultural and morphologic characteristics of members of the family Adenoviridae. 2 The serologic reactivity, restriction endonuclease cleavage pattern, and hexon amino acid sequence indicate that the isolates are OAdV-7 and are virtually identical to strain OAV 287, which was isolated from a sheep in Australia and is the proposed prototype for OAdV-7. 2,3 These isolates are the first of this serotype to be obtained from sheep in the United States.
The lesions and finding of adenoviral inclusions in multiple tissues of the lambs in this study and the lack of other pathogens support the hypothesis that the adenoviral infections caused death in these lambs. Adenovirus infections in sheep are often described as mild or inapparent. However, losses of approximately 30–40% have been reported in lambs at 2–12 weeks of age, with approximately 10% of the losses associated with acute adenoviral infection and 90% of the loss associated with secondary bacterial complications. 1 Experimental Mannheimia (Pasteurella) haemolytica infections were more severe in adenovirus-infected lambs than in lambs not infected with adenovirus. 4,7 There was no evidence of secondary bacterial complications in the lambs in this study.
The adenovirus infecting the lambs in this study had a tropism for endothelial cells, based on the location of inclusions and immunohistochemical staining. Other ruminant adenoviruses also have endothelial cell tropism, including OAdV serotype 4 (OAdV-4), 11 bovine adenovirus serotype 10, 12,13 and a deer adenovirus isolated in California. 14 Necrotizing hepatitis has been previously described as a result of experimental infection of lambs with OAdV-4 11 but not with other serotypes of OAdV. In the present study, the presence of adenoviral antigen within the necrosuppurative lesions in the liver is strong evidence that these lesions were caused by the adenovirus infection. Severe lesions were present in kidneys of two of the lambs, supporting the putative high virulence of the virus with which these lambs were infected.
Experimental infection of lambs with the Australian isolate, OAV 287, which is nearly identical to the virus isolated in the present study, did not result in clinically apparent disease, 3 although infection was indicated by virus isolation and serologic response. The age of the lambs may have precluded the development of clinical disease in that study, and lambs were not examined for gross or histopathologic signs of disease. Infection of lambs with WV757, a New Zealand isolate of OAdV-7, resulted in mild disease with very minor gross and microscopic lesions, lacking identifiable viral inclusions. 7 A longitudinal serologic survey for antibody to WV757 in New Zealand lambs suggested that this virus may have a role in development of ovine pneumonia. 5
Adenoviral infection was the probable cause of death of the lambs in the flock in the present study. Significant morbidity was not observed in other lambs within this or other lambing groups of the season on this farm, indicating that the adenoviral infections were either limited to these few lambs in the group or that other lambs had inapparent infections.
