Abstract
Clinicopathologic and immunophenotypic characteristics of large granular lymphocyte (LGL) neoplasia in 21 cats were examined. All cats were domestic short (19) or long hair (2) with a mean age of 9.3 years at diagnosis. Increased peripheral blood LGL counts were present in 18/21 cats. Neutrophilia (12/21 cats) and increased serum liver enzymes (7/12), total and direct bilirubin (7/13), BUN (5/14), and creatinine (2/14) were observed. Cats usually presented with advanced disease and none survived longer than 84 days (mean 18.8 days) postdiagnosis. Cytologically, LGLs had a mature (6/21), immature (13/21), or mixed (2/21) morphology. Necropsy lesions consisted of neoplastic lymphoid infiltrates in the jejunum, ileum, and duodenum in decreasing order of frequency. In the small intestine, mucosal ulceration (9/13) and epitheliotropism of neoplastic cells (9/13) were common. Neoplastic infiltrates were also present in the mesenteric lymph nodes (13/13), liver (12/13), spleen (8/13), kidneys (5/ 7), and bone marrow (5/7). A T cell phenotype (CD3∊+) characterized LGL neoplasia in 19/21 cases. A CD8αα+ cytotoxic/suppressor phenotype was present in 12/19 T cell tumors, 2 had a CD4+CD8αα phenotype, 3 had a CD4-CD8- phenotype, and 2 were CD4+ helper T cells. CD8β chain expression was not detected in any instance. In two cats, a B or T cell origin could not be established. CD103 was expressed by 11 of 19 (58%) of the lymphomas tested. The immunophenotypic features shared by neoplastic LGLs in the cat and feline intestinal intraepithelial lymphocytes (IELs) support a small intestinal IEL origin for feline LGL lymphoma.
Large granular lymphocytes (LGLs) are a morphologically distinct subset of lymphocytes characterized by intracytoplasmic azurophilic granules. LGLs are a minor population of peripheral blood lymphocytes 1, 40, 47, 66 but represent a large subset of intestinal intraepithelial lymphocytes (IELs) in most species, including cats. 1, 45, 56, 58, 67 In humans, LGLs are grouped into two major lineages based on phenotype and T-cell receptor (TCR) gene rearrangement: T-cell LGLs rearrange TCR genes and express surface CD3 and TCR, while natural killer (NK) LGLs are CD3 surface negative and have a germline configuration of the TCR genes. 44, 53
From a clinical perspective, the spectrum of LGL disorders documented in humans and dogs consists of reactive benign expansions, acute or chronic leukemias, and primary lymphomas. 40, 44, 47, 59, 63– 65 Chronic leukemias are the most frequent LGL disorder in both humans and dogs. 40, 44, 47, 63 Reactive forms of LGL disease and LGL chronic leukemia frequently express a CD8αβ cytotoxic T cell phenotype and have a nonprogressive to slowly progressive clinical course in these two species. 40, 44, 47, 63
In humans, acute LGL leukemias have an aggressive behavior and are characterized by prominent peripheral blood LGL lymphocytosis with massive bone-marrow infiltration. 40, 44, 63 These cases may derive from T cells 25, 41 or, more frequently, from NK cells. 41, 44 Primary LGL lymphomas are extremely rare in humans. 41, 44 However, intestinal LGL lymphomas are the most frequently reported type of primary LGL lymphoma. 19, 24, 35, 50, 52 This disease has a particularly aggressive biological behavior and is usually of NK origin. 19, 24, 50, 52 Similarly, aggressive intestinal lymphoma with or without a leukemic profile appears to represent the most prevalent LGL disorder reported in cats. 10, 15, 20– 22, 26, 30, 32, 37, 48, 66
Phenotypic and molecular studies have assigned feline LGL lymphoma either to the T cell lineage 15, 20 or to a non-B non-T phenotype most consistent with NK cells. 10, 20, 26 Primary intestinal T cell lymphomas account for only 5% of all gastrointestinal lymphomas in humans and they are believed to arise from gut-associated lymphoid tissue. 11, 18 In humans, the cytological features, presence of CD103 expression, and the phenotypic similarities with lymphoid cells residing in the intestinal mucosa suggest that most human primary intestinal T cell lymphomas represent the malignant transformation of various subsets of intestinal IELs. 60, 71
In this report, we evaluated cytomorphologic, clinicopathologic, and immunophenotypic features of LGL neoplasia in 21 cats. Most cats had a peripheral blood LGL lymphocytosis associated with small intestinal LGL lymphoma. A CD3ε+, CD8αα+ cytotoxic T cell phenotype predominated and CD103 was expressed in 57% of the lymphomas. The unusual phenotype of most feline LGL lymphomas closely resembled that expressed by the majority of normal feline intestinal IELs. 56 Clinicopathologic and immunophenotypic findings of this study are therefore indicative of an intestinal IEL origin for the majority of LGL neoplasms in cats.
Materials and Methods
Case selection criteria
Cases were accessed largely from IDEXX Veterinary Services (Sacramento, CA) during the period 1996–1998 (cat Nos. 1, 3–13, 17–21). The remaining cases were accessed from UC Davis (cat No. 2) and University of Milan (cat Nos. 14–16) prior to 1996. Cases were included based on absolute elevation of LGLs in blood (reference interval 0–350/µl, unpublished observation) (cat Nos. 1, 2, 3–13, 20, and 21), or presence of LGLs in fine-needle aspirates of abdominal masses (cat Nos. 17–19), or abdominal effusion (cat No. 1). For the IDEXX cases, blood, abdominal and thoracic effusions, or fine-needle aspirate (FNA) smears from all cats that met the inclusion criteria were referred to UC Davis during the study period.
Blood collection and clinical evaluation
Venous whole blood from 18 cats with absolute elevation of LGLs was utilized for leukocyte morphological evaluation, differential cell counts, flow cytometry, and immunocytochemistry. Complete blood count (CBC) data and biochemical profiles were also obtained. Serum of 13/21 cats was tested for feline immunodeficiency virus (FIV) and feline leukemia virus (FeLV) infection by enzyme immunosorbent assay (Petcheck FIV antibody and FeLV antigen detection kits, IDEXX Laboratories).
Intestinal involvement was assessed clinically by evaluation of history, clinical signs, palpation, ultrasound and/or radiographic examination of the abdominal cavity in all cats. Clinical data and follow-up information were collected from attending veterinarians.
Preparation of blood smears and cytological specimens
Morphologic evaluation and differential cell counts (300 cells) were performed on blood smears stained with May-Grünwald-Giemsa or Wright-Giemsa. For immunocytochemistry, blood smears from each patient were air dried and fixed in acetone for 3 minutes. Abdominal and thoracic effusions were centrifuged and resuspended at a concentration of 50 cells/µl of PBS with 10% v/v heat-aggregated horse serum and 0.1% sodium azide. Aliquots of 100 µl were centrifuged at 1,000 rpm for 5 minutes using a cytocentrifuge (Shandon, Pittsburgh, PA). Bone marrow and imprint smears of fresh organs and biopsies were obtained in some cases to confirm or exclude the presence of LGLs. All cytological specimens were air dried and stained with May-Grünwald-Giemsa.
Tissue samples
Tissue samples from 13 cats were evaluated. Tissues were fixed in 10% neutral buffered formalin, paraffin embedded, sectioned at 5 µm, and stained with hematoxylin and eosin (HE). Fresh tissue samples from 11 cats were snap frozen in isopentane cooled to the freezing point in liquid nitrogen. For immunohistochemistry, 5-µm cryosections were cut, air dried, fixed in acetone (3 minutes) and stored at −70°C.
Monoclonal antibodies
A panel of feline leukocyte-specific monoclonal antibodies was used for flow cytometry, immunocytochemistry of blood smears, and immunohistochemistry of frozen sections (Table 1). Two of these monoclonal antibodies, which recognize intracytoplasmic epitopes (anti-CD79a and anti-CD3ε), were used for immunocytochemistry and immunohistochemistry in all cases. Monoclonal antibodies specific for CD8α and CD8β were distinguished by cell-distribution studies, antigen immunoprecipitation, and reactivity with cloned and expressed recombinant feline CD8α and CD8β (M. Shimojima, Veterinary Microbiology, Faculty of Agriculture, University of Tokyo).
Monoclonal antibody panel used for immunophenotyping of feline LGL lymphoma.
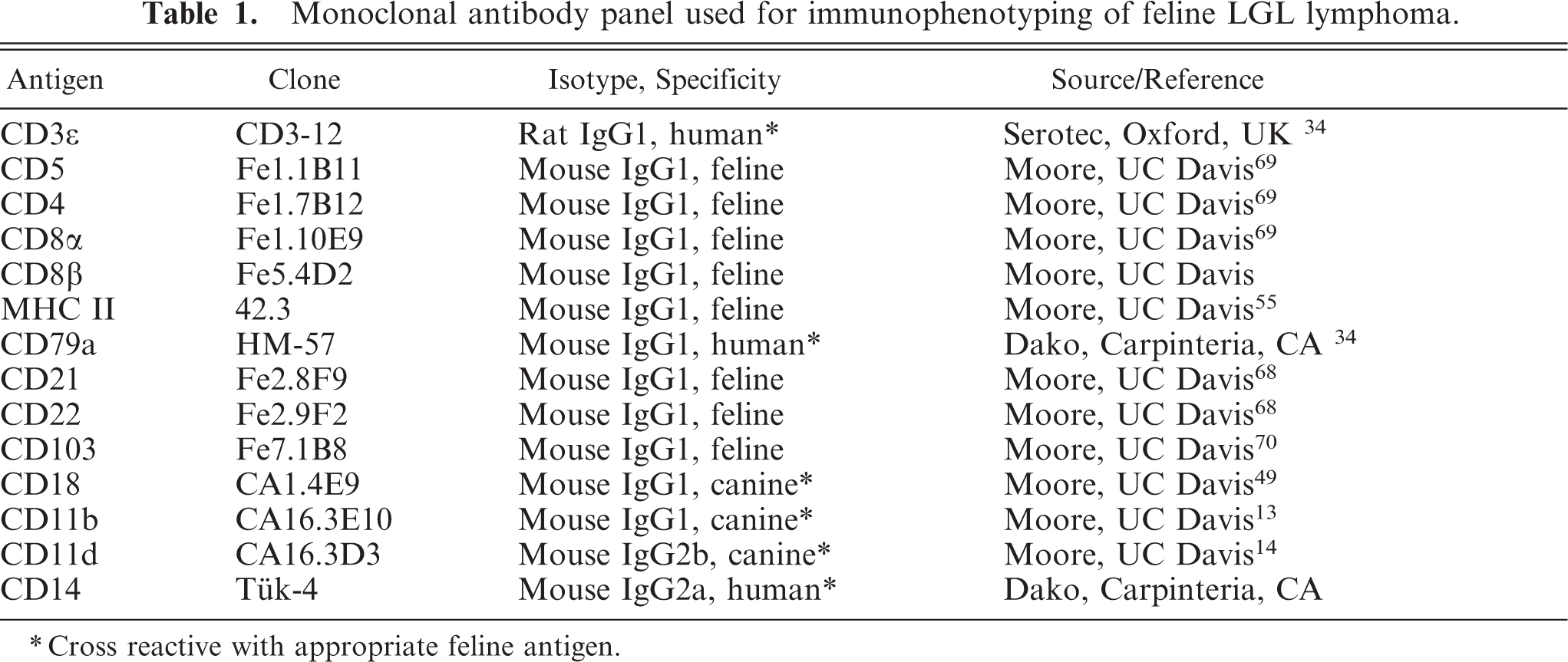
∗Cross reactive with appropriate feline antigen.
Flow cytometry
For single-label studies, 50-µl aliquots of whole blood were incubated with 25 µl of undiluted tissue culture fluid (TCF) of primary antibodies followed by red blood cell lysis and incubation with horse anti-mouse fluorescein isothiocyanate (FITC) (Vector Laboratories, Burlingame, CA) as described previously. 13 Assessment of intracytoplasmic CD3ε in cat Nos. 11 and 20 was also achieved by using anti-CD3ε-FITC in conjunction with cell-surface permeabilization (Fix & Perm kit, Caltag Laboratories, Burlingame, CA) using the manufacturer's protocol. A nonspecific mouse myeloma IgG1 hybridoma supernatant and an irrelevant FITC-conjugated mouse IgG served as negative controls. Stained cell suspensions were analyzed with a FACScan analytical cytometer (Becton Dickinson, San Jose, CA) equipped with Lysis II, Cell Quest (Becton Dickinson) and FlowJo software (Ashland, OR).
Immunocytochemical and immunohistochemical studies
Blood smears of cats with absolute increase of LGLs (18/21) were stained with anti-CD79a and anti-CD3ε chain antibodies to assess cytoplasmic expression of these molecules, and also with anti-CD4, anti-CD8α, anti-CD8β, anti-CD11d, and anti-CD103 antibodies to compare flow cytometry results with immunocytochemistry results. Frozen sections and cytologic specimens were stained by a streptavidin-horseradish peroxidase method according to the manufacturer's instructions (Zymed, South San Francisco, CA) and previously described methods. 49 Monoclonal antibodies specific for feline leukocyte antigens listed above were applied to sections as TCF at a 1 : 10 dilution. Negative controls consisted of substitution of specific monoclonal antibodies with isotype matched nonspecific monoclonal antibodies. Frozen sections of normal feline spleen were used as positive controls. A limited panel of antibodies (CD3ε, CD79a, CD18) reactive in formalin-fixed tissue sections was utilized in the two cases in which only formalin-fixed tissue was available.
Results
Clinical summary
A total of 21 cases of LGL neoplasia were studied. Of these, 18 cats were evaluated because of increased numbers of peripheral blood LGLs. After the diagnosis of LGL leukemia, intestinal thickening and probable intestinal involvement was confirmed by attending veterinarians using ultrasound or abdominal radiographs in all leukemic cats (18/21). In three cats, intestinal LGL lymphoma was not associated with a leukemic profile (cat Nos. 17–19). LGL lymphoma was diagnosed in these cats by FNA cytology of renal (cat Nos. 17, 18) or intestinal (cat No. 19) masses.
All cats were domestic short (19) or long hair (2). Age of affected cats ranged from 2 to 15 years with a mean of 9.3 years. There were 16 female cats with a female : male ratio of 3.2 (Table 2). Cats were presented to the attending veterinarian with a variety of clinical signs, including anorexia (14 cats), weight loss (12 cats), depression and lethargy (4 cats), vomiting (3 cats), jaundice (2 cats), diarrhea (2 cats), constipation (1 cat). Hepatomegaly (17 cats), palpable abdominal mass (12 cats), mesenteric lymphadenopathy (12 cats), splenomegaly (10 cats), and renomegaly (7 cats) were common clinical findings. Other clinical findings included abdominal effusion (3 cats), thoracic effusion (2 cats), peripheral lymphadenopathy (3 cats), septic peritonitis secondary to intestinal rupture (2 cats), and third eyelid enlargement (1 cat). Seven cats had a previous history of inflammatory bowel disease.
Demographics, leukogram, organ involvement and survival of cats with LGL lymphoma.∗
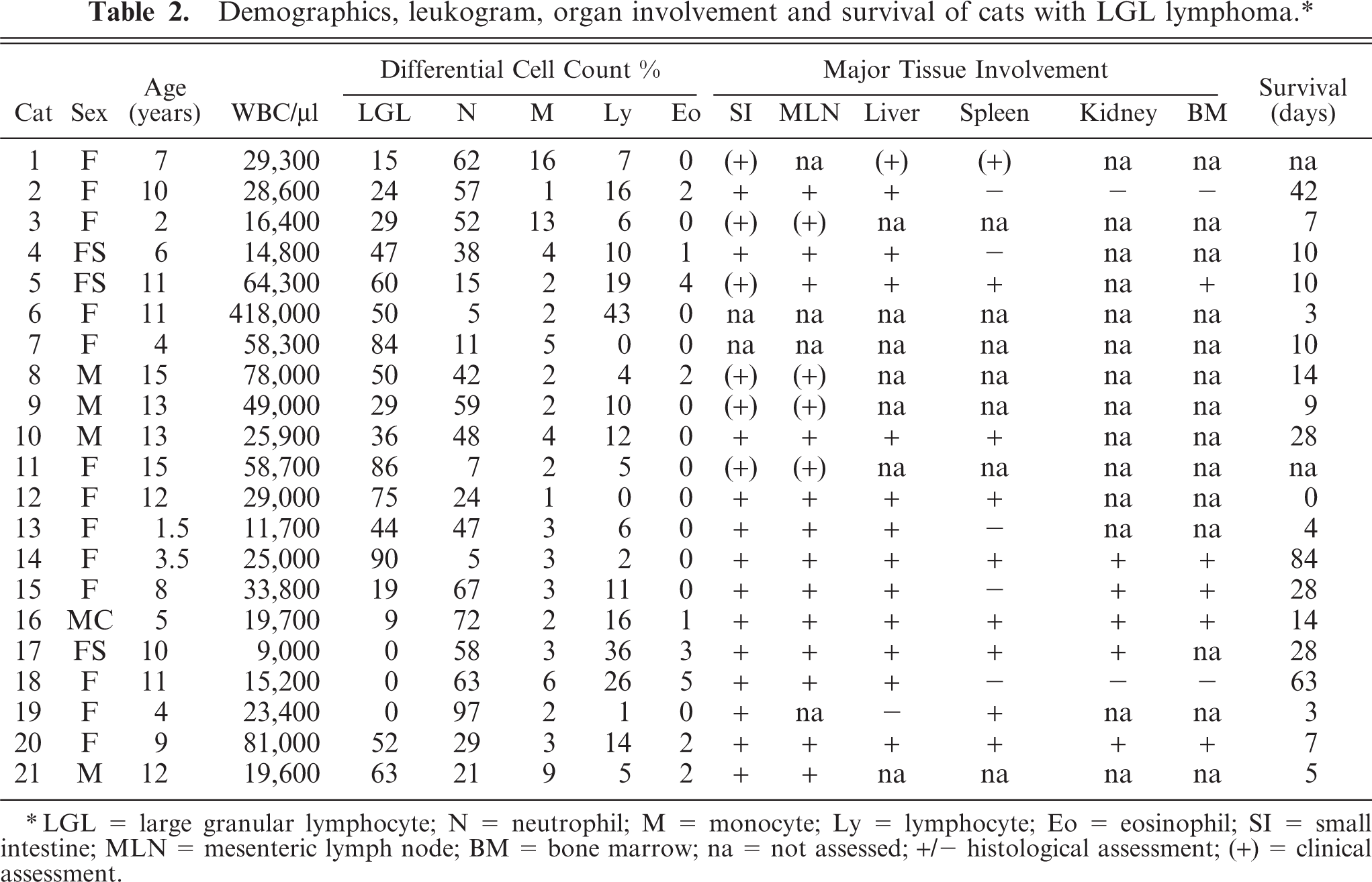
∗LGL = large granular lymphocyte; N = neutrophil; M = monocyte; Ly = lymphocyte; Eo = eosinophil; SI = small intestine; MLN = mesenteric lymph node; BM = bone marrow; na = not assessed; +/− histological assessment; (+) = clinical assessment.
The white blood count was within reference limits (4,500–14,000/µl) in 2 cats and was increased in the remaining cats (range 15,200–418,000/µl). LGLs were increased in 18/21 cats (1,773–209,000/µl; reference interval 0–350/µl). Neutrophilia (14,184–32,760/µl; reference interval 2,000–9,000/µl) was present in 12 cats (57%). Normocytic, normochromic anemia (5 cats), or hypochromic anemia (2 cats) was also present in some instances. Bone marrow was mildly to moderately infiltrated in 5/7 cats. FeLV and FIV serology was performed in 13 cats. One cat had a positive FeLV test; all were FIV negative.
Increased serum alanine amino transferase activity was present in 7/12 cats (range 136–786 IU/l; reference interval 5–130 IU/l). Total bilirubin (range 0.8–8.8 mg/dl; reference interval 0.1–0.4 mg/dl) and direct bilirubin (range 0.4–4.8 mg/dl; reference interval 0.0–0.1 mg/dl) were frequently increased (7/13). Blood urea nitrogen (BUN) was elevated in 5/14 cats (range 49–78 mg/dl; reference interval 17–35 mg/dl) while serum creatinine was increased in 2/14 cats (range 1.9–2.5 mg/dl; reference interval 0.8–1.8 mg/dl).
All patients presented with advanced disease and did not survive more than 84 days (mean 18.8 days). Survival times were only approximate because most cats were euthanized due to severe deterioration of their physical condition (15/18).
Cytology of LGL disorders
LGL neoplasia was confirmed in all cats by cytologic evaluation of peripheral blood smears and/or imprints of internal organs. In six cats, LGLs predominantly had a mature morphology (Fig. 1). Cells varied in size (8–15 µm) and were characterized by round, frequently indented nuclei with coarsely clumped chromatin and inapparent nucleoli. These cells had an incomplete rim of pale-blue cytoplasm with fine granules often grouped close to the nuclear indentation. In 13 cats, LGLs predominantly had an immature morphology (Figs. 2, 3). Cells were large (15–35 µm) with pleomorphic nuclei, dispersed chromatin, prominent nucleoli, and a variable volume of cytoplasm containing round, fine to large, irregularly distributed cytoplasmic granules. In 2 cats, an even mixture of both cell types was present. LGLs contained cytoplasmic granules that ranged from 1 to 20 per cell and were usually small (Figs. 1, 2). Coarse, large (1–2 µm) cytoplasmic granules were occasionally present (Fig. 3). Abdominal (three cats) and thoracic (two cats) effusions contained neoplastic LGLs that had a similar morphology. In many instances, lymphocytes with otherwise identical cytological features to adjacent LGLs lacked discernible azurophilic cytoplasmic granules. These were counted as lymphocytes in the differential cell counts, and not LGLs, despite the high probability that they were members of the neoplastic population. Marked variation in LGL granularity has been previously described in humans and cats. 44, 66 Cytologic appearance of LGLs did not appear to correlate with clinical behavior or subsequent immunophenotype.
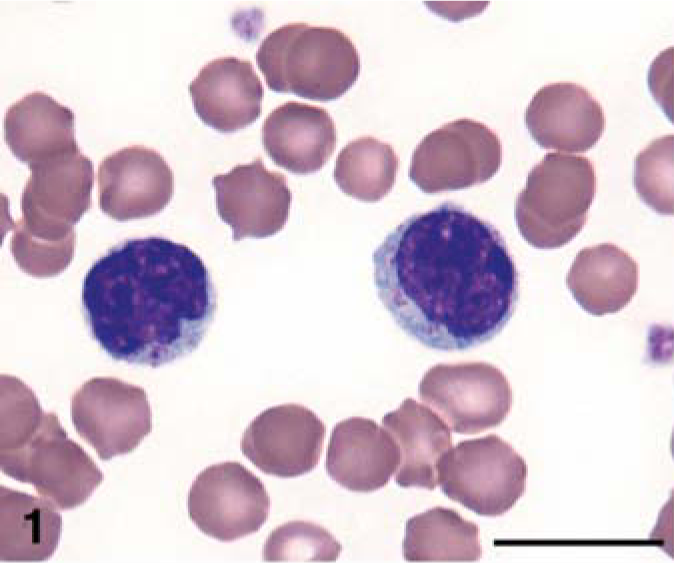
Venous blood; cat No. 10. Granular lymphocytes with mature morphology and uniform, fine, cytoplasmic granules. Wright-Giemsa stain. Bar = 10 µm.
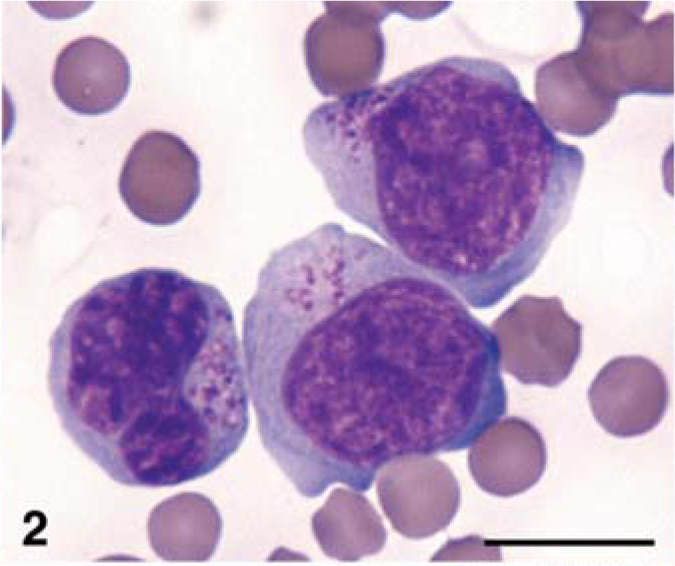
Venous blood; cat No. 11. Granular lymphocytes with immature morphology and heterogeneous cytoplasmic granules. Wright-Giemsa stain. Bar = 10 µm.
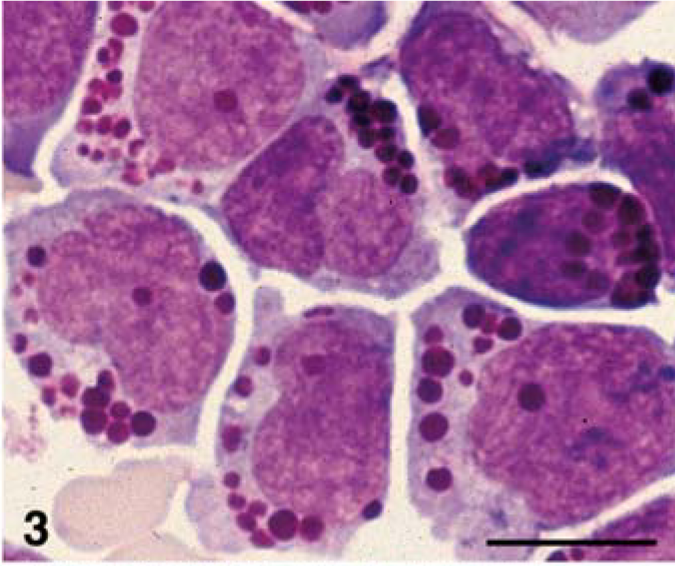
Venous blood; cat No. 15. Granular lymphocytes with immature morphology and coarse cytoplasmic granules up to 2 µm in diameter. Wright-Giemsa stain. Bar = 10 µm
Macroscopic findings
Segmental thickening or multiple masses were present in the small intestine of all cats necropsied. The jejunum, ileum, ileo-ceco-colic junction, and duodenum were affected in decreasing order of frequency. On cut section, ampulliform enlargement of the intestinal lumen with centrifugal thickening of the intestinal wall characterized all masses (Fig. 4). Areas of necrosis and hemorrhage were also present in the thickened intestinal wall (Fig. 4). Mesenteric lymph nodes in proximity of the intestinal lesions were frequently enlarged. On cut section, loss of cortico-medullary architecture was evident in most instances.
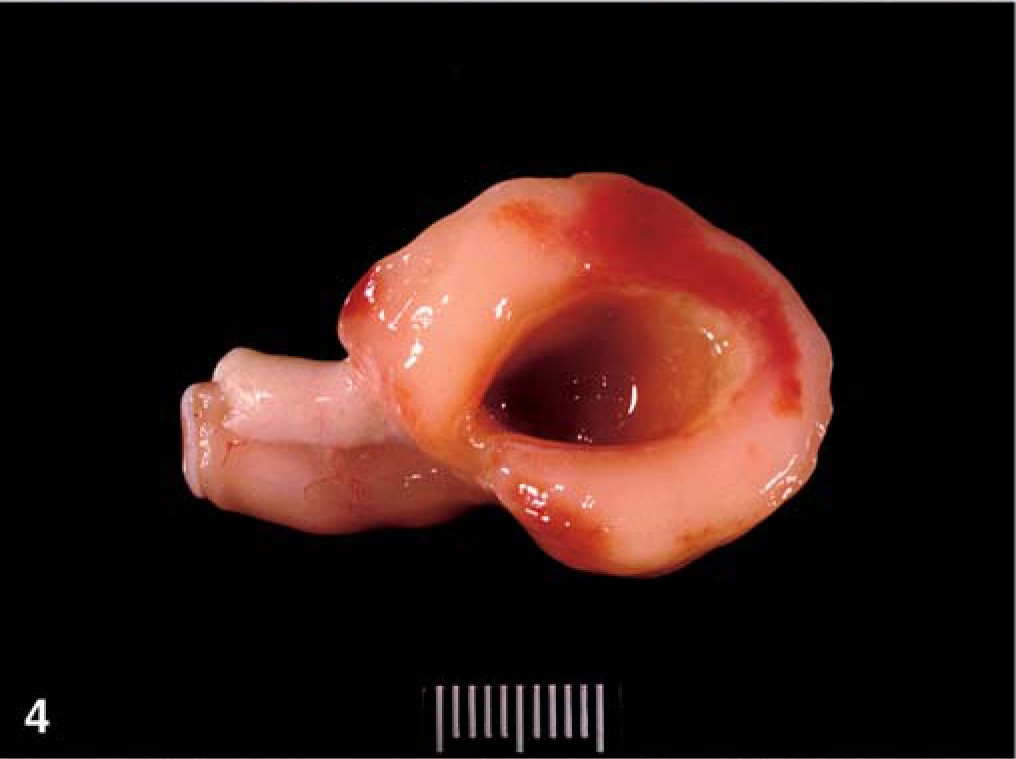
Jejunum; cat No. 19. Cross-section of an intramural, circumferential mass, which is associated with luminal dilation. Ruler = 1 cm.
Moderate to severe diffuse hepatomegaly was observed in all cats for which tissues were available. Multifocal to coalescing, white neoplastic foci were frequently observed on cut section. Hepatic lipidosis was a consistent finding. Splenomegaly with diffuse effacement of splenic architecture was evident in 8/13 cats. Renal involvement (5/7) was usually bilateral. Kidneys were enlarged and pale with multiple round to coalescing white cortical foci. In another cat, mural thickening (1.5 cm) of the urinary bladder was associated with severe bilateral cortical renal involvement. Three cats had peripheral lymph node involvement; one of these cats also had a neoplastic nodule (1 cm in diameter) in the third eyelid.
Microscopic findings
Multiple sections of small intestine were evaluated in 13 cats. Lesions were similar in the duodenum, jejunum, and ileum. The lamina propria was moderately to severely expanded by neoplastic lymphocytes in all cases. Neoplastic cells often extended to the submucosa (11 cats) and frequently invaded muscular layers (seven cats) (Fig. 5). Serosal erosion and mesenteric fat invasion were also noted (five cats). Multifocal to diffuse intestinal ulceration was present in nine cats. Epitheliotropism of neoplastic cells was prominent in nine cats (Figs. 6, 7). In three cats, ulceration was so widespread that epitheliotropism could not be accurately evaluated. In two of these cats, crypts were severely infiltrated by tumor cells. Neoplastic infiltrates were usually monomorphic and the cells ranged from small and mature with minimal cytoplasm, in five cats, to highly pleomorphic with more abundant cytoplasm in eight cats. Eosinophilic cytoplasmic granules were occasionally visible, but only on histologic preparations where neoplastic cells had abundant cytoplasm.
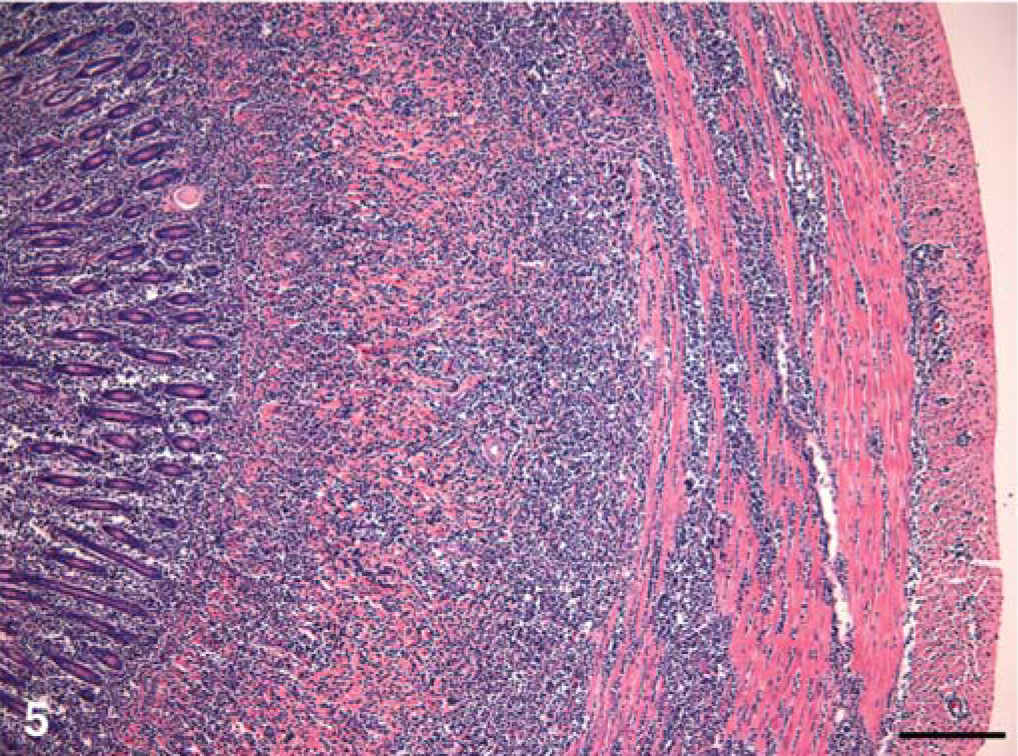
Jejunum; cat No. 20. Transmural lymphoid infiltrate. HE. Bar = 200 µm.
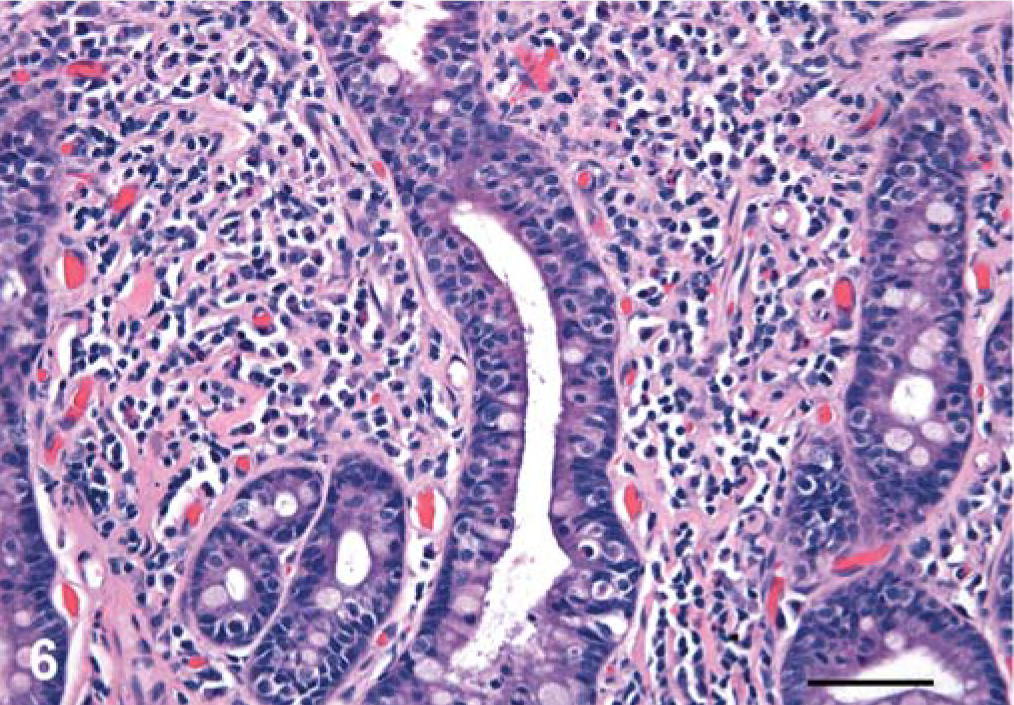
Jejunum; cat No. 21. Epitheliotropic lymphoid infiltrate in the intestinal crypts. HE. Bar = 50 µm.
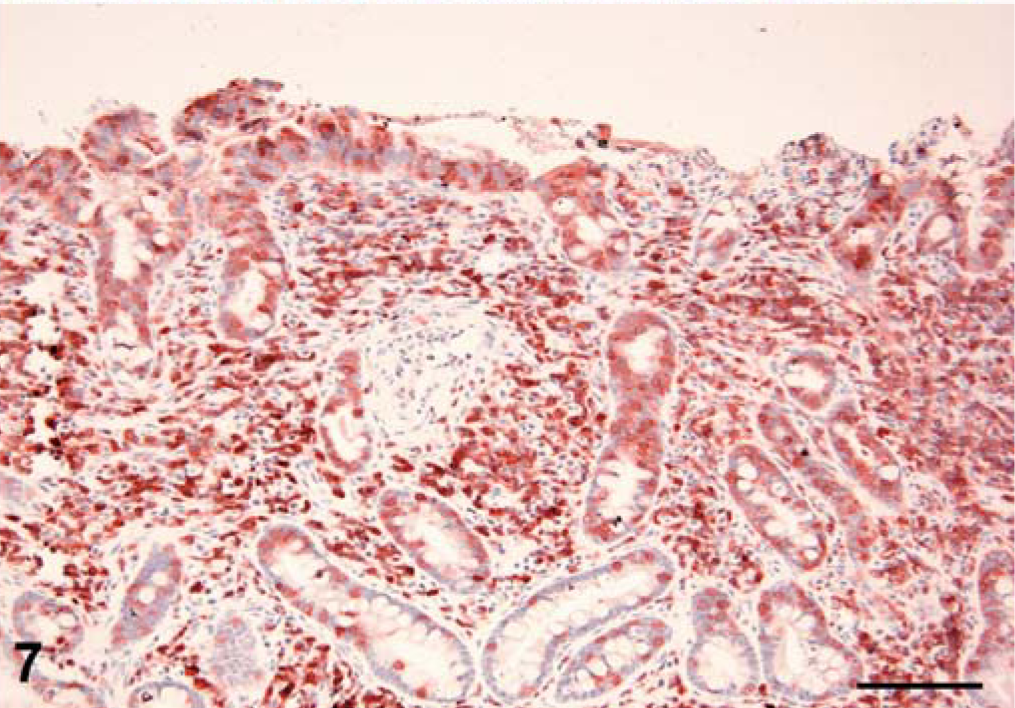
Jejunum; cat No. 21. CD3 expression by lymphocytes within the lamina proprial and intraepithelial compartments. Immunohistochemical staining: Streptavidin-peroxidase method; amino-ethyl carbazole substrate; hematoxylin counterstain. Bar = 100 µm.
Mesenteric lymph nodes were diffusely infiltrated in 13/13 cats. In six cats, lymph node architecture was completely effaced. The hepatic parenchyma contained neoplastic aggregates in 12/13 cats. Variably sized portal and centrilobular infiltrates were observed. Multinodular to diffuse infiltration of the hepatic parenchyma was also common. Additional findings in the liver were moderate to severe lipidosis (10/13), and intracytoplasmic and/or canalicular bile stasis (6/13).
The spleen was examined in 13 cats. Neoplastic infiltrates expanded splenic red pulp or effaced splenic architecture in eight cats while the spleen was spared in five cats.
The renal cortex was diffusely infiltrated by neoplastic lymphocytes in 5/7 cats. The cortex was effaced and residual glomeruli and degenerated tubules were entrapped among neoplastic lymphocytes.
Phenotype of LGL disorders
The leukocyte origin of LGL neoplasia was confirmed by expression of the leukointegrin CD18 (100%). A T cell immunophenotype (CD3ε+) was established in 19 tumors (Table 3, Figs. 7, 8). CD8αα+ cytotoxic/suppressor immunophenotype was observed in 13 tumors, two had a CD4+ helper T cell origin, two had a double-positive (CD4+CD8αα+) immunophenotype, and two had a double negative (CD4-CD8-) immunophenotype. Cytoplasmic CD8α expression only was detected in two tumors, and CD8β expression was not detected in any of the LGL tumors. CD5 expression was observed in only six tumors. In two tumors, a T cell immunophenotype could not clearly be established (CD3ε-). The leukointegrin αE (CD103) was expressed in 11/19 of the LGL tumors. MHC class II was detected in 15/20 tumors. Neoplastic LGLs did not express B cell markers (CD21, CD22, and CD79a), nor did they express CD14. Lymphocytes were phenotypically homogeneous by flow-cytometry analysis of peripheral blood, despite the variation in cytoplasmic granularity and the apparent lack of granules in some cells (Fig. 9). In all instances, neoplastic tissue infiltrates had the same phenotype as the corresponding peripheral blood LGLs. The phenotype did not appear to correlate with cytological appearance or with clinical behavior.
Leukocyte antigen expression in feline LGL lymphoma.∗
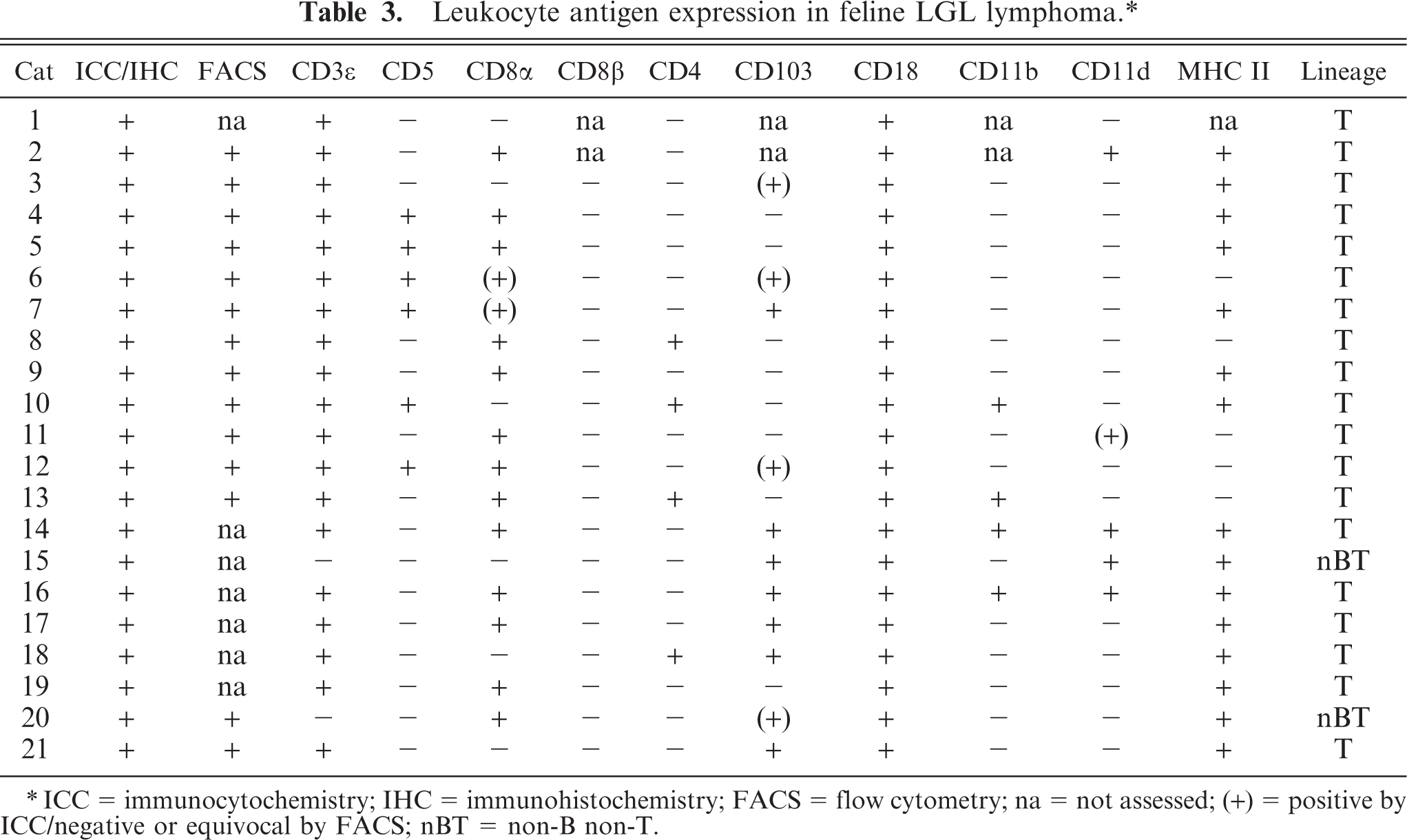
∗ICC = immunocytochemistry; IHC = immunohistochemistry; FACS = flow cytometry; na = not assessed; (+) = positive by ICC/negative or equivocal by FACS; nBT = non-B non-T.
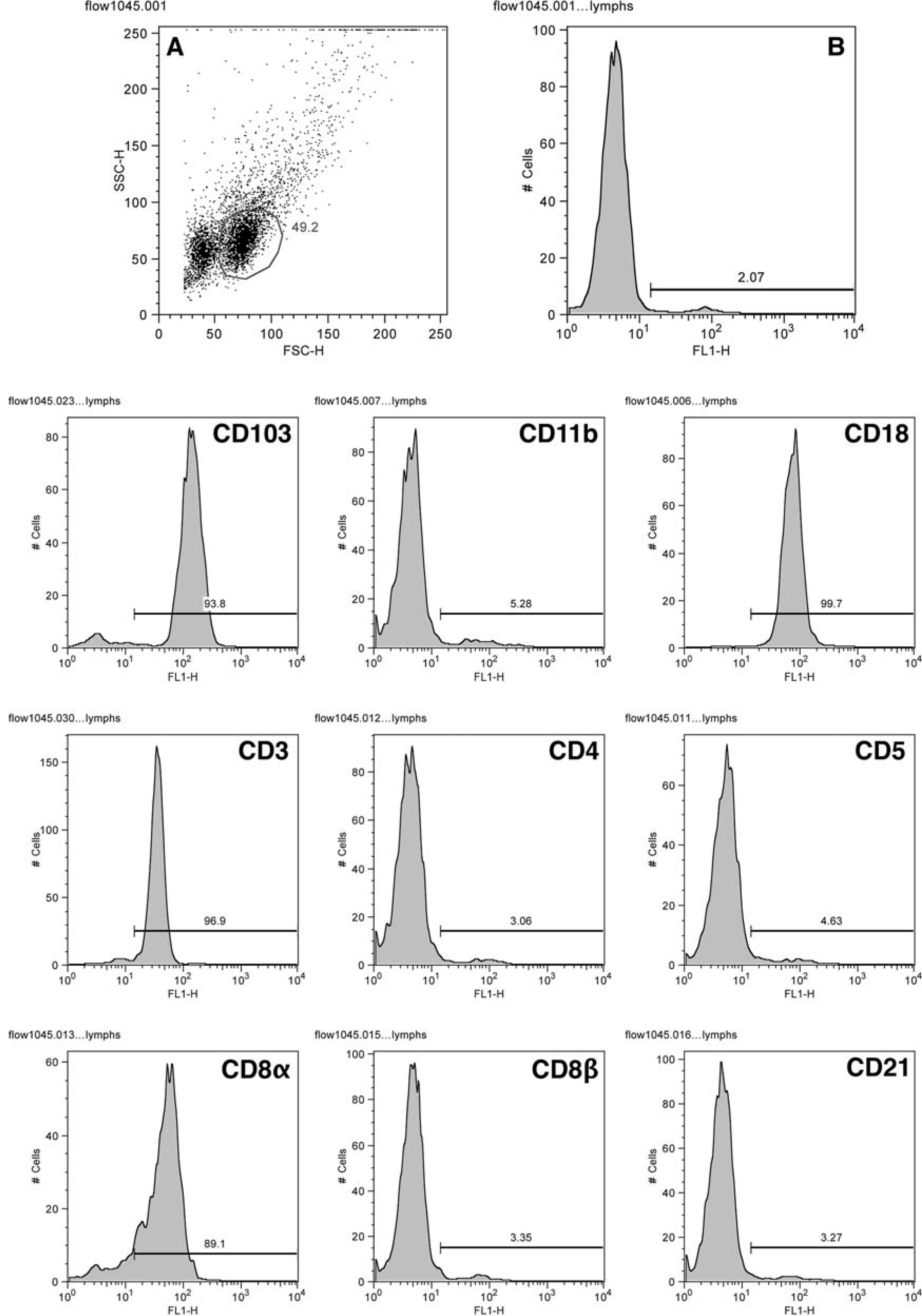
Venous blood; cat No. 11.
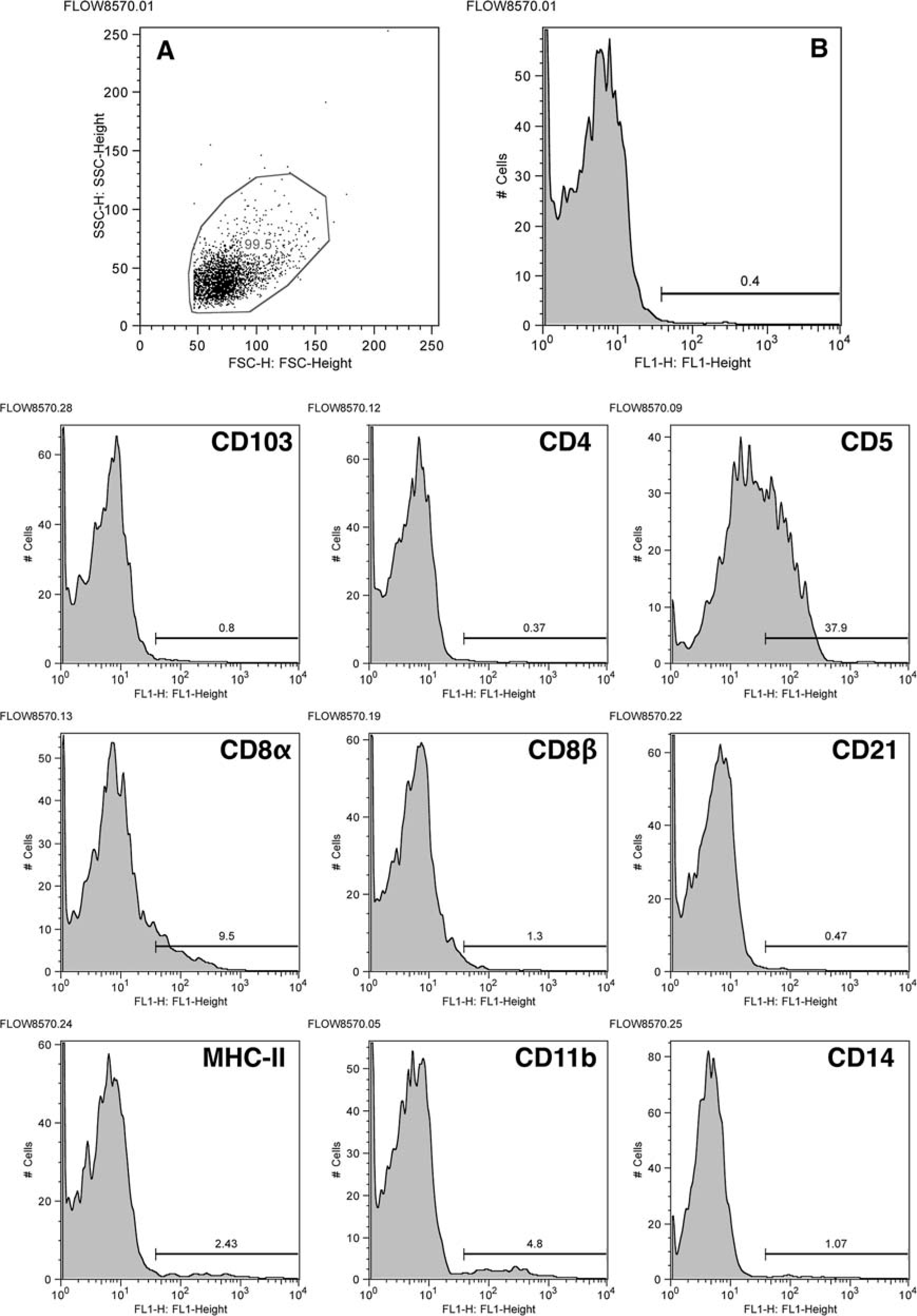
Venous blood; cat No. 6.
Discussion
Feline LGL neoplasia presented as an aggressive disease that was characterized by primary small-intestinal involvement and aggressive biologic behavior with an attendant poor prognosis. From a clinicopathologic perspective, our findings paralleled previous reports of feline LGL lymphoma. 10, 15, 20, 22, 26, 32, 37, 66 This differed from LGL neoplasia in humans and dogs, where a predominance of indolent forms have been described. 40, 44, 47, 63 Feline LGL tumors more closely resembled rare forms of human intestinal LGL lymphomas of NK origin and enteropathy associated intestinal T cell lymphoma (EATCL). 17, 19, 24, 40, 44, 50, 52, 71
Interestingly, seven cats with LGL lymphoma had a previous history of chronic IBD. In humans, EATCL is a well known consequence of chronic IBD. 2, 17, 71 EATCL seems to arise from clonal transformation of intestinal intraepithelial T cells after prolonged antigenic stimulation. 3, 4, 33 T cell receptor gene rearrangement studies have demonstrated the same clone of T cells in concurrent celiac disease, ulcerative jejunitis, refractory sprue, and subsequent EATCL. 3, 4, 33 A similar progression of the lesions could be hypothesized in cats with intestinal LGL lymphoma. If this were the case, feline LGL lymphomas may actually have a more chronic clinical course, but are diagnosed at advanced stage of disease due to lack of early clinical signs.
Feline LGL lymphomas have generally been associated with negative FeLV and FIV serology. 20, 22, 37, 66 Analysis of six LGL intestinal lymphomas at the molecular level detected FeLV insertion only in one cat. 20 In cats, T cell lymphomas are the most common FeLV-associated lymphomas but are usually of the multicentric and mediastinal type in juvenile cats. 28 In feline LGL neoplasia, only a single FIV-positive case has been described. 5 In this study, all cats tested were FIV negative by serology, and only one cat had positive FeLV serology. Similar to human and simian lentiviral infections, 42, 54 cats with natural or experimental FIV infection usually develop high-grade B cell lymphomas of the miscellaneous or alimentary type. 6 Hence, a role for FIV in the development of feline LGL lymphoma is considered unlikely.
Major clinical signs in the cats in our series were either nonspecific or were related to gastrointestinal disease and included anorexia, weight loss, vomiting, and diarrhea. Also, a palpable abdominal mass was frequently reported. These observations parallel findings of previous reports 10, 15, 20, 22, 26, 32, 37, 66 and contrast with LGL disorders reported in dogs and humans. Dogs and humans are generally asymptomatic to paucisymptomatic when diagnosed with LGL neoplasia. In these species, LGL neoplasia is typically indolent and not associated with intestinal infiltration. 40, 63 In contrast, cats with LGL neoplasia in this study generally presented with intestinal disease and an aggressive and rapidly progressing course.
Feline LGL lymphocytosis was associated with intestinal LGL lymphoma in this study. However, the majority of cats in this series were identified by screening feline peripheral blood for LGL lymphocytosis, which resulted in a significant bias for cases of intestinal lymphoma with secondary leukemia. The major issue in diagnosing primary intestinal LGL lymphoma when a leukemic blood profile is lacking is the difficulty in observing cytoplasmic granules on HE-stained sections. In this study, cytologic assessment was crucial in the diagnosis of feline LGL neoplasia. Eosinophilic cytoplasmic granules were occasionally observed in HE sections of tumors composed of immature LGLs with abundant cytoplasm. Giemsa stain and other special stains, such as periodic acid schiff and phosphotungstic acid hematoxylin, for enhanced visualization of granules have been applied with discordant results. 15, 21, 22, 48, 66 We believe that routine hematologic stains of peripheral blood and cytologic specimens are more sensitive for the recognition of LGLs and should be performed in the assessment of cats in which intestinal lymphoma is suspected. Both mature and immature LGLs were observed by cytology, but cytologic appearance did not appear to correlate with clinical presentation or prognosis. This observation parallels what has previously been described in cats, 5, 30, 37, 66 but differs from dogs in which an immature LGL morphology is associated with a more aggressive clinical course, often related to the development of acute leukemia. 63
Anemia and neutrophilia were common hematologic abnormalities. Neutrophilia has been reported as the most common leukogram abnormality in feline LGL lymphoma 66 and is in marked contrast with the consistent neutropenia that accompanies LGL disorders in humans. 40 While two cats in this report developed peritonitis secondary to intestinal rupture, inflammation was not a feature in the tissues of other cats and neutrophilia could not readily be explained. In a previously reported case of feline LGL lymphoma with severe neutrophilia, production of IL-1 and IL-3 by the neoplastic cells was demonstrated. 36 Thus, neutrophilia may represent a paraneoplastic condition associated with feline LGL lymphoma. However, this mechanism would need to be confirmed by evaluation of the cytokines produced by neoplastic LGLs in a larger series of cases.
LGL lymphoma in cats was characterized by consistent small-intestinal involvement with predominance of jejunal lesions. This distribution parallels previous descriptions 15, 22, 48, 66 and contrasts with the predominant duodenal location of epitheliotropic intestinal lymphoma (EIL) recently described in cats. 7 Distribution of other gross and microscopic lesions closely resembled previous cases and smaller series of LGL lymphoma reported in cats. 10, 15, 20– 22, 26, 30, 32, 37, 48, 66 However, compared with these reports, a higher frequency of renal involvement was noted in this series. The major difference observed between feline EIL and feline LGL leukemia/lymphoma is the tendency for feline EIL to have a prolonged clinical course with a better prognosis. 7 However, these authors also describe occasional feline EILs with aggressive behavior and poor response to therapy, similar to LGL tumors described here. 7 Cytologic specimens of intestinal lesions from cats with EIL were not available. 7 Thus, an LGL origin of these more biologically aggressive EILs cannot be excluded.
Feline LGL lymphoma was an epitheliotropic process and, in this respect, is similar to human EATCL. 17, 33, 71 LGLs have been observed in some cases of human EATCL. 60, 71 Also, EATCL usually develops in the jejunum as a complication of celiac disease or ulcerative jejunitis, and is usually characterized by epithelial ulceration. 17, 33, 71 Mucosal ulceration was also a frequent finding in the cats in this report.
In humans, primary intestinal LGL lymphomas have been described and have similar clinical behavior to feline LGL lymphomas. 19, 24, 35, 50, 52 In humans, intestinal LGL lymphoma presents as a rapidly progressive disease often associated with ascites. 40 Similar to cats in this study, hepatic sinusoid and portal infiltration, splenic invasion, bone marrow infiltration, occasional ascites, and an aggressive clinical course unresponsive to chemotherapy are observed in human LGL lymphoma. 24, 46, 50
Feline LGL lymphomas in this series had a T cell phenotype in 90% of cases. A T cell origin was assigned to LGL tumors that expressed T cell-associated markers, such as CD3ε, CD5, CD8α, and/or CD4. In our study, CD3ε expression was detected by an antibody specific for an intracytoplasmic epitope, and T cell receptor expression was not assessable. Hence, we were unable to confirm surface expression of CD3 in association with a T cell receptor, which is a characteristic of T cells. Intracytoplasmic CD3ε positivity does not completely rule out an NK origin. 61 However, evidence of feline NK cell expression of CD3ε is not available. Also, human NK cells do not express CD4 or CD5, and CD8α is expressed in a minor subpopulation of NK cells and NK cell tumors. 44 Furthermore, human LGL leukemias of NK cell origin usually lack expression of CD3. 44 Therefore, all things considered, CD3 expression is most indicative of a T cell origin. More recently, an antibody that recognizes the extracellular domain of feline CD3ε has been produced, and if available for flow cytometric assessment of feline LGL lymphocytosis, would directly address this issue. 51
Feline LGL lymphomas predominantly expressed CD8α, presumably as a homodimer, because CD8β was not expressed by LGLs in any cats. CD8 is a dimeric molecule present on cytotoxic T cells and is expressed as a CD8 αβ heterodimer, which is the prevalent isoform observed in peripheral lymphoid compartments, or as the CD8αα homodimer, which is most commonly expressed by intestinal IELs in humans and cats. 23, 56 In this study, CD8αα was expressed alone (62%) or in combination with CD4 (9%) in LGL lymphoma. CD8αα expression has not been reported previously in cats with LGL neoplasia, and its high prevalence seems unique to this species, as T cell LGL leukemias in dogs 47, 63 and humans 44, 53 more frequently express the CD8αβ heterodimer, likely reflecting the different tissue origin. A CD8αα phenotype has been observed only in a minor subset of human and canine LGL expansions. 16, 63 Intestinal CD8αα intraepithelial T cells may have a specific immunoregulatory function related to their intraepithelial location that is characterized by enhanced cytokine production and protection of epithelial cell layers. 23, 29 Also, CD4-CD8- double-negative cells are rare in the systemic circulation but may represent 10% of human intestinal IELs 23, 29 and are a large subpopulation (up to 45%) of intestinal IELs in the cat. 56 In this study, 14% of LGL lymphomas had this double-negative phenotype. Also, 9% of LGL lymphomas expressed a CD4+ T helper cell phenotype. This phenotype is rarely reported in human LGL leukemias 39, 72 and intestinal LGL lymphomas. 27, 43 Two cases in this series were not clearly assigned to the T cell lineage, as they lacked expression of CD3ε; they could represent NK cell lymphomas. However, specific markers to identify feline NK cells are not available to confirm this. Despite the different phenotypes observed in LGL lymphoma in this series, differences in clinical course were not apparent. The disease was always aggressive, regardless of phenotype, and generally was the cause of death or euthanasia of the patients.
The intestinal leukointegrin, αEβ7 (αE = CD103), was expressed by circulating LGLs in almost 60% of cats tested. Strikingly, αEβ7 is only expressed by about 1% of lymphocytes in the blood of normal cats. αEβ7 is expressed by 59–88% of feline intestinal IELs and 28–46% of lamina proprial lymphocytes. 70 αEβ7 is present on 90% of CD8+ T cells and 40–50% of CD4+ T cells in the human intestinal mucosa. 38, 57 E-cadherin, which is present on the basolateral surface of epithelial cells, is a ligand for αE (CD103) and mediates T lymphocyte adhesion to epithelial cells of several mucosal tissues, including the intestine. 8, 62 EATCL and intestinal intraepithelial T cell lymphomas in humans may also express CD103. 9, 12, 31, 71 Dominant expression of CD103 and the associated expression of CD8αα in feline intestinal LGL lymphoma are important immunophenotypic features underscoring the likely IEL origin of this tumor.
Canine LGL leukemia, which most often manifests as chronic lymphocytic leukemia (CLL), is noteworthy by its consistent expression of CD11d. 47, 63 CD11d is the αDβ2 leukointegrin and is expressed by macrophages and T cells of the splenic red pulp and by peripheral blood LGLs. In dogs, the splenic red pulp has the greatest CD11d expression compared with other canine lymphoid compartments. 14 This finding has been considered indicative of a primary splenic origin of canine LGL CLL. 63 In contrast, CD11d was only expressed by 24% of feline LGL lymphomas, consistent with their intestinal origin.
In conclusion, LGL lymphomas in cats are usually clinically aggressive neoplasms, which may manifest with an associated secondary leukemia. We contend that accurate diagnosis of LGL neoplasia requires cytologic assessment of both intestinal lesions and peripheral blood because of the difficulty in recognizing LGLs in routinely stained tissue sections. The key immunophenotypic similarities shared by feline LGL lymphomas and intestinal IELs indicate that LGL lymphomas in cats most often arise from the lymphocytes residing in the intestinal epithelium. Furthermore, a large subpopulation (25–35%) of normal feline intestinal IELs have LGL morphology, a feature not shared by lamina proprial lymphocytes. 56 From a clinical perspective, middle-aged cats presenting with anorexia, weight loss, increased peripheral blood LGLs, and abnormal liver function should always be investigated for primary intestinal LGL lymphoma.
Footnotes
Acknowledgements
We especially thank Dr. Sonjia Shelly and other clinical pathologists and pathologists at IDEXX Veterinary Services (Sacramento, CA) for providing peripheral blood and fresh tissues from numerous cats with LGL lymphoma.
