Abstract
Neuronal vacuolation and spinocerebellar degeneration in young Rottweiler dogs is a neurodegenerative condition characterized by neuronal vacuolation of several nuclei in the central nervous system and degeneration of the spinal cord white matter. Here, we describe the morphologic and ultrastructural findings in laryngeal muscles and peripheral nerves of a 16-week-old female and a 32-week-old female Rottweiler dog affected by progressive ataxia and tetraparesis associated with laryngeal paralysis. Lesions were characterized by neurogenic muscle atrophy of the intrinsic laryngeal muscles, and a loss of large myelinated fibers in the recurrent laryngeal nerve, accompanied by demyelinating/remyelinating features affecting the small myelinated fibers. No significant changes were detected in the cranial laryngeal, vagus, phrenic, ulnar, or peroneal nerves. These findings were indicative of a selective distal neuropathy of the recurrent laryngeal nerve with early severe axonal degeneration, mainly of the large myelinated fibers.
A neurodegenerative disorder characterized by selective neuronal vacuolation, diffuse myelopathy involving all funiculi of the spinal cord, and selective atrophy of intrinsic laryngeal muscles has been reported in young Rottweiler dogs in the USA, Europe, Brazil and Australia.
1,
7,
10,
11,
13,
14
The condition, designated as
A 16-week-old female (dog No. 1) and a 32-week-old female (dog No. 2) Rottweiler dog were admitted to the Clinica Valdinievole (Monsummano Terme, Italy) and to the Faculty of Veterinary Medicine, Pisa University, respectively. The dogs originated from different litters. Dog No. 1 presented with an 8-week history of progressive pelvic limb ataxia. Clinical examination revealed dyspnea, cough, inspiratory stridor, failure to bark, difficulty in swallowing, and mild tetraparesis with propriocetive deficits more severe in the pelvic limbs. Bilateral microphthalmus with protrusion of the third eyelids was also present. Dog No. 2 presented with a 16-week history of inspiratory stridor and dyspnea. Clinical examination revealed dyspnea, cough, spinal ataxia, and tetraparesis with proprioceptive deficits more severe in the pelvic limbs. Bilateral decreased menace response, microphthalmia, and immature bilateral cataracts were also observed.
Because of poor prognosis, both dogs were euthanized and a complete necropsy was performed. The entire central nervous system (CNS) was fixed in 4% buffered formalin and routinely processed for histology. Samples of RLN, cranial laryngeal nerve, intrathoracic vagus, phrenic, ulnar, and peroneal nerves were fixed in 2.5% glutaraldehyde in 0.1 M phosphate buffer at 4°C, postfixed in osmium tetroxide, dehydrated in ethanol, and embedded in epoxy resin. A distal portion of RLN was sampled approximately 5 cm from the larynx and a terminal portion of the nerve was sampled inside the larynx after removal of the thyroid cartilage. Semithin sections were stained with toluidine blue, and ultrathin sections were stained with uranyl acetate and lead citrate. Samples of intrinsic laryngeal muscles (cricoarytenoideus dorsalis, thyroarytenoideus and cricothyroideus) and thyrohyoideus muscle were frozen in isopentane precooled in liquid nitrogen. Frozen sections were stained with hematoxylin and eosin, periodic acid-Schiff, Gomori trichrome stain, Oil Red O, and myosin adenosine triphosphatase (ATPase) at pH 4.3 and 9.8. Morphometric analysis was performed on nerve and muscle samples using a computerized video-display image analysis system for quantitative studies (MacIntosh Image 1.49®). Mean external diameters of myelinated nerve fibers were determined from a random field fiber count of 500 for each nerve sample. Laryngeal nerves and fresh-frozen laryngeal muscles from an 8-month-old, neurologically normal Rottweiler dog were used as controls.
Histologic examination of the CNS in both dogs revealed bilateral and symmetrical vacuolation of neurons in several brainstem nuclei, especially the cerebellar and vestibular nuclei. Ambiguus nuclei showed no degenerative change or neuronal loss. Lesions in the spinal cord, especially affecting the dorsolateral and ventral funiculi, were characterized by different degrees of axonal degeneration and secondary demyelination with swollen axons, edema, and vacuolation of myelin sheaths. A mild astrogliosis was associated with spinal cord white matter degeneration. The cricoarytenoideus dorsalis (Fig. 1) and thyroarytenoideus muscles had large groups of small fibers with an angular outline and some hypertrophic fibers. Both type 1 and type 2 fibers were affected by angular atrophy and no fiber grouping was evident. The cricothyroideus and thyrohyoideus muscles were normal. The cricothyroideus and the thyroarytenoideus showed a mild and pronounced predominance of type 2 fibers, respectively. Samples of the RLN showed a loss of large-caliber myelinated fibers with a prevalence of small myelinated fibers. The distal portion of the RLN in both affected dogs and in the control was composed of two different fascicles (Fig. 2). In the control, one fascicle had a prevalence of large-caliber myelinated fibers, whereas the other one had a prevalence of small-caliber myelinated fibers. Fiber diameter distribution in the control RLN had a bimodal distribution with peaks at 3 µm and 8 µm. In affected dogs, the two different fascicles were still recognizable, but morphometric analysis demonstrated a unimodal distribution with the majority of fibers having a diameter less than 4 µm. The fascicle composed of large-caliber fibers showed an almost total loss of myelinated fibers and the remaining medium-caliber myelinated fibers showed inappropriately thin myelin sheaths compared to the axonal caliber. No clear evidence of active ongoing axonal degeneration was observed. Moderate loss of large-caliber myelinated fibers and occasional large rounded cells with eccentric nuclei and granular or finely vacuolated cytoplasm were evident in the fascicle with small myelinated fibers. These cells were interpreted as macrophages. Sometimes groups of three or more closely apposed thinly myelinated axons were present, which could be interpreted as regenerative clusters because of axonal sprouting (Fig. 3). Lesions were more prominent in the distal portion of the RLN. Ultrastructural examination revealed a lack of large-caliber myelinated fibers and scattered myelin-like debris in Schwann cell cytoplasm, but little evidence of active axonal degeneration. Some smaller myelinated fibers had axons with 3–5 lamellae and were adjacent to myelinated fibers that had axons of similar caliber but thicker myelin sheaths. Reduplication of Schwann cell basal lamina surrounded some of these fibers (Fig. 4). The cranial laryngeal, vagus, phrenic, ulnar, and peroneal nerves revealed no abnormalities and were morphologically and morphometrically similar to control nerves.
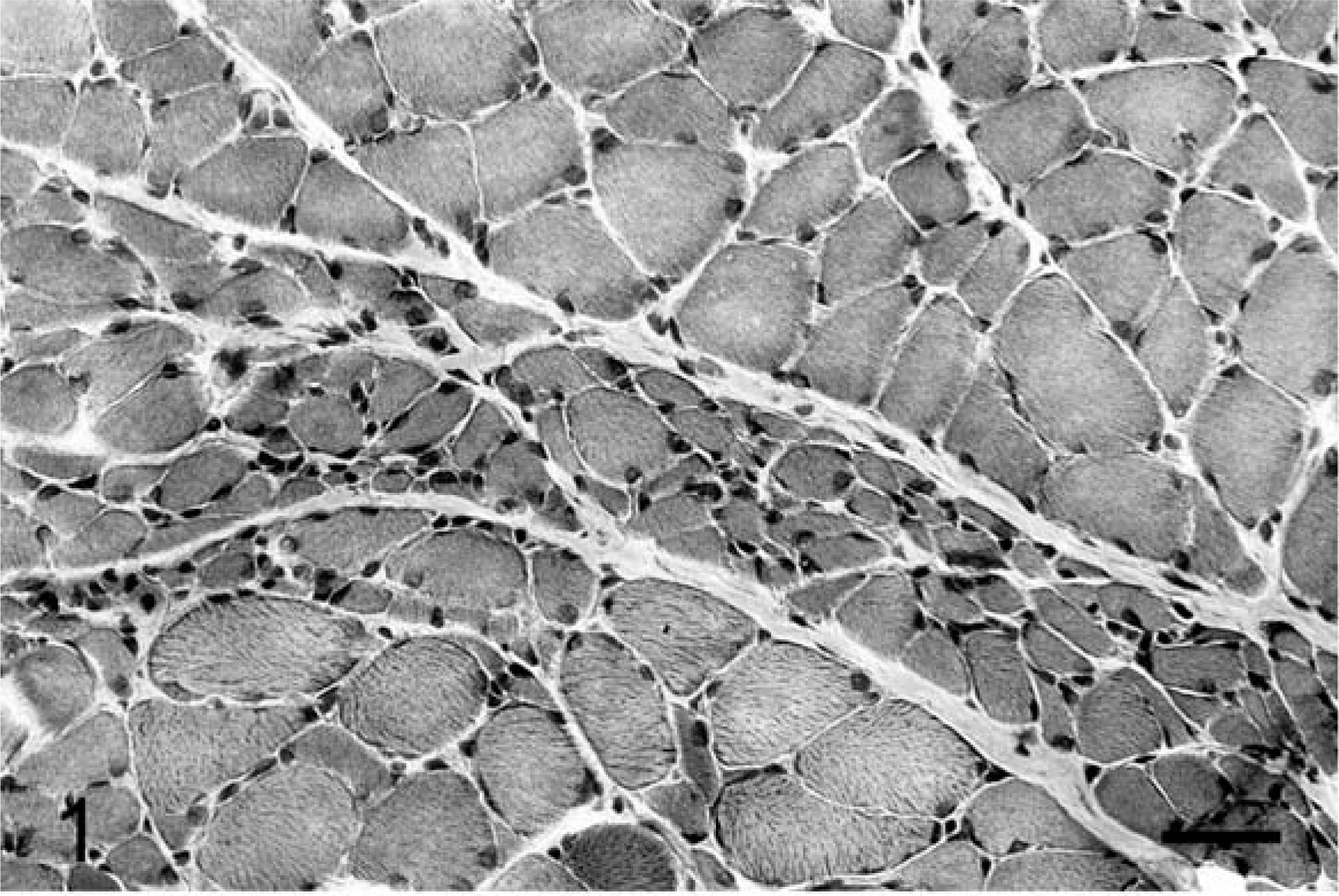
Cricoarytenoideus dorsalis muscle; dog No. 1. A group of many angular atrophic fibers (large grouped atrophy) is evidentGomori trichrome stain. Bar = 35 µm.
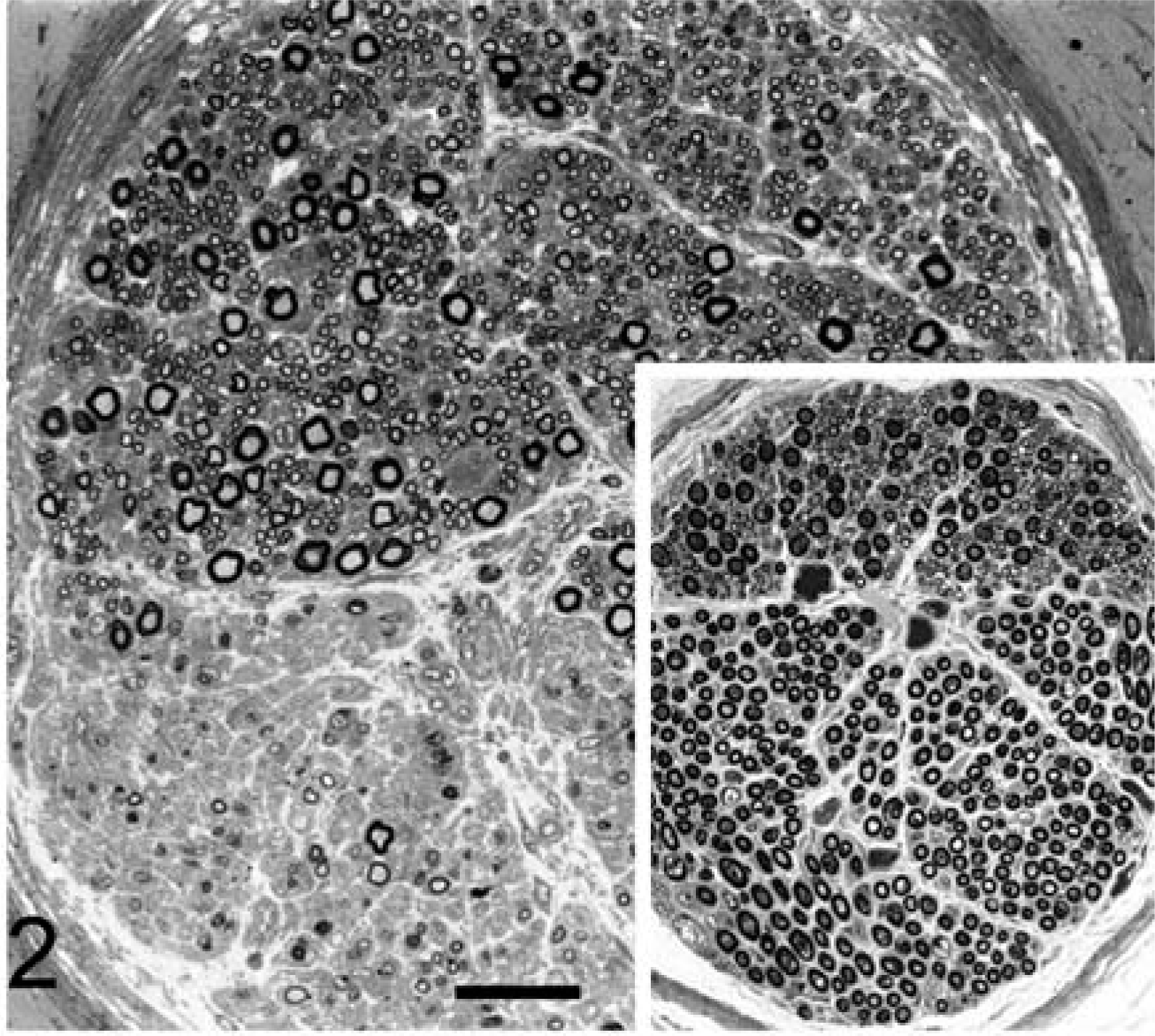
Distal recurrent laryngeal nerve; dog No. 2.
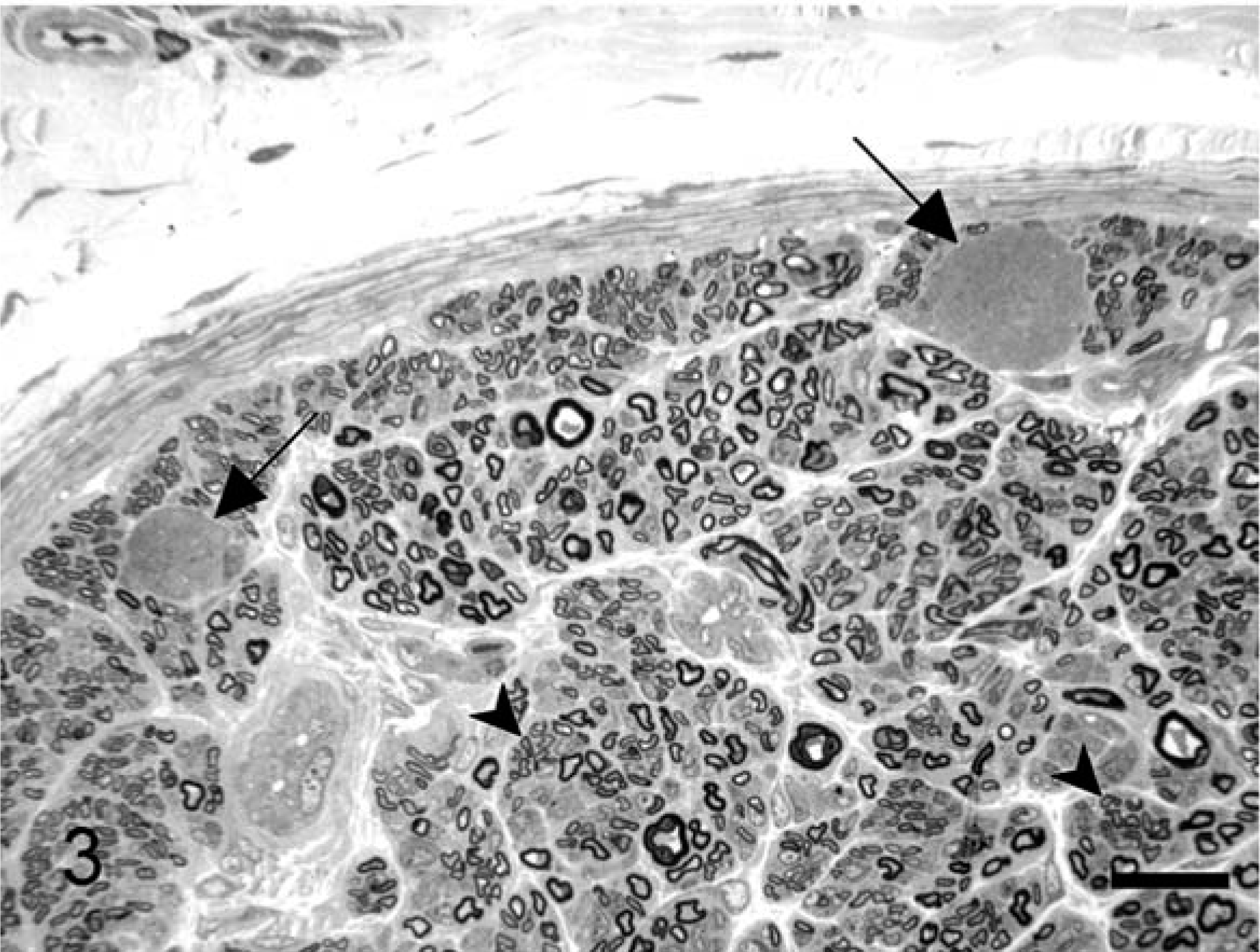
Distal recurrent laryngeal nerve; dog No. 1. Two large rounded cells (arrows) with microvacuolation of the cytoplasm are present in the endoneurium. Loss of large-caliber fibers and thinly myelinated fibers sometimes grouped in regenerative clusters are evident (arrowheads). Toluidine blue stain. Bar = 15 µm.
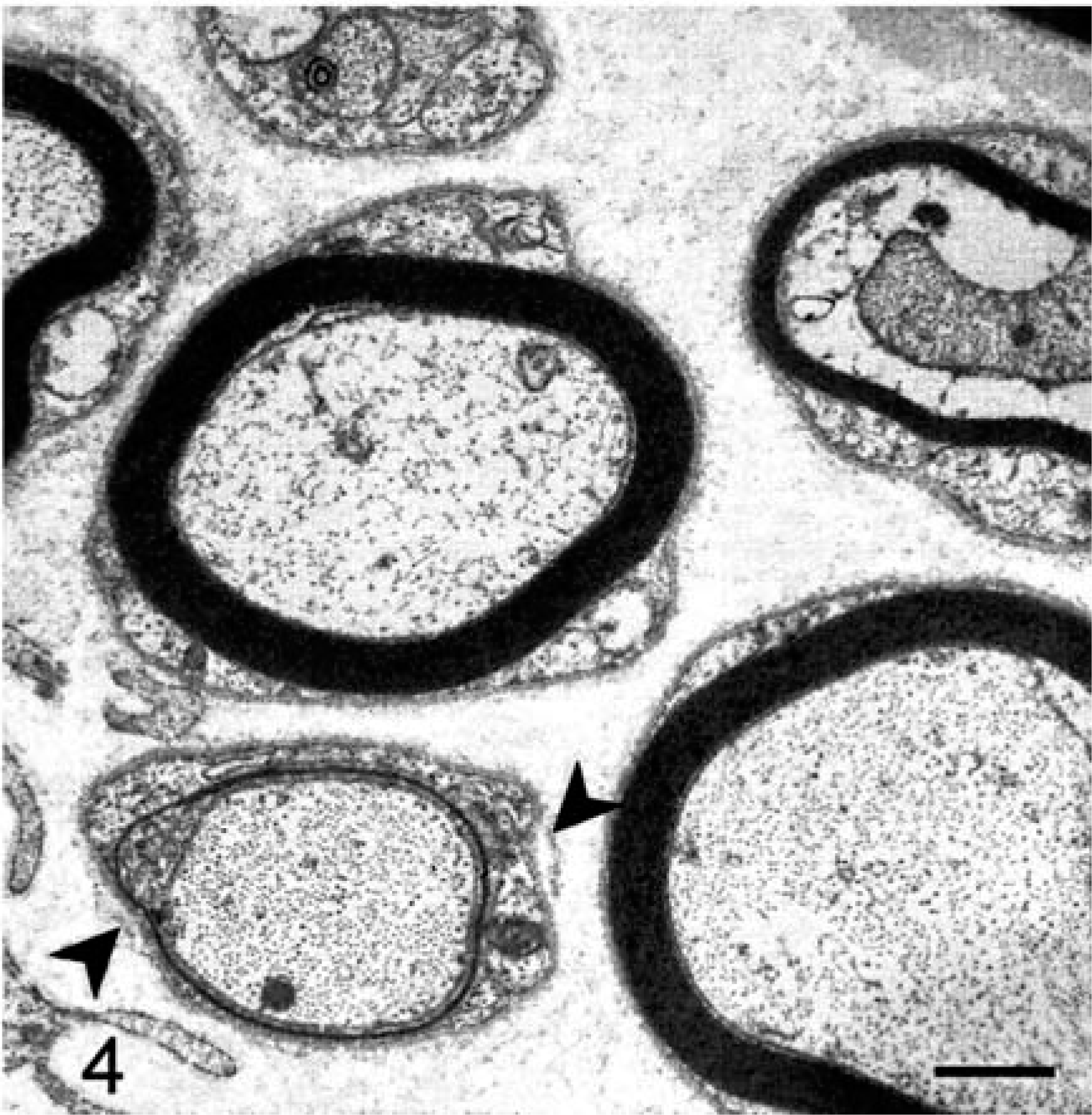
Distal recurrent laryngeal nerve; dog No. 2. Two small myelinated fibers with inappropriately thin myelin sheaths are evident at lower left and upper right corners. Redundant bits of basal lamina surround a thinly myelinated fiber (arrowheads) at lower left. Uranyl acetate and lead citrate. Bar = 2.5 µm.
Our clinical and pathologic results are consistent with the findings described in previous reports of young Rottweiler dogs affected by neuronal vacuolation and spinocerebellar degeneration. 1, 7, 10, 11, 13, 14 The clinical and neuropathologic features of this syndrome have been extensively described and reviewed. 1, 5, 7, 10, 11, 13, 14 A dying back–type neuropathy has been suspected as the cause of laryngeal paralysis. 5, 7
Cricoarytenoideus dorsalis and thyroarytenoideus muscles, which are innervated by the RLN, showed features of active neurogenic atrophy, without changes in the ratio of type 1 to type 2 fibers. Indeed, a pronounced type 2 fiber predominance in the thyroarytenoideus has been described in clinically normal dogs. 3 No fiber grouping was detected, indicating an absence of reinnervation. The muscles innervated by the cranial laryngeal nerve (cricothyroideus and thyrohyoideus) were normal. The pathologic changes seemed to selectively affect the distal portion of the RLN, and loss of axons in intramuscular nerve fascicles has been described. 10 Because the RLN originates from the vagus nerve 6 and no vacuolation or pathologic changes affected the nucleus ambiguus, the lesions in the laryngeal nerves are suggestive of a selective peripheral neuropathy.
In affected dogs, the loss of myelinated fibers in both fascicles of distal RLN was striking. Areas devoid of myelinated fibers are regarded as normal features of the canine proximal RLN. 4 However, the fiber distribution of the RLN in affected dogs was different from the control nerves and from the data reported in the literature. 4 Indeed, in clinically normal dogs, the myelinated fiber diameter distribution has been reported to show a bimodal distribution with peaks at 3 µm and 6 or 7 µm, whereas the affected dogs had an unimodal distribution. Although no active axonal degeneration was detected by electron microscopy, the lack of large-caliber myelinated fibers and the presence of scattered axonal sprouts suggest a past degenerative process. Moreover, thin myelin sheaths associated with reduplicated Schwann cell basal lamina likely represent the remaining sprout of a regenerative cluster or an individual regenerating fiber. However, these profiles are also consistent with demyelination and subsequent remyelination involving smaller myelinated fibers that frequently show inappropriately thin myelin sheaths. Taken together, these observations support a neuropathic process consisting of a primary axonopathy with abortive regeneration affecting larger fibers that is accompanied by a demyelinating component involving smaller myelinated fibers.
Hereditary, idiopathic, and acquired forms of laryngeal paralysis have been noted in several dog breeds. 12 In young Rottweiler dogs, laryngeal paralysis has been described associated with neuroaxonal dystrophy 2 and in the laryngeal paralysis–polyneuropathy complex. 12 In the latter cases, the described lesions involved motor and sensory limb nerves as well as autonomic nerves, and were characterized by multifocal axonal degeneration and nerve fiber loss that was more severe in the distal portions. In our report, no lesions were detected in limb nerves or other autonomic nerves. However, a slight axonal degeneration has been described in the ischiatic nerves in a previous report. 14 The laryngeal neuropathy in the neuronal vacuolation and spinocerebellar degeneration affecting young Rottweiler dogs documented here is indicative of a distal neuropathy where early, severe axonal degeneration of the large-caliber myelinated fibers may be accompanied by a demyelinating process with subsequent remyelination involving small myelinated fibers.
The pathologic features of recurrent laryngeal neuropathy in Rottweiler dogs are rather similar to the distal neuropathy of equine idiopathic laryngeal hemiplegia that also has an early age of onset. In the equine disease, the adductor branch of the left RLN shows marked distal loss of the medium/large-caliber myelinated fibers with swollen axons, regenerating clusters, onion bulbs and endoneurial fibrosis. 9 Moreover, features of axonal degeneration, characterized by paranodal accumulation of dense lamellar bodies and mitochondria with axonal swelling, are accompanied by chronic demyelination and remyelination. 9 The etiology of equine laryngeal hemiplegia is unknown, but several causes, such as nerve compression, lead intoxication, injections, and guttural pouch mycosis, have been recognized. 8
