Abstract
A 6-month-old, female, intact Rottweiler dog was presented to the Iowa State University Veterinary Teaching Hospital for a progressive history of abnormal behavior and generalized ataxia. At necropsy, there was eosinophilic infiltration of the brain and spinal cord, most severe in the medulla oblongata, cerebellum, and cervical spinal cord. Infiltrates of eosinophils were also present in the liver and small intestines. The dog was diagnosed with idiopathic eosinophilic meningoencephalomyelitis based on cerebrospinal fluid analysis, histopathology, and special stains to exclude etiologic agents.
Eosinophilic meningoencephalitis is a rare condition in veterinary medicine; the etiology in the majority of canine cases is often undetermined. Protozoan and nematode parasites can occasionally cause eosinophilic encephalitis in dogs. 5 In humans, the most common cause of eosinophilic meningitis is a result of infection with a rat lungworm (Angiostrongylus cantonensis). This infection is usually nonfatal and occurs after ingesting third-stage larvae of the lungworm in contaminated produce or inadequately cooked snails. 7 In the past 2 decades, there have been several reports of an idiopathic form of eosinophilic meningitis that has been shown to affect dogs, cats, and cattle. 1–3,6,8–10 In these cases, no infectious etiology has been identified; however, Rottweiler and Golden Retriever dogs appear to be overrepresented, indicating a possible breed predisposition. Dogs with eosinophilic meningoen-cephalitis often have signs consistent with both brain and spinal cord disease; however, no report has histologically documented the spinal cord changes. In the current report, a young Rottweiler dog with an acute onset of a severe and fatal eosinophilic meningoencephalomyelitis is described.
A 6-month-old, 20 kg, female, intact Rottweiler presented to the referring veterinarian with a 6-day history of mild ataxia, lethargy, and decreased appetite. Blood work was performed, including complete blood cell count (CBC) and biochemical profile. The following abnormalities were noted: hypercholesterolemia (328 mg/dl, reference interval: 125-260 mg/dl) and moderate eosinophilia (4,200/μl, reference interval: 0-600/μl). A urinalysis was also performed, which revealed the presence of white blood cells, red blood cells, and bacteria in the sediment. The dog was given clindamycin (300 mg orally, every 12 hr), kept for a few hours for observation, and then sent home. The following day, the dog presented to Iowa State University Veterinary Teaching Hospital (ISU-VTH, Ames, Iowa) with worsening tetraparesis and proprioceptive ataxia.
On presentation to the ISU-VTH, the dog was in lateral recumbency, but was quiet, alert, and responsive. On physical examination, the dog had a heart rate of 100 beats per min, a rectal temperature of 40.6°C, and was panting. Generalized muscle tremors were present along with apparent hypersensitivity to touch and sound. On neurological evaluation, the dog had appropriate mentation but was quiet. Anisocoria was present with a dilated nonresponsive left pupil. Mildly contracted facial muscles were present on the right in conjunction with deviation of the head to the right, and torticollis to the right. All 4 limbs were held in rigid extension, and pain was elicited with manipulation of the front limbs. Conscious proprioception was delayed in the hind limbs and absent in the front limbs. Withdrawal reflexes were decreased in both front limbs and normal in the hind limbs. Pain was elicited on palpation of the cervical spine. Based on the neurological examination, the lesion was determined to be multifocal with signs of cerebral disease and a C6-T2 myelopathy. Additional medical history stated that the dog was allowed to roam free over an acreage unattended and had only been vaccinated once for Canine distemper virus and had never been vaccinated for Rabies virus.
The dog was hospitalized and treated with acepromazine, hydromorphone, penicillin, metronidazole, and intravenous fluids. The following day, the dog was anesthetized, and a computerized tomography of the brain was performed. The results of the scan were within normal limits, with no evidence of space-occupying or contrastenhancing lesions. Following the scan, a sample of cerebrospinal fluid (CSF) was collected from the cisterna magna and submitted for routine analysis (cell count, cytology, and protein analysis). The results revealed a white blood cell count of 1,251 cells/μl (reference interval: 0-5 cells/μl), red blood cell count of 45 cells/μl (reference interval: 0-5 cells/μl), and protein concentration of 90.2 mg/dl (reference interval: 10-25 mg/dl). The fluid cytology was highly cellular with a marked increase in eosinophils and fewer large mononuclear cells; a differential count was not performed. No etiologic agent was observed. The CSF results were consistent with severe eosinophilic meningitis.
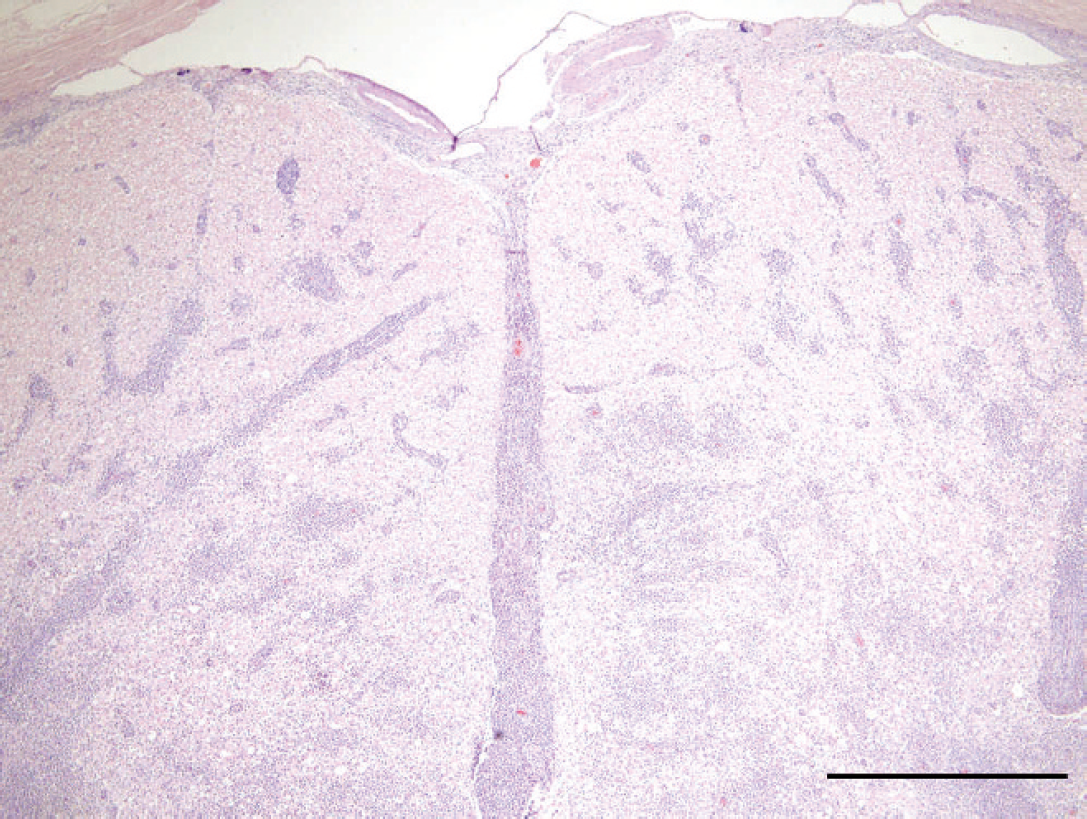
Spinal cord; dog. Large perivascular accumulations of eosinophils are present throughout the cervical spinal cord. Hematoxylin and eosin. Bar = 0.5 mm.
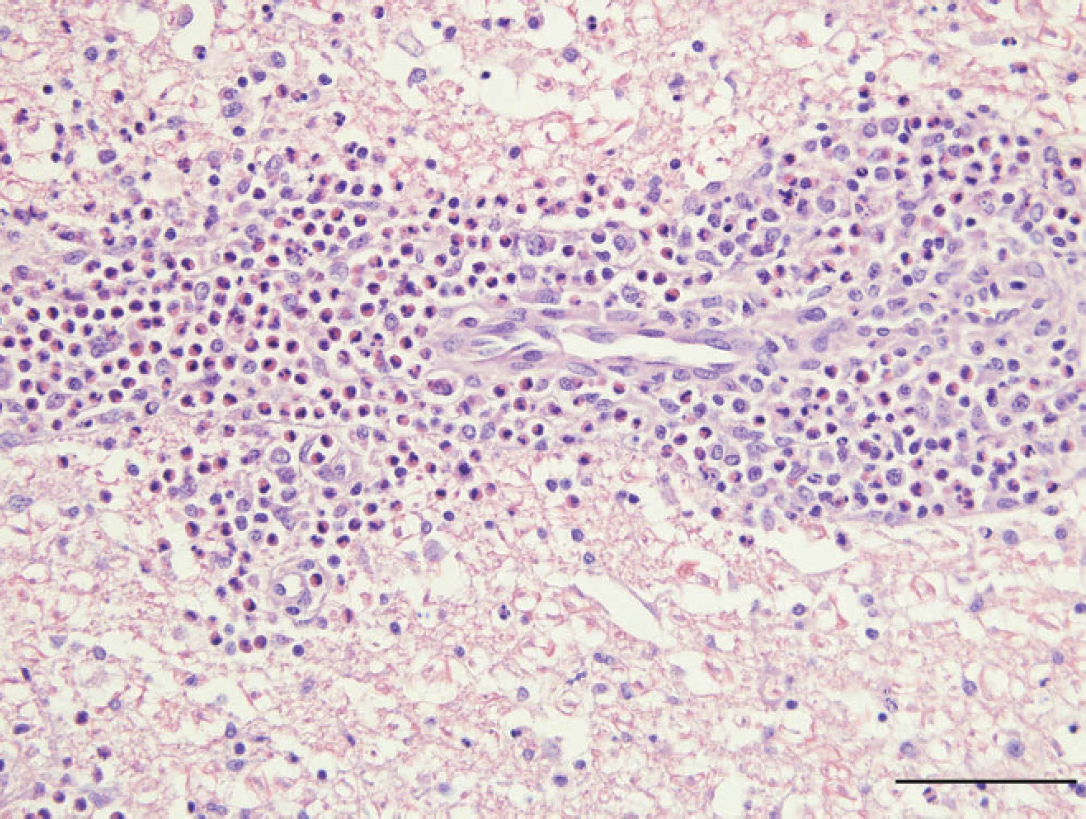
Cervical spinal cord; dog. Virchow-Robin spaces are markedly expanded by large numbers of eosinophils admixed with fewer lymphocytes, plasma cells, and macrophages. Inflammatory cells extend into the neuropil. Hematoxylin and eosin. Bar − 50 μm.
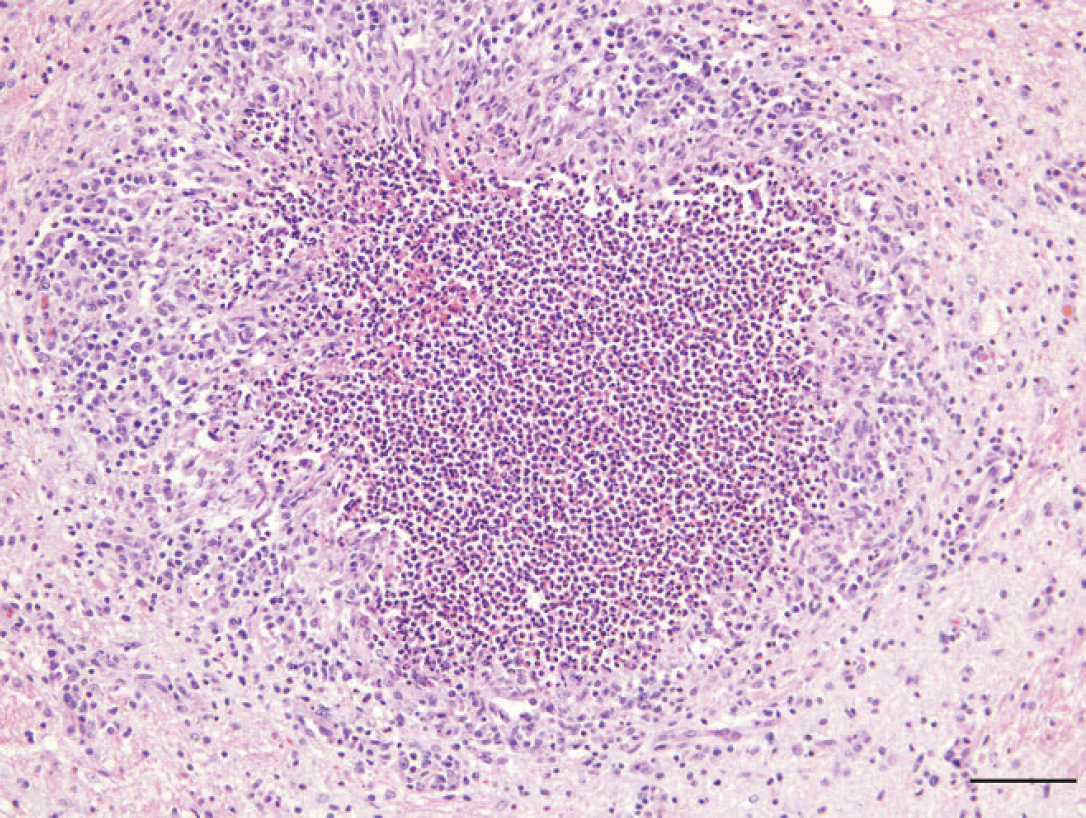
Medulla oblongata; dog. Locally extensive parenchymal loss is replaced by eosinophils and cellular debris. This area is surrounded by reactive astrogliosis and scattered lymphocytes and plasma cells. Hematoxylin and eosin. Bar = 100 μm.
The dog was given intravenous corticosteroids and supportive care. The dog deteriorated neurologically, and because of the poor prognosis for return to function, the owners elected euthanasia. Following euthanasia, the dog's body was submitted for a full necropsy including ancillary rabies testing.
A complete necropsy was performed, and a complete set of tissues were sampled for histopathology, including brain and spinal cord. Tissues were fixed in 10% neutral buffered formalin, processed routinely, sectioned at 5μm, and stained with hematoxylin and eosin. Replicate tissue sections of brain and spinal cord sections were negative for Canine distemper virus, Toxoplasma gondii, and Neospora caninum via immunohistochemistry.
Microscopically, the leptomeninges of the brain and spinal cord were moderately infiltrated by eosinophils, lymphocytes, plasma cells, and macrophages. Marked expansion of the Virchow-Robin spaces by large numbers of eosinophils admixed with fewer lymphocytes, plasma cells, and macrophages was present throughout all central nervous system (CNS) sections but was most severe in the medulla oblongata, cerebellum, and cervical spinal cord. Cellular infiltrates often extended into the neuropil (Figs. 1, 2). The macrophages often contained eosinophilic globular cellular debris. In the medulla oblongata and cervical spinal cord, locally extensive areas of parenchymal loss were characterized by large aggregates of eosinophils and cellular debris surrounded by astrogliosis with adjacent areas of vacuolation and swollen axons (Fig. 3).
Microscopically, the liver had moderate infiltrates of eosinophils, lymphocytes, and plasma cells within several portal tracts. A moderate infiltrate of eosinophils, lymphocytes, and plasma cells was also present in the lamina propria of the small intestine.
In the present report, the CSF evaluation and histologic findings confirmed a diagnosis of eosinophilic meningoencephalomyelitis. The clinical signs in the dog were multifocal, which was not uncommon based on previous reports. 1,2 Extensive areas of parenchymal loss caused by severe eosinophilic infiltration were present in the cervical spinal cord and correlated with the clinical signs of decreased spinal reflexes in both thoracic limbs and the decreased motor activity and conscious proprioception identified in all 4 limbs.
In addition to the spinal cord involvement, moderate eosinophilic infiltration was also observed in the liver and small intestine. Peripheral eosinophilia with eosinophilic infiltration of the respiratory tract, gastrointestinal tract, spleen, and bone marrow has been reported in hypereosinophilic syndrome. 4 Thus, idiopathic eosinophilic meningoencephalitis may represent a continuum of the hypereosinophilic syndrome. A Swedish study of dogs with eosinophilia found that 34% of the cases involved Rottweiler dogs. 3 Such data suggest that this breed may have a predisposition for hypereosinophilic disorders.
Common causes of hypereosinophilia in dogs include allergic disease, parasitic infections or other infectious diseases, and neoplastic disorders. The dog described herein had no evidence of neoplasia, and immunohistochemical staining was negative for Canine distemper virus, T. gondii, and N. caninum. Another differential diagnosis is fungal disease, but no evidence of fungi was found on microscopic examination. As no infectious agents were apparent after extensive evaluation, the eosinophilic inflammation in the present case was considered idiopathic.
Research has shown that the presence of eosinophils in the CNS can lead to eosinophil-induced neurotoxicity of neurons and myelinated axons. 11 In addition, free radicals produced by various enzyme pathways can lead to further cellular injury. 9 One could therefore conclude that with large numbers of eosinophils in the CNS, the prognosis for recovery would be decreased, even following appropriate therapy. However, there is not a clear association between eosinophil number and disease severity, likely because of the small number of cases available for study.
In the current case, treatment with corticosteroids was begun after the tentative diagnosis of eosinophilic meningitis was made. Unfortunately, the dog's neurological status deteriorated in spite of therapy. In the veterinary literature, the response to treatment with corticosteroids is quite variable. 1,4,9 In a recent retrospective report, the majority of dogs (8/12) diagnosed with idiopathic eosinophilic meningoencephalitis and treated with corticosteroids had resolution of clinical signs. 12 More cases will need to be followed before an accurate prognosis for this disease can be determined.
