Abstract
Sixty-three cases of benign and malignant canine mammary tumors were analyzed to define the alteration of exons 5–8 for the p53 tumor suppressor gene using polymerase chain reaction direct sequence analysis with paraffin-embedded tissues. Four missense mutations were found in 38 benign mammary tumors (11%), and five missense (one tumor had two missense mutations) and one nonsense mutations were found in 25 mammary carcinomas (20%). These data suggest that the p53 gene alterations might be initiated at an early stage of canine mammary carcinogenesis and p53 mutations might be associated with malignancy. However, there was no evidence of any relationship between the p53 alterations and the histologic types of tumors or breeds of dogs.
The p53 tumor suppressor gene plays an important role in the carcinogenesis of various organs through the regulation of cell proliferation, genomic stability, and programmed cell death. 7 Mammary tumors are the most common tumor type in female dogs. 4 , 15 In human breast tumors, p53 mutations are the most common acquired genetic abnormality and are found in 15–34% of the breast tumors analyzed. 1 , 3 , 16 The deduced amino acid sequence of p53 protein of dogs was nearly 80% homologous to that of the human proteins. 3 , 20 In the past, mutations of canine p53 have been identified in a small number of spontaneous tumor groups: one of 23 (4%) thyroid tumors, 5 one oral papilloma, 13 four of 17 (24%) 18 and seven of 15 (47%) 9 osteosarcomas, one of 12 (8%) circumanal gland tumors, 12 and three of eight (38%) lymphomas. 20 Moreover, three cases of p53 mutations (33%) have been found in canine mammary carcinoma cell lines. 19 Although there have been two reports concerning p53 mutation in spontaneous canine mammary carcinomas, 3 , 11 surveys of p53 mutations in benign tumors have been limited. In this study, we detected some alterations of exons 5–8 for the canine mammary tumors, including benign tumors and carcinomas, using polymerase chain reaction (PCR) direct sequence analysis. 17
Materials and Methods
Tissues acquisitions and DNA preparation
Samples from 63 primary canine mammary tumors, diagnosed during 1997–1998, were obtained at Azabu University School of Veterinary Medicine. The average age of the dogs was 11.8 years (range, 7–15 years). Dogs evaluated were 21 mixed breeds, 12 Dachshunds, 9 Malteses, 8 Shih Tzus, 6 Shibas, 4 Yorkshire Terriers, and 3 Poodles. None of the dogs had been neutered at the time of the diagnosis of the mammary tumors. The tissue samples were routinely fixed in 10% buffered formalin, embedded in paraffin, and stored at room temperature. Serial sections, one 4 µm thick and three 8 µm thick, were cut and mounted individually on glass slides. One section from each tumor was stained with hematoxylin and eosin (HE). HE-stained slides from each case were reviewed by two pathologists (S. Wakui, M. Furusato) to confirm the original diagnoses of the neoplasms according to criteria proposed previously. 8 Tumors were classified as benign (38/63; 60%) or malignant (25/63; 40%) (Table 1). No metastases were found. Microdissection method 17 was applied prior to tumor DNA preparation. The outline of the entire tumor area was directly marked on the HE-stained slides using fine felt-tip pen under the light microscope at low magnification (20×). The 8-µm-thick unstained section was overlaid on the HE-stained slides, then each tumor outline was adjusted, and only the tumor area was dissected using a sterile razor blade under the stereoscopic microscope. Sterile procedures were also applied independently for each case to avoid cross-contamination between the tissue specimens. Following deparaffinization with xylenes and ethanol, tissues were digested in Tris–ethylenediaminetetraactic acid (TE) buffer (pH 8.0) containing proteinase K, added every 24 hours. Following a standard series of phenol/chloroform extractions, DNA was precipitated and resuspended in TE buffer.
Histological diagnoses of canine mammary neoplasms used for p53 mutation analysis.

PCR direct sequence analysis
p53 at exons 5, 6, 7, and 8 was analyzed by the PCR direct sequence analysis method. The intron-based PCR primers, as used in a previous study, 3 were applied. PCR reactions were carried out at a volume of 50 µl containing 50 ng of genomic DNA, 1.5 mM MgCl2, 50 mM KCl, 10 mM Tris-HCl (pH 8.3), 200 µM each of dATP, dCTP, dTTP, dGTP, each primer at 1 µM, and 2.5 units of Taq polymerase (ABI/PE, Foster City, CA). Amplification was performed using 35 cycles in a Perkin-Elmer 9600 thermal cycler, each consisting of 3 minutes at 94 C, 1 minute at 55 C, and 1 minute at 72 C, followed by a 3-minute extension period at 72 C after the last cycle. The PCR products were analyzed with 2% NuSieve/Agarose gel electrophoresis.
Amplification resulted in a single discrete band and nonspecific bands were not observed. Following PCR, products were purified and concentrated into 40 µl using SUPREC™-01 (Takara Co., Tokyo) to remove salts, primers, and dNTPs. Purified PCR products were sequenced on an Applied Biosystem Prism 377 DNA Sequencer using an ABI PRISM™ dye terminator cycle sequencing ready reaction kit (ABI/PE) and Centri-Sep Spin Columns (ABI/PE) according to the manufacturer's instructions. Electrophoresis was performed at 2500 V for 14 hours, and DNA sequences were analyzed using ABI sequencing analysis software. Exons amplified from normal skin or lymphocytes, corresponding to the tumor tissues, were also sequenced to identify possible germline defects and to eliminate the possibility of pseudogene amplification or breed-associated polymorphisms. For confirmation of mutation, nucleotide sequences were determined on both strands and were repeated three times for each PCR reaction.
Statistical analysis
The chi-square test was employed for statistical analysis using statistical analysis computer system Stat View J4.02 (Abacus Concepts, Cary, NC).
Results
Exons 5–8 of p53 were screened individually for mutations in 63 dog mammary tumors, and 10 mutations were found in 9 mammary tumors (14%): four missense mutations (11%) in 38 benign mammary tumors, and one nonsense and five missense (one tumor had two missense mutations) (20%) in 25 mammary carcinomas. Although the incidence rate of mutation in mammary carcinomas was higher than that of benign mammary tumors, there was no significant incidence difference between the two groups (P = 0.293). A summary of the mutations of the two groups is shown in Table 2. Examples of mutations are shown in Fig. 1, and some corresponding tumor photomicrographs are shown in Figs. 2 and 3. In Fig. 1, smaller low peaks are present at mutations sites. These peaks reflect contamination noises with DNA of normal cells and/or with normal p53 alleles. This type of contamination is expected owing to the method that was used. A silent mutation was also found at codon 272 (GTA > GTT) (Fig. 1). Silent mutations were excluded in the calculation for presence of p53 mutations. All of the mutations were somatically acquired and tumor specific; sequence analyses of DNA from each corresponding normal tissues revealed only wild-type sequence in each case. There was no evidence of any relationship between the p53 mutations and the histopathologic types of the tumors or the breeds of dogs. A breed-associated polymorphism was not identified.
Mutations in p53 exons 5–8 in canine mammary neoplasms: benign tumors (n = 38) and carcinomas (n = 25).
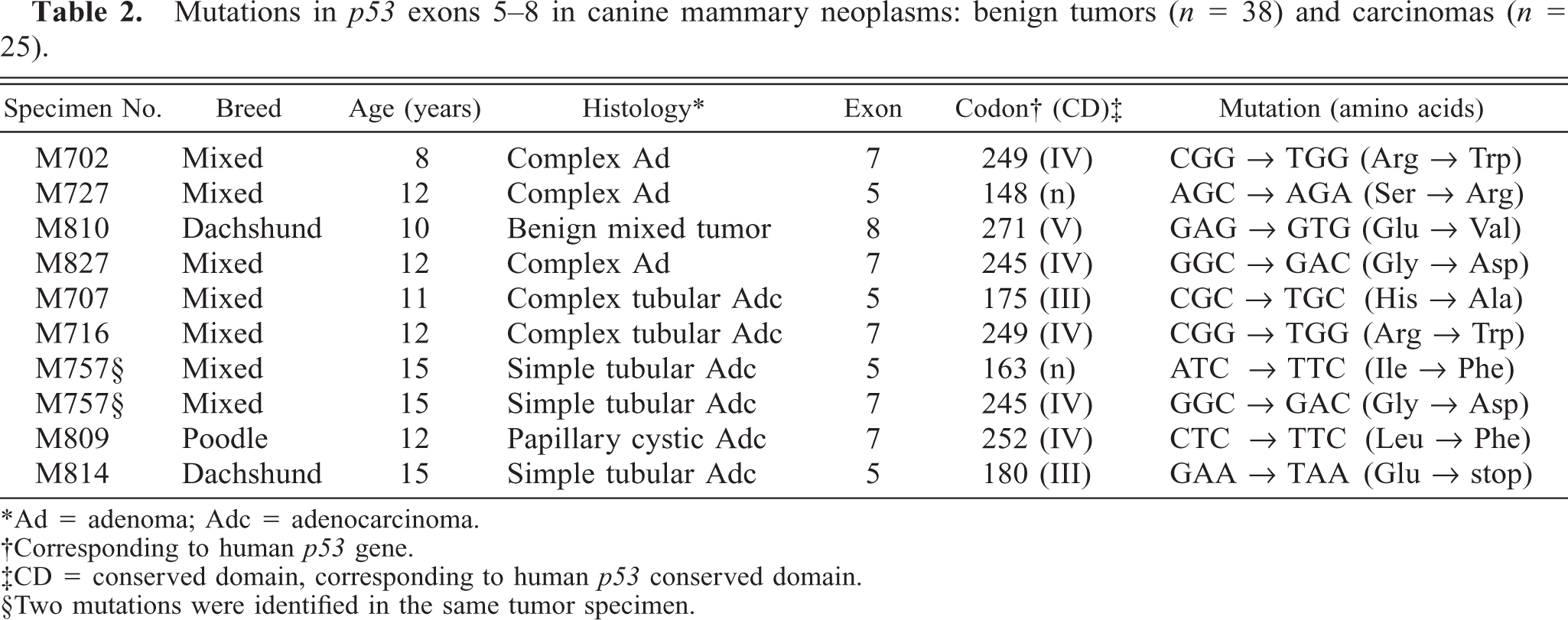
∗Ad = adenoma; Adc = adenocarcinoma.
†Corresponding to human p53 gene.
‡CD = conserved domain, corresponding to human p53 conserved domain.
§Two mutations were identified in the same tumor specimen.
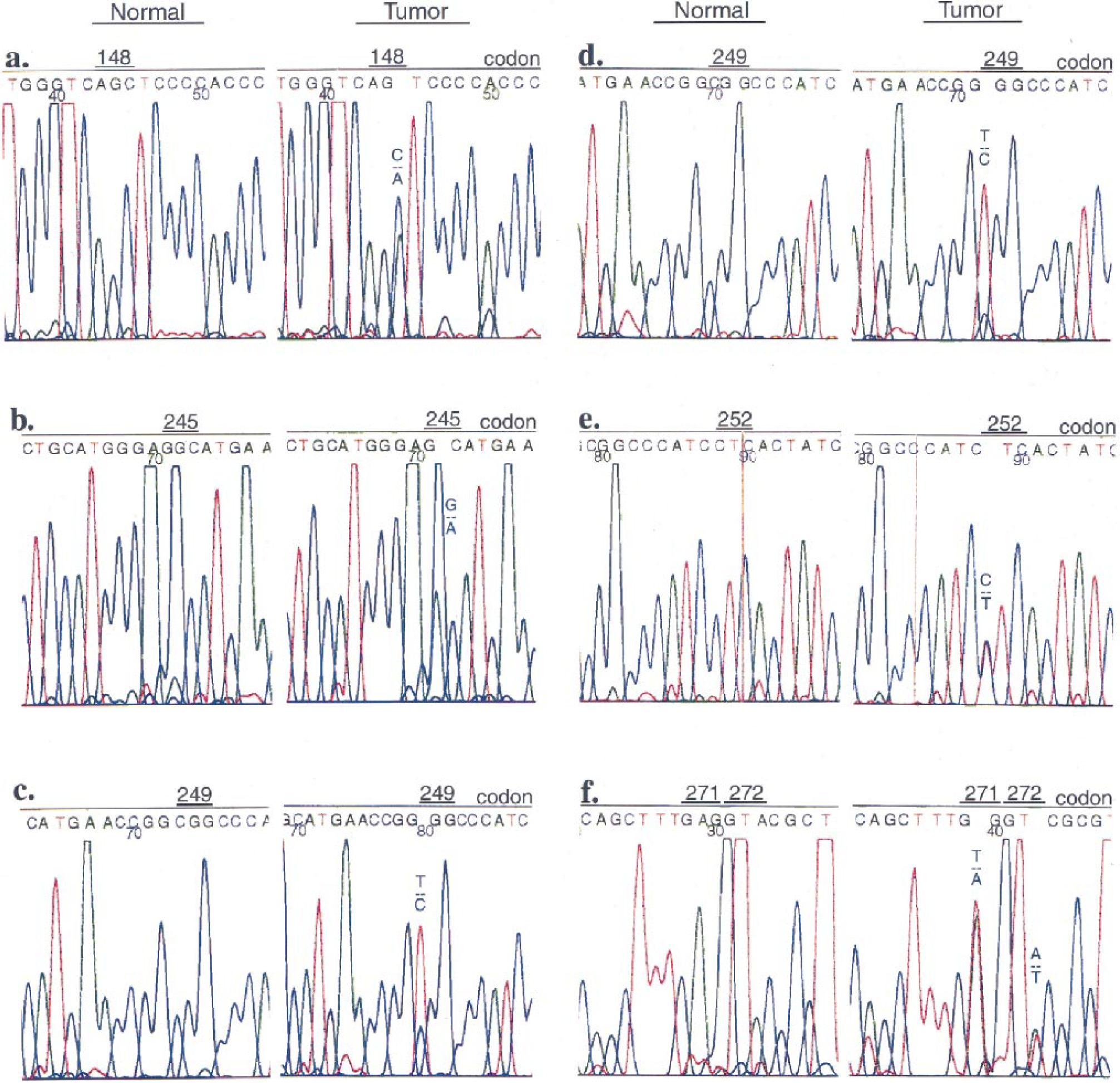
p53 Mutations in dog mammary tumors. Sequence analysis of p53 Exon 5–8 PCR products from normal tissue genomic DNA (Normal) and tumor genomic DNA (Tumor) reveal point mutations. Presence of wild type sequences result from DNA of normal tissues. Fig. 1a. Tumor M727, benign. A point mutation is present at codon 148 of p53 exon 5 showing the presence of C and A peaks. Fig. 1b. Tumor M827, benign. A point mutation is present at codon 245 of p53 exon 7 showing the presence of G and A peaks. Fig. 1c. Tumor M702, benign. A point mutation is present at codon 249 of p53 exon 7 showing the presence of T and C peaks. Fig. 1d. Tumor M716, malignant. A point mutation is present at codon 249 of p53 exon 7 showing the presence of T and C peaks. Fig. 1e. Tumor M809, malignant. A point mutation is present at codon 252 of p53 exon 7 showing the presence of C and T peaks. Fig. 1f. Tumor M810, benign. A point mutation is present at codon 271 of p53 exon 8 showing the presence of T and A peaks, and a silent mutation is present at codon 272 showing the presence of A and T peaks.
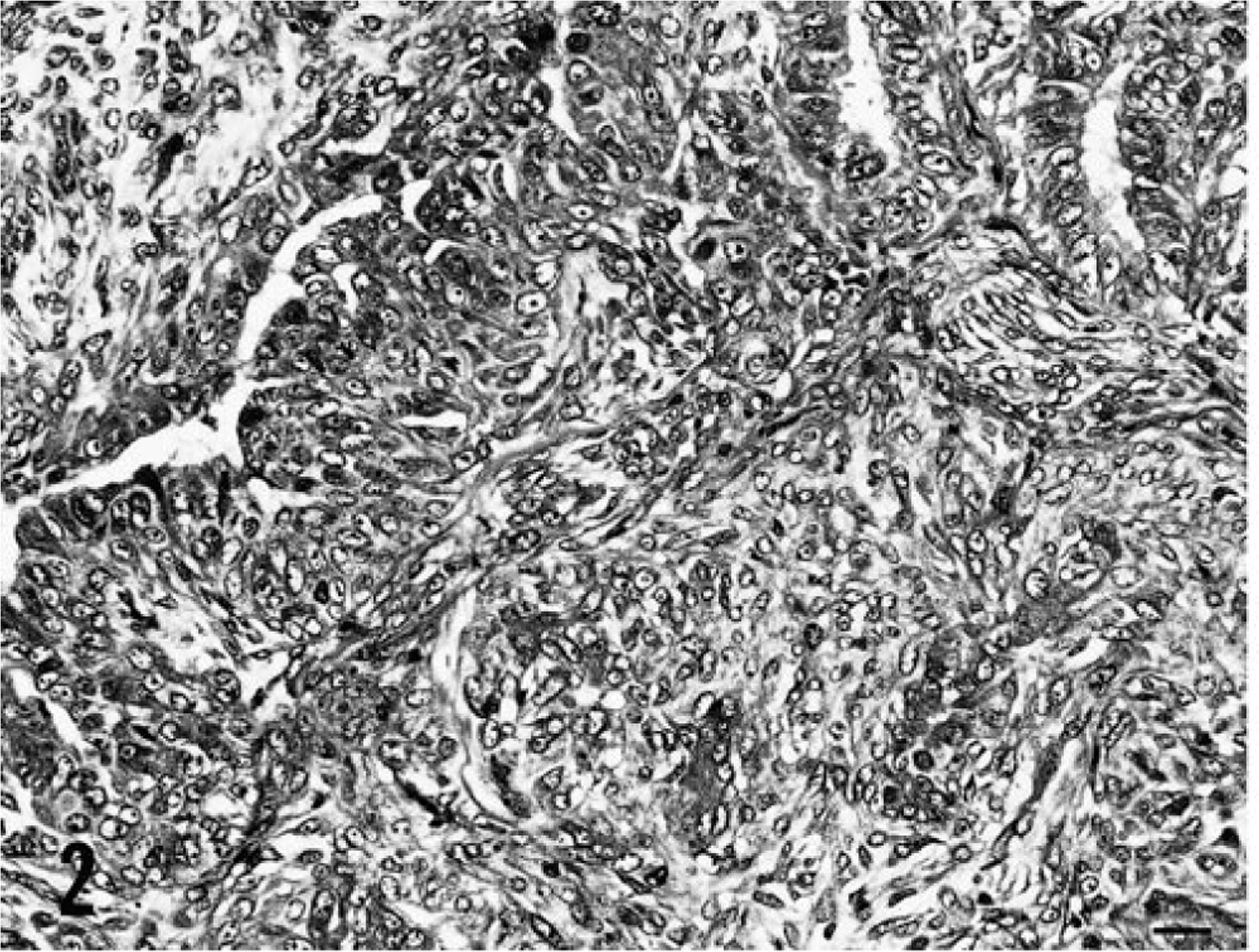
Benign mammary tumor M727; dog. Complex adenoma is composed of proliferated secretory epithelial cells and myoepithelial cells. HE. Bar = 20 µm.
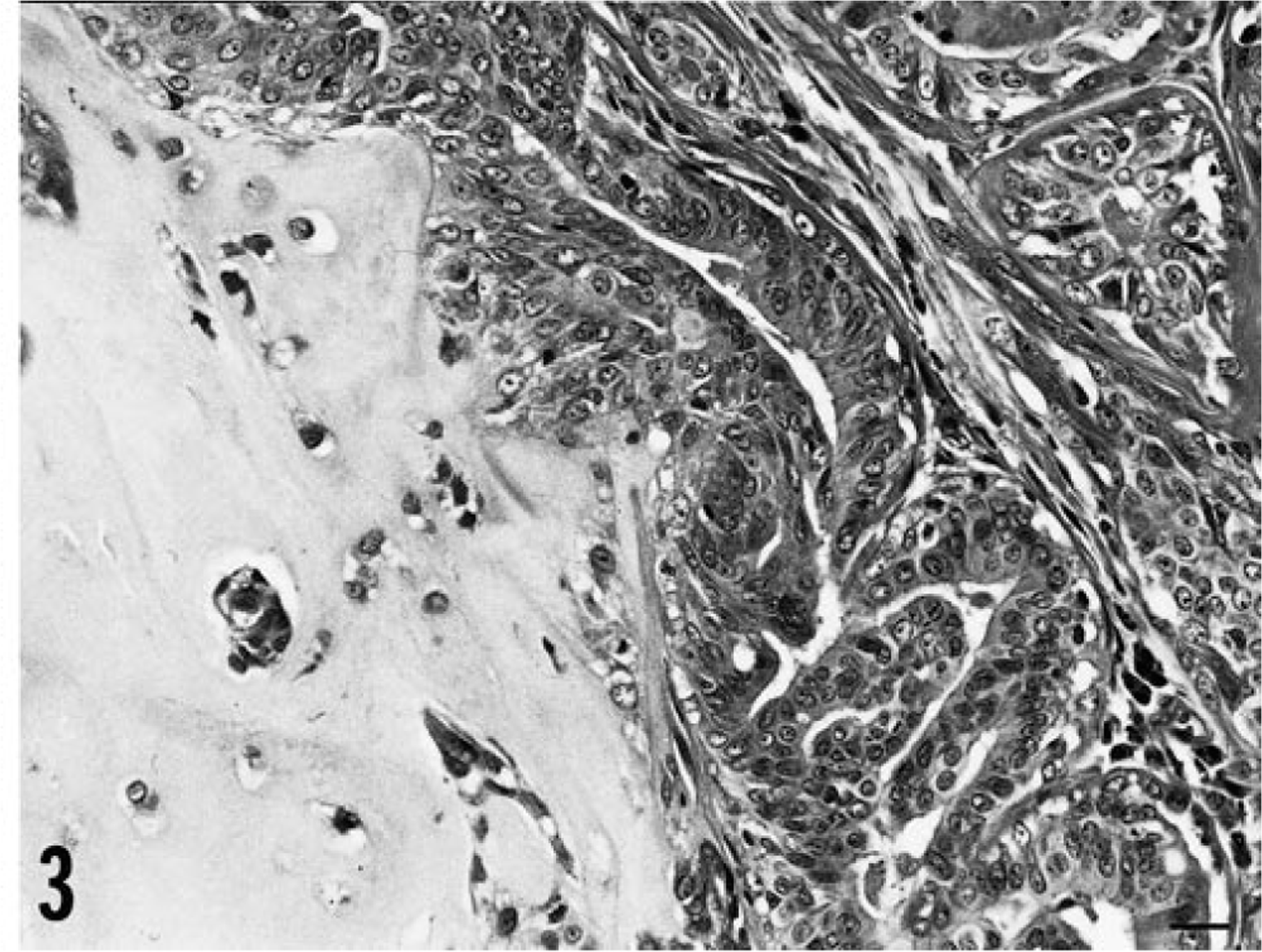
Benign mammary tumor M810; dog. Benign mixed tumor is composed of proliferated secretory epithelial cells and myoepithelial cells and cartilage. HE. Bar = 20 µm.
Discussion
There is a limited number of studies concerning p53 mutations in spontaneous malignant canine mammary tumors. In one study, six mutations were found in 40 (15%) carcinomas, 3 and in another study one mutation was found in 10 carcinomas (10%). 11 In the present study, the frequency of p53 mutation (20%) in spontaneous canine mammary carcinomas is comparable to that of human breast carcinomas. 1 The frequency of mutations in this study is relatively high compared with that of the previous studies. 3 , 11 This difference might be due to methodologic differences, especially concerning DNA acquisition. Previous authors noted that tumor DNA samples from tumors were contaminated with abundant DNA from normal tissues. 3 , 11 To avoid possible contamination by normal tissues, we employed a microdissection method for DNA acquisition. 17
One p53 mutation has been found in a canine mammary tumor cell line, which was established from a benign mixed tumor. 14 However, it was unclear whether the p53 alteration in this benign mammary tumor cell line had occurred during the process of establishment of the cell line or whether it was already present in the cells of the original benign tumor. 19 The incidence of p53 mutations in the original tumors is actually much lower than that seen in tumor cells in culture. 22 We first detected p53 missense mutations in the spontaneous canine benign mammary tumors. In human beings, mutations in p53 have also been found in benign breast tumors and preneoplastic breast lesions. 14
The nature of the mutations reported here is also noteworthy. G:C → A:T transitions represent the majority of all p53 point mutations identified in human cancers, including breast tumors. 6 In this study, in benign tumors, the mutations were G:C → T:A (25%) and A:T → T:A (25%) transversions and G:C → A:T transitions (50%). In carcinomas, the mutations were G:C → T:A (17%) and A:T → T:A (17%) transversions and G:C → A:T (67%) transitions. In general, G:C → A:T transitions at CpG sites represent endogenous DNA damage resulting from deamination of 5-methylcytosine. 6 , 21 In this study, G:C → A:T transitions occurred at CpG sites in benign tumors (100%) and in carcinomas (75%). However, G:C → T:A transversions are generally associated with carcinogen exposure. 6 , 21
The majority of p53 mutations reported in human cancers are clustered in four of the five evolutionarily highly conserved domains, the so called hot spot regions of p53 mutation, localized in exons 5–8 (Fig. 4). 2 , 6 , 21 In this study, two mutations (at codons 148 and 163) occurred outside the highly conserved domains. The remaining mutations (80%) were found within highly conserved domains III, IV, and V (Fig. 4). Thus, mutations in the conserved domains of p53 appear to play a significant role in mammary carcinogenesis in both the human 6 and the dog. Moreover, two identical mutations (at codons 245 and 249; Fig. 4) here located at the mutational hot spots described in human beings. 16 , 21 The identical p53 mutations in this study at codon 245 had also been described in the canine mammary carcinoma cell lines, 19 and the mutation at codon 249 was previously reported in canine mammary carcinoma. 11
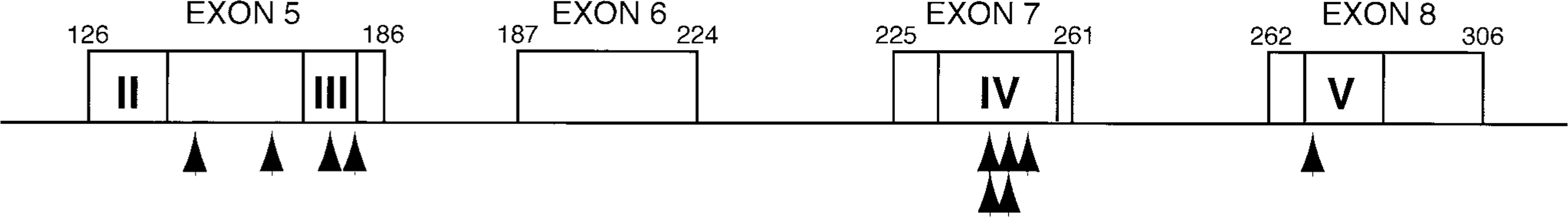
Summary of p53 mutations in canine mammary neoplasms. Location of mutations is indicated with an arrow. Detail of each mutation is provided in Table 2. The evolutionarily conserved domains are II–V.
Previously, the canine wild-type p53 sequence of codon 227 was described as TTT 11 , 19 or TCT, 3 , 10 , 12 , 20 and that of codon 301 was described as CCA 13 or CCC. 3 , 10 , 19 , 20 In the present study, the canine wild-type p53 sequence of codon 227 was TCT, and that of 301 was CCC; no polymorphisms were found (data not shown).
The results of this study indicate that p53 alterations are involved in mammary carcinogenesis in the dog. Moreover, p53 alteration could be an early event in canine mammary carcinogenesis; mutations have been detected not only in mammary carcinomas but also in benign tumors. Although several studies show that p53 mutations are an indicator of increased malignant potential and a poor prognosis in human breast cancer patients, 4 the clinical significance of the observed p53 mutations in canine mammary tumors cannot be determined without further investigation.
Footnotes
Acknowledgements
This study was supported by Grants-in-Aid 05856059, 06856036, 07806042, and 09660332 from the Ministry of Education, Science and Culture, Japan. We are grateful to Miss H. Sudo, Miss M. Takagi, Miss M. Arima, and Miss K. Morita for technical assistance.
