Abstract
Progressive respiratory failure and pulmonary fibrosis in West Highland White Terriers (WHWT) is an apparently genetic disorder of unknown pathogenesis. This study characterizes the light microscopic, ultrastructural, and immunohistochemical features of affected WHWT in comparison with lesions in usual interstitial pneumonia (UIP) of humans. Lesions in WHWT were confined to the expansion of the interstitial space of alveolar septa by extracellular matrix (ECM) determined to be mixtures of type-I and -III collagens. Features of UIP such as intra-alveolar fibroblastic foci, subpleural distribution, and honeycombing were not observed in six WHWT. Comparison with normal dogs showed no apparent increase in septal myofibroblasts. Ultrastructually, the ECM in alveolar septa consisted of large aggregates of periodic collagen filaments underlying alveolar capillaries that were surrounded by thick bands of amorphous to fine fibrillar matrix. This study suggests that chronic pulmonary disease of WHWT is a result of aberrant collagen regulation.
Keywords
Interstitial pneumonia is a broad category of inflammatory disease defined by the primary involvement of alveolar septa in the pathogenesis of the lesions. Histologically, however, the interstitium of the alveolar septa is defined as the space between the alveolar epithelial cells and the capillaries. 3 , 13 A variety of alterations in injury and repair in interstitial pneumonia may lead to a common progression toward interstitial fibrosis. 4 The process of fibrosis in the alveolar parenchyma often has an insidious onset and uncertain etiology. 5 The diagnosis and treatment of chronic interstitial fibrosis can be quite challenging because irreversible loss of pulmonary function has often occurred before clinical symptomology is recognized. The significance of the morbidity and mortality of this process drives the interest in experimental and animal models of chronic pulmonary parenchymal disease. A classification scheme for idiopathic interstitial pneumonia in humans has been modified by several authors since the original description by Liebow and Carrington and generally encompasses at least four different categories with a spectrum of histologic appearances. 12 These include usual interstitial pneumonia (UIP) characterized by a patchy to subpleural distribution of interstitial inflammation and fibrosis with multifocal foci of proliferating fibroblasts; desquamative interstitial pneumonia characterized by the presence of large numbers of macrophages in the alveolar spaces; acute interstitial pneumonia or diffuse alveolar damage characterized by a diffuse active proliferation of fibroblasts and mixed inflammatory infiltrate subsequent to an exudative phase with hyaline membrane formation; and organizing pneumonia, cryptogenic organizing pneumonia, or idiopathic bronchiolitis obliterans characterized by peribrochiolar inflammation and Masson bodies in the lumen of alveoli and bronchioles. 8 , 12 Inflammation and fibroblast proliferation are common features of all interstitial fibrosis syndromes. The source of the fibroblast precursors is also an area of active research in these diseases, with much emphasis placed on the role of the myofibroblasts. 5 , 12 In the process of lung repair, myofibroblasts are localized to intra-alveolar “fibroblast foci.” 5 Myofibroblasts are also thought to be responsible for collagen synthesis in UIP. 14
Although interstitial fibrosis is evident in a variety of animal diseases, most are associated with infectious or toxic etiologies, and the nomenclature has remained much less complicated. Increased awareness of respiratory compromise in small-animal practice has allowed recognition of clinical syndromes of respiratory failure associated with idiopathic interstitial fibrosis in dogs and cats. 13 At the present time, predisposition for idiopathic interstitial disease is limited to the terrier breeds with a higher incidence in the West Highland White Terrier (WHWT). 4 This syndrome is characterized by significant dyspnea, exercise intolerance, and hypoxia in the face of relatively subtle increase in pulmonary fibrosis. 13 The WHWT has been suggested as a potential model for human UIP.
This article compares the WHWT lesions with those reported in human UIP by characterizing the nature and distribution of extracellular matrix (ECM) in the alveolar septa of the WHWT by determining the collagen subtypes present and evaluating the potential role of the myofibroblast. We examined archival specimens of affected dogs by light microscopy using collagen and elastin stains in concert with immunohistochemical characterization of collagen subtypes and smooth muscle actin–positive cells. We further characterized the alveolar septa of a subset of these cases by transmission electron microscopy.
Materials and Methods
Case selection
The medical records at the University of California Veterinary Medical Teaching Hospital were searched between the years 1996 and 2002 for cases of WHWT that had a pathologic diagnosis of pulmonary interstitial fibrosis (Table 1). Clinical criteria for case inclusion were a history of respiratory distress and cough in addition to radiographic interpretations of an interstitial lung pattern and hypoinflation. Additional criteria were the exclusion of patients with evidence of cardiac disease, thoracic radiographs of the cardiac silhouette and electrocardiograph. Six cases with available lung tissue that met these criteria were selected; three were biopsy specimens, and three were specimens from routine necropsies. The WHWT age range was between 6 and 16 years. Two clinically normal dogs were chosen for controls. One of the control samples (Labrador Retriever) was a lung biopsy from a middle-aged dog, and the other sample (Shetland Sheepdog) was a necropsy specimen taken from a juvenile dog. These controls were chosen to represent different ages and potential incidental age-related changes.
Age, sex, and clinical finding in six WHWT.
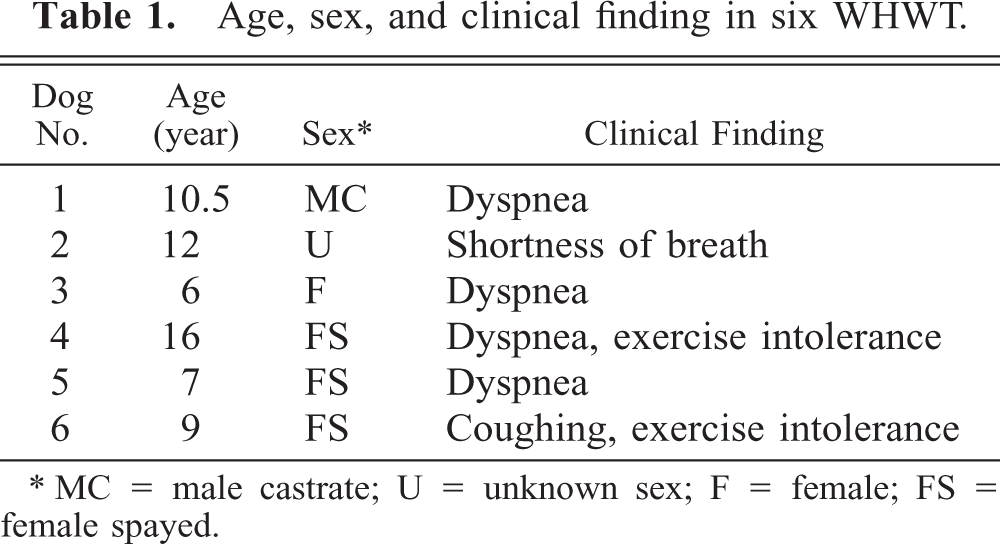
∗MC = male castrate; U = unknown sex; F = female; FS = female spayed.
Histology
Necropsy specimens and one of the biopsy specimens were sampled from multiple lung lobes. In the remaining two biopsy specimens, only one lung lobe was available. Two to four lung sections were examined for each case. All tissue had been fixed in 10% neutral buffered formalin and embedded in paraffin wax through routine histologic processing. Five-micrometer sections were stained with hematoxylin and eosin. Each specimen was also stained with Masson's trichrome to identify collagen deposition by using the Luna technique 2 and stained with Verhoeff's Van Gieson to identify elastin by using the Mallory technique. 2
Immunohistochemistry
Immunohistochemistry was performed on serial sections using the following antibodies: smooth muscle alpha actin (clone 1A4 1 : 200, BioGenex Corp., San Ramon, CA), collagen I (NCL-Coll-Ip 1 : 200, Vector Laboratories Inc., Burlingame, CA), collagen III (Chemicon AB768 1 : 200, Vector Laboratories Inc.), and collagen IV (clone CIV 22 1 : 350, Dako Corp., Carpinteria, CA) and matrix metalloproteinases 1 and 8 (rabbit anti-human MMP-1 and MMP-8, 1 : 200, Cedarlane Laboratories, Hornby, Ontario, Canada). Standard immunohistochemical protocols established by the Veterinary Medical Teaching Hospital, University of California–Davis, were followed with the following modifications. 1 After deparaffinizing, the slides for collagen III staining were heat pretreated at 37 C for 30 minutes in Glyca pH 3.3 buffer (BioGenex Corp.) and the slides for collagen I, collagen IV, and MMP-1 and MMP-8 were heat pretreated at 98 C for 30 minutes in Citra pH 6.0 buffer (Dako Corp.) before application of the primary antibody.
Light microscopic evaluation
All tissue were examined with an Olympus BX light microscope for criteria of pulmonary interstitial fibrosis, i.e., increased numbers of fibroblasts and collagenous matrix that obliterates normal architecture and extends into alveolar spaces. 12 Controls were determined to have no underlying lung disease. The distribution of the ECM in the alveolar septa was analyzed subjectively and categorized as either “concentric” (surrounding the capillary endothelium and widening the distance between the capillary and alveolar epithelium) or “eccentric” (widening the interstitium but displacing the capillary so that it remains closely apposed to one side of the alveolar epithelium). 3 The amount of trichrome-stained collagen was subjectively evaluated and ranked on a scale of 1–5 with five being the most severe. The same scale was used to score the amount of elastin-stained septal tissue. Immunostains for collagen and smooth muscle actin were scored from 1 to 5 with a score of one representing the expression present in control lungs.
Ultrastructure analysis
Formalin-fixed tissue from two of the six WHWT cases were evaluated by electron microscopy and compared with the juvenile control sample. Three- by five-millimeter sections of affected lung tissue were embedded in epon-arylidite plastic in JB4 molds and 1-µm-thick sections examined by light microscopy. Representative regions of affected alveolar septa were selected and prepared for ultrathin sectioning. Ultrathin sections were stained with lead citrate and uranyl acetate according to standard techniques. Specimens were viewed and photographed on a Ziess EM10 electron microscope.
Results
The ages of WHWT in this study ranged from 6 to 16 years. The signalment and clinical diagnosis, are shown in Table 1. All the WHWTs had a history of dyspnea or coughing for a year or less. Clinical data were not complete enough to evaluate blood gases for hypoxia. There was a broad spectrum of progressive histopathologic change in these specimens. All cases had thickening of the interstitium by an eosinophilic ECM that varied in thickness from few fine parallel fibers to numerous dense tightly packed fibers that formed a broad sheet of matrix (Fig. 1). The distribution of the alveolar septal thickening was variable. In some cases, the process appeared to be diffuse, whereas in other cases it was multifocal or regional. In the most severely affected cases, there were multifocal areas of type-II pneumocyte hyperplasia. In all cases, there was a cellular infiltrate present in the interstitium and alveolar space; however, the quantity of the cellular infiltrate varied between samples. This infiltrate was composed of lymphocytes, plasma cells, and macrophages. Masson's trichrome stained the matrix expanding the interstitial space as collagen. In the alveolar septa from control lungs, there was very little trichrome-stained collagen. In all affected WHWT, there was an increase in trichrome-positive material throughout the septa. In the less severely affected cases, the trichrome staining was discontinuous. In the more severely affected cases, the trichrome staining was abundant and formed multiple thick coarse short bands that in some cases expanded the interstitium more than normal (Figs. 2, 3). Individual trichrome scores ranged from 2 to 5, indicating that all cases had an increased amount of interstitial collagen in comparison with the normal tissue. Four of six cases had a concentric distribution of collagen, which supported the concept that the blood-air barrier was thickened by the deposition of ECM. The distribution of elastin stain did not vary between the WHWT and normal lungs with scores of 1 (normal) and included fine fibers throughout the interstitium as well as the walls of the airways and vessels.
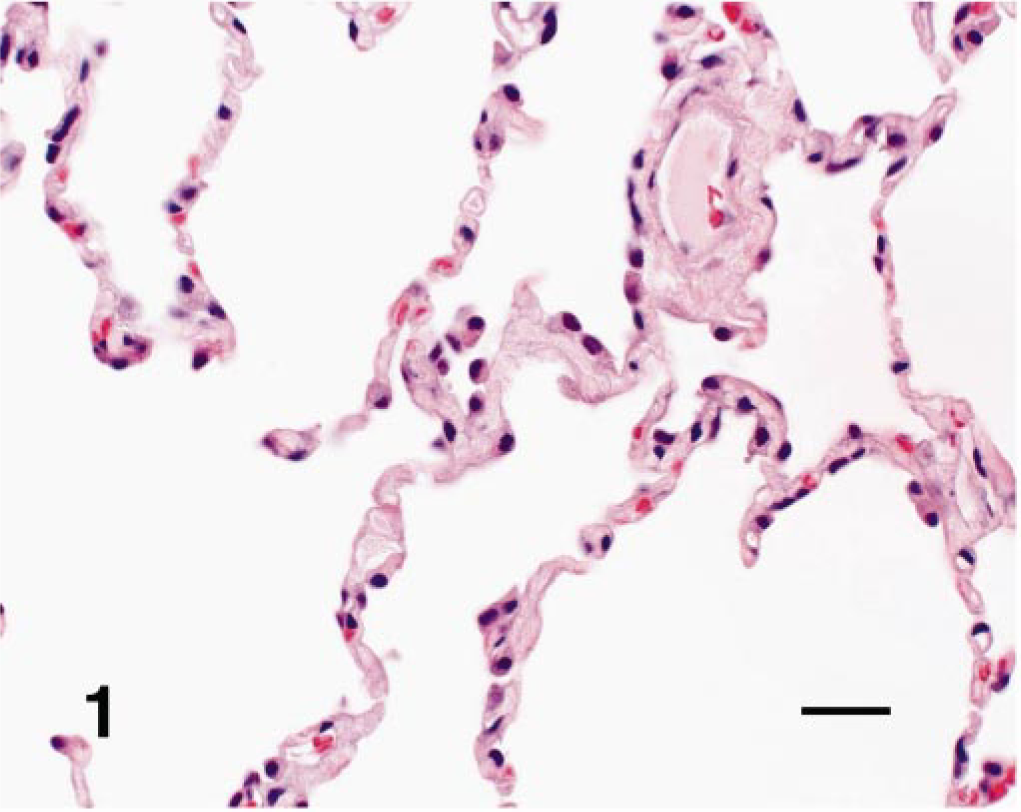
Lung; dog. Alveolar septa and spaces from an affected WHWT. The septa are diffusely expanded by an eosinophilic matrix that isolates capillaries and widens the distance between the septal epithelium and endothelium. HE. Bar = 60 mM.
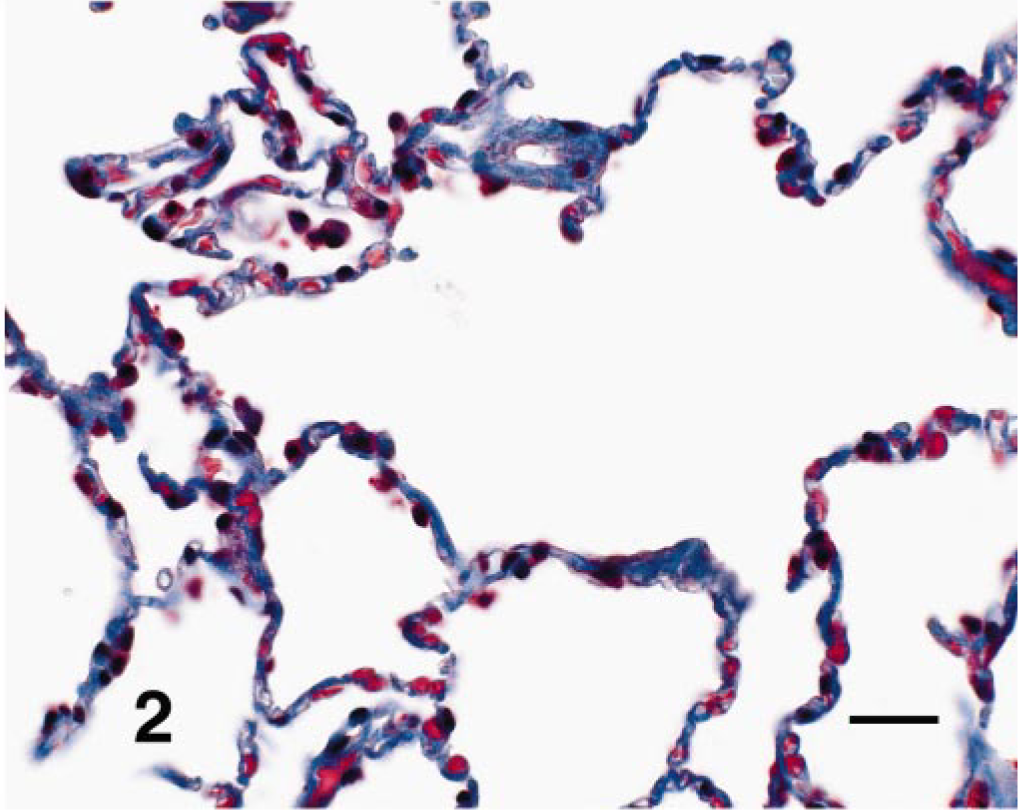
Lung; dog. Alveolar septa and spaces from an affected WHWT. The septa show diffuse trichrome-positive collagen staining and concentric distibution around capillaries. Masson's trichrome. Bar = 60 mM.
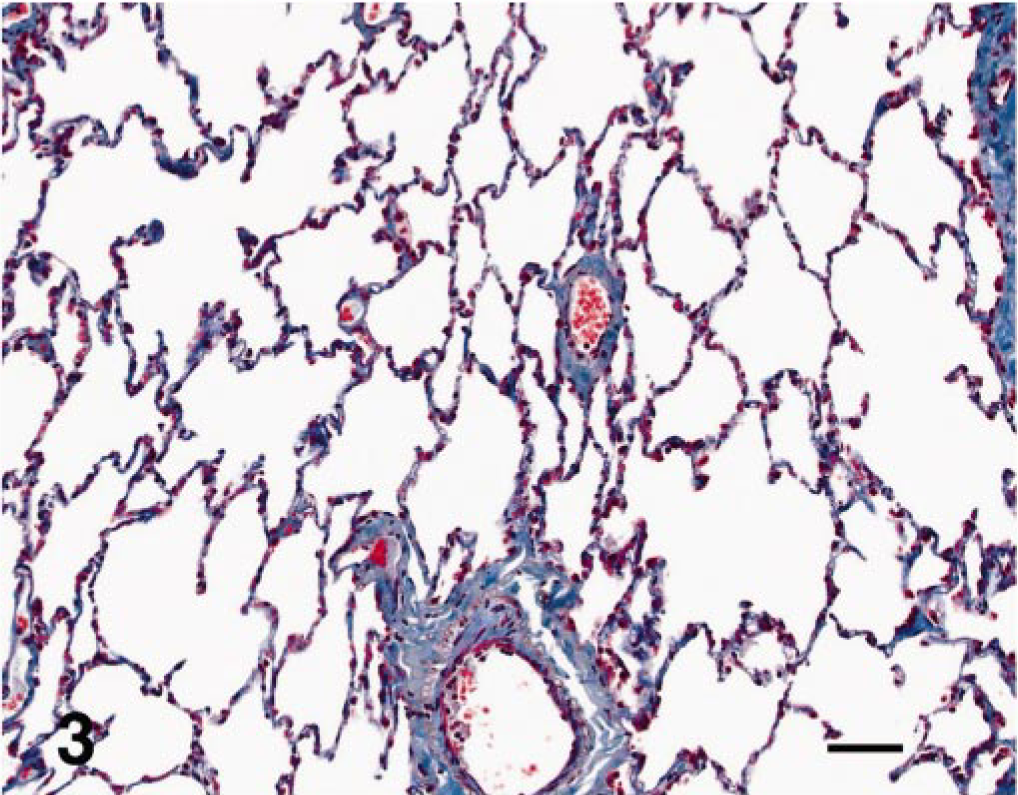
Lung; dog. Low magnification of the positive trichrome staining throughout the alveolar septa of an affected WHWT. Masson's trichrome. Bar = 20 mM.
In control dogs, immunostains for smooth muscle actin labeled smooth muscle cells concentrically around blood vessels, muscle in the airways (larger airways to bronchioles), and scattered contractile tips of the alveolar septa in alveolar ducts. In the WHWT, positive staining for the smooth muscle actin was detected in the same areas as present in the controls (Fig. 4). The distribution of collagen I in both normal lungs and WHWT had variably light staining throughout the septa (Fig. 5). Immunostaining for collagen III was denser in the WHWT than in the controls (Fig. 6). In general, the ECM throughout the septa of the WHWT stained more intensely and abundantly with collagen III than with collagen I. Immunoreactivity for collagen IV in the WHWT and control samples had inconsistent reactions with interstitial matrix, basal lamina, and alveolar macrophages.
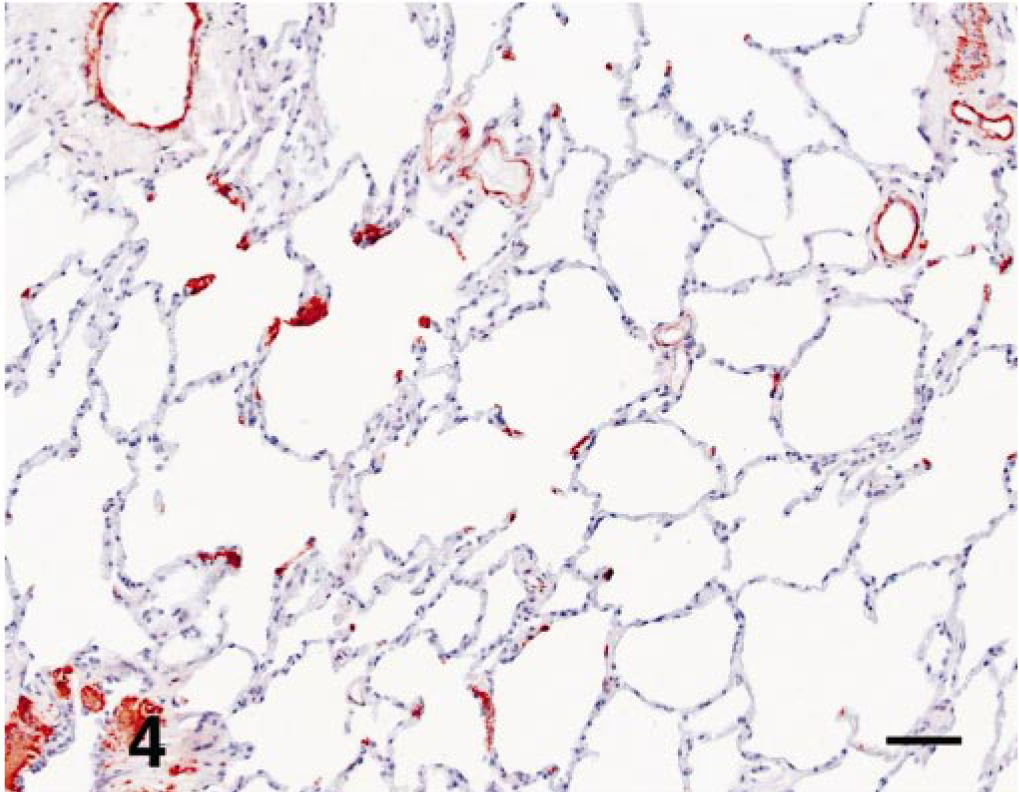
Lung; dog. Smooth muscle actin immunoreactivity of interstitial contractile cells and vascular and bronchiolar walls in an affected WHWT. Note the absence of immunoreactivity throughout the septa and alveolar space. Smooth muscle actin. Bar = 20 mM.
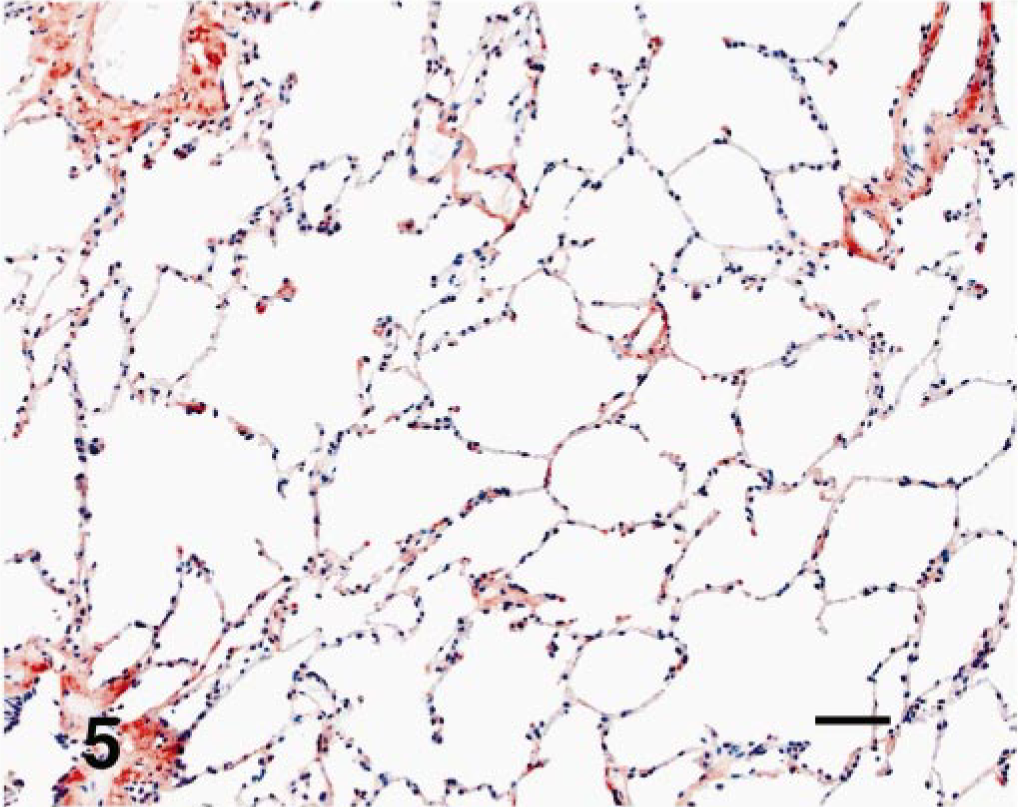
Lung; dog. Multifocal pale immunoreactivity of type-I collagen throughout the interstitium of an affected WHWT. There was also normal immunoreactivity around vascular and bronchiolar walls. Type-I collagen. Bar = 20 mM.
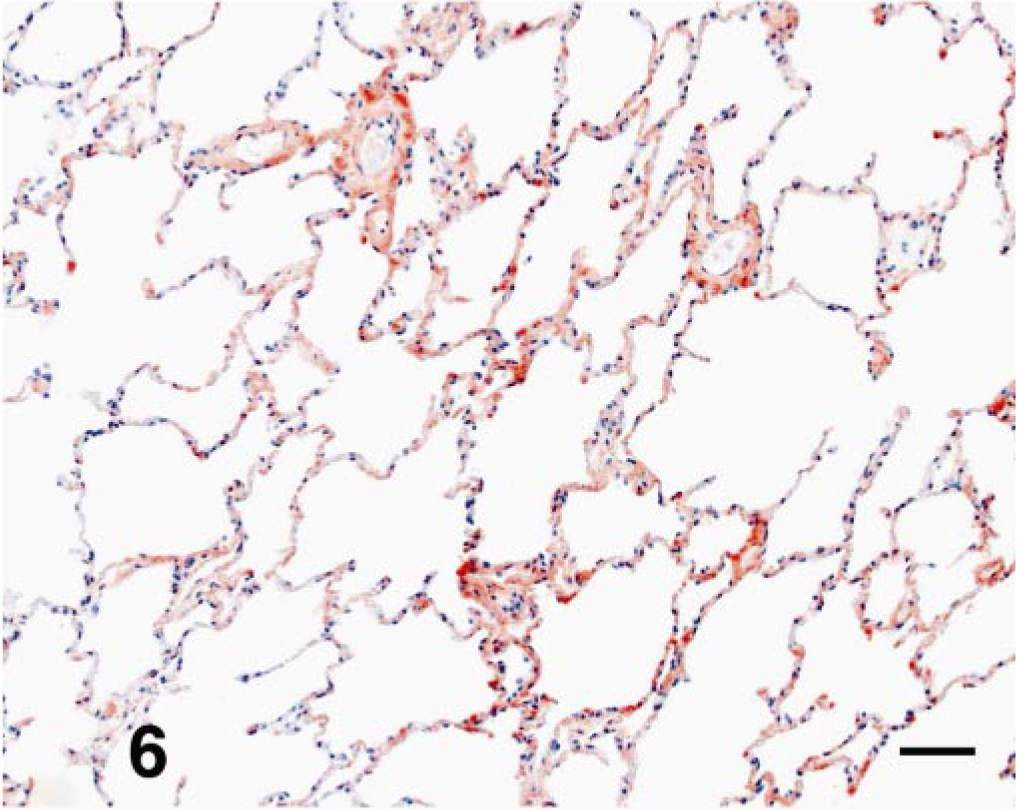
Lung; dog. More diffuse immunoreactivity of type-III collagen throughout the intersitium of an affected WHWT. The immunoreactivity for type-III collagen, when compared with the type-I collagen, is stronger and more abundant. Type-III collagen. Bar = 20 mM.
In preliminary studies with MMP-1 and MMP-8, immunoreactivity was evident in paraffin sections of lung from one control, two WHWTs, and an additional dog diagnosed with chronic right left failure. Differences in staining between the control and WHWT were equivocal. In all three categories of dogs, MMP-8 was observed primarily in the cytoplasm of bronchial and bronchiolar epithelium with lesser immunoreactivity detected in glands surrounding bronchi and perivascular connective tissue. One distinct feature of the MMP-8 immunoreactivity was the strong expression in alveolar macrophages and neutrophils in the heart failure dog. The immunoreactivity of MMP-1 was scarce in all three groups, with detection in the connective tissue of some alveolar septa being most prominent in the WHWTs. As with the expression of MMP-8, there was some weak immunoreactivity of alveolar macrophages with MMP-1 in the heart failure dog.
The last method used to characterize the interstitial matrix was the examination and comparison of the alveolar septa from two WHWTs and two control samples by transmission electron microscopy. In comparing the control samples with the WHWTs, it was evident that the interstitium in the WHWT was markedly expanded by numerous bundles of electron-dense fibrils (Fig. 7). On longitudinal section, these fibrils were aligned parallel to one another. The individual fibrils had even-spaced band periodicities characteristic of callagen 6 (Fig. 8). The deposition of these fibrils resulted in increased ECM expanding the interstitium and separating the capillary endothelial cell from the alveolar epithelium. 6 These parallel fibrils were not present in the interstitium of control dogs. In addition to fibrils of periodic collagen, most capillaries were surrounded by amorphous moderately electron-dense matrix. This matrix resulted in additional separation between epithelial and endothelial cell cytoplasm at the blood-air interface.
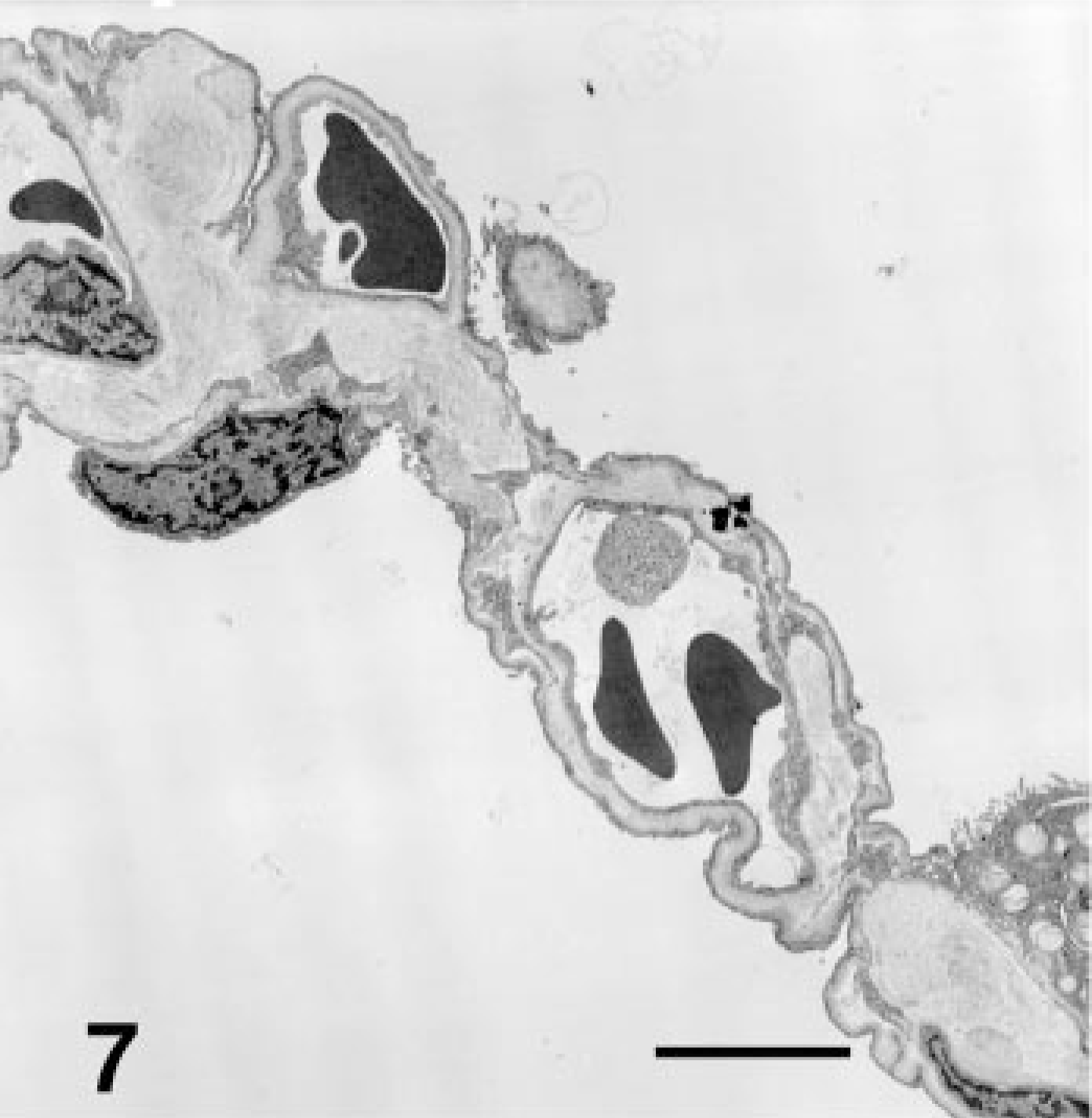
Lung; dog. Alveolar septa from an affected WHWT showing a generalized increase in ECM widening the septal wall and isolating alveolar capillaries. Type-I and type-II alveolar epithelial cells retain normal structure. TEM. Bar = 25 mM.
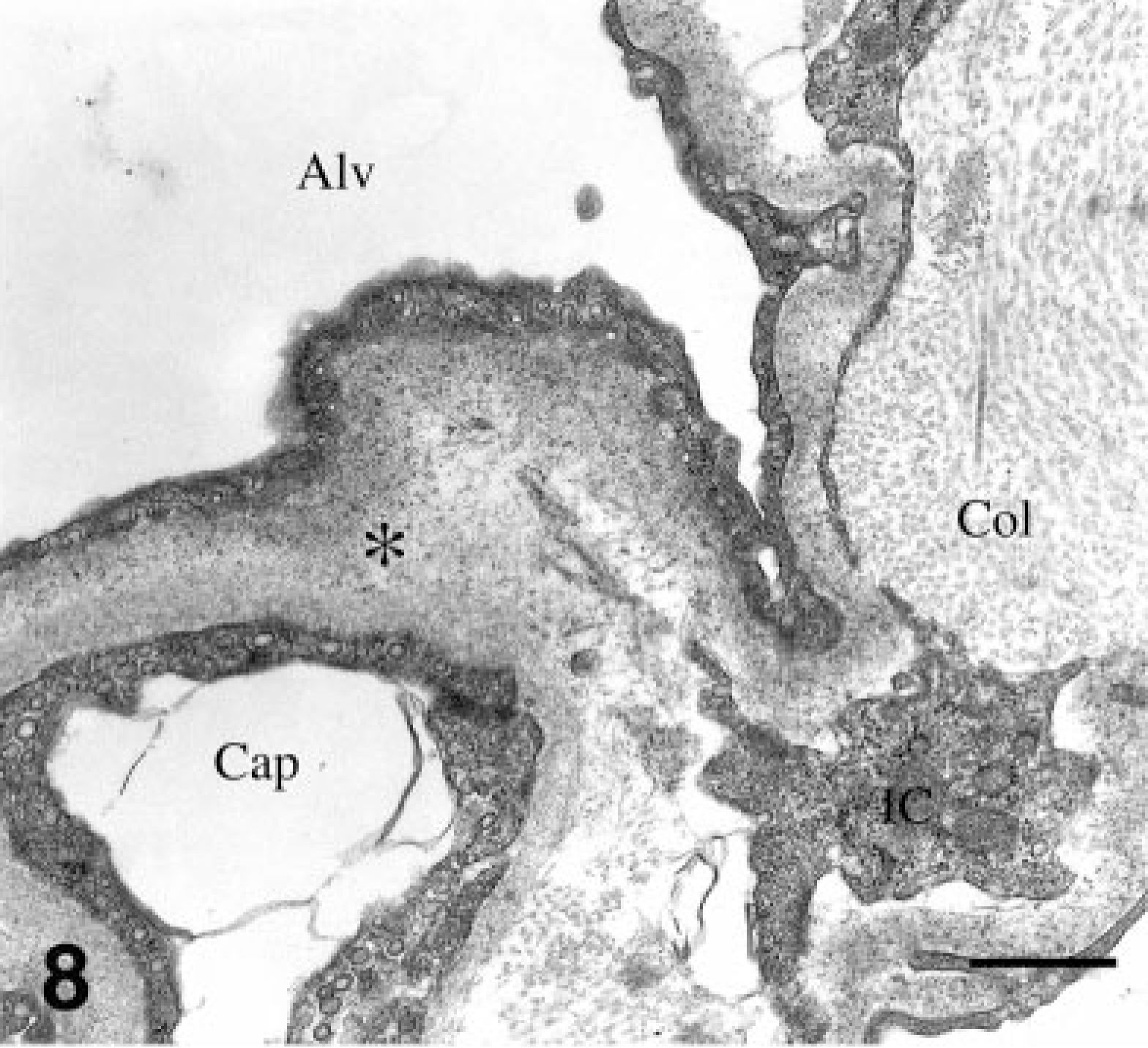
Lung; dog. Capillary (Cap) from an affected WHWT with bundles of periodic collagen (Col) that underlie the capillary and are partially compartmentalized by processes of interstitial cells (IC). Endothelial cells are separated from type-I epithelium and alveolar space (Alv) by a contiguous layer of amorphous ECM (∗). TEM. Bar = 0.5 mM.
Discussion
The original characterization of chronic pulmonary disease in WHWT described histologic features of multifocal to regional deposition of interstitial ECM, multifocal epithelial squamous metaplasia, and an increased number of alveolar and circulating macrophages as well as occasional lymphocytes and plasma cells. 4 Lesions in this study encompass a spectrum from generalized deposition of septal collagen with a scant inflammatory cell infiltrate in some cases to marked septal thickening by collagen, a more abundant cellular infiltrate of predominately macrophages and lymphocytes and hyperplasia of alveolar-lining type-II pneumocytes in more severely affected dogs. Squamous metaplasia of alveolar epithelium or larger airways was not evident in this study. The most consistent light microscopic feature in the cases evaluated in this study was the concentric deposition of ECM around alveolar capillaries. In the normal alveolar septum, ECM is woven eccentrically about capillaries, allowing alternating protrusions of the vascular lumen to be separated from the airspace by the thin layers of the basement membrane, type-I pneumocyte, and endothelial cytoplasm. On histology, the blood-air barrier in affected WHWTs appeared to be significantly compromised by the separation of epithelial and endothelial cells by the concentric deposition of ECM that expanded the interstitium. In four of six cases, increased concentric distribution of ECM was identified with light microscopy. Based on results of trichrome and immunohistochemical stains, the ECM represents mixtures of type-I and -III collagens with type-III collagen being the most prominent type. Elastin does not appear to be altered in this disease.
It has been reported in the normal lung that collagen types I and III are present throughout the interstitium and have different morphologic patterns. However, in disease, the morphologic patterns of collagens I and III change and the ratio is shifted. 14 A predominance of type-III collagen has been reported in the interstitium of humans with early pulmonary fibrosis. Conversely, type-I collagen has been observed in late disease of pulmonary fibrosis. 9 , 14 It has also been shown that collagen synthesis and degradation in adult life is constant. 14 To achieve this constant concentration of collagen in the ECM, shifts must occur in both the rate of synthesis and degradation of types I and III. The presence of both types I and III in the WHWT with the predominance of type III in most of the cases may suggest an earlier stage of disease or release of a factor that promoted type-III synthesis.
Transmission electron microscopy demonstrated two forms of ECM, one demonstrating periodic fibrils characteristic of collagens I and III and a second amorphous matrix more characteristic of thickened basal lamina. The latter separated the alveolar epithelium from the endothelium, thus thickening the blood-air barrier. The combination of histochemical, immunohistochemical, and ultrastructural evaluation suggested that the periodic fibrils of the ECM deposition were predominately type-III collagen. To the author's knowledge, this is the first description of collagen immunoreactivity in the canine lung. Further characterization of the immunologic reagents for collagen types has potential application for a variety of canine interstitial lung diseases.
There are several histologic features that characterize human UIP. Most recent reviews characterize the definitive feature of UIP as active zones of proliferating fibroblasts and myofibroblasts (fibroblast foci) closely apposed to areas of mature fibrosis commonly observed beneath the pleura. 5 , 7 , 12 These fibroblast foci have become the key histologic feature and research focus for understanding and characterizing this disease. 7 In UIP, these foci are immunoreactive for smooth muscle actin and vimentin and negative for desmin, classifying these cells as myofibroblasts and distinguishing them from smooth muscle cells. 10 , 11 Proliferative foci were not present in the WHWT, and smooth muscle actin immunoreactivity was limited to individual cells in the septa (presumably contractile interstitial cells). Cells in the septa that demonstrated positive actin staining were individual cells and not observed to be arranged in bundles or located on the luminal side of the basal lamina in areas of basal lamina or epithelial damage, as is characteristic for UIP. 10
In an immunohistochemical study performed on human cases of pulmonary fibrosis, collagen was observed predominately in the alveolar lumen or deposited subepithelially. This was suggestive of fibroblastic organization of an exudative process and implied damage to the basal lamina. In other human cases, the matrix within the alveolar lumen contained type-III collagen. 9 In the WHWT, extracellular collagen was limited to the interstitium and there was no evidence of luminal or subepithelial matrix accumulation that would indicate damage to the basal lamina. Thus, the interstitial changes in the WHWT are distinct from fibrosis in human UIP. These differences make it unlikely that the WHWT would be an appropriate model for UIP of humans.
Alternatives for a genetic predisposition for noninflammatory collagen accumulation include abnormally increased synthesis or defects in matrix lysis. The predominance of type-III collagen might suggest an abnormal signaling pathway associated with the earlier stages of wound repair. Recent research in exploring the pathogenesis for UIP has better characterized the role of IL-1β and transforming growth factor β(TGF-β). 5 In the cases of ECM accumulation without much inflammation, increased expression of TGF-β linked to fibrosis has been observed in humans. Because the WHWT cases exhibit very mild inflammation, it may be critical to determine the role of TGF-β and other such growth factors or cytokines.
The augmentation of both collagenous and other forms of ECM in affected WHWT could also be a consequence of decreased matrix turnover. Studies in humans with UIP have focused on different therapeutic strategies targeting tissue MMPs and MMP inhibitors. 5 In a preliminary study, we found no differences from the control in the immunohistochemical expression of either MMP-1 or MMP-8 in the affected WHWT. More specific evaluation of collagen synthesis and degradation require unfixed tissue specimens and should be considered in future prospective studies of this disease. The balance between MMPs and their inhibitors could be an important aspect of the future characterization of the accumulation of ECM in WHWT.
Footnotes
Acknowledgements
We thank Esi Djan and Drs. Lynelle Johnson and Carol R. Norris for the histological specimens and case contributions. This study was supported by the Center for Companion Animal Health at the University of California-Davis.
