Abstract
Tumors at the cranial base in 2 cats (a 9 1/2-year-old, castrated male Chinchilla and a 7-year-old, castrated male American shorthair) were diagnosed as malignant craniopharyngioma. The tumor lesion was histopathologically divided into four parts: 1) a small acinus part, in which relatively large cells with a pale cytoplasm composed small acini; 2) a duct part, in which small cuboidal cells composed ducts; 3) a cyst part, in which there were large cysts lined with flat cells; and 4) a pavement part, in which large multiangular-shaped cells proliferated in a pavement pattern. The epithelial cells of some parts were positive for keratin by immunohistochemistry. Histopathologic findings of the present feline cases were identical to those of malignant craniopharyngioma in other animal species.
Keywords
Craniopharyngioma is a generally benign tumor that develops over the diaphragm of sella or sometimes in the sella turcica, and consists of neoplastic cells with characteristics of epithelium. This tumor comprises about 3% of all intracranial tumors in humans 2 . However, the tumor is rare in animals and has been reported only in the dog, 3, 8 rat, 9, 10 mouse 6 and Mongolian gerbil. 5 We report here two cases of malignant craniopharyngioma in the cat.
Case 1 was a 9.5-year-old, castrated male Chinchilla cat, referred to an animal hospital because of anorexia, rhinor-rhea, and wheezing. The cat had been treated for an allergy; subsequently, the color of his eyes changed, mydriasis appeared, and he became blind. At the Veterinary Medical Center of the University of Tokyo (VMC), magnetic resonance imaging (MRI) examination revealed an oval-shaped mass about 3 cm in diameter in the area of the hypothalamus to the optic chiasm (Fig. 1). The cat's condition had been maintained by dexamethasone for a while, but corneal trepanation and iridoptosis of the right eye occurred and circling and dysgnosia also appeared gradually. The cat was then euthanatized and a necropsy was performed immediately.
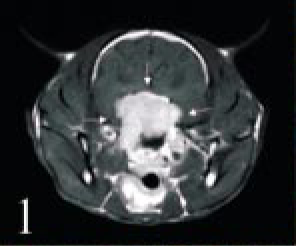
Head; cat No. 1. MRI of an oval-shaped mass at the cranial base (arrows). The white opaque mass is located in the area of the hypothalamus to the optic chiasm.
At necropsy, a white solid mass, 3 × 3 × 1 cm in size, was observed on the skull base (Fig. 2). The mass forced the cerebrum upward, and involved the optic nerves. Although the bone of the skull base around the right ocular opening was lysed by the tumor, the brain and the right eye were not invaded. The hypophysis could not be observed. The other organs looked normal.
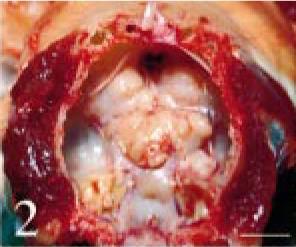
Intracranial tumor mass; cat No. 1. There is a white solid mass on the skull base. Brain was removed. Bar = 1.5 cm.
Tissues were fixed in 10% neutral buffered formalin. Four-micrometer paraffin sections were made and stained with hematoxylin and eosin (HE). The tumor was composed of small multiangular- or spindle-shaped neoplastic cells proliferating in a solid mass. These cells were moderately anaplastic, with a pale-stained nucleus and prominent nucleolus (Figs. 3–6). Many mitotic figures were seen. The lesion was divided into four parts, namely, 1) a “small acinus” part, in which relatively large cells with a pale cytoplasm composed small acini (Fig. 4), 2) a “duct” part, in which small cuboidal cells composed ducts (Fig. 5), 3) a “cyst” part, in which there were large cysts lined with flat cells (Fig. 3), and 4) a “pavement” part, in which large multi-angular-shaped cells proliferated in a pavement pattern (Fig. 6). The cyst and pavement parts were often observed to be adjacent. Free tumor cells, eosinophilic materials, and cholesterol crystals were occasionally seen in the cyst lumina. Interstitial collagen proliferation and melanocytes were also seen.
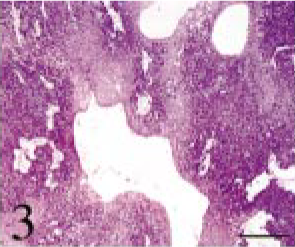
Intracranial tumor mass; cat No. 1. The tumor is composed of small multiangular- or spindle-shaped neoplastic cells proliferating in a solid mass. There are large cysts lined with flat cells. HE. Bar = 700 μm.
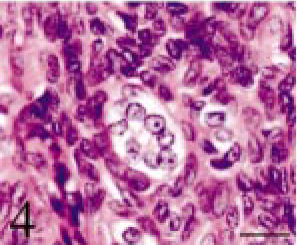
Intracranial tumor mass; cat No. 1. The small acinus part. Relatively large cells with pale cytoplasm compose small acini. HE. Bar = 12 μm.
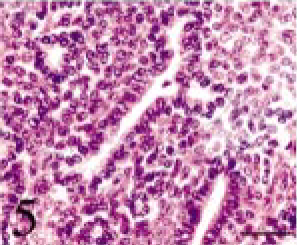
Intracranial tumor mass; cat No. 1. The duct part. Small cuboidal cells compose ducts. HE. Bar = 25 μm.
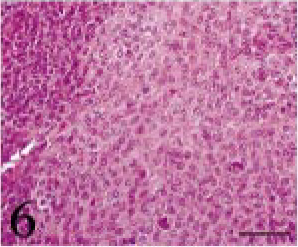
Intracranial tumor mass; cat No. 1. The pavement part. Large multiangular-shaped cells proliferate in a pavement pattern. HE. Bar = 50 μm.
Immunohistochemistry was performed by the ABC method (Vector Laboratories, Inc., Burlingame, CA, USA) using primary rabbit polyclonal antibodies against keratin (wide spectrum screening; Dako, Carpinteria, CA, USA), pituitary hormones (TSH, ACTH, GH, FSH, LH, prolactin; Dako), and mouse monoclonal antibodies against vimentin (Dako), α-smooth muscle actin (α-SMA; Dako), and S-100 protein (Dako). The secondary antibodies used were goat serum against rabbit IgG (KPL, Gaithersburg, MD, USA) and mouse IgG (KPL). The epithelial cells of the small acinus part, the cyst part and the pavement part were positive for keratin (Fig. 7), but those of the duct part were negative. The spindle-shaped cells surrounding the small acini were positive for vimentin and α-SMA. All the tumor cells were negative for S-100 protein and pituitary hormones.
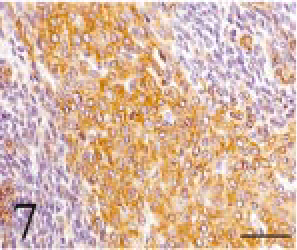
Intracranial tumor mass; cat No. 1. The tumor cells are positive for keratin. Immunostain counterstained with hematoxylin. Bar = 50 μm.
Formalin-fixed tumor tissues were refixed in 2% glutal-dehyde and 1% osmium tetroxide, and embedded in Epon 812. Ultrathin sections were double stained with 5% uranyl acetate and 5% lead nitrate, and observed using a JEM 1200 electron microscope. Microvilli were sometimes observed at the free margin of flat cells lining the cyst. Tonofilaments in the cytoplasm and desmosome-like structures were also observed. The cells were bound to the basement membrane by hemidesmosome-like structures (Fig. 8). Neoplastic cells in the other parts showed almost the same features.
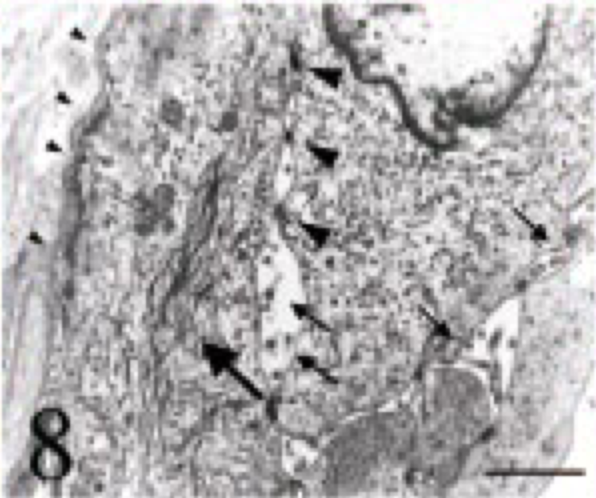
Intracranial tumor mass; cat No. 1. Electron microscopy of a tumor cell. Note the microvilli at the free margin (small arrows), tonofilaments in the cytoplasm (large arrow) and desmosome-like structures (large arrowheads). The cell binds to the basement membrane by hemidesmo-some-like structures (small arrowheads). Bar = 1.2 μm.
Case 2 was a 7-year-old, castrated male American Short-hair cat, and was referred to an animal hospital because of mouth breathing and abnormal breathing sounds. Clinical signs did not improve in spite of medication with prednisolone, ofloxacin, antihistamine drugs and interferon. The cat was therefore referred to the VMC for a detailed examination. X-ray and endoscope examinations revealed a mass in the pharynx, which was removed by operation. Histopathologic findings of the biopsy specimen were almost the same as in case 1. The tumor tissue was composed of small cuboidal anaplastic epithelial cells, often making ducts and small or large cysts (Fig. 9). The acinus pattern seen in case 1 was not observed. Immunohistochemical findings indicated that the ductular epithelial cells were positive for keratin and mesenchymal cells for both vimentin and α-SMA. Taking the tumor site into consideration, this tumor was diagnosed as craniopharyngioma.
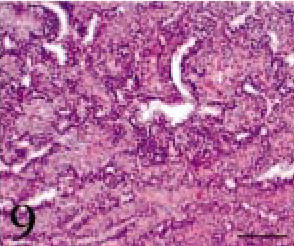
Intracranial tumor mass; cat No. 2. The tumor tissue is composed of small cuboidal anaplastic epithelial cells, often making ducts and small or large cysts. HE. Bar = 100 μm.
MRI examination after the operation revealed that the tumor was located beneath the hypothalamus and extended to the right nasal cavity and bulla tympanica. Radiotherapy and removal of residual tumor were performed, but the tumor recurred repeatedly. Because the cat's general condition gradually worsened and blindness appeared, he was euthanatized according to the owner's wish. Autopsy was not permitted. Observation of the sample obtained at the last operation revealed invasion of the tumor cells into the skull bone, meninges, and neighboring peripheral nerves. On the basis of the clinical signs and histopathologic findings, this tumor was finally confirmed as malignant craniopharyngioma.
Craniopharyngioma in humans is classified into ameloblastomatous type and papillomatous type. 1 In the former type, multiangular- or stellate-shaped tumor cells form focal or cord-like structures, and the nuclei of cells surrounding the structure are arranged as a palisade. Many cysts are lined with thin epithelial cells, and keratinization can be seen. In the papillomatous type, squamous cells proliferate in papillary form. Electron microscopic findings of human cranio-pharyngioma have revealed that tumor cells and interstitial tissues are separated by the basement membrane, and that hemidesmosomes, desmosomes, and assembly of tonofilaments are present. 4
In the present two feline cases, the histopathologic findings were similar to those reported in previous canine 1 and murine 6, 9, 10 cases, in which solid proliferation of cuboidal, columnar, and squamous epithelial cells, and formation of cysts lined with the epithelial cells, were seen. In other reports, occasional keratinization and calcification were observed, but these were not present in the current cases. The immunohistochemical and electron microscopic findings, which supported the epithelial origin of the tumor, were also identical to those in other animals. 5, 9 The present feline cases were, therefore, diagnosed as craniopharyngioma. Pituitary adenoma/adenocarcinoma, chordoma, meningioma and granular cell tumor were considered as differential diagnoses of the present cases, but all were ruled out by morphologic differences. Pituitary adenoma/adenocarcinoma was denied because of negative immunohistochemical reaction to pituitary hormones. Chordoma usually consists of large-sized cells and massive intercellular matrices. Meningioma and granular cell tumor are characterized by a whirl-like arrangement of tumor cells and fine periodic acid Schiff-positive cytoplasmic granules, respectively. The present feline cases were, in addition, diagnosed as malignant because of the severe anaplasia of the tumor cells and the invasion into the surrounding bones. Accordingly, the present cases were diagnosed as feline malignant craniopharyngioma. Though craniopharyngioma is not an extremely rare tumor in humans and dogs, only a few malignant cases have been reported in humans, 7 rats, 9 and mice. 6 As far as we know, no feline cases, either benign or malignant, have been reported up to the present.
