Abstract
Pregnant mares were experimentally exposed to whole caterpillar or exoskeleton of the Processionary caterpillar (Ochrogaster lunifer) via gavage. Tissues were collected from resulting abortions and near or full-term pregnancies consisting of 13 aborted fetuses, 3 fetuses from treated euthanized mares, membranes of 5 foals, and organs from 3 foals. Three control membranes and 1 control fetus and membranes were examined. Caterpillar setal fragments were present in the allantochorion of the 3 fetuses from the euthanized mares and 11 of 12 aborted fetuses (92%) embedded in the chorion (villi or stroma) or allantois (vasculature or stroma). Placental locations of fragments ranged from the cervical pole region to the body encompassing the umbilical insertion and pregnant horn. Numbers in each fetus ranged from 1 to 7 fragments. Setae were present in the allantochorion from 2 to 22 days after the initial treatment. Acute to chronic active inflammation was present in all aborted fetuses, all euthanized fetuses, and within at least 1 tissue level (chorion, allantois, umbilical cord, or amnion) of the membranes from full-term foals. Amnionitis, funisitis, and allantoitis were present in 95% of the examined membranes. Pneumonia was present in 95% of the specimens, and bacteria were present histologically in 90.5% of the specimens with or without accompanying inflammation. The rapid migration of setae within 2 days after mare exposure suggests that direct setal migration into the fetal membranes is a likely initiating factor for equine amnionitis and fetal loss (EAFL).
Equine amnionitis and fetal loss (EAFL) is a recently recognized cause of abortion in Australia associated with the ingestion of processionary caterpillars (PC) (Ochrogaster lunifer) or their exoskeleton by pregnant mares. 10,11,34 Caterpillar-related abortion in mares has also been reported in the United States, where it is known as mare reproductive loss syndrome (MRLS) and is caused by the Eastern tent caterpillar (Malacosoma americanum) (ETC). 38 Inflammation of the amnion and umbilical cord with some degree of fetal pneumonia occurs in both conditions, with isolation of atypical bacteria in the form of environmental or enteric organisms not commonly isolated in other forms of bacterial abortion. 27,30,34
Equine amnionitis and fetal loss is characterized by acute abortions with absent to minimal gross lesions and abortions with chronic lesions, including marked edema and hyperemia of the fetal membranes, increased vessel tortuosity, and diphtheritic membranes on the umbilical cord, amnion, and umbilical insertion. 34 Histologically, inflammation in the lungs, allantois, amnion, and umbilical cord were the most common findings in confirmed and suspect field cases along with the isolation of atypical bacteria from the lungs and/or stomach contents. 34 As in other causes of bacterial placentitis, fetal death is attributed to overwhelming bacterial infection compromising the fetus either through placental insufficiency, inflammatory mediators, or sepsis. 40
A number of aspects of the etiology and pathogenesis of caterpillar-induced abortion have yet to be fully elucidated. The exposure of mares to PC is most likely explained by accidental consumption of shed caterpillar exoskeleton while grazing, since it is unlikely that mares would consume whole caterpillars. 11,25 However, the most intriguing aspect of this condition is the manner in which caterpillars facilitate fetal infection by essentially nonpathogenic bacteria in apparently healthy mares. It has been proposed that ingested caterpillar setae (hairs) penetrate the gut wall through mechanical action and carry along bacteria that are present in the mare’s intestinal tract, allowing these bacteria to then spread hematogenously. 33 Others suggest that these caterpillars may contain toxin(s) that facilitate the transfer of bacteria across the gut wall to the mare’s circulation. 30 Moreover, it has been proposed that septic material (encompassing bacterial emboli and/or septic setal fragments) crossing the gut wall is a likely explanation for the range of MRLS-related disease, including unilateral uveitis, pericarditis, and encephalitis in the mare. 33 These 3 conditions have not been associated with EAFL. 34
Experimental investigation into the cause of EAFL in which pregnant mares were gavaged with emulsified suspended PC found variable amounts of PC setal fragments at all levels of the gastrointestinal tract, within mesenteric connective tissue, mesenteric lymph nodes, liver, and uterus to the level of the endometrium. 11,35 These findings are detailed in the companion article 35 and support the idea that the initiation of EAFL involves the attachment of environmental or enteric bacteria to caterpillar setae within the gastrointestinal tract and direct inoculation into the fetal membranes by setal migration and/or bacterial translocation via a pathway made by migrating setae into the uterus of the mare and fetus. 10,35 The objective of this study was to assess the histological changes in fetuses and fetal membranes of pregnant mares experimentally exposed to PC to further delineate the pathogenesis of EAFL.
Materials and Methods
The Standardbred mares from which the fetuses and fetal membranes where obtained for this study participated in an experimental program that sought to establish the relationship between PC ingestion and the occurrence of EAFL. Three experiments were undertaken using whole caterpillar or caterpillar exoskeleton, emulsified in 500 ml distilled water in a laboratory blender, and gavaged directly into the stomach of the mares. The mares were mated with the same Australian Stockhorse stallion and were all exposed to PC in mid-gestation (128–252 days). The experiments were approved by the University of Queensland Animal Ethics Committee and were reported in part previously. 10,11
Briefly, in experiment 1, 2 groups of 4 mares were gavaged by nasogastric tube with either 500 ml of distilled water (controls) or 100 g whole PC emulsified in 500 ml of distilled water once daily for 5 days. Experiment 2 consisted of 4 groups of 3 mares gavaged with distilled water (controls) or 1 g, 2 g, or 5 g of caterpillar exoskeleton emulsified in 500 ml of distilled water once daily for 5 days. Experiment 3 consisted of 3 groups (A, B, C) of mares undergoing gavage with 500 ml distilled water (controls; group A) or 50 g whole PC (group B) emulsified in 500 ml distilled water once daily for 5 days. Groups A and B both consisted of 6 pregnant mares that had never been exposed to caterpillars. In contrast, in their previous pregnancy, group C (4 mares from experiment 2) mares had been exposed to caterpillar exoskeleton, rebred, and were back in foal when recruited for experiment 3. All mares in each experiment were housed and managed the same and monitored daily by clinical examination, hematology, and ultrasonography. 11
Collection of Abortuses
Aborted fetuses and the associated fetal membranes were collected during the expulsion process or within 1 hour if the abortion was unexpected. Full-term placentae were collected immediately after parturition. Seven mares (Table 1), including 1 pregnant control mare, were euthanized during the experiments either for humane reasons (2 mares developed severe colic, 1 mare developed suppurative endometritis and laminitis) or as a structured euthanasia to determine the effects of caterpillar ingestion in the pregnant mare after gavage with PCs (4 mares). If the euthanized mares had not aborted, their fetuses were collected at the time of postmortem. Details of these mares and results are described in the companion article on mare histopathology. 35
Histopathological Lesions in Fetuses and Fetal Membranes From Pregnant Mares After Experimental Gavage With Processionary Caterpillars.a
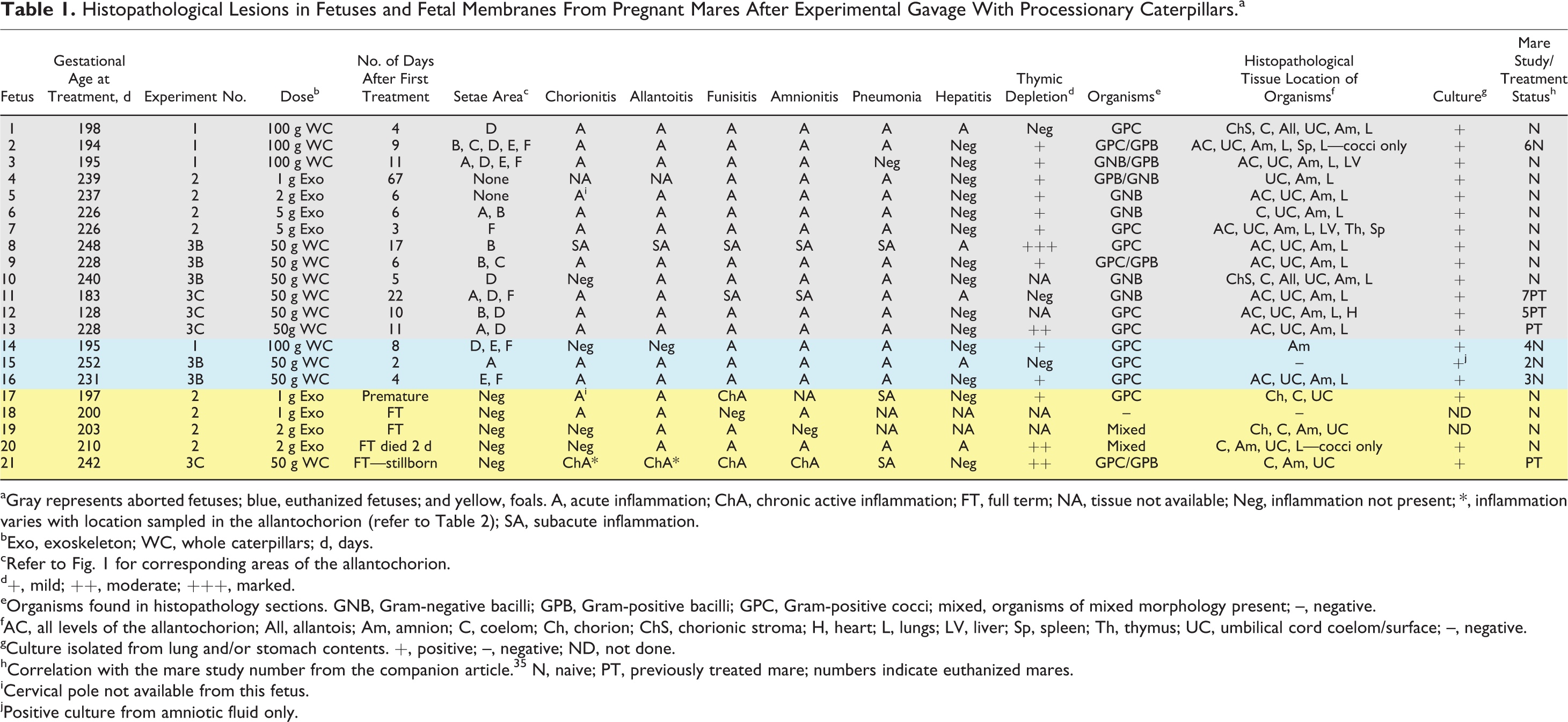
aGray represents aborted fetuses; blue, euthanized fetuses; and yellow, foals. A, acute inflammation; ChA, chronic active inflammation; FT, full term; NA, tissue not available; Neg, inflammation not present; *, inflammation varies with location sampled in the allantochorion (refer to Table 2); SA, subacute inflammation.
bExo, exoskeleton; WC, whole caterpillars; d, days.
cRefer to Fig. 1 for corresponding areas of the allantochorion.
d+, mild; ++, moderate; +++, marked.
eOrganisms found in histopathology sections. GNB, Gram-negative bacilli; GPB, Gram-positive bacilli; GPC, Gram-positive cocci; mixed, organisms of mixed morphology present; –, negative.
fAC, all levels of the allantochorion; All, allantois; Am, amnion; C, coelom; Ch, chorion; ChS, chorionic stroma; H, heart; L, lungs; LV, liver; Sp, spleen; Th, thymus; UC, umbilical cord coelom/surface; –, negative.
gCulture isolated from lung and/or stomach contents. +, positive; –, negative; ND, not done.
hCorrelation with the mare study number from the companion article. 35 N, naive; PT, previously treated mare; numbers indicate euthanized mares.
iCervical pole not available from this fetus.
jPositive culture from amniotic fluid only.
Examination of Abortuses
All postmortems were carried out in a systematic manner for each fetus and fetal membranes regardless of the method of collection. Samples required for other studies (fluids and tissues for culture and EHV-1 exclusion) were taken prior to any tissues for histopathology. The allantochorion was laid out in the reverse F to ensure completeness and any gross lesions recorded after examination of both surfaces. The amnion and umbilical cord were similarly laid out and any gross lesions recorded. Samples for histopathology were taken from each tissue corresponding to areas of the allantochorion, amnion, and umbilical cord as depicted in Fig. 1.
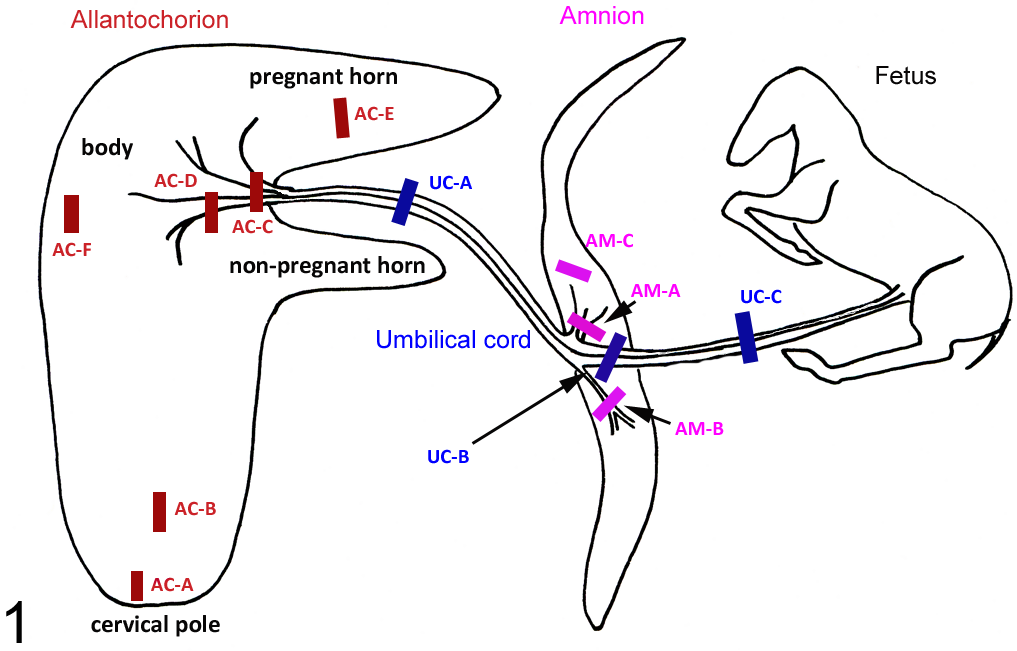
Diagram of areas sampled for histopathological examination from the fetal membrane, fetuses, and foals of pregnant mares after experimental gavage with processionary caterpillars. AC-A, allantochorion cervical pole; AC-B, allantochorion 10 cm from cervical star; AC-C, allantochorion umbilical insertion; AC-D, allantochorion 10 cm from umbilical insertion; AC-E, allantochorion pregnant horn; AC-F, allantochorion body; Am-A, amnion at the umbilical insertion; Am-B, amnion 10 cm from the umbilical insertion; Am-C, amnion 30 cm from the umbilical insertion; UC-A, umbilical cord midway between the amnion and allantochorion; UC-B, umbilical cord at the amnion; UC-C, umbilical cord between the amnion and fetus.
All fetuses were necropsied in right lateral recumbency, and samples for bacterial culture were taken in situ, before organ systems were removed. Approximately 1-cm sections of tissue were taken from the thymus, liver, spleen, lung, kidney, and heart. Collected tissue samples were fixed in phosphate-buffered 10% formalin for 48 hours or longer. Thin (3–4 mm) sections of tissue were embedded in paraffin, sectioned at 6 μm, and stained with Wright’s modification of the hematoxylin and eosin (HE). Additional specific sections were stained with toluidine blue (TB) for mast cell presence within the tissues or with Gram stain to confirm microorganism morphology if detected. Areas of the fetal membranes examined were divided into the chorion, consisting of the villi, intervillous trophoblast epithelium, and chorionic stroma; the allantois, consisting of the extraembryonic coelom (EEC), allantoic vessels, stroma, and allantoic epithelium; umbilical cord; and amnion.
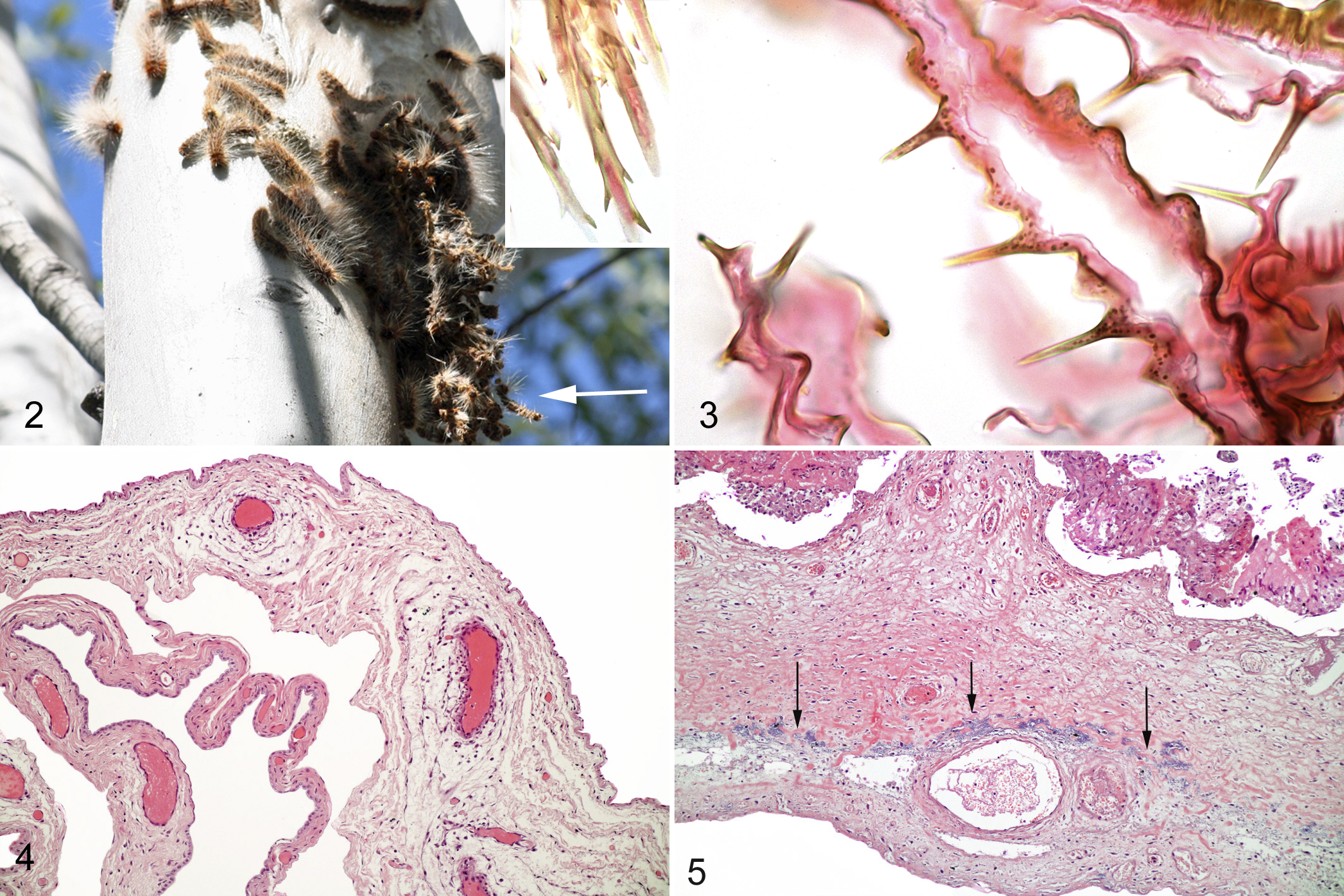
For comparison, a whole PC (Fig. 2) collected for use in the above experiments and caterpillar exoskeleton were placed in 10% formalin for 48 hours. The caterpillar was selected beginning at the head in sequential 3-mm sections and processed as described above. Exoskeleton was processed intact. Slides were stained using HE and TB and used as a standard for setae morphology and staining characteristics.
Results
Caterpillar
Sections of the caterpillar showed large numbers of true setae present within multiple ventral depressions suggestive of the described mirrors. These setae were golden and refractile with sharp tapered ends measuring approximately 120 μm in length. Short angled barbs were present along the shaft, longer at the anterior end to barely present at the posterior end (Fig. 2, inset). Setae diameter ranged from 3 to 10 μm with a hollow interior. Many setae were fragmented and lost during processing. The exoskeleton consisted of thin convoluted remnants of the cuticle with regularly spaced smooth sharp spines (Fig. 3). The area of the mirrors could not be identified in the tangled mass of the cuticle and spines.
Experimental Overview
Across the 3 experiments, 23 mares were gavaged with caterpillar suspension, but only 21 fetuses, fetal membranes, and foals were available for examination. Specimens examined consisted of 13 aborted fetuses, 3 fetuses from treated euthanized mares, and membranes of 5 foals from a total of 21 treated mares. Results are summarized in Tables 1 and 2. Membranes from an additional 2 normal full-term foals were not collected and therefore excluded from the study. Organs were available from 3 of 5 foals: 1 foal (F17) was born premature at 311 days’ gestation and euthanized 2 days after birth, 1 full-term foal (F20) died at 2 days of age, and 1 foal (F21) was stillborn. The remaining 2 full-term foals were healthy and only their membranes were examined. Out of the study population, 1 fetus was missing the entire allantochorion (F4), 1 fetus (F5) was missing the cervical pole region of the allantochorion, and 1 premature foal (F17) was missing the cervical pole and umbilical insertion regions and the amnion. Nine of the aborted fetuses (9/13) were from mares that received whole caterpillars and 4 mares received caterpillar exoskeleton. Euthanized mares received whole caterpillars. Of the foals, 4 mares received exoskeleton and 1 received whole caterpillars. Controls consisted of 3 sets of membranes from full-term foals from control mares participating in the experiments and the membranes and tissues of 1 control fetus euthanized 5 days after the first control treatment of distilled water to coordinate with the same gestational age of the aborted fetuses. Detailed results of bacteria isolated during the experiments will be published elsewhere, but morphology, histologic tissue location of organisms, and presence in culture are detailed in Table 1. The gross pathology found in the fetuses has been previously reported. 10,11
Controls
Tissues of the fetal membranes were mildly to moderately autolyzed with variable amounts of intracoelomic and/or stromal hemorrhage more prominent in the cervical pole region. Increased cellularity was confined to the allantoic membrane attached to the umbilical cord with a uniform diffuse infiltration of the allantoic stroma with mononuclear cells in low numbers. Mild hypertrophy of the allantoic epithelium was present without evidence of inflammation. The amniotic surface of control membranes showed mild superficial necrosis in association with rare infiltration of neutrophils at the epithelial basement membrane. A focal plaque of squamous metaplasia was present on the amniotic surface of 1 control near the insertion of the umbilical cord without associated inflammation. Neutrophils were not associated with the EEC in any tissues. Organs of the control fetus were normal for the gestational age.
Treated
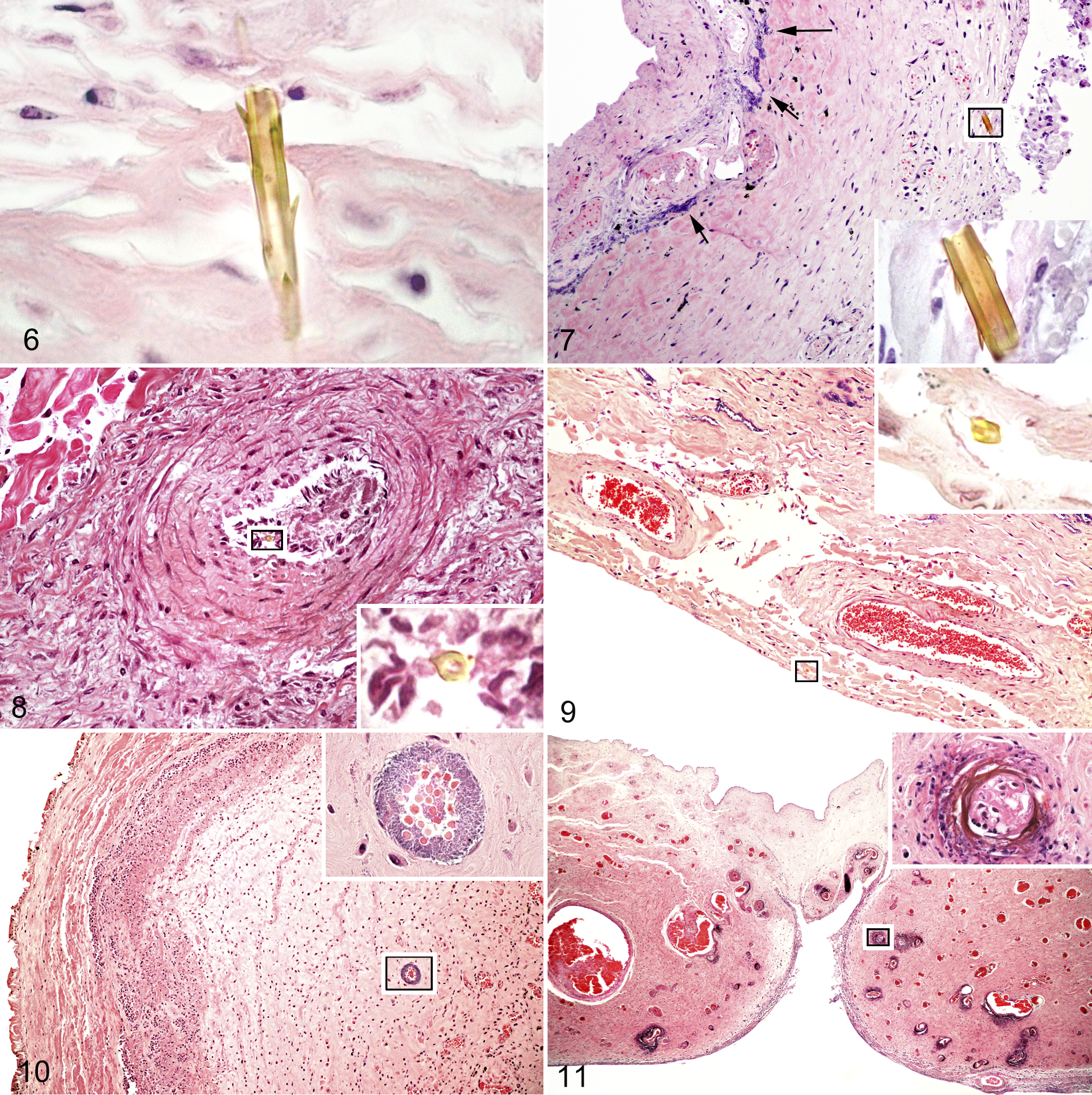
Autolysis was moderate to severe in the membranes of all aborted fetuses and 1 full-term foal (F20). Perivascular edema at all tissue levels (Fig. 4) was the most common finding affecting all aborted and euthanized fetuses as well as 4 foal membranes (F17, F18, F19, F21).
Bacteria
Bacteria were found histologically in 19 of 21 fetuses and foals (90.5%; Table 1). Culture was positive in all 19 of the fetuses and foals from which tissues were cultured (100%; Table 1). Of the 2 full-term foals not cultured, bacteria were observed histologically within the fetal membranes of 1 foal (F19) but not in the membranes of the other foal. Bacteria were present histologically in most regions sampled in the examined membranes with the exception of 1 fetus (F14) from a euthanized mare in which bacteria were found within only 1 section of the amnion. All aborted fetuses as well as 1 fetus (F16) from 1 euthanized mare and 1 full-term foal (F17) born prematurely had bacteria present histologically with associated inflammation. This correlated with positive bacterial cultures from the lung and/or stomach contents of these fetuses (Table 1). Bacteria were not present histologically but were isolated from the amniotic fluid in 1 fetus (F15) from a mare euthanized within 2 days of the first treatment. Bacteria were present histologically with only subtle inflammation in the umbilical cord of 1 full-term foal (F19). Bacterial colonization confined to the coelom and directly adjacent tissue of the chorionic stroma and allantois (Fig. 5) was found in all allantochorionic samples from 3 fetuses (F1, F6, F10) aborted within 6 days of the initial treatment and 2 full-term foals (F20, F21). The remaining aborted fetuses had bacterial colonization throughout all tissue levels in all sampled areas of the allantochorion, umbilical cord, and amnion. Detailed results of specific bacteria from positive cultures will be expanded in another publication.
Setal Fragments
All 3 fetuses from the euthanized mares and 11 of 12 aborted fetuses (92%) in which allantochorion was available had setal fragments embedded in the allantochorion (Fig. 6) ranging from the villi of the chorion, within the subvillous chorionic stroma (Fig. 7), within allantoic vasculature (Fig. 8), and protruding through the allantoic stroma (Fig. 9). Cervical pole tissue was not available in the aborted fetus (F5) in which setae were not found. Setal fragments were found in all sampled regions of the allantochorion (AC), ranging from the cervical pole region (A) to the body (F) encompassing the area of umbilical insertion (C) and pregnant horn (E) (see Fig. 1). Numbers of setal fragments in each fetus ranged from 1 to a maximum of 7 fragments in various locations (Table 2). Time from the first treatment of the mare to detection of setal fragments in the allantochorion of fetuses ranged from 2 to 22 days. Setae were not found in any of the full-term fetuses or control membranes. Inflammation was not generally associated with the embedded setal fragments other than focal necrosis around the setae or rare solitary neutrophils in proximity to the fragment.
Distribution and Type of Inflammation in Fetal Membranes of Pregnant Mares After Experimental Gavage With Processionary Caterpillars.a
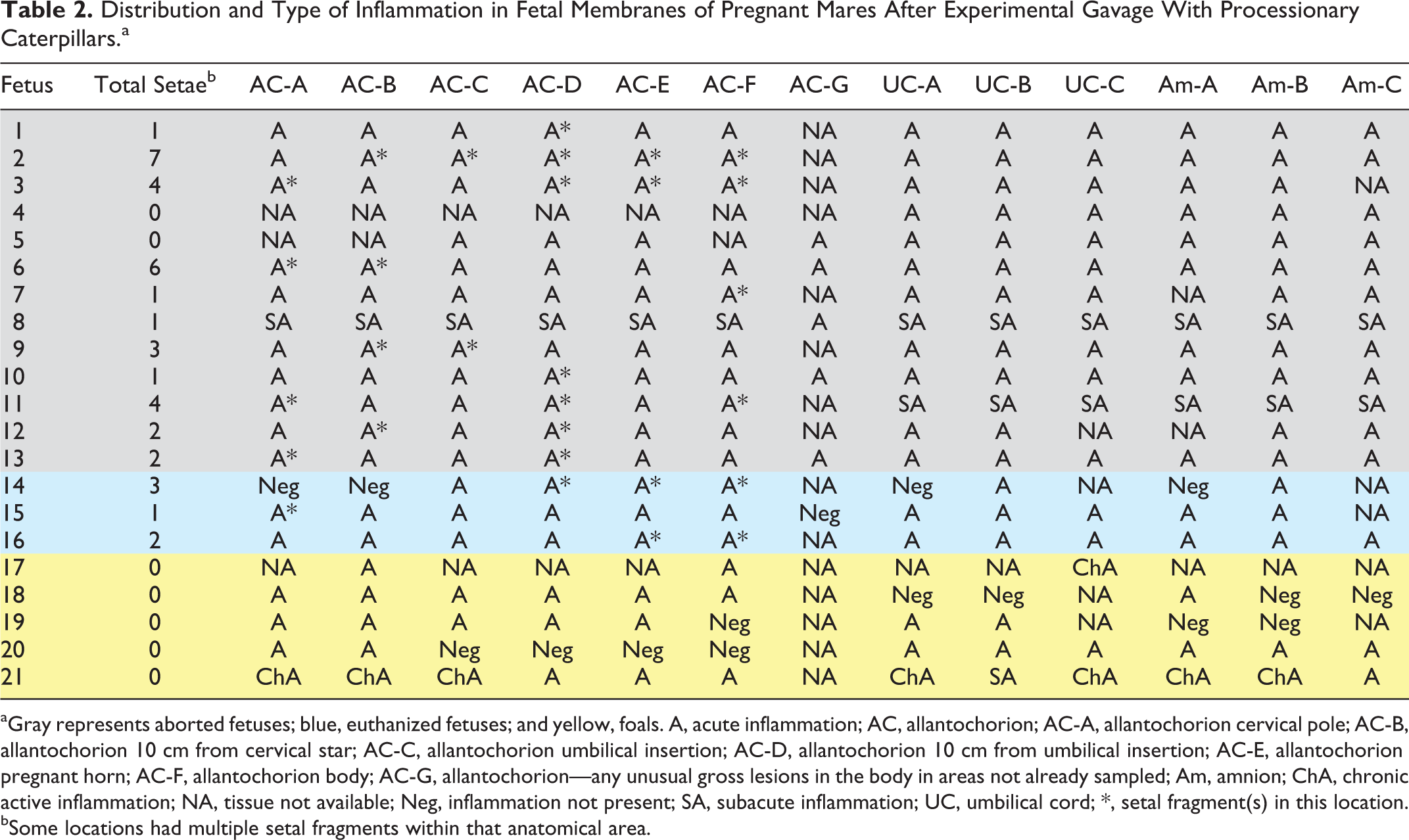
aGray represents aborted fetuses; blue, euthanized fetuses; and yellow, foals. A, acute inflammation; AC, allantochorion; AC-A, allantochorion cervical pole; AC-B, allantochorion 10 cm from cervical star; AC-C, allantochorion umbilical insertion; AC-D, allantochorion 10 cm from umbilical insertion; AC-E, allantochorion pregnant horn; AC-F, allantochorion body; AC-G, allantochorion—any unusual gross lesions in the body in areas not already sampled; Am, amnion; ChA, chronic active inflammation; NA, tissue not available; Neg, inflammation not present; SA, subacute inflammation; UC, umbilical cord; *, setal fragment(s) in this location.
bSome locations had multiple setal fragments within that anatomical area.
Inflammation
Acute to chronic active inflammation was present in all aborted fetuses, all euthanized fetuses, and within at least 1 tissue level (chorion, allantois, umbilical cord, or amnion) of the membranes from foals (Table 1). Inflammation if present was generally diffuse throughout all sections of the sampled areas (Table 2). Nineteen of 20 fetuses/foals had some degree of amnionitis and/or allantoitis (95%), and 20 of 21 fetuses/foals had some degree of funisitis (95%). Pneumonia was present in 18 of 19 fetuses/foals (95%), and 19 of 21 fetuses/foals (90.5%) had bacteria within the tissues microscopically with or without inflammation present. Chorionitis was present in 16 of 20 fetuses/foals (80%). Three previously treated mares (F11, F12, F13) aborted within 22 days of the first treatment and had acute inflammation within the fetal membranes and fetal lungs with positive cultures and bacteria evident histologically. The remaining previously treated mare (F21) had a stillborn full-term foal with chronic active inflammation within the fetal membranes and fetal lungs with a positive bacterial culture and mixed bacteria present histologically.
Chorion
Acute inflammation characterized predominantly by neutrophils was present in the chorion of 10 of 12 aborted fetuses (allantochorion missing for 1 aborted fetus) and 2 fetuses from euthanized mares (Table 1). Inflammation ranged from small clusters of neutrophils in intervillous spaces to infiltration of the villous lamina propria with low to high numbers of neutrophils and/or neutrophil margination and migration through vessels of the chorionic stroma and lamina propria. Mononuclear cell inflammation in the form of low to moderate numbers of lymphocytes and/or macrophages infiltrating the chorionic stroma either diffusely or at the base of the villous stalks was present in 3 foals (F17, F18, F21). Early to advanced mineralization of villous tips or vasculature of the lamina propria of the chorion was present in 6 aborted fetuses, 1 euthanized fetus, and 1 full-term foal. Subacute inflammation was present in the remaining aborted fetus (F8) with expansion of the chorionic stroma by infiltrating neutrophils, eosinophils, and macrophages that were more prominent in areas adjacent to the villous stalks. In addition, in the area adjacent to the cervical pole of this fetus, there was a well-demarcated focal area of neutrophil infiltration in the chorionic stroma with intact villous tips and a large amount of amorphous eosinophilic exudate in the intervillous areas. In the area of umbilical attachment, there were multifocal areas of localized neutrophilic vasculitis and infarction with colonization of the resulting degenerate villous tissue by bacteria.
Allantois
Acute inflammation characterized by mild to marked fibrin and neutrophil exudation within the EEC, neutrophil margination and migration within the allantoic vessels, and/or neutrophil infiltration of the allantoic stroma was present in 11 of 12 aborted fetuses, 2 euthanized fetuses, and 5 foals (Table 1). More intense subacute inflammation characterized by areas of fibrinoid vasculitis and infarction of the allantoic vessels, expansion of the allantoic stroma by mixed inflammatory cells, and stage I allantoic epithelial hyperplasia was present in 1 aborted fetus (F8). 20 Chronic active inflammation in 1 full-term foal (F21) was characterized by predominantly mononuclear cells with marked fibrin exudation within the EEC, moderate exudation of lymphocytes and plasma cells followed by lesser numbers of macrophages and neutrophils, and scattered low numbers of neutrophils within the allantoic stroma. These changes were more marked in the cervical pole and less prominent in the area of umbilical insertion of this fetus (Table 2).
Umbilical Cord
Acute funisitis in the form of mild to marked fibrin and/or neutrophil exudation within the EEC, neutrophil margination and migration within the superficial stromal vessels, and/or neutrophil infiltration of the vascular stroma was present in 11 of 13 aborted fetuses, all 3 euthanized fetuses, and 2 full-term foals. Subacute inflammation was present in the remaining 2 aborted fetuses (F8, F11), and chronic active inflammation was present in 2 full-term foals (F17, F21) (Table 1). In the aborted fetuses, subacute inflammation was characterized by marked infiltration of superficial surfaces by neutrophils and macrophages as well as heavy exudation of fibrin, neutrophils, and macrophages within the EEC. Fetus No. 8 had vasculitis of superficial vessels within the stoma and amniotic attachment with patchy areas of infarction. Bacterial colonization of the umbilical cord vasculature was present in fetus No. 11 (Fig. 10). The umbilical cord of the premature foal (F17) exhibited mixed inflammation of lymphocytes, neutrophils, and macrophages in perivascular and superficial areas; moderate stromal fibrovascular proliferation; and intraluminal abscess formation with mixed bacterial colonization. The other full-term stillborn foal (F21) had patchy areas of squamous metaplasia with underlying fibroplasia as well as dense areas of squamous debris on the surface with marked infiltration of these areas by macrophages, neutrophils, and scattered multinucleated giant cells. The EEC was obliterated by large numbers of lymphocytes, plasma cells, and neutrophils.
Amnion
Acute amnionitis was present in 12 of 13 aborted fetuses, all 3 euthanized fetuses, and 2 full-term foals (Table 1). Inflammation consisted of mild to marked neutrophil margination and migration in amniotic vessels, perivascular infiltration by neutrophils, and/or mild to marked accumulation of fibrin and neutrophils within the coelom. One aborted fetus (F13) had extensive fibrinoid vasculitis and infarction of the amnion with mineralization of the tunica media in small to medium superficial vessels (Fig. 11). The remaining aborted fetus (F8) had subacute inflammation with multifocal areas of vasculitis, infarction, and distension of the coelom with fibrin, neutrophils, and macrophages. The allantoic surface of the amnion exhibited multifocal areas of stage I epithelial hyperplasia with the stroma expanded by edema, neutrophils, and macrophages. The stillborn foal (F21) had severe extensive chronic active inflammation of the amnion with a mixed inflammatory infiltrate, marked capillary proliferation in surface areas and around large vessels, and diffuse stromal fibroplasia. There were multifocal thick layers of squames on the amniotic surface with frequent focal mineralization and associated multinucleated giant cells in low numbers. Multifocal areas of amniotic epithelial necrosis were also present.
Lungs
Acute pneumonia characterized by multifocal to diffuse accumulations of neutrophils mixed with squamous epithelial cells and proteinaceous material was present in alveoli and bronchioles of 11 aborted fetuses, 3 euthanized fetuses, and 1 of 3 examined foals (Table 1). Subacute pneumonia characterized by macrophages and neutrophils in alveoli, bronchioles, and the interstitial tissue was present in 1 aborted fetus (F8) and 2 foals (F17, F21). Multinucleated giant cells were present in alveoli of 1 aborted fetus (F8) and 1 premature foal (F17). One aborted fetus (F3) had bacterial presence in the alveoli and bronchioles without obvious inflammation.
Liver
Three aborted fetuses, 1 euthanized fetus, and 1 full-term foal had mild acute hepatitis ranging from focal periportal infiltrates of neutrophils to more diffuse presence of neutrophils within the sinusoids (Table 1).
Thymus
There was mild thymic lymphoid depletion in 7 aborted fetuses, 2 euthanized fetuses, and 1 premature foal characterized by mild increases in medullary macrophages and/or decreased medullary lymphocyte cellularity. Moderate lymphoid depletion was present in 1 aborted fetus (F13) and 2 full-term foals (F20, F21) with increased numbers of both cortical and medullary macrophages, active macrophage phagocytosis, decreased lymphocyte cellularity in either the cortices or medullary areas, and prominent medullary reticular epithelium. Severe lymphoid depletion was present in 1 aborted fetus (F8) with blurring of the corticomedullary junction, marked increase in both cortical and medullary macrophages, diffuse decrease in lymphocyte numbers, and prominent reticular epithelium in both the cortical and medullary areas. Bacterial colonies were present in the vasculature of the thymus in 1 aborted fetus (F7).
Other Tissues
Splenic lymphoid depletion was variably present in many of the fetuses but was not graded due to a lack of a published defined grading scale.
Discussion
Identification of caterpillar setal fragments in the allantochorion has not been described previously. Setal fragments were found at all tissue levels in the gastrointestinal tract from the mucosa to the serosa in selected mares from which the abortuses in this study were gathered. 35 In addition, fragments were found within hyperplastic serositis of the small and large intestines in these mares as well as within lymph nodes, livers, and uteri from the serosa to the endometrium. 35 The presence of the setae in the allantochorions in this study indicates setal migration through the uteri and across the fetomaternal interface from the gastrointestinal tracts of the mares.
Setal fragments were found throughout the allantochorion encompassing the cervical pole to the pregnant horn, umbilical attachment, and body. These sites correspond to the many areas in the uterus where setae were found in mares euthanized on sequential days after gavage with PC. 35 In the mares, the largest number of setae were found in the large and small colon. 35 The nonpregnant uterus is in constant contact with the small colon and in variable contact with the small and large intestine depending on the amount of ingesta, parity of the mare (pendulous uterus), and gut motility. 4 As gestation progresses, contact between the pregnant uterus and gastrointestinal tract becomes more constant as the available space for expansion is reduced. This close proximity at many points during pregnancy could result in the wide dispersal of migrating setae into any area of the allantochorion, and thus any area of the allantochorion could be involved in the inflammation associated with EAFL. The significance of the experimental finding of setal presence in the allantochorion to the pathogenesis of caterpillar-induced abortion is supported by the recent finding of caterpillar setae fragments in the placenta of a field case of EAFL. 36
The presence of setal fragments in the equine allantochorion within 2 days of initial caterpillar exposure indicates that caterpillar setal migration can be very rapid, in contrast with the slower rate of weeks and months described in human tissue. 8,43 The rate of migration is likely influenced by many variables. Ocular migration of caterpillar setal fragments has been thought to be caused by the synchronous movements of the eye where these movements along with the barbs on the setal fragments produce progressive inward propulsion of the setae. 32 The streamlined shape and orientation of the barbs of the setae, stretching of organ walls due to ingesta and pregnancy, and the muscular contractions of the gastrointestinal tract and uterus may explain the rapid movement of setae through the gastrointestinal tract, uterus, and into the membranes of the equine fetus. Slower or incomplete migration may also occur if any of those factors are changed, including blunting of the setal fragment, denser tissue, gut stasis, or early stage of gestation. Setae were found in all of the placentas from the euthanized mares up to 22 days from the first treatment but were not found in late abortions (>22 days from the first treatment) or in the membranes from near- or full-term foals. This may be due to degradation of setae from the long-term presence within the tissue, full migration of setae into the allantoic or amniotic fluid and loss at parturition, or migration and lodgement in an area of the membranes not sampled. However, the presence of acute inflammation within the membranes in these foals with neonatal compromise suggests some setae migration did occur after 22 days from the first exposure. Slow setal migration into the retina after human ocular penetration can occur years after exposure. 8 This allows speculation that setal migration does not have to take place immediately after exposure but can occur slowly at any stage of gestation after exposure, including near term.
Relative setal numbers (Table 2) within the anatomical locations in the chorioallantois were higher in 2 of the fetuses (F2, F3) from mares given 100 g whole caterpillars than those given 50 g of whole caterpillar or caterpillar exoskeleton. The mare aborting fetus 2 (Table 1) had 11 setal fragments in various locations in the uterus. 35 Large numbers of setal fragments in small uterine tissue sections may indicate that massive numbers of setal fragments are migrating in a virtual shower through the uterus and into the fetal membranes. The high dose of whole caterpillar given to this mare with subsequent high numbers of setal fragments in the chorioallantois of her fetus suggests the exposure dose does play a role in the numbers of setae migrating and possibly in the pathologic outcome.
Dendrolimiasis, pararamose, and ocular lodgement of setal fragments from caterpillars have been reported to cause localized acute to granulomatous inflammation with a lymphocytic and multinucleated giant cell reaction similar to the granulomas in the gastrointestinal tract of experimentally exposed animals and mares. 21,30,32,35 Processionary caterpillars are members of the Lepidopteron family Notodontidae, subfamily Thaumetopoeidae and have setae composed of chitin. 3,15 Chitin degradation in mammalian tissues is primarily by chitotriosidase, a chitinase-like protein present in macrophages. 3 Presence and breakdown products of chitin can stimulate a proinflammatory response in humans, and this response in the form of macrophage stimulation may be responsible for the mononuclear and granulomatous reactions seen around setal fragments in the gut, hyperplastic serosal lesions, and uterus of the mares. 3 A general lack of reaction around many of the setal fragments in the allantochorion, similar to findings in some locations within the mare, may reflect the continued migration of the fragments that have not been blunted, eroded, or otherwise halted in their migration paths. 35 Alternatively, stationary setae may exist in tissues without causing an inflammatory reaction, which has been reported in cases of ocular setae penetration in people. 23
Greater than 90% of the tissues examined from fetuses, including aborted, euthanized, and full-term foals, had evidence of allantoitis, amnionitis, funisitis, and pneumonia that was generally diffuse. The inflammation was likely a response to the bacterial infection introduced by the setae. The distribution of the inflammation and the presence of setae within the chorionic stroma, allantoic vessels, and protruding from the allantois into the allantoic cavity suggest one route of bacterial entry is via direct inoculation or seeding from caterpillar setal migration into the allantoic EEC. 16 The EEC is a remnant of the space that develops between the allantoic endoderm and the connective tissue containing the allantoic vessels. Much of this space is eliminated by day 90 of gestation, with only distinct spaces overlying larger blood vessels of the allantochorion remaining. 16 Morphological studies of normal fetal membranes of the foal found that this space communicates with the infundibulum of the allantoic portion of the umbilical cord and with the remnant of the EEC within a rectangular portion of the amnion at the umbilical insertion. 40,41 Once bacteria invade the EEC, organisms can rapidly multiply and colonize the allantois and spread into the umbilical cord, amnion, and ultimately the fetus. 39 This communication is the likely reason for the occurrence of the gross lesions within a 30-cm radius from the umbilical insertion in EAFL field cases. 34 A similar distribution of inflammation and mechanism of bacterial spread can be seen in other types of infective placentitis, such as ascending and peripheral placentitis where organisms have been identified in 1 or more of the EEC compartments within the allantochorion, amnion, or infundibulum. 26,39 In ascending placentitis, bacteria originate in the cervical area, initially affect the chorion where there is no attachment to the endometrium, and then spread superficially along the body and deeply via the communication between the EEC of the allantochorion, umbilical cord, and amnion. However, experimentally induced ascending placentitis results in amnionitis much less frequently (28% of cases), likely because bacteria must invade across multiple tissue layers and are not inoculated directly into the EEC. 9
Bacteria were evident only in the EEC and adjacent tissue in 3 aborted fetuses aborted less than 6 days from the first treatment and in 3 full-term foal membranes. Two full-term foals exhibited mild acute inflammation in some areas of the membranes (F18, F19). The remaining foals were compromised (1 premature, 1 stillborn, and 1 dead after 2 days). Presence of bacterial colonies throughout the EEC within the allantochorion, umbilical cord, and amnion without a significant inflammatory response in these cases might suggest peracute inflammation with rapid proliferation of bacteria initially through the EEC followed by spread to the adjacent stroma, vasculature, and fetus. From 3 days after initial treatment, organisms and acute inflammation were seen histologically in aborted fetuses and euthanized foals. Thereafter, abortions had bacteria at all levels of the allantochorion, umbilical cord, and amnion, supporting the likelihood that the initial infection often originates in the EEC and suggesting that bacterial multiplication and spread is rapid after gaining entry to the tissues. Presence of bacteria in the EEC of the near- or full-term foals may reflect a variable infection time due to slower setae migration and/or a different initial inoculation site (eg, amnion) with the variable range or lack of inflammation in the membranes of these foals and the different perinatal outcomes.
Complete rapid setal migration through the allantochorion and amnion with inoculation of the amniotic fluid may explain the very acute changes within the fetal membranes of fetus No. 15 at 2 days postexposure but with only a positive culture from the amniotic fluid. Rapid setal migration may also explain the pathologic finding of advanced amnionitis with the absence or minimal involvement of the chorion in field cases of EAFL. In contrast to clinical cases, chorionitis was detected in this study after experimental exposure to caterpillar setae. It was less prevalent than amnionitis and funisitis in the examined membranes and, with the exception of one 17-day posttreatment abortion, generally acute. Five of the 6 fetuses from euthanized mares (see Table 1) had acute inflammation in the chorion. This corresponded to acute inflammation within the endometrium of their dams in which setal fragments were also found. 35 Fetus No. 14 did not have inflammation within the allantochorion; however, the uterus from its dam was not available for correlation. 35 Acute inflammation at the fetomaternal interface manifested by exudate and inflammatory cells in the intervillous spaces are a maternal response to localized or diffuse inflammation on the surface of the endometrium or chorionic villi and may be one cause of the acute chorionitis in these fetuses. 39 However, acute inflammation in the chorion could also be initiated following migration of setal fragments through the chorion, reactive to any advanced inflammation in the allantois, or due to incomplete penetration of setae into the intervillous space between the endometrium and chorion. The different manifestations of chorionitis from acute to subacute inflammation (as in 1 aborted fetus) could depend on the number of setae migrating (up to 7 were found in 1 fetus), presence and type of associated bacteria, the rate of success of bacterial or setal dispersal, and allantochorionic location of the setae migration, involvement of the mare’s endometrium, and stage of gestation.
Eosinophils were found within the chorionic stroma in the cervical pole region of 1 aborted fetus with subacute inflammation. Eosinophils are an uncommon finding in equine placentas but have been associated with pneumovagina and fungal infections in endometrial biopsies of reproductively challenged mares. 31 In humans, large numbers of eosinophils in the amniotic fluid of women in preterm labor may indicate a previous type I hypersensitivity reaction, and an eosinophilic presence in the chorioamnion may indicate myeloid depletion of the fetus. 28,29 Eosinophilic vasculitis has been found in ovine and bovine fetuses aborting from Coxiella burnetii. 42 Fungal elements and eosinophilic vasculitis were not detected in this equine placenta. Hypersensitivity reactions were noted in some mares receiving whole caterpillars via gavage, and although this mare was not noted to have any discernable reaction during treatment (A. J. Cawdell-Smith, 2009, personal communication), a hypersensitivity reaction cannot be ruled out. 10,11 Combinations of factors, including the location of eosinophilic infiltration (cervical pole area), duration of inflammation (subacute), or production of inflammatory cytokines that stimulate eosinophil chemotaxis, may all be involved. 2
Perivascular edema is one of the most common findings in acute clinical and experimental EAFL cases. 11,34 While this can be a relatively nonspecific common finding in equine placentas, it is more severe in EAFL and is generally seen both grossly and histologically in affected membranes. Perivascular edema was not a feature of the full-term control membranes or the euthanized control in these experiments. In EAFL cases, perivascular edema may reflect a change in intravascular pressure of the chorioallantoic vasculature and/or hypoxic damage to capillaries. 29 Increased vascular permeability is one of the first signs of inflammation, allowing leakage of protein-rich fluid, fibrinogen conversion to fibrin, and diapedesis or migration of leucocytes into the affected tissues. 1 The migration of neutrophils may take 12 hours to 2 days from the initiation of inflammation. 1 This lag time from infection to inflammatory response may explain the presence of perivascular edema in the allantois of the fetus euthanized at 2 days posttreatment with only rare neutrophils and a positive culture but without obvious microorganisms histologically.
Vascular lesions within the fetal membranes consisted primarily of early neutrophil margination and migration in the allantoic vasculature, larger amniotic vessels, and superficial vessels within the tunica media and adventitia of the umbilical cord. In human chorioamnionitis, bacteria and bacterial toxins were found to cause the production of proinflammatory cytokines such as interleukin-1β (IL-1β) and tumor necrosis factor–α (TNF-α) that stimulate the margination and migration of neutrophils during acute inflammation. 17,29 Endothelial cells can be directly damaged by these proinflammatory cytokines as well as by activating neutrophils, which can lead to endothelial cell apoptosis and increased vascular permeability. 13 Endothelial damage by TNF-α was suggested as the cause of the vascular luminal platelet adhesion, thrombosis, and infarction in the placenta of sheep aborting from experimental chlamydial infection. 7 A similar mechanism may be a cause of the vasculitis and infarction seen in the amnion of fetus Nos. 13 and 8 and in the umbilical cord and allantochorion of fetus No. 8. Both fetuses had Gram-positive cocci evident within the allantochorion, amnion, and umbilical cord with a positive culture. Gram-positive bacteria have been found to induce twice as much production of TNF-α by human macrophages compared with Gram-negative bacteria, and their presence may lead to more severe vascular lesions. 19
The additional findings of thymic lymphoid depletion may reflect fetal stress caused by inflammation within the fetal membranes or fetal sepsis. Chorioamnionitis in humans has been shown to elevate cortisol levels as a stress response to inflammation and induce thymic lymphoid depletion in fetuses with and without sepsis. 18,37 In these experiments, bacterial presence in the fetal membranes induced by exposure to processionary caterpillars resulted in variable inflammation with a high risk of sepsis in these fetuses and full-term foals. Fetal bacterial exposure in these experiments may have been via direct association with infected amniotic fluid or due to a bacteremia secondary to setal fragment penetration of allantoic vasculature as found in 2 fetuses.
To the best of our knowledge, the migration of caterpillar setal fragments from the gastrointestinal tract of any species to the membranes of the fetus has not been previously documented. Other foreign objects of similar shape to caterpillar setae, including grass awns, porcupine quills, toothpicks, and sewing needles, have been found in aberrant locations, including the brain, joints, urinary bladder, and uterus of animals, generally far distant from the point of entry. 5,6,14,22,24 In humans, caterpillar setae migration has been documented in the chronic syndrome of ophthalmia nodosa, an ocular manifestation of urticating hair exposure in the eye and phalangeal microgeodic syndrome in a child after playing with a processionary caterpillar, but the effects after ingestion are not known. 8,43
Conclusion
The current findings provide several new insights into the effects of caterpillar exposure via ingestion by pregnant mares:
Setae from PC can rapidly migrate through maternal and fetal membranes from 2 days after exposure onward. The likelihood of finding setae in the membranes from abortions >22 days postexposure and near- or full-term compromised foals is low.
Setal migration can potentially occur in any location where the gastrointestinal tract is in close contact to the uterus, including the areas of the cervical pole, umbilical insertion, and body. Thus, the presence of cervical gross pathology may not necessarily imply an ascending origin for infection.
Inflammation at any level of the allantochorion is possible, ranging from the intervillous space, chorionic villi, subvillous chorionic stroma, allantoic vessels, and allantoic epithelium.
Bacterial colonization of the extraembryonic coelom of the allantois either through direct inoculation by setal fragments or seeding by migrating setal fragments and subsequent spread into the umbilical cord and amnion is a likely mechanism for the bacterial abortions that characterize EAFL.
Exposure to PC may have a detrimental effect on equine fetuses and foals from early placentation to full term through acute or chronic active inflammation within the fetal membranes. 12,35
Exposure of pregnant mares to PC is emerging as a major cause of acute and chronic placentitis in Australia. There are a range of lesions associated with caterpillar setae migration, encompassing focal chronic active placentitis from early gestation exposure, acute and chronic EAFL abortion in mid-gestation, and compromise of full-term foals from late gestation exposure. 12 Awareness of these lesions allows accurate diagnosis and aids development of practices for managing pregnant broodmares to prevent or minimize the effects of caterpillar exposure. Further investigations on preventing exposure through environmental management, broodmare monitoring, and treatment of early detected placentitis are under way to minimize foal losses due to caterpillars.
Footnotes
Acknowledgement
We thank the staff of the Equine Research Unit for their technical support during the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by the Horse Program of the Rural Industries Research and Development Corporation, Canberra, Australia.
