Abstract
Chronic wasting disease (CWD) is a unique transmissible spongiform encephalopathy (TSE) of mule deer (Odocoileus hemionus), white-tailed deer (O. virginianus), and Rocky Mountain elk (Cervus elaphus nelsoni). The natural history of CWD is incompletely understood, but it differs from scrapie and bovine spongiform encephalopathy (BSE) by virtue of its occurrence in nondomestic and free-ranging species. CWD has many features in common with scrapie, including early widespread distribution of disease-associated prion protein (PrPd) in lymphoid tissues, with later involvement of central nervous system (CNS) and peripheral tissues. This distribution likely contributes to apparent efficiency of horizontal transmission and, in this, is similar to scrapie and differs from BSE. Clinical features and lesions of CWD are qualitatively similar to the other animal TSEs. Microscopically, marked spongiform lesions occur in the central nervous system (CNS) after a prolonged incubation period and variable course of clinical disease. During incubation, PrPd can be identified in tissues by antibody-based detection systems. Although CWD can be transmitted by intracerebral inoculation to cattle, sheep, and goats, ongoing studies have not demonstrated that domestic livestock are susceptible via oral exposure, the presumed natural route of exposure to TSEs. Surveillance efforts for CWD in captive and free-ranging cervids will continue in concert with similar activities for scrapie and BSE. Eradication of CWD in farmed cervids is the goal of state, federal, and industry programs, but eradication of CWD from free-ranging populations of cervids is unlikely with currently available management techniques.
Keywords
The transmissible spongiform encephalopathies (TSEs) are unusual infectious diseases of animals and humans. The TSEs, including chronic wasting disease (CWD), are designated prion diseases because of their association with aberrantly refolded isoforms of the prion protein, a normal cellular glycoprotein (PrPC). 120 CWD-associated prion protein (PrPCWD or PrPd) 82 is widespread in the lymphoid tissues and the CNS from early in the incubation phase until death. 98, 132, 137 Scrapie, the first TSE identified, was the focus of considerable research and even controversy concerning its origin and nature (genetic versus infectious) for many years. 2, 114, 115 However, it was the recognition that prion diseases were transmissible and affected humans as well as animals that stimulated much of the current scientific interest in the nature of these diseases.
Scrapie has been recognized for hundreds of years, transmissible mink encephalopathy (TME) for more than 50 years, CWD for more than 30 years, and bovine spongiform encephalopathy (BSE) for fewer than 20 years. Although CWD is now well known, it was just a few years ago considered an obscure disease of mule deer (Odocoileus hemionus) and elk (Cervus elaphus nelsoni) in a limited area of North America. The first published reports of CWD were in the early 1980s, 157, 158 but well before that time, biologists working in facilities in which this disease occurred recognized CWD as a syndrome that limited the lifespan of the captive mule deer they were studying. It was years before it was determined that this wasting syndrome of deer was contagious. Since the BSE epidemic, followed by recognition of the associated variant Creutz-feldt-Jacob disease (vCJD) of people, there has been much greater scientific and public attention paid to TSEs in general and concordant increased interest in understanding CWD. This has resulted in studies of CWD that now span the spectrum from pathogenesis to spatial epidemiology.
CWD occurs among two very different populations of cervids: deer and elk that are raised and managed in much the same way as other ruminants in agricultural systems and free-ranging deer and elk populations that are under much less human control. Understanding differences in epidemiology and management strategies that can be employed to control or eradicate CWD for these two populations of animals is critical. In this review, I will survey the rapidly accumulating knowledge about CWD with a focus on information most relevant for veterinary pathologists.
Clinical and Epidemiologic Features
Descriptions of clinical CWD are based primarily on observations of captive animals; the progression of CWD in free-ranging animals is less well known because of the inherent difficulties in their study. Features of CWD in terminal clinical stages are readily appreciated, even by untrained observers and have been described. 155, 159 Qualitatively, they are similar to sheep with terminal scrapie 40 or cattle with BSE, 151 although disruptions and alterations of locomotion can be more subtle in CWD. The salient clinical features of progressive CWD disease in adults is weight loss and behavioral changes that typically span weeks or months. In addition to these general nonspecific features, signs that might only be present in some cases include odontoprisis; sialorrhea, or excess salivation because of difficulty swallowing; ataxia and head tremors; esophageal dilation and regurgitation; and aspiration pneumonia. Terminal physiologic and behavioral alterations can include polydipsia and polyuria; syncope; periods of lack of awareness; fixed stare; changes in interaction with herdmates and handlers (decreased or increased flight distances); altered stance, often with lowered head; repetitive walking of perimeters of enclosures; and hyperexcitability when handled. In general, the signs of terminal CWD are more subtle in elk than in deer. Elk more often have disturbances in locomotion and less often display polydipsia than deer.
Pruritus with hair loss, commonly observed in terminal sheep scrapie, is not a feature of advanced CWD. However, the hair coat of affected animals can be rough and dry, 158 with patchy retention of the winter hair coat in summer. 10, 159 This appears to be a reflection of poor body condition rather than a primary effect of the disease.
No specific clinical diagnostic feature of CWD appears during the early to midphase of disease. Behavioral changes can be subtle and fall within the normal repertoire of cervids. Similarly, normal seasonal changes in body mass occur in free-ranging and captive cervids; thus, evaluation of body condition must take into account time of year, nutritional or habitat quality, and reproductive status.
Polydipsia and consequent polyuria in terminal cases of CWD are probably associated with damage to the supraoptic and paraventricular nuclei and diabetes insipidus. 157 With the exception of low urine specific gravity, found in terminally affected cervids with free access to water, 157, 158 clinical chemistry and hematology are not useful in diagnosis of CWD. Alterations from normal cervid values reflect nonspecific changes from poor body condition, aspiration pneumonia, or another intercurrent disease process. Specific physiologic studies have not been conducted on CWD-affected cervids.
Deer with subclinical or early clinical CWD are susceptible to sudden death after handling 100 and are more likely to die following immobilization or misadventure than unaffected deer 155 (L. Wolfe et al., personal communication; W. Cook et al., personal communication). Although the mechanism of this posthandling mortality has not been identified, functional lesions in the parasympathetic innervation of the heart have been identified in BSE; 7 similar processes might occur in CWD, accounting for acute mortality following significant stress.
Aspiration pneumonia can occur early or late in the clinical course of CWD 155, 159 and could be responsible for apparent rapid death, even in the absence of other signs of CWD. Thus, CWD should be considered in any adult cervid with aspiration pneumonia. 156 Aspiration pneumonia presumably follows loss of effective motor control over swallowing associated with regurgitation or rumination due to central damage. 159
Subclinical or clinically apparent changes in behavior could increase the likelihood of an affected free-ranging deer or elk being harvested during the fall hunt, 35 succumbing to collisions with vehicles, 90, 100 or being killed by predators 90 (M. Miller, personal communication).
Duration of clinical disease is extremely variable and in part might reflect difficulty in determining the onset of clinically apparent CWD. Astuteness in detection of subtle clinical signs might require considerable familiarity with the individual animal; this is not possible in many captive herds and is essentially impossible in free-ranging cervids. Sudden death might occur rarely in captive deer, 100 and short duration of disease (days) is occasionally observed in elk (W. Schultz and T. Kreeger, personal communication). More often, a slowly progressive clinical course of “ain't doing right” is noted over weeks or months. Death typically occurs within 4 months, although a few animals survive as long as a year. 155 Death of clinically affected animals might follow environmental stresses, such as periods of extreme cold. Because of a compromised ability to forage, find water, and possibly avoid predators, clinical duration is likely to be shorter in free-ranging cervids compared with cervids maintained in captivity.
Because of prolonged incubation periods, which in the natural disease is a minimum of approximately 16 months, fawns do not develop clinical CWD, although evidence of infection can be detected by a variety of methods through much of the incubation period. 111, 132 Under experimental conditions, disease-associated prion protein (PrPd) was found in lymphoid tissues of the alimentary tract in mule deer by 42 days postoral exposure. 132 Only rarely are yearlings clinically affected. Maximum incubation periods of naturally exposed free-ranging animals are difficult to determine because it is impossible to determine exactly when an animal is exposed, but average incubation periods probably range from 2 to 4 years. However, CWD has been diagnosed in an elk >15 years of age 155, a mule deer >12 years of age (unpublished data), and in white-tailed deer of >5 years of age 111 residing in CWD endemic facilities with high prevalence of the disease. These cases might represent animals exposed to the CWD agent as adults or could indicate prolonged incubation periods. The influence of dose of agent on duration of incubation is not clear in CWD, but preliminary observations of experimentally and naturally exposed captive elk suggest that, to a degree, higher doses result in shortened incubation periods (E. S. Williams, unpublished data; T. Kreeger, personal communication). Incubation periods in rodents exposed to scrapie 48, 88, 121 are inversely related to dose of TSE agent, and a similar trend appears to be present in cattle orally exposed to BSE agent (G. Wells, personal communication).
Seasonality is not a significant feature of CWD at the individual level because animals can develop clinical CWD at any time of the year. However, on a population level, affected animals are more commonly reported in the fall, perhaps because of an observational bias, or in the winter, possibly reflecting increased environmental stress.
Epidemiologic features should be considered when investigating possible cases of CWD. In farmed herds naturally affected by CWD, rarely will more than one animal be affected at a time, 87, 117, 134 although there is often herd history of ill thrift in adult animals. Deer and elk from captive herds that have a history of frequent purchases from unmonitored sources or from herds in known CWD endemic areas (Fig. 1) are at greater risk for CWD than animals from closed and monitored herds and from herds outside CWD endemic areas.
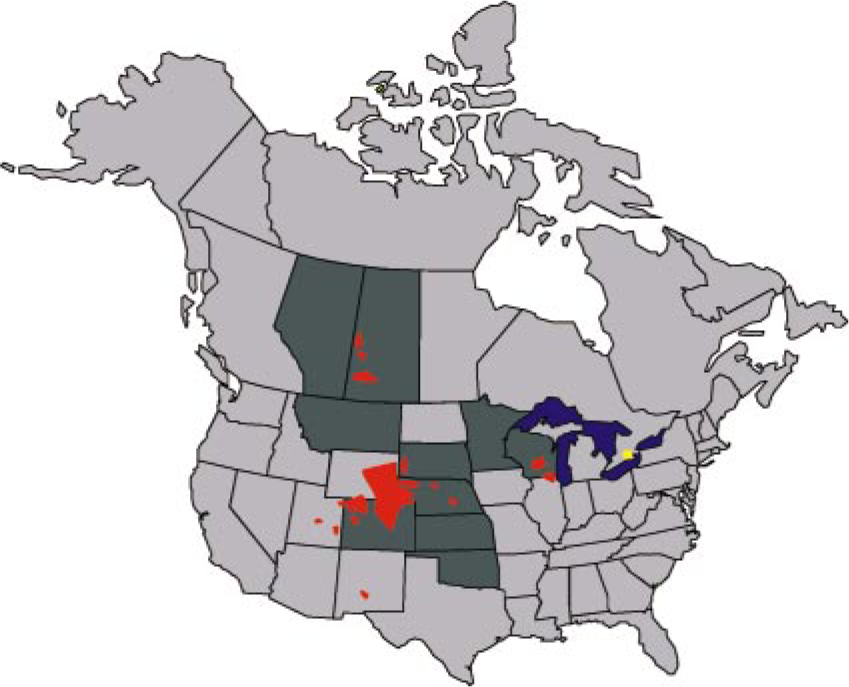
Known historic distribution of chronic wasting disease in farmed and free-ranging cervids in North America, 1977–2004. Dark gray jurisdictions indicate where CWD has been identified in farmed cervids; to date all but a few identified farmed cervid herds known to have had CWD have been depopulated. Red foci are areas where CWD has been diagnosed in free-ranging cervids. The yellow focus indicates occurrence of CWD in mule deer in a zoological garden. In March–May 2005, the New York State Department of Agriculture and Markets announced five positive cases of chronic wasting disease in captive white-tailed deer from two closely connected herds in central New York (Oneida County) and two positive cases in wild white-tailed deer from the same area. 104 .
Within CWD endemic areas, the vast majority of CWD cases in free-ranging deer and elk detected in the course of surveillance activities are subclinical (>97%). 98 Clinically affected free-ranging cervids are relatively rare, and even within endemic areas only about a third to half of clinical suspects examined in diagnostic laboratories have CWD (E. S. Williams, unpublished data). However, surveillance programs in free-ranging cervids that target deer and elk that show clinical signs compatible with CWD have been successful at identifying CWD in a more efficient and more cost effective manner than widespread hunter-harvested animal surveillance. 100, 103, 156
Prevalence of clinical or subclinical CWD infection as detected by immunohistochemistry (IHC) of lymphoid tissue or brain in captive herds varies considerably from <1% in some farmed herds with recent introduction of the disease to essentially 100% in CWD endemic research facilities. 111, 117, 157 Likewise, prevalence varies widely in free-ranging populations from <1% in deer and elk to ∼30% in some local populations of deer 103 (W. Cook, personal communication; Wyoming Game and Fish Department and Colorado Division of Wildlife, unpublished data). Prevalence of CWD in elk is lower than in sympatric deer. 101
Gross Pathology
As in the other animal TSEs, individual gross lesions of CWD are nonspecific, but in combination, they may be helpful in diagnosis of CWD. Lesions that might be present reflect the clinical signs: rough, dry hair coat that might not have been appropriately shed and megaesophagus. It is important to realize that body condition of animals in subclinical or early clinical stages of CWD can be normal; good body condition does not rule out CWD. In the terminal stages of disease, typically the animals are emaciated; might or might not have aspiration pneumonia; often have watery rumen contents, which can be frothy or contain increased amounts of sand and gravel; and have dilute urine if water was freely available. 155, 159 Carcasses of free-ranging deer and elk might be dehydrated, presumably due to a lack of ready access to water. Other potential causes of emaciation and pneumonia should be ruled out during the course of postmortem examination of cervids suspected of having CWD.
Histopathology and Electron Microscopy
The histopathology of CWD in clinically affected animals is typical of the ruminant TSEs; 53, 138, 153, 160 specific histologic lesions are only observed in gray matter of the CNS. Qualitatively, lesions observed in brains of natural and experimental (intracerebral or oral routes of exposure) cases of CWD in natural hosts are the same; duration of clinical disease does not significantly affect lesion distribution or severity, 160 and lesions of CWD in free-ranging and captive mule deer are similar. 137 Some variation in severity can occur among individuals but the anatomic distribution of lesions is quite constant. CNS lesions are bilaterally symmetrical and spongiform change is obvious; vacuolization occurs in neuronal perikarya and neuronal processes. Neuronal degeneration and astrocytic hyperplasia and hypertrophy occur 53, 160 but are not prominent features. As with the other TSEs, inflammatory cell response is not apparent unless associated with intercurrent disease.
Microscopic lesions are most striking in the dien-cephalon, olfactory cortex, and nuclei of the medulla oblongata, prominently the dorsal vagal nucleus, although milder lesions are widespread in brain and spinal cord, with relative sparing of the basal ganglia, cerebral cortex, and hippocampus. 137, 160 In clinically affected cervids, examination of well-fixed medulla oblongata at the level of the obex is considered sufficient for diagnosis of CWD, 160 and sections at this level were used for CWD surveillance before availability of immunohistochemistry.
Amyloid plaques are relatively common and can be detected on hematoxylin and eosin (HE)–stained brain sections, most prominently and with decreasing frequency, in white-tailed deer, mule deer, and elk (E. S. Williams, unpublished data). These appear as slightly pale fibrillar eosinophilic areas of neuropil in HE preparations (Fig. 2a) and can be surrounded by vacuoles (“florid plaques”). 94, 153, 160 These are more easily detected with silver stains, 160 with Congo red staining, 8 or, most dramatically, by immunohistochemistry 51, 52, 94, 95 for detection of PrPd (Fig. 2b). Amyloid plaques have been reported in a variety of other TSEs, including kuru, 56 sporadic CJD, 13 murine scrapie, 29 BSE in macaques (Macaca fascicularis), 93 and rarely in BSE and scrapie, 14, 53, 82, 165 and they are a salient lesion of vCJD in humans. 152 Thus, the presence of amyloid plaques is not reflective of a particular strain of agent, but rather, it presumably reflects the interplay between host and pathogen.
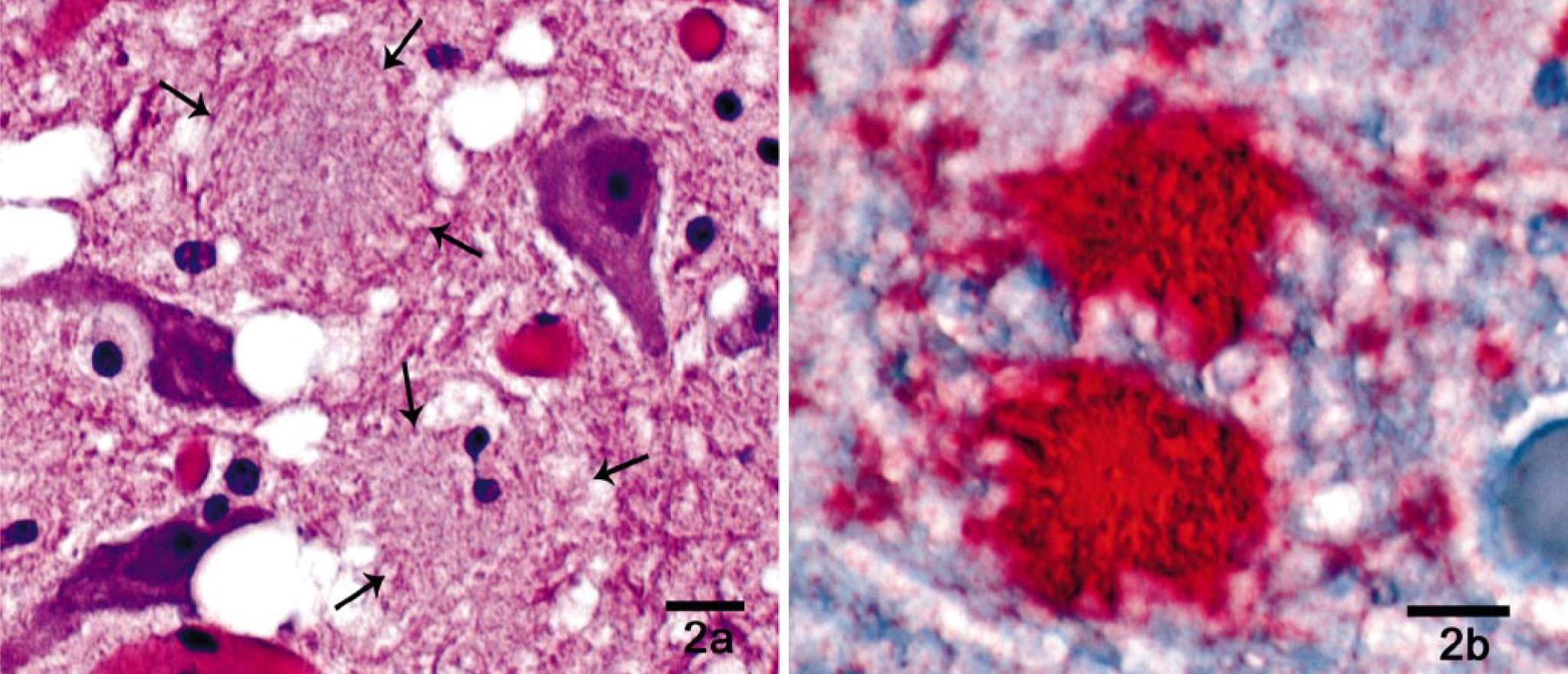
Thalamus; white-tailed deer with chronic wasting disease. Amyloid plaques.
Ultrastructurally, the lesions of CWD are similar to the other TSEs. 49, 50, 95 Membrane-bound vacuoles, some containing secondary vacuoles and curled membrane fractions, are present in neurons and neuronal processes. Dystrophic neurites can contain degenerating mitochondria, pleomorphic membrane-bound structures, and electron-dense bodies.
Usefulness of IHC in diagnosis of TSEs, for study of natural and experimental pathogenesis of these diseases and for strain typing, is well established 51, 65, 82, 97, 146, 149 and offers a sensitive method for detection of PrPd while maintaining structural context. Obtaining brains from free-ranging and extrinsically managed captive cervids free of significant autolytic change can be difficult which often precludes use of histopathology alone for diagnosis of CWD. In addition, histopathology is only sensitive for diagnosis of clinically affected cervids; it is insensitive in subclinical CWD 98 because spongiform encephalopathy occurs at about the same time clinical signs develop (E. S. Williams, unpublished data).
Patterns of PrPd deposition in CWD-affected cervid brains include perineuronal accumulation, extracellular plaques and granular deposits, perivascular accumulation, and subependymal and subpial deposition. 52, 94, 137 In clinical CWD, widespread staining of PrPd occurs throughout the brain in susceptible species (Table 1). 136, 138, 153 Typically, correlation is good between deposits of PrPd and regions of spongiform change (Fig. 3a, b); 136 however, PrPd can be found in areas of the brain with no spongiform change. Spraker et al. 137 exhaustively documented deposition of PrPd in mule deer brain in various stages of CWD infection.
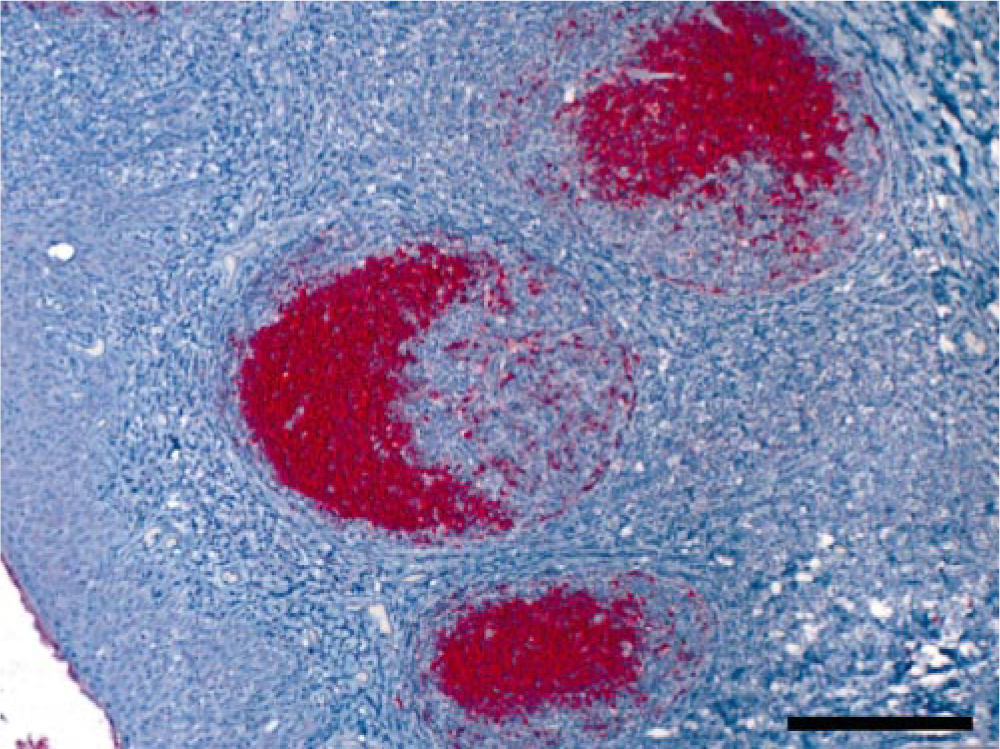
Tonsil; mule deer with chronic wasting disease. Formalin-fixed, paraffin-embedded tissue. Immunohistochemical stain demonstrating accumulation of disease associated prion protein in germinal centers. Bar = 100 μm.
Published reports of tissue distribution of histologic lesions, PrPd, or infectivity detected in cervids with CWD by histopathology, immunohistochemistry, enzyme-linked immunosorbent assay, western blot immunoassay, or bioassay.

∗ IHC = immunohistochemistry; WB = western immunoblot; ELISA = enzyme-linked immunosorbent assay.
Deposition of PrPd occurs widely in lymphoid tissues during CWD incubation in the absence of histologic lesions in these tissues. 112, 132, 153 In this way, CWD closely resembles scrapie. 4, 68, 69, 146, 149 Lymphoid tissues therefore are very useful for diagnostic purposes and surveillance. Only rarely have deer been found to have brain but no lymphoid PrPd deposition 136 (<1%; E. S. Williams, unpublished data); this occurs more frequently in elk (∼10–15%; 134 E. S. Williams, unpublished data). Thus, use of lymphoid tissues alone for surveillance of CWD in elk could miss a significant number of infected animals 134 but is useful in surveys designed to detect CWD-infected cervid populations. 71
Specific granular PrPd labeling can be present in the light and dark zones of germinal centers (Fig. 4), but staining is less often identified in the follicular mantle. In studies of tonsils of mule deer early and late in incubation by triple label immunofluorescence and confocal microscopy, PrPd was detected primarily in extracellular locations in association with follicular dendritic and B-cell membranes. 130 Aggregates of PrPd also were found in tingible body macrophages within follicular germinal centers.
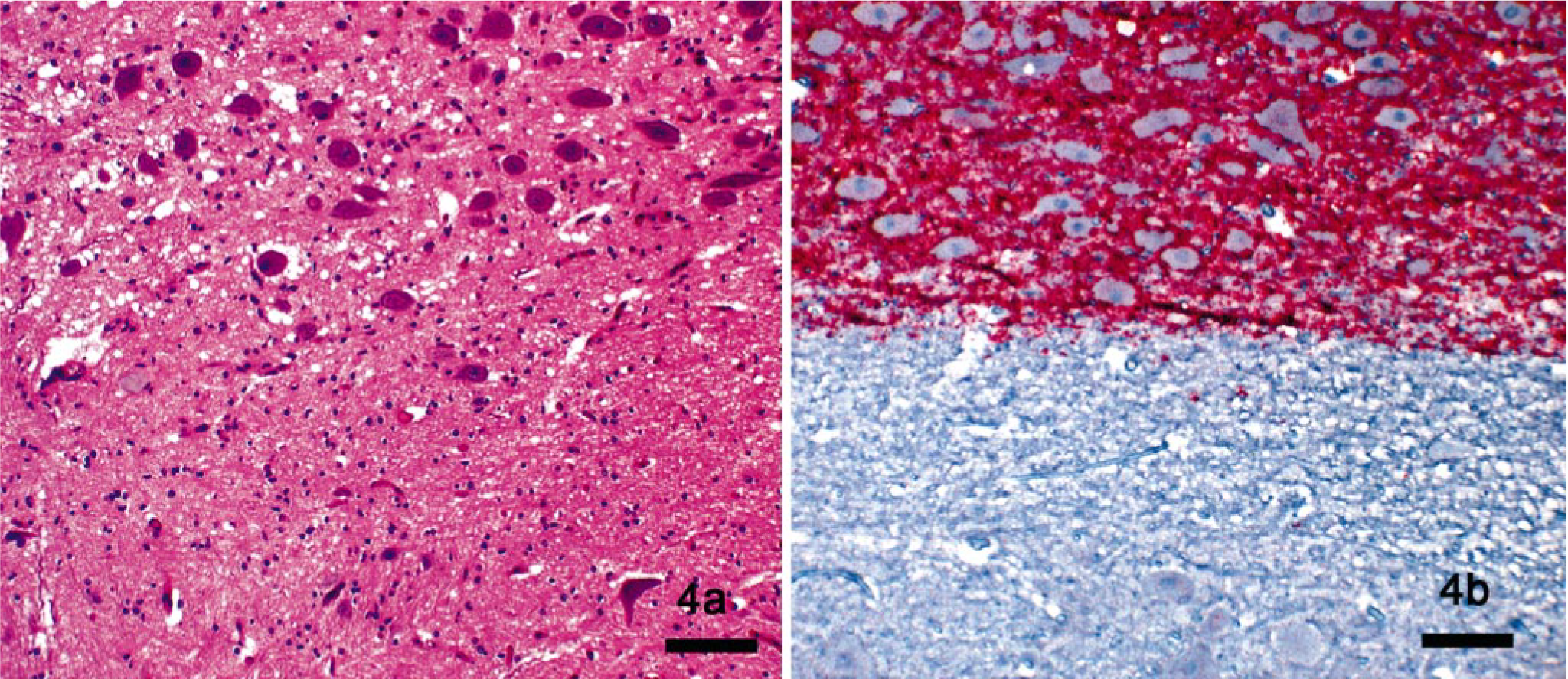
Dorsal vagal nucleus and hypoglossal nucleus in obex region of the medulla oblongata; mule deer with chronic wasting disease.
Disease-specific prion protein is detected by IHC in a variety of tissues of subclinically and clinically affected cervids 131, 137 (E. S. Williams, M. Miller, K. Fox, and J. Jewell, unpublished data; Table 1). In peripheral tissues other than lymphoid tissues, PrPd deposition appears as scattered coarse granules to relatively diffuse staining. Coarsely granular staining can be found in and around myenteric and submucosal plexus neurons, around nerve fibers, and in satellite cells. 131 Staining occurs in islets of Langerhans in the pancreas, in the adrenal medulla, and in the pars nervosa and intermedia of the pituitary gland 131 (E. S. Williams, unpublished data).
Recent studies in laboratory rodent models of scrapie demonstrated PrPd in skeletal muscles 22, 103, 141, 142, and two studies found infectivity for rodents in muscle. 22, 142 PrPd has also been demonstrated in skeletal muscles of humans with CJD 45 and of sheep with scrapie, 6 suggesting skeletal muscles as a source of infectivity for TSEs. No evidence of PrPd was found in sections of skeletal muscle from deer with natural 138 and experimental 60 CWD by IHC, but additional studies are warranted and are underway.
Classification systems for the stages of CWD infection in naturally affected mule deer and elk based on histologic lesions and deposition of PrPd have been proposed 98, 134, 137 . The essence of these systems is that, during the course of incubation, PrPd is typically detected first in lymphoid tissues followed by deposition in the lateral aspect of the dorsal vagal nucleus in the medulla oblongata in the absence of spongiform change. Spongiform change then occurs in the vagal nucleus, probably at about the onset of clinical disease, followed by more widespread deposition of PrPd and spongiform change in the brain. Variations in this pattern occur in individual animals. The usefulness for management purposes of such stage description systems has yet to be shown, but they have been used in attempting to evaluate the influence of genetics on pathogenesis. 111
Diagnostic Techniques
The search for simple and reliable antemortem tests for TSE agents continues. Biopsy of the lymphoid tissue of the third eyelid 105, 107, 109 or the tonsil 129, 146 and examination of the tissue by IHC for PrPd have proven useful in diagnosis of scrapie in live sheep. Tonsil biopsy of live mule deer and white-tailed deer for CWD testing has been useful in a research context 112, 150, 163 (W. Cook, D. Edmonds, and E. S. Williams, unpublished data; N. Mathews, personal communication) and, more recently, in evaluation of management strategies (test and removal) for a local population of free-ranging mule deer in Colorado. 164 Tonsil biopsy is not practical on a large scale because it requires anesthesia of the animal before sampling. Sampling lymphoid tissue from the nictitating membrane of deer and elk has not proven to be as useful as in sheep (K. O'Rourke, personal communication; L. Wolfe, personal communication).
Immunohistochemistry is considered the “gold standard” for diagnosis of CWD. 112, 145 Protocols for IHC employ various techniques for antigen retrieval and a variety of antibodies (Table 2) and chromagens. 106, 130, 136 For regulatory purposes, standard IHC protocols are used in US Department of Agriculture–approved laboratories and include hydrated autoclaving, formic acid treatment of sections, specified antibody, and automated staining of retropharyngeal lymph node samples from deer and retropharyngeal lymph node or obex samples, or both, from elk and deer. Retropharyngeal lymph nodes are used in deer because of early lymphoid deposition of PrPd at this location, making testing more sensitive than if brain alone was used. 71, 98 For maximum sensitivity, multiple sections of lymph node should be examined because of patchy PrPd deposition in early infection. 71 Retropharyngeal lymph node is positive in the majority of elk with CWD infection; however, 10–15% of elk have detectable PrPd in brain but not retropharyngeal lymph node 134 (E. S. Williams, J. Kreeger, and H. Edwards, unpublished data), leading to recommendations to test both lymph node and obex samples in elk. Rapid screening tests for the TSEs, including CWD, are in increasingly wide usage. Five rapid tests have been licensed by the US Department of Agriculture, Center for Veterinary Biologics, for CWD testing (enzyme-linked immunosorbent assays [ELISAs]: Bio-Rad Chronic Wasting Disease Test, BioRad Laboratories, Hercules, CA; HerdCheck CWD Antigen EIA Test, IDEXX Laboratories, Westbrook, ME; Enfer TSE Test, Abbott Laboratories, Abbott Park, IL; and CWD Dot Blot ELISA Test, VMRD Inc., Pullman, WA; and a strip test: PDL CWD Rapid Antigen Test, Prion Development Laboratory, Buffalo Grove, IL). Sensitivity of IHC compared with these rapid detection methods depends on the techniques and tissues used, and there are few controlled direct comparisons. In a large study of hunter-harvested cervids in Colorado and with IHC results as the gold standard, one ELISA (Bio-Rad) was 98.3% and 100% sensitive on retropharyngeal lymph nodes from mule deer and elk, respectively. 71 Specificity was essentially 100% for both species.
Antibodies and epitopes used for diagnosis of CWD by immunohistochemistry or immunoblotting.
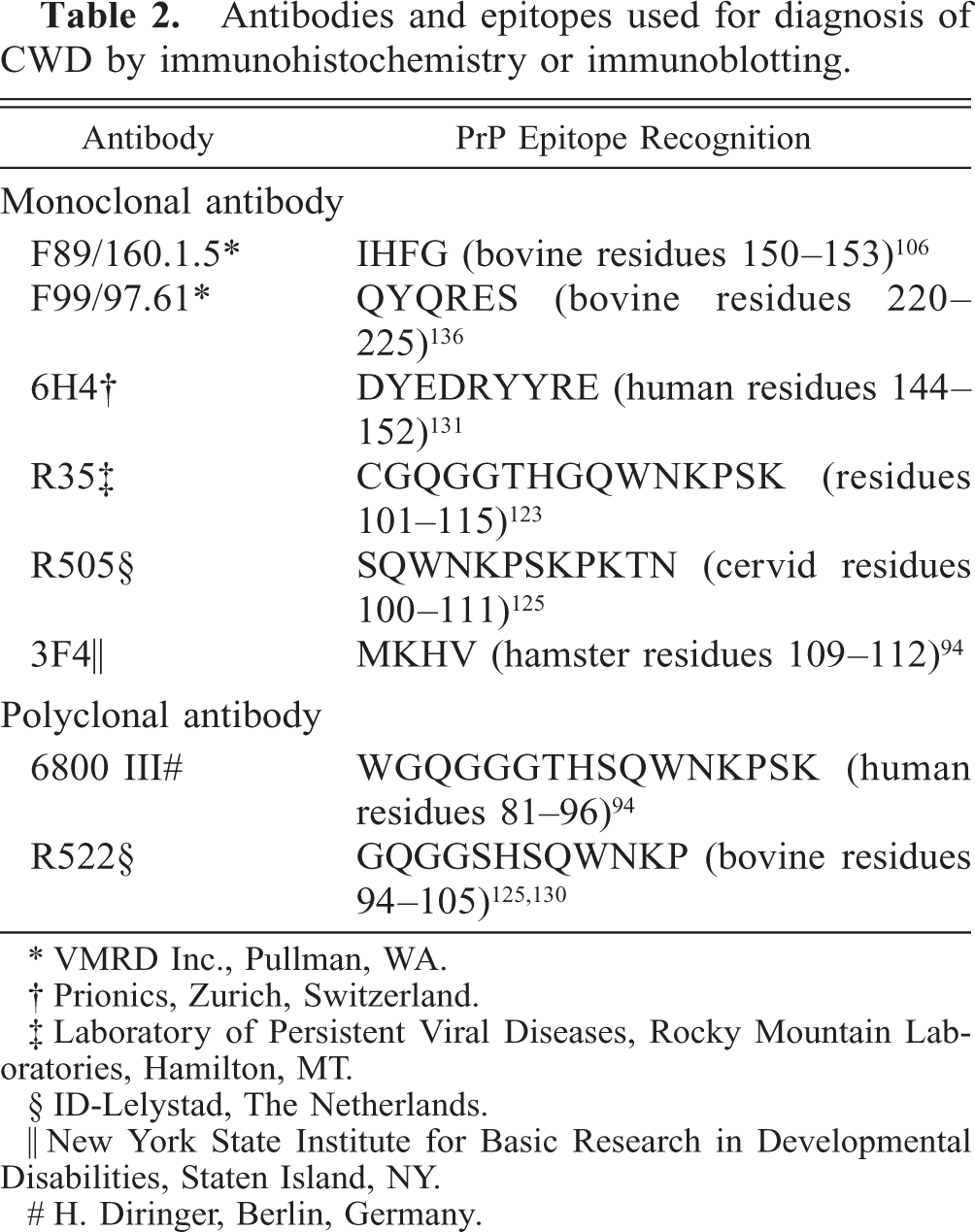
∗ VMRD Inc., Pullman, WA.
† Prionics, Zurich, Switzerland.
‡ Laboratory of Persistent Viral Diseases, Rocky Mountain Laboratories, Hamilton, MT.
§ ID-Lelystad, The Netherlands.
‖ New York State Institute for Basic Research in Developmental Disabilities, Staten Island, NY.
# H. Diringer, Berlin, Germany.
Differential Diagnoses
Describing the full range of differential diagnoses for CWD is beyond the scope of this review, but many diseases should be considered when attempting to determine cause of illness or death of cervids with signs compatible with CWD. As for other infectious diseases, epidemiologic aspects, as well as clinical and pathologic findings, should be brought to bear in determining the cause of cervid illness and mortality. Important infectious diseases of cervids have been reviewed. 154
In much of North America, hemorrhagic disease (epizootic hemorrhagic disease and bluetongue) can cause clinical signs compatible with CWD. Occasionally, encephalitis can occur in white-tailed deer and elk with acute hemorrhagic disease, leading to clinical neurologic manifestations that could slightly resemble signs of CWD. Chronic hemorrhagic disease can result in emaciation of white-tailed deer and, rarely, mule deer, and affected animals are occasionally observed in late fall or winter. The lesions and epidemiology of epizootic hemorrhagic disease and bluetongue in cer-vids have been reviewed 74 ; histopathology, IHC, virus isolation, and serology can be used to distinguish these diseases.
In eastern North America, meningeal worm (Parelaphostrongylus tenuis) infection is relatively common in populations of white-tailed deer, the reservoir host; although uncommon, CNS disease occasionally occurs in this species. Mule deer and elk can be seriously affected. The nematodes migrate through spinal cord and brain, resulting in local traumatic and inflammatory damage. The lesions and epidemiology of disease caused by this parasite have been reviewed. 91 Meningeal worm only occurs in a few areas in which free-ranging populations of elk and mule deer exist, but farmed elk and mule deer are at risk of infection in areas in which this parasite is present.
Loss of condition and behavioral changes occur in elk with locoweed intoxication, 162 and these signs can resemble CWD. The microscopic lesions of locoism in the CNS 1 are easily differentiated from CWD. Naturally occurring locoism has not been described in deer.
Pathogenesis
Studies of the pathogenesis of scrapie in natural hosts and in laboratory rodents have proven to be good models for investigating CWD. Before the availability of immunoassays for tracking the presence of PrPd, bioassay of tissues for scrapie agent was by intracerebral inoculation of mice, 55, 89 an expensive and time-consuming method. More recently, elegant studies on scrapie agent by IHC have tracked accumulation of PrPd throughout the body during incubation. 4, 15–17, 67–70, 146–149 Scrapie agent in naturally and orally exposed animals enters via the alimentary tract; accumulation of PrPd occurs in gut-associated lymphoid tissue—in particular, in germinal centers innervated by sympathetic fibers and in myenteric and submucosal plexuses—and moves to the CNS via sympathetic and parasympathetic nerves to the intermediolateral columns of the spinal cord and the parasympathetic vagal nucleus (dorsal nucleus of the vagus nerve) in the obex region of the medulla oblongata. From there, PrPd can be found in increasing amounts and widening distribution in lymphoid tissues and CNS. A similar pattern appears to occur in deer with CWD, with involvement first of tonsil and gut-associated lymphoid tissues, presence of PrPd in the enteric nervous system, followed by initial involvement of the CNS at the vagal nucleus and thoracic spinal cord and concurrent distribution in peripheral lymphoid tissues 98, 131, 132, 137 (Williams et al., unpublished data). More detailed studies of CWD pathogenesis in all three naturally susceptible species are underway.
Recent studies have detected prion infectivity in blood of sheep with scrapie and BSE 73, 75 and has been suspected in humans with vCJD. 96, 116 Occurrence of the agent in blood might account for the widespread distribution of PrPd that occurs in lymphoid tissues in scrapie and possibly in CWD.
Genetics
The influence of genetics on scrapie susceptibility, resistance, and incubation period has been recognized, investigated, and debated for decades. 2, 14, 76–80, 110, 114, 115 At least 10 amino acid polymorphisms in the protein encoding region of the sheep PrP gene have been reported, 23 and alleles at codons 136 and 171 strongly influence susceptibility and resistance. Now determination of PrP genotype and selection for resistant genotypes form an important aspect of programs to control this disease in North America (http://www.aphis.usda.gov/vs/nahps/scrapie/) and in some countries in Europe (http://www.defra.gov.uk/animalh/bse/othertses/scrapie/nsp/index.htmlh). 39
Investigations of possible genetic influences on CWD susceptibility are less extensive but ongoing (Table 3). The first such studies reported nucleotide sequences of the PrP gene and deduced amino acid sequences and compared them to those of domestic livestock or humans. 33, 66, 128 O'Rourke et al. 108 determined that elk with methionine homozygosity at codon 132 (132MM) were overrepresented among elk with CWD compared with elk heterozygous for methionine and the minor allele, leucine (132ML) or homozygous for leucine (132LL) at that locus. Although naturally acquired CWD has been diagnosed in elk with these three genotypes 132 (J. Jewell, personal communication), evidence indicates that incubation period might be lengthened in elk with 132ML or 132LL genotypes (T. Kreeger et al., personal communication; E. S. Williams, unpublished data). If these observations are confirmed and expanded, some degree of genetic manipulation of farmed elk herds might be useful in management of CWD.
Substitution polymorphisms in the protein coding region of the prion gene (Pnrp) in deer and elk.
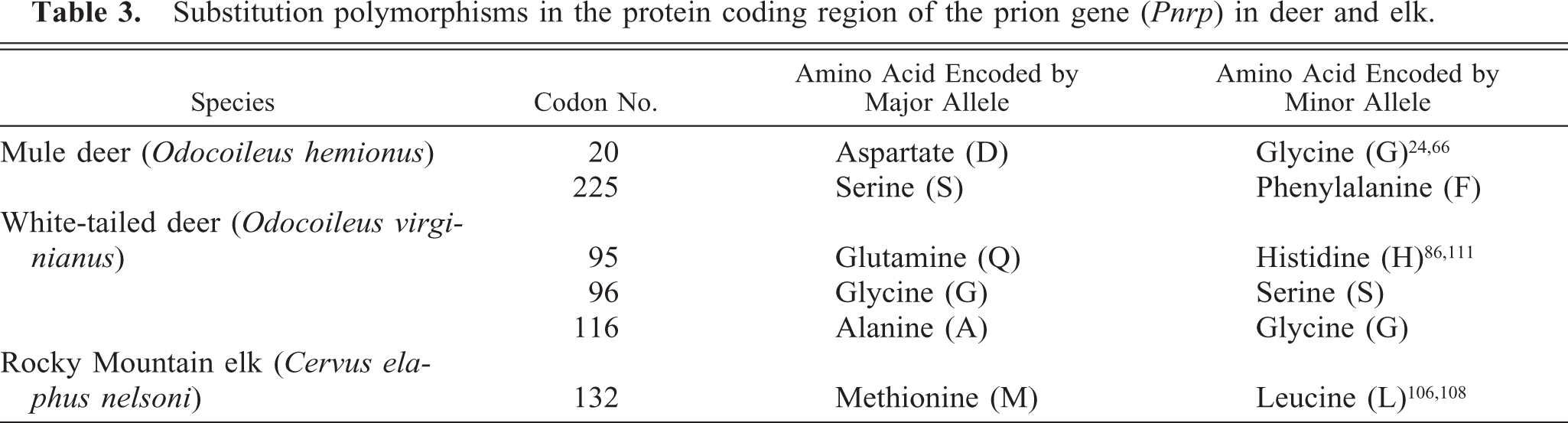
Experimental studies of elk exposed to scrapie agent by intracerebral inoculation resulted in somewhat longer incubation periods in elk of the 132ML genotype compared with elk homozygous for methionine. 62 However, influence of genotype on brain and lymphoid tissue distribution of PrPd was not detected in elk. 132
Three substitution polymorphisms occur in white-tailed deer PrP (Table 3) and two in mule deer, of which one (codon 20) is removed during processing and one (codon 225) remains in the mature polypeptide. The existence of a processed pseudogene of high sequence identity to the functional PrP gene in all mule deer and both black-tailed deer subspecies (O. hemionus columbianus and O. hemionus sitkensis) 24, 85 (J. Jewell, personal communication) and about 25% of white-tailed deer 111 (J. Jewell, personal communication) complicated initial sequence studies of the deer prion protein. Thus, some deduced amino acid sequences reported before recognition of the pseudogene list a polymorphism at codon 138 (serine/asparagine); only serine is encoded at that locus by the functional deer PrP gene. In one population of white-tailed deer with high prevalence of CWD, presence of the pseudogene did not appear to influence presence or absence of disease. 111 Pseudogenes are rarely expressed, but functional pseudogenes have been described, 9 warranting more investigation into the role that pseudogenes might play in CWD.
Results to date indicate that deer of all common PrP genotypes are susceptible to CWD. CWD was diagnosed in all major genotypes of white-tailed deer studied in Wisconsin and Nebraska; 86, 111 however, fewer deer homozygous for serine at codon 96 were found with CWD than statistically expected. Captive mule deer heterozygous for serine/phenylalanine at codon 225 (225SF) are susceptible to experimental oral exposure but have prolonged CWD incubation periods compared with homozygous (225SS) deer. 84 A study of free-ranging mule deer in the CWD endemic areas of Wyoming and Colorado found that the 225SF genotype is less frequent among CWD-positive animals than in the overall population 84 ; thus, 296 positive animals included one SF and 295 SS genotypes, whereas 22 SF samples were expected statistically. In one Colorado mule deer herd sampled extensively, about 18% were 225SF. However, only 4% of the CWD-infected mule deer from the population that included this herd were of the SF genotype (J. Jewell, personal communication; M. Miller, personal communication).
Thus, in all three natural cervid hosts, the influence of genetics on CWD incubation periods and pathogenesis requires further understanding. Although no cer-vid genotype may provide complete resistance to CWD, if incubation periods and pathogenesis of CWD are significantly affected by genotype, as occurs in sheep scrapie, this might influence CWD epidemiology and have implications for development of management strategies for this disease.
Host Range
The natural and experimental host ranges of CWD are shown in Table 4. Only three species—mule deer, white-tailed deer, and elk—are known to be naturally susceptible to CWD. Black-tailed deer (O. hemionus columbianus), a subspecies of mule deer, are susceptible to CWD. 157 It is likely, because of close taxonomic relationships, that all subspecies of white-tailed deer, mule deer, and elk (which includes red deer, C. elaphus elaphus) would be susceptible to CWD if appropriately exposed.
Natural and experimental host range for the transmissible spongiform encephalopathies of cervids.

Because of concerns that CWD could spread to cattle on ranges shared by deer and elk, collaborative studies were begun in 1997 to evaluate susceptibility of cattle. Five of 13 (38%) mixed breed beef cattle intracerebrally inoculated with a pool of brains from mule deer with CWD developed evidence of disease transmission approximately 2–5 years postinoculation. 58, 59 Microscopically, spongiform encephalopathy was equivocal, but PrPd was identified by IHC in many parts of the brain except the cerebellum PrPd was found by immunoblotting, and scrapie-associated fibrils were detected by negative stain electron microscopy. Deposits of PrPd were multifocal and centered around or in astrocytes; neurons were seldom involved. Occasional small plaques occurred in the neuropil. The lymphoid tissues examined were negative for PrPd staining. No evidence of transmission was found in the remaining intracerebrally inoculated cattle killed 7 years postinoculation. In contrast, transmission of scrapie occurred in nine of nine (100%) cattle inoculated intracerebrally and maintained for more than 1 year. 37, 38 Lesions of scrapie in cattle in this study were subtle, spongiform change was minimal, and PrPd staining was within neurons. In another study of scrapie transmission to cattle, three of 10 (30%) animals inoculated with scrapie agent had incubation periods between 2 and 4 years. The lesions in these affected animals also were very subtle. 126 No transmission of scrapie to cattle occurred when they were exposed orally to the scrapie agent in rendered feed, 36 and there is no epidemiologic evidence that scrapie can be horizontally transmitted to cattle. These observations suggest that if CWD is similar to scrapie, it is unlikely that cattle will contract CWD by natural routes, even though a small percentage might be susceptible by intracerebral inoculation. Cattle orally exposed to a high dose of brain from mule deer with CWD and cattle living in CWD endemic facilities remain clinically healthy more than 7 years postinoculation or -exposure (Williams et al., unpublished data). In addition, a survey of 262 adult culled cattle that resided in CWD endemic areas for at least 4 years was negative for evidence of a TSE. 47
Domestic sheep are susceptible to CWD agent by intracerebral inoculation; Suffolk sheep of a genotype highly susceptible to scrapie (homozygous for glutamine [Q] at codon 171 [171QQ]) developed a disease indistinguishable from scrapie following inoculation (A. Hamir, personal communication). Susceptibility of domestic sheep to CWD agent by oral exposure has not been studied. One domestic goat developed CWD approximately 6 years after intracerebral inoculation of deer CWD brain. 159 Clinical signs included intense pruritis and loss of body condition. Microscopically, widespread spongiform encephalopathy resembled that of scrapie.
Currently, no widely available laboratory animal model exists for the study of CWD. Mink and ferrets are susceptible to CWD by intracerebral inoculation but have only been used in a limited number of CWD studies 11, 92 (R. Marsh, personal communication; C. Sigurdson et al., personal communication; E. S. Williams, unpublished data). Conventional wild-type laboratory mice and hamsters are poor hosts for CWD agent; it transmits with low efficiency on first passage in mice 27 and only to hamsters following passage in domestic ferrets. 11 Lack of a suitable laboratory model for CWD has hampered study of some aspects of CWD biology. However, transgenic mice expressing normal cervid cellular prion protein (PrPc) are being developed and validated by several laboratories 26 (M. Oldstone, personal communication; R. Rubenstein, personal communication) and availability of these animals in the future will be very useful in the study of CWD.
Based on epidemiologic investigations, there is currently no evidence that humans are susceptible to CWD, 18–20, 167 although it is impossible to prove that humans are not susceptible to this agent. 21 A few squirrel monkeys (Sciurus saimiri) were susceptible to CWD by intracerebral inoculation (R. Marsh and S. Young, personal communication), and expanded studies of nonhuman primate susceptibility to CWD are underway (R. Race, personal communication).
Deer and elk in CWD endemic areas are hunted for venison and consumed by local as well as nonresident families. Because it is not possible to provide absolute assurance that venison is without risk, hunting orders and Web sites in states and provinces with CWD in cervid populations provide information and guidance for the public about CWD (e.g., http://www.dnr.state.wi.us/org/land/wildlife/whealth/issues/CWD/, http://wildlife.state.co.us/CWD). Hunters are advised to avoid harvesting deer and elk that appear ill, to debone meat during processing, to wear latex or rubber gloves when dressing the carcass, and to avoid contact with brain, spinal cord, and lymphoid tissues. Regulations concerning requirements for harvested deer and elk to be tested for CWD vary among jurisdictions; in most locations, this is voluntary and available through state laboratories. Recommendations are that animals testing positive for any prion disease should not be consumed by humans or other animals. 167
Origin and Strain Typing
The origin of CWD is not known and might never be known. Three hypotheses have been suggested. Perhaps the most plausible is that CWD derives from scrapie. Support for this hypothesis comes from the moderate ability of PrPd from CWD-affected deer and elk to convert PrPc from sheep to the abnormal isoform PrPd 125 and from comparisons of glycoform patterns on western blots of PrPd from CWD-affected deer and elk and sheep scrapie. 123 In addition, scrapie agent inoculated intracerebrally causes a disease in elk similar to scrapie. 61, 62 However, CWD is unlike any characterized scrapie strain in rodent models; 27, 31 it does not appear to be transmissible to raccoons (Procyon lotor) as is scrapie, 63 and incubation period in an intracerebrally inoculated goat 159 was longer (6 years) than expected for scrapie. Alternatively, CWD might be a disease of mule deer after spontaneous alteration of PrPc resulting in formation of an infectious prion protein. Although spontaneous generation of prions might be theoretically possible and has been suggested as the origin of some cases of sporadic CJD of humans, 44, 119, 120 it would be exceedingly difficult if not impossible to prove retrospectively. A third possibility is that CWD is derived from another, currently un-identified, source of infection. Although it seems highly unlikely, a human source of CWD can not be completely dismissed; Hadlow 54 reminds us that, at least experimentally, TSE agents are transmissible from humans to animals.
The epidemiology of CWD is most compatible with a single strain that originated in mule deer and then infected elk and white-tailed deer. 159 CWD was first recognized as a clinical syndrome in mule deer many years before it was detected in elk housed in facilities with CWD-affected mule deer. 159 The first recognition of CWD in white-tailed deer was in free-ranging deer sympatric with CWD-affected mule deer and elk by retrospective examination of tissues when IHC for PrPd became available. 155
Characteristics of histologic lesions have been used for decades to distinguish forms of human and natural and experimental animal TSEs. 31, 42, 113 Typing TSE strains in rodents is based on intracerebral inoculation of genetically characterized mice with the agent of interest and then comparing lesion profiles on the basis of vacuolation scores in specified areas of the brain of affected mice and incubation periods. 42 Multiple strains of scrapie have been characterized in mice. 28, 30, 43 One mule deer source of CWD (captive mule deer from Colorado) was tested in this system, and it was found to be unlike any scrapie strains, BSE, TME, vCJD, or strains of sporadic CJD. 27, 31 The mule deer CWD source examined differed from other TSE sources studied in mice, giving a unique lesion profile and presence of marked perivascular amyloid deposits. 27 Microscopic characterization of BSE and scrapie strains in sheep has been evaluated recently with patterns of PrPd deposition by IHC 46, 83 and a panel of antibodies recognizing different PrP epitopes, providing yet another way of potentially evaluating strains of the TSE agents.
Another proposed system for distinguishing animal TSE strains uses intracerebral inoculation of raccoons. 57, 64 Incubation time of TME in raccoons was very short (5–6 months), incubation time of scrapie was moderate (about 2 years), and at the time the study was published, raccoons had not developed evidence of CWD infection. However, many more sources of TSE agents would need to be tested in raccoons before this could be considered a useful system for strain typing.
Recently, comparison of the relative amounts of di-, mono-, or unglycosylated PrPd detected by immunoblotting, or glycoform pattern analysis, is receiving considerable attention as a method of determining strains of TSE agents and as a potential epidemiologic tool, although a plea for caution in the use of this technique alone was recently published. 118 Multiple forms of PrPd appear to be produced by different cell types after infection with the same TSE agent, suggesting that structural differences in PrPd do not invariably code for different strains of scrapie; 82 this could also be the case for CWD. The diglycosylated forms of PrPd predominate in cervids with CWD 123 (Fig. 5).
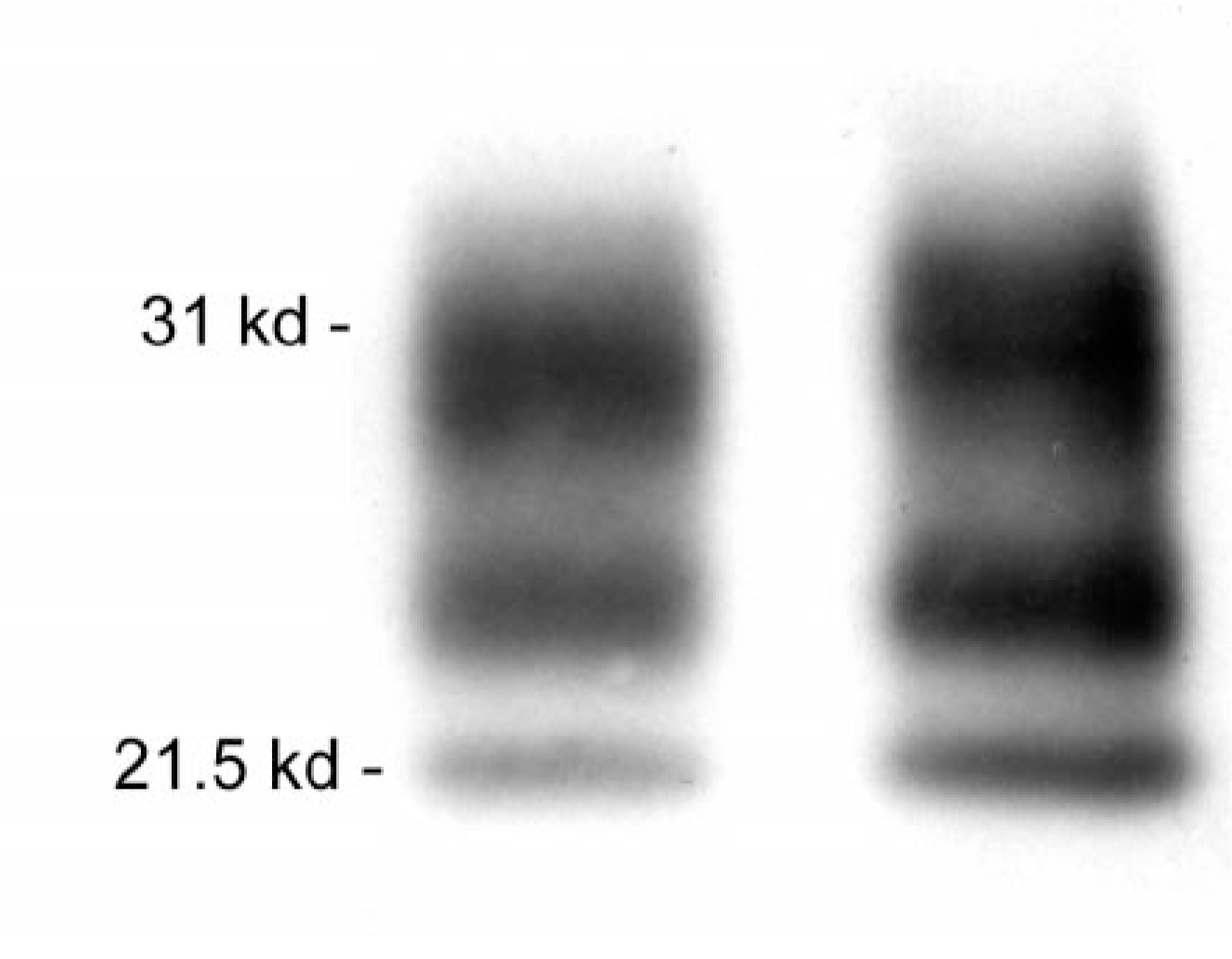
Western immunoblot of chronic wasting disease–associated prion protein; brain from mule deer (left) and white-tailed deer (right). The strongest signal is in the upper diglycosylated band and the weakest in the lower unglyco-sylated band.
Glycoform pattern analysis of PrPd was one of the techniques used to help determine the relationship between BSE and vCJD 34, 72 and is a component used in characterization of forms of classical CJD. 81, 113 Comparison of PrPd from a small number of mule deer, white-tailed deer, and elk with CWD, sheep with scrapie, and cattle with BSE, did not reveal glycosylation patterns that could be used to reliably distinguish these agents; however, some differences found between PrPd from individual mule deer and elk suggested the possibility of different CWD strains. 123 No differences were found in PrPd derived from six distinct regions of brain from clinically affected mule deer or elk. 123 Although the origin of CWD remains obscure, the similarity of glycoform patterns in scrapie-affected sheep and CWD-affected cervids was felt to provide support for the hypothesis that CWD was derived from sheep scrapie. 123
Other molecular techniques for investigation of TSE strains have been applied to CWD. In vitro analysis of the ability of cervid PrPd to convert PrPc of various species to the abnormal isoform indicated a greater degree of conversion of ovine PrPc to PrPd than either bovine or human PrPc but less than conversion of homologous PrPc. 125 Conformation-dependent immunoassay, an assay that recognizes PrPd without the need for proteinase treatment, has also been applied to PrPd from a small number of CWD-affected cervids. 127 In these analyses, PrPd from elk could be distinguished from that of mule and white-tailed deer, suggesting possible strain differences; the explanation for these differences has not been determined. Links between scrapie and CWD have been proposed on the basis of similarity of ribosomal DNA sequences from Spiroplasma mirum in sheep with scrapie and elk with CWD; 12 however, the evidence provided was weak and association of S. mirum with the TSEs is not widely accepted.
Transmission
Although the exact method of transmission of CWD is not known, horizontal and indirect transmission appear to be the most important routes of spread. 99 Epidemiologic observations suggest that in addition to transmission within species, CWD can be transmitted from elk to mule deer and white-tailed deer, from mule deer to elk, and from mule deer to white-tailed deer. 156 Maternal transmission, if it occurs, does not appear to play a significant role in the epidemiology of the disease. 99 To date, PrPd and infectivity have not been identified in placentas of deer and elk. This is in contrast to scrapie, in which high levels of infectivity reside in the placenta, 5, 122, 143, 144 and epidemiologic investigations have found that transmission at the time of lambing appears to be important in maintaining scrapie in flocks.
Results of a series of studies investigating CWD transmission have recently been published. 102 Direct transmission of CWD between animals occurred, although the exact mechanism was not determined. The role of environmental contamination in maintaining infectivity is not entirely understood; however, controlled studies showed infectivity remained on pastures in which CWD-affected deer resided approximately 2 years previously. These studies were conducted in pastures presumed to be highly contaminated. Thus, extrapolation to field situations should be done with care, but these data suggest caution in managing pastures or paddocks that have housed CWD-affected cervids. In addition, mule deer were infected by contact with skeletal remains of CWD-affected deer and surrounding ground and vegetation. This information bolsters the need for caution when moving portions of harvested cervids that contain the highest amount of infectivity, such as the head and the spine, to areas where CWD does not exist.
Regulatory Considerations
Concomitant with growing concerns about CWD has been an increase in guidelines and regulations governing farmed cervids and management of free-ranging deer and elk. Following recognition of CWD in farmed elk, states, provinces, and federal animal health regulatory agencies developed voluntary and/or mandatory programs to control or manage the disease, with the ultimate goal of eradication of CWD from these industries. Details of these programs are available online at the various state and provincial Web sites and from the Canadian Food Inspection Agency (http://www.inspection.gc.ca/english/anima/heasan/disemala/cwdmdc/certnorme.shtml) and the US Department of Agriculture (http://www.aphis/usda.gov/vs/nahps/cwd/farmed-cwd.html).
These programs are based on individual animal identification, annual census and visual inspection, and testing of animals dying on the farm, when harvested, or at slaughter, to establish certified herds after years (typically 5 years) of monitoring. If CWD is identified, herds are quarantined, they might be depopulated with indemnity, and herd and premises plans are developed. At the time of writing, only two elk herds (Colorado) and one white-tailed deer herd (Wisconsin) were under quarantine for CWD.
Management of CWD in free-ranging herds is more difficult than in farmed cervids and varies depending on jurisdiction and history of CWD in that location. For example, an ambitious program of white-tailed deer population reduction is being pursued in the relatively small CWD endemic area in Wisconsin (www.dnr.state.wi.us/org/land/wildlife/whealth/issues/cwd/index.htm). In the much larger geographic area in which CWD occurs in Wyoming, Colorado, and contiguous states, eradication is impossible with currently available techniques. Management of CWD in these areas varies among states and can include surveillance, attempts to prevent geographic spread, and efforts to reduce prevalence through population reduction. Most states or provinces provide guidance to hunters or have restrictions concerning transport from CWD endemic areas of tissues of highest infectivity from harvested deer and elk, particularly skull containing brain and vertebral column containing spinal cord. Most jurisdictions recommend transporting only deboned meat, cleaned skull cap and antlers, and hides. A list of state and provincial regulations related to CWD has been compiled and is available online (http://www.cwd-info.org/index.php/fuseaction/policy.regulations).
Concerns about safe handling of animals and tissues suspected of having CWD or scrapie led the American Association of Veterinary Laboratory Diagnosticians (AAVLD) to develop best management practices for handling animal TSE agents classified as biosafety level 2 agents, including CWD, scrapie, and TME. Details are available to members on the AAVLD Web site (http://www.aavld.org/aavld-3/Current_news.jsp) 3 or to the public on the Wyoming State Veterinary Laboratory web site (http://wyovet.uwyo.edu/WSVL/updates/UPDATES_BY_YEAR/Updates_2004.htm) and should be consulted if working with these agents in diagnostic laboratories. The primary features of these guidelines are shown in Table 5.
Summary of best management practices for handling chronic wasting disease carcases and tissues in animal health laboratories (adapted from the American Association of Veterinary Laboratory Diagnosticians Best Management Practices 3 ).

∗ See Brown et al., 25 CDC and NIH, 32 Ernst and Race, 41 Race and Raymond, 124 Taylor, 139 Taylor et al., 140 and WHO. 166
† Environ LpH, Steris Corporation, Mentor, OH.
Formic acid is used in many laboratories for pre-treatment of tissues possibly containing TSE agents after cutting into blocks but before processing into formalin. 25 This treatment greatly decreases or abrogates infectivity before sectioning of tissue blocks. Because treatment with full-strength formic acid can result in brittleness of the tissues, difficulty in sectioning, and alteration of staining qualities, adequate washing is important. However, good quality histologic preparations can be obtained readily.
Summary
CWD is a unique TSE and has inherent challenges beyond those of the domestic animal TSEs. Transmission readily occurs among cervids; no complete genetic resistance has been identified, and environmental contamination might be important in maintenance of the disease. Although CWD probably can be controlled in farmed cervid industries, management of CWD in free-ranging populations of deer and elk is problematic, and it is likely the disease will continue to spread. Veterinary pathologists need to be aware of the features of CWD and will be important participants in surveillance programs and investigation of CWD natural history and biology in the future.
Footnotes
Acknowledgements
I gratefully acknowledge the mentorship of Dr. Stuart Young, and this review is dedicated to his memory. Through many exciting discussions and untold hours of working on the microscope, he was an inspiration. He will be missed. I also acknowledge a long-standing collaboration and innumerable discussions about CWD with Dr. Mike Miller with the Colorado Division of Wildlife and Dr. Tom Thorne with the Wyoming Game and Fish Department. Discussions with Dr. Jean Jewell about genetics and molecular biology have been particularly helpful. Collaboration and discussion with too many individuals working with the TSEs to mention individually are gratefully acknowledged. Partial support was provided by the Department of Veterinary Sciences, University of Wyoming; National Science Foundation–National Institutes of Health Grant DEB-0091961; Department of Defense Grant DAMD 17-03-1-0542; and the National Cattlemen's Beef Association.
1Deceased December 29, 2004. This paper was in revision at the time of Dr. Williams's death. Final edits were completed by her collaborators, Dr. Jean Jewell at the University of Wyoming and Dr. Mike Miller of the Colorado Division of Wildlife.
