Abstract
A study of the lesions of beaked whales (BWs) in a recent mass stranding in the Canary Islands following naval exercises provides a possible explanation of the relationship between anthropogenic, acoustic (sonar) activities and the stranding and death of marine mammals. Fourteen BWs were stranded in the Canary Islands close to the site of an international naval exercise (Neo-Tapon 2002) held on 24 September 2002. Strandings began about 4 hours after the onset of midfrequency sonar activity. Eight Cuvier's BWs (Ziphius cavirostris), one Blainville's BW (Mesoplodon densirostris), and one Gervais' BW (Mesoplodon europaeus) were examined postmortem and studied histopathologically. No inflammatory or neoplastic processes were noted, and no pathogens were identified. Macroscopically, whales had severe, diffuse congestion and hemorrhage, especially around the acoustic jaw fat, ears, brain, and kidneys. Gas bubble-associated lesions and fat embolism were observed in the vessels and parenchyma of vital organs. In vivo bubble formation associated with sonar exposure that may have been exacerbated by modified diving behavior caused nitrogen supersaturation above a threshold value normally tolerated by the tissues (as occurs in decompression sickness). Alternatively, the effect that sonar has on tissues that have been supersaturated with nitrogen gas could be such that it lowers the threshold for the expansion of in vivo bubble precursors (gas nuclei). Exclusively or in combination, these mechanisms may enhance and maintain bubble growth or initiate embolism. Severely injured whales died or became stranded and died due to cardiovascular collapse during beaching. The present study demonstrates a new pathologic entity in cetaceans. The syndrome is apparently induced by exposure to mid-frequency sonar signals and particularly affects deep, long-duration, repetitive-diving species like BWs.
The possibility that exposure to sonar stimuli could lead to the stranding of cetaceans is an issue of increasing interest because of recent strandings temporally and geographically coincident with naval sonar operations. 1, 11, 17, 61, 66, 67 BWs (family Ziphiidae), one of the deepest-diving whales, are of particular interest because of a notable relationship between military operations employing midfrequency sonar and BW mass strandings. 1, 11 Typically, strandings of BWs prior to 1963, when certain types of midfrequency sonar equipment began to be employed, involved single individuals. 1 Subsequently, mass strandings of BWs linked with naval exercises have been described in several locations: Bonaire in 1974, the Canary Islands in 1985, 1988, and 1989, Greece in 1996, and the Bahamas in 2000. 1, 11, 17, 61, 66, 67
A mass stranding of BWs in the northern Bahamas Islands causally incriminated the use of tactical, mid-frequency sonar because of its close temporal association with such naval maneuvers. 1, 11 Hemorrhage in the brain, ears, and acoustic fat was reported as the main lesion found in some BWs in the Bahamas stranding. A number of acoustically mediated behavioral modifications and pathophysiologic pathways were proposed to have caused these lesions; however, conclusive evidence linking a mechanism to the lesions was lacking. 11
Past research efforts on the potential for anthropogenic sound to affect marine mammals have focused on auditory effects and behavior modifications following sound exposure. 11, 19, 28, 53 Nonauditory consequences of exposure to sound have received less attention. 6, 23 A hypothesized, nonauditory link between strandings and sonar exposure is proposed to occur when tissues are supersaturated with dissolved nitrogen gas, and bubble growth–facilitated diffusion is stimulated within tissues. 6, 23 Bubble growth could result in emboli-induced tissue separation and increased localized pressure in tissues, the presumed cause of decompression sickness (DCS) in human divers. 16, 31, 33, 46, 47 Following the initial stimulation of bubble growth in tissue that has been highly supersaturated with gas, growth could continue in the absence of a persistent acoustic stimulus. It is believed that marine mammals have evolved adaptations to prevent deleterious, nitrogen bubble formation. 34, 50, 57 However, to our knowledge, no studies have specifically addressed whether nitrogen bubble formation in tissue occurs in diving marine mammals. Recently, researchers have presented strong evidence of chronic, gas bubble lesions in the liver and kidney of different stranded cetacean species. 25 These lesions suggest that gas bubbles formed in vivo can persist and generate fibrosis in diving cetaceans. 14, 25 Such emboli presumably cause ischemia and would explain the formation of chronic, bony lesions, consistent with those of dysbaric osteonecrosis (DON), described in sperm whales (Physeter macrocephalus). 40
DCS is the result of the supersaturation of body tissues with nitrogen gas and the subsequent release of nitrogen gas bubbles. 16, 26 In human divers, DCS is typically caused by rapid decompression following dives using compressed air 8, 16 and also has been reported during repetitive, breath-hold dives. 15, 33, 47, 52 Until the lungs collapse during a dive, alveolar gases are absorbed into the blood proportional to hydrostatic pressure. The amount of gas dissolved in specific tissues depends on dive depth and duration, descent and ascent rates, lipid content of the tissue, and surface time between successive dives. 16, 26, 42
A number of anatomic, physiologic, and behavioral adaptations of marine mammals have been proposed to guard against nitrogen bubble formation. 12, 34, 50, 54–57 Due to alveolar collapse, gas exchange does not occur at depths greater than 70 m in dolphins 56 or at depths greater than 30–50 m in seals. 34 Pinnipeds and shallow-diving cetaceans are thought to be at low risk of developing a high nitrogen load, either because lung collapse obviates gas uptake at deeper depths or because their typical dive profiles result in only modest tissue nitrogen accumulation. 23 BWs 22 are theoretically at a greater risk of developing deleterious tissue–nitrogen levels because their deep prolonged dives increase during the period in which nitrogen uptake can occur prior to lung collapse. 23 Thus, deep-diving cetaceans may be more susceptible to the action of high-intensity acoustic energy on preexisting gas nuclei. 6, 23
The presence of gas emboli is an important finding in human DCS, but pulmonary fat emboli have also been reported with DCS-related, severe cardiorespiratory disturbances. 26, 29 Systemic fat embolism is a secondary effect of the abrupt pressure changes observed with DON, a condition initiated by the evolution of gas bubbles in nitrogen-supersaturated fatty marrow after inadequate decompression. 26, 29, 30 The clinical manifestations of fat emboli depend on the volume of fat reaching the lungs and other affected tissues. Fat emboli are not reversible with recompression therapy. The severity of fat emboli–induced clinical disease may be progressive as survival time after trauma increases. 10, 31, 43, 49, 62
Forensic pathologists associate fat emboli with bone fractures, diabetes mellitus, burns, acute pancreatitis, fat and soft tissue injury, and DCS with acute DON. 10, 26, 30, 31, 43, 49, 62 These entities are underdiagnosed clinically and at postmortem examination. 10, 31, 49 The pathogenesis of fat embolism is not fully understood, and it is likely multifactorial. 24 Two mechanisms have been proposed for the development of fat emboli. First, direct entry of fat emboli into the bloodstream after trauma may cause direct, toxic injury in the lung and produce respiratory insufficiency when free fatty acids are released from fat tissues. 24, 62 A second mechanism involves the generation of fat emboli from plasma lipoprotein disruption and coalescence of lipid at the intravascular gas bubble interface. 24
Fat emboli have not been reported in stranded cetaceans, and reports of gas bubble lesions are uncommon. 13, 14, 25 This report describes the pathologic findings in BWs involved in a mass stranding that occurred coincidentally with naval exercises in the Canary Islands. Both gas bubble lesions and fat emboli are documented in these whales, and a hypothesis is presented to explain the association of mass stranding of BWs and sonar exposure.
Materials and Methods
The southeastern coast of Fuerteventura (Canary Islands) is well known for BW aggregations due to its ocean depths greater than 1,000 m within a few hundred meters of the coast. On 24 September 2002, 14 BWs were stranded on the beaches of Fuerteventura and Lanzarote islands. Seven whales (one female Mesoplodon densirostris, one female Mesoplodon europaeus, and five male Ziphius cavirostris) died. The remaining seven live whales were returned to deeper waters. During the next 3 days, three male BWs and one female BW (Z. cavirostris) were found dead either on the coast or floating offshore (Table 1).
Epidemiology of beaked whales from a mass stranding in the Canary Islands.
The strandings were temporally associated with naval exercises involving numerous surface warships and several submarines (maneuvers called Neo-Tapon 2002). The exercise area extended from the coast to up to 40 km away. On 24 September at 9:00 a.m., while examinations of animals were ongoing, warships were readily visible from 1 km to several kilometers from the beaches. Spanish naval sources indicated that tactical, midrange frequency sonar was used during the exercises from 3:00 a.m. to 10:00 a.m. on 24 September 2002.
Postmortem examinations were performed 35 on the beach on 10 animals. External findings were recorded, and internal organs were sampled. The heads were preserved at 4 C and delivered to the Veterinary School at Gran Canaria for detailed dissection. The first six stranded animals (BW-1 through BW-6) were examined postmortem on 24 September, within 12 hours of stranding. Heads were dissected and sampled the next day. BW-7 was found dead offshore on the morning of 25 September. On 25 September, BW-8 and BW-9, both mature males, were found stranded on Lanzarote, and BW-9 was found beached with multiple, postmortem shark bites. BW-10 was found floating close to Fuerteventura on 26 September and was given a postmortem examination on 27 September. All the animals were judged to have died within 24 hours of the first stranded BW that was sighted at 7:00, 24 September 2002. One other whale was recovered but not examined postmortem because of advanced autolysis.
Tissues were collected and stored in buffered 10% formalin immediately or frozen and were later formalin fixed before processing for routine light microscopy. Tissues from stranded animals included: brain, hypophysis, choroid plexus, cervical spinal cord, liver, lung, kidney, heart, lymph nodes, digestive tracts, reproductive tracts, and perilarygeal tissues, including the trachea and thyroid (Table 2). The eyes from BW-4 were collected.
Detection of fat emboli in tissues of BWs.∗

∗ NS = not sampled.
† Formalin postfixed frozen samples. The other samples were directly fixed in 10% formalin during postmortem examination. + = positive for fat emboli; − = negative for fat emboli.
Histologic stains used on frozen, formalin-fixed tissues to detect fat emboli were oil red O stain 22 (ORO) and Sudan black B 23 stain. 2, 51 Osmium tetroxide postfixation and paraffin embedding was also used to detect fat emboli. 7 A picric acid technique was used to reduce pigment artifacts when necessary. 5 The best technique for demonstrating fat emboli was the postfixation with the osmium of paraffin-embedded tissue. Frozen sections of fixed tissue stained with ORO and Sudan black B stained fat emboli well, but tissue morphology was inferior to osmium postfixation-stained, paraffin-embedded sections. The picric acid technique removes formalin pigment from fixed tissues and demonstrated fat better, but it washed out some of the lipid from tissue sections. Because fat emboli are not commonly assessed in stranded cetaceans, 86 archived samples of lungs of single-stranded cetaceans found on the coasts of the Canary Islands were used as fat emboli controls.
Results
The ambient temperatures during the period of the mass stranding ranged from daytime highs of 24°C to nighttime lows of 17°C. All stranded animals belonged to the family Ziphiidae, commonly named BWs. Macroscopically, no systemic, inflammatory, or neoplastic processes were noted in any animal. In BW-1 through BW-7, the lack of external trauma and the lack of marked dermal bruising indicated that blunt trauma was probably not involved in the stranding (Fig. 1). A large amount of fresh, undigested gastric contents was noted in the whales, suggesting a rapid onset of disease and death. Subconjunctival hemorrhages were observed in several animals.

A freshly stranded beaked whale (Ziphius cavirostris) without cutaneous lesions that would be an indication of collision trauma, terminal struggling, or predation.
The first (BW-1 to BW-7) carcasses showed marked congestion of all organs, particularly the neck (including the larynx, thyroid gland, and trachea). Multifocal petechiae or severe, extensive ecchymoses were observed in the acoustic fat around the jaws (Fig. 2). Melon fat was examined in only two whales, and some petechiae were observed in each of these whales. Head and neck lymph nodes were enlarged and congested. The lungs were congested and had multiple pleural hemorrhages. A moderate amount of froth was present in the tracheas. The peribullar and pterygoid sinuses were full of white froth. Large cardiac blood vessels were distended. Macroscopic descriptions of the abdominal organs from the first six animals were brief, because they were necropsied rapidly so that sample collection could be finished in natural light. The abdominal lesions included some liver fibrosis, general organ congestion, and marked congestion of large and midsize veins. Hemorrhages were seen in congested kidneys. Parasites (Crassicauda sp.) were observed in the kidneys of all animals. Diffuse vascular congestion and some petechiae were observed on the intestinal serosa. Postmortem examinations of BW-7 to BW-10 showed similar macroscopic lesions, and tissues were sampled with the exception of central nervous system (CNS) tissues (Table 2). No detailed macroscopic examination for gas bubbles was conducted in the portomesenteric veins of any whale.
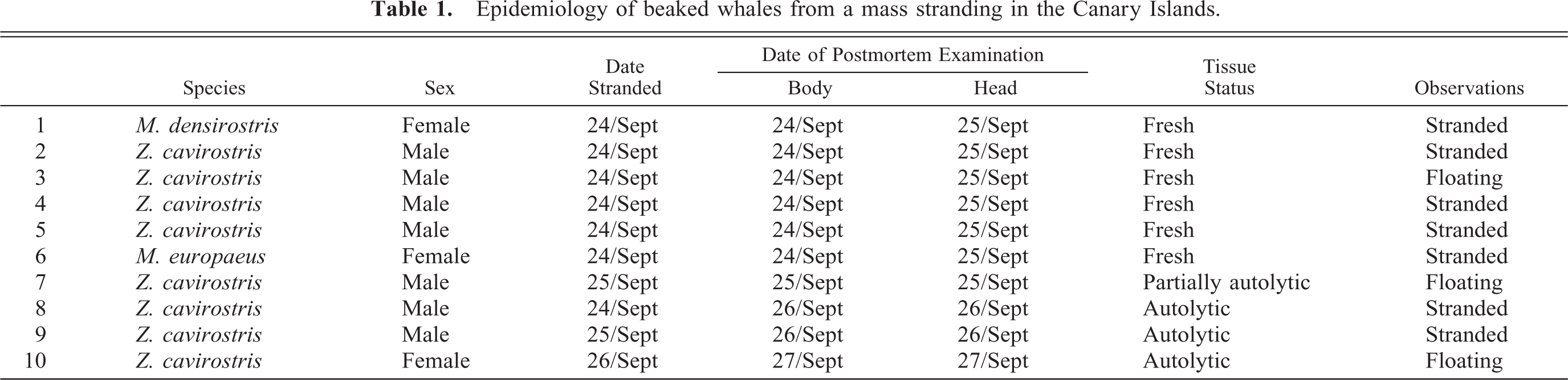
Mandible; beaked whale (BW-3). Areas of hemorrhage are in the acoustic fat of the jaw. Inset: Photomicrograph of perivascular hemorrhage in the acoustic fat with nonstaining, intravascular bubbles/emboli (arrow). HE. Bar = 25 μm.
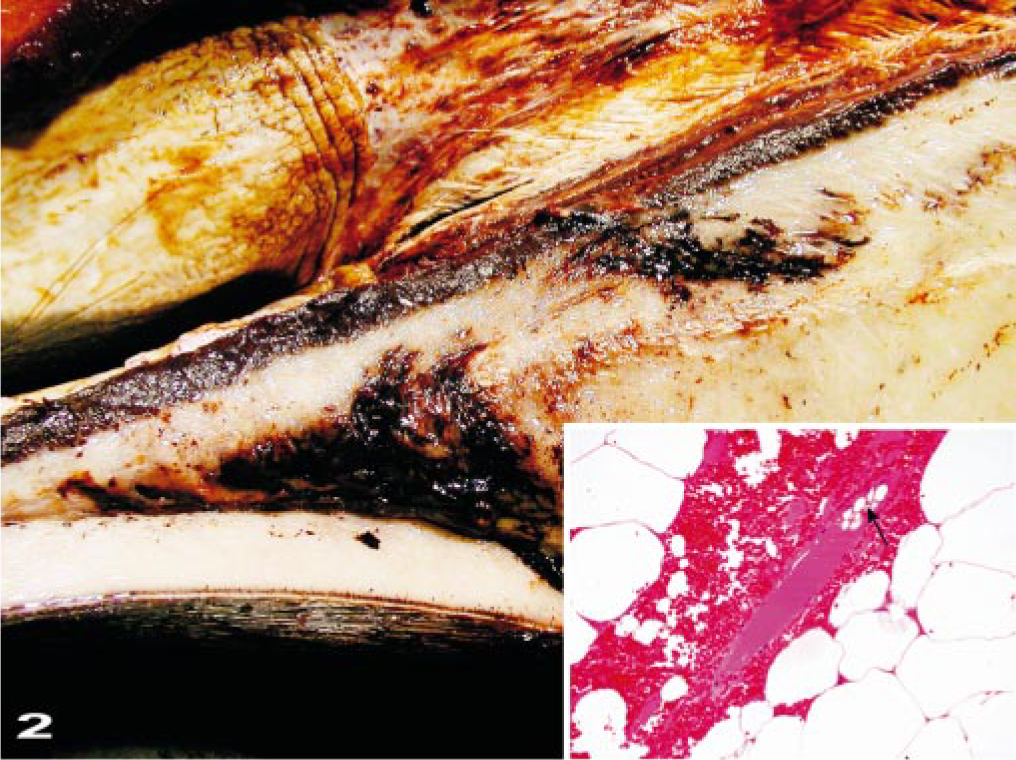
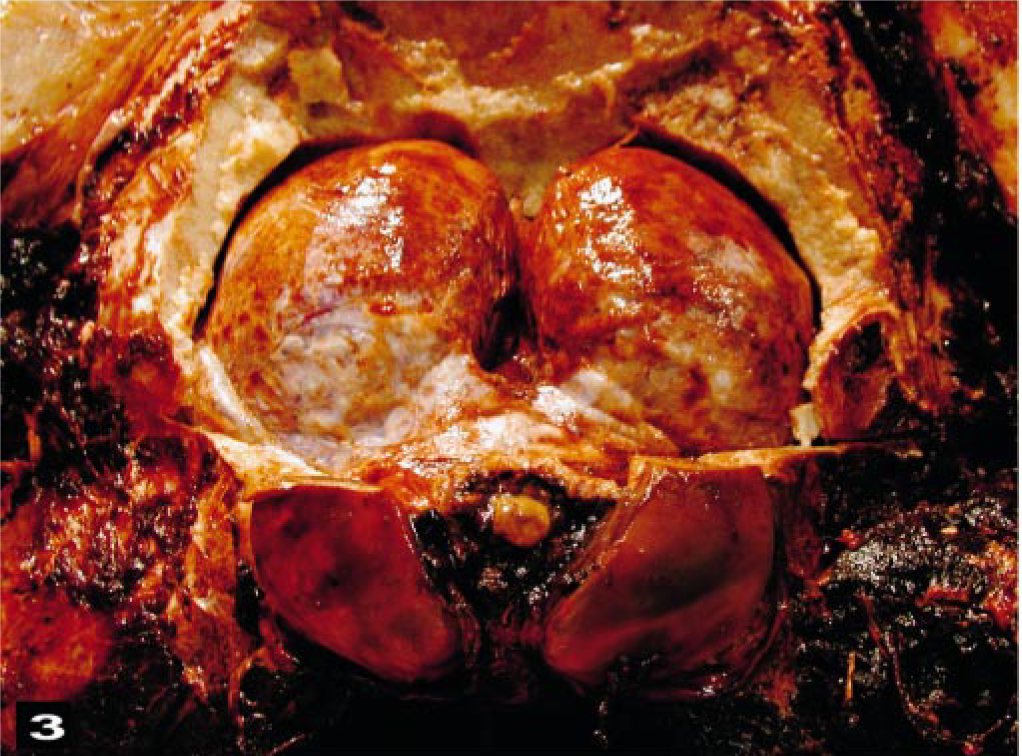
The primary macroscopic cerebral lesions were severe, diffuse, vascular congestion with multifocal-to-extensive subarachnoidal hemorrhages (Fig. 3). Clotted blood was observed bilaterally in the lateral ventricles of some brains, along with severe hemorrhaging around the proximal cervical spinal cord. The gyri of the brain were swollen and moist. Multifocal hemorrhages were observed in the meninges. Macroscopic gas bubbles that were observed retrospectively in brain photographs taken during the dissections (Fig. 4) were prominent within superficial, midsize, cerebral vessels in some areas.
The exposed caudal aspect of the brain and spinal cord shows congestion of the meninges and severe congestion and hemorrhage in the epidural, vascular plexus around the spinal cord.
Cerebral cortex; beaked whale (BW-2). The gyri of the cortex are swollen and have focal, subarachnoidal hemorrhaging. Inset: Intravascular gas bubbles (arrows) are prominent within a meningeal vessel.
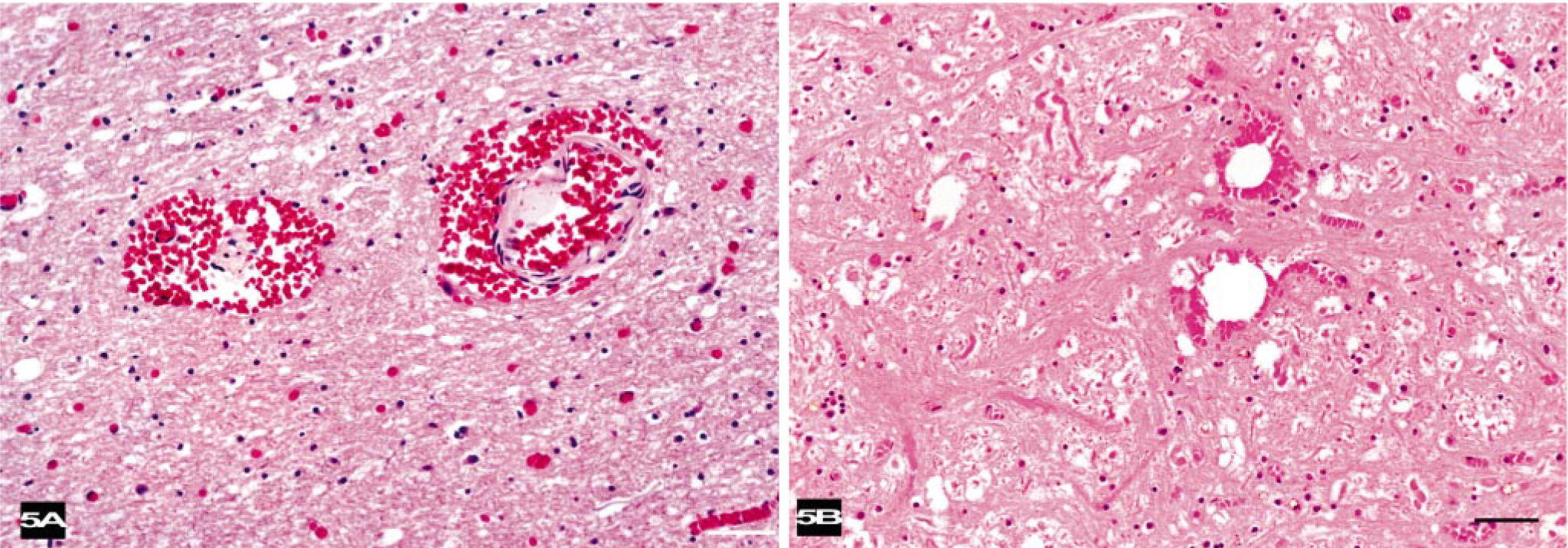
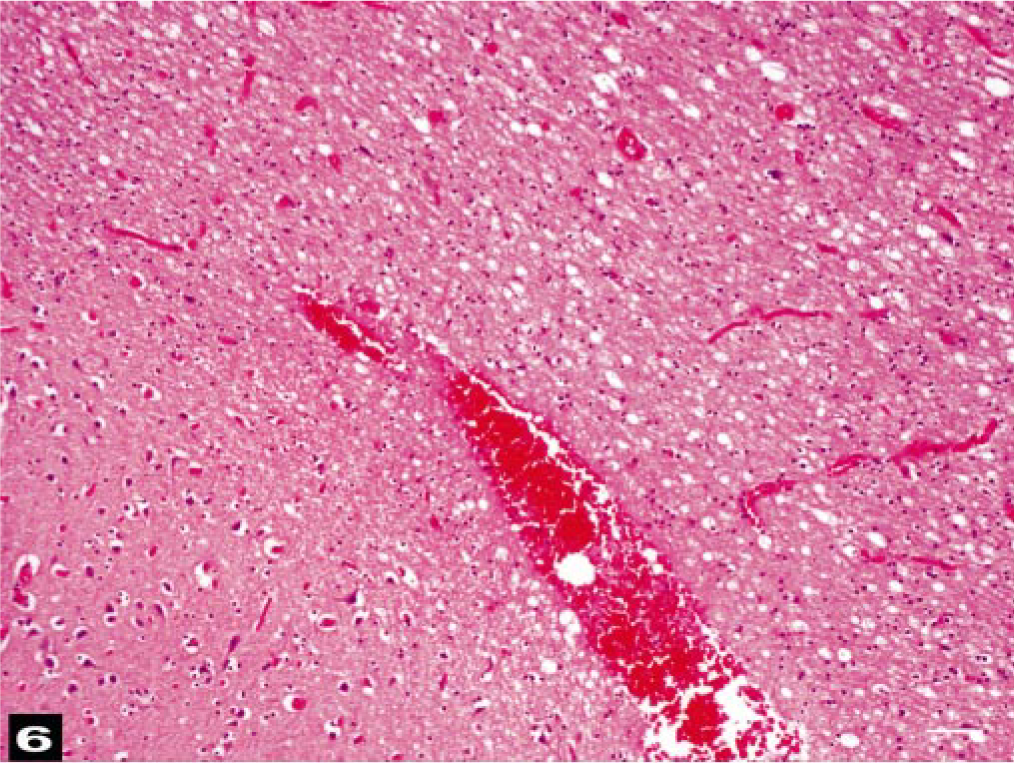
Histologic lesions in the first seven animals (BW-1 to BW-7) were similar. Severe, diffuse congestion was observed in the large and small cerebral and pial vessels of the gray and white matter of the cerebral hemispheres. Varying severity and extensions of hemorrhaging were observed in the subarachnoid space. In the brainstem and around the proximal cervical spinal cord, severe hemorrhaging was observed in the perivascular spaces associated with congested midsize vessels (Fig. 5a). Large unstained vacuoles that did not stain as fat were observed in some veins and within hemorrhages in the parenchyma or disrupted columns of the erythrocytes in perivascular spaces (Fig. 5b). Intravascular air vacuoles were most numerous in BW-1. Hemorrhages were observed in the white matter of most of the animals, and the adjacent parenchyma often had a spongiotic appearance (edema, Fig. 6). Proteinaceous, perivascular edema fluid and swollen glia surrounded some vessels. Severe, diffuse hemorrhaging was observed consistently around the cervical medulla and associated with the epidural, vascular plexus. Less severe hemorrhaging was observed around the ophthalmic rete of BW-4. Locally extensive conjunctival hemorrhaging sometimes dissected the scleral connective tissue of BW-4. In the proximal, cervical spinal cord, vascular congestion and perivascular edema affected both the white and gray matter.
Brainstem; beaked whale (BW-1).
Brain; beaked whale (BW-3). Congested vessels, some of which have perivascular hemorrhage, are associated with spongiotic parenchyma. HE. Bar = 25 μm.
No cardiac lesions were observed. Pulmonary vascular congestion, vacuoles in midsize veins, atelectasis, and hemorrhage varied from mild to severe among the whales examined. Multifocal, interstitial, interlobular, and subcapsular hemorrhages were prominent in the kidneys. Often, renal interlobular connective tissue was disrupted by venous vacuoles associated with hemorrhaging (Fig. 7). Moderate congestion of the glomeruli and vasa recta with some microhemorrhages were observed in sections of the renal medulla. Multifocal granulomatous nephritis and pyelitis were associated with the parasites observed grossly in all of the kidneys.
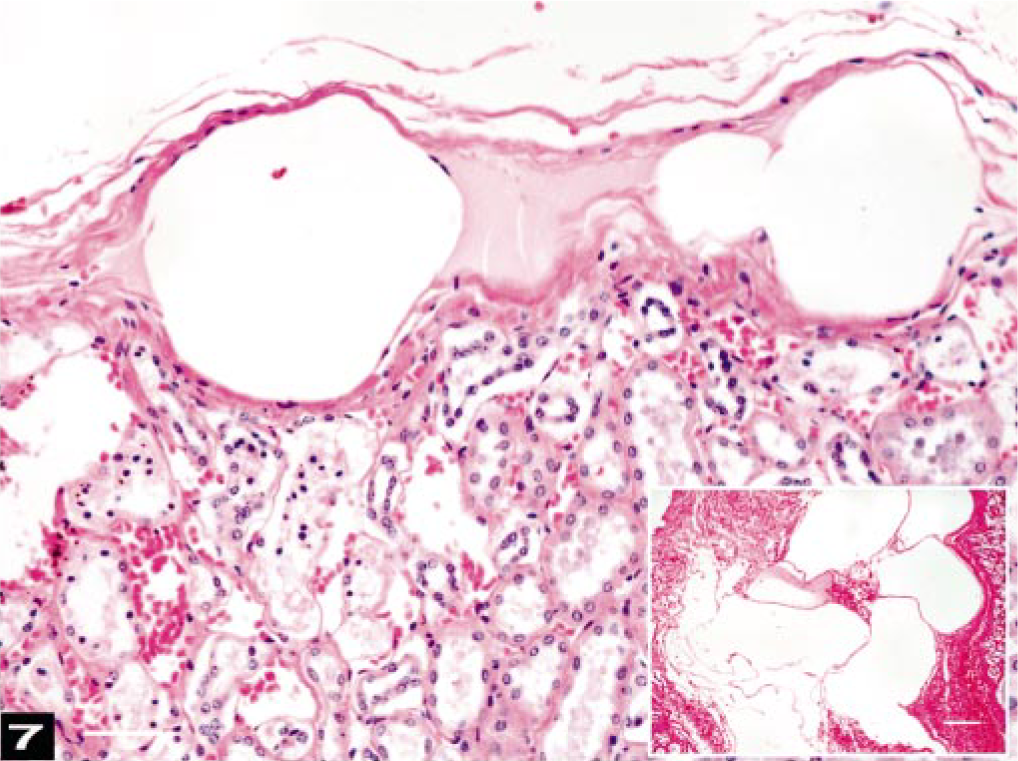
Kidney; beaked whale (BW-1). Subcapsular kidney veins are distended by round, fat-negative gas emboli. Bar = 25 μm. Inset: Renal interlobular connective tissue is disrupted by large, round-to-oval air emboli in veins associated with hemorrhage. HE. Osmium tetroxide-postfixation. Bar = 25 μm.
All lymph nodes of the head and neck contained large amounts of erythrocytes in the subcapsular and sinusoidal areas. Enlarged subcapsular sinuses had many round-to-oval unstained vacuoles (Fig. 8). Macroscopically, visceral lymph nodes that drained the areas of hemorrhaging had similar changes. Spleens were congested. Livers had moderate-to-severe portal vein congestion, and all livers contained variable numbers of sinusoidal vacuoles (Fig. 9). Gastric and intestinal submucosal and serosal vessels were congested. Some large vacuoles were observed in the submucosal veins. Intestinal and reproductive tract serosae had focal hemorrhages.
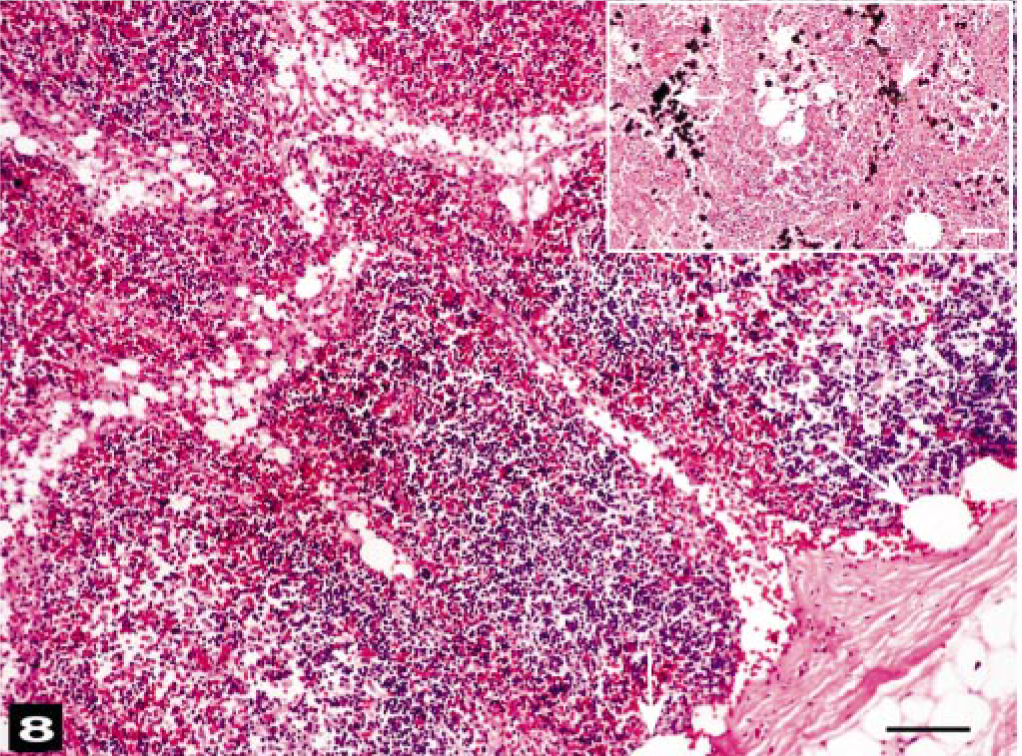
Lymph node of the head; beaked whale (BW-1). Many erythrocytes are in the subcapsular and medullary sinuses. Subcapsular sinuses contain round-to-oval, fat stain–negative, unstained air bubbles (arrows). HE. Bar = 25 μm. Inset: Fat vacuole (arrow) staining in a lymph node sinusoid. HE. Osmium tetroxide postfixation. Bar = 25 μm.
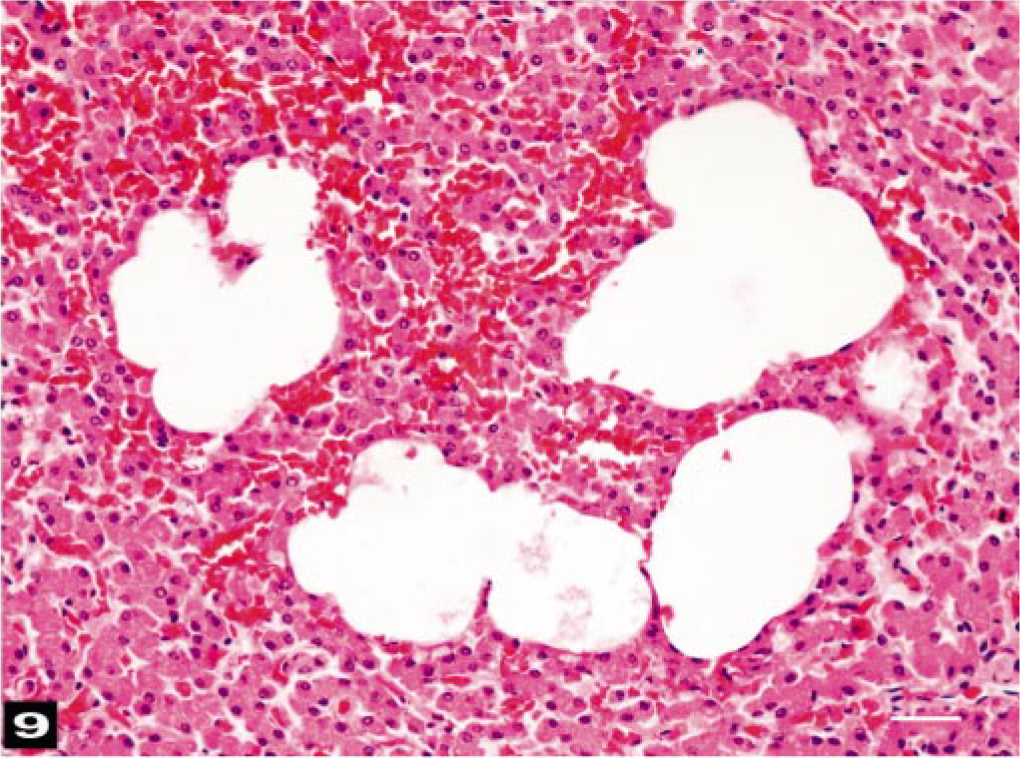
Liver; beaked whale (BW-1). Sinusoids are dilated by gas bubble–like dilatations. HE. Bar = 25 μm.
No fat emboli were detected in the vessels of the cerebral or spinal gray matter, free in the subarach-noidal hemorrhages, or within the choroid plexuses (Table 2). However, many fat emboli were observed within the veins and lymphatics of the epidural retia surrounding the spinal cord. Epidural adipocytes associated with hemorrhaging were enlarged or ruptured. Adipocytes adjacent to the ophthalmic plexus, thyroid capsule, ovaries, and intestinal serosae were similarly affected.
Histologically, all lungs studied had fat emboli widely distributed within the small veins and capillaries (Figs. 10, 11). The fat emboli varied considerably in shape, with rounded and branching profiles. The size of fat emboli depended on the vessels in which they were located. Lymph nodes of the head and neck contained many fat emboli in the subcapsular areas sometimes in large numbers. Variably sized, fat stain–negative vacuoles, which were presumed to be air emboli, were observed in some lymph nodes, together with fat emboli. The kidneys had only a few fat emboli in small medullary veins. With the exception of one fat emboli in a cardiac arteriole (BW-10), fat emboli were observed only in veins.
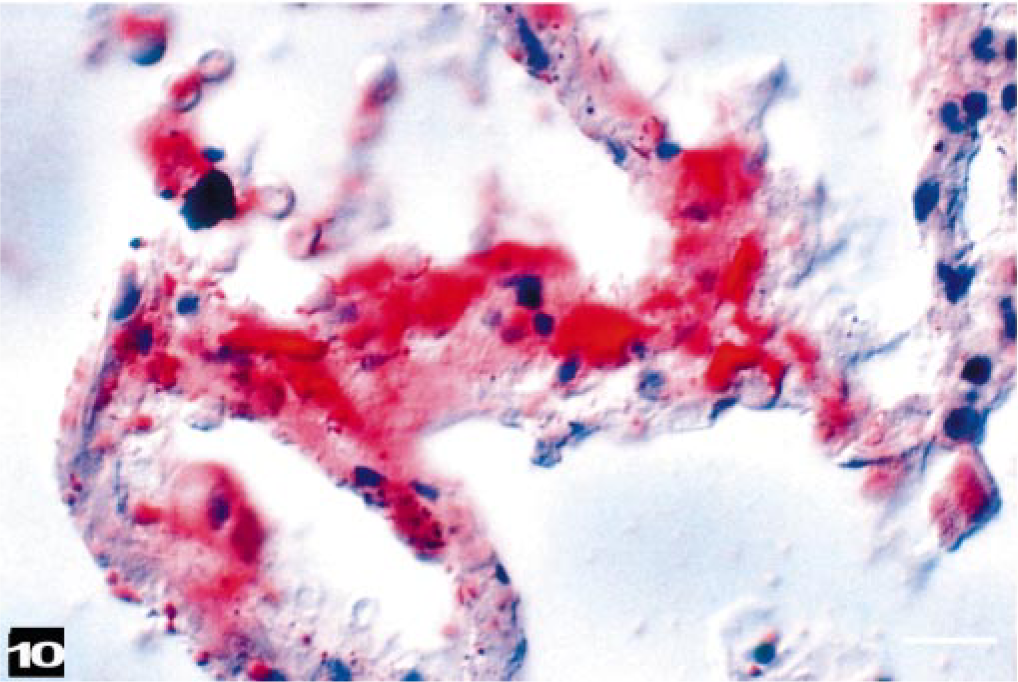
Beaked whale (BW-7). Red-staining fat emboli are in vessels of the alveolar septa, and erythrocytes are in an adjacent alveolus. Phase-contrast microscopy. Oil red O stain, phase-contrast photomicroscopy. Bar = 25 μm.
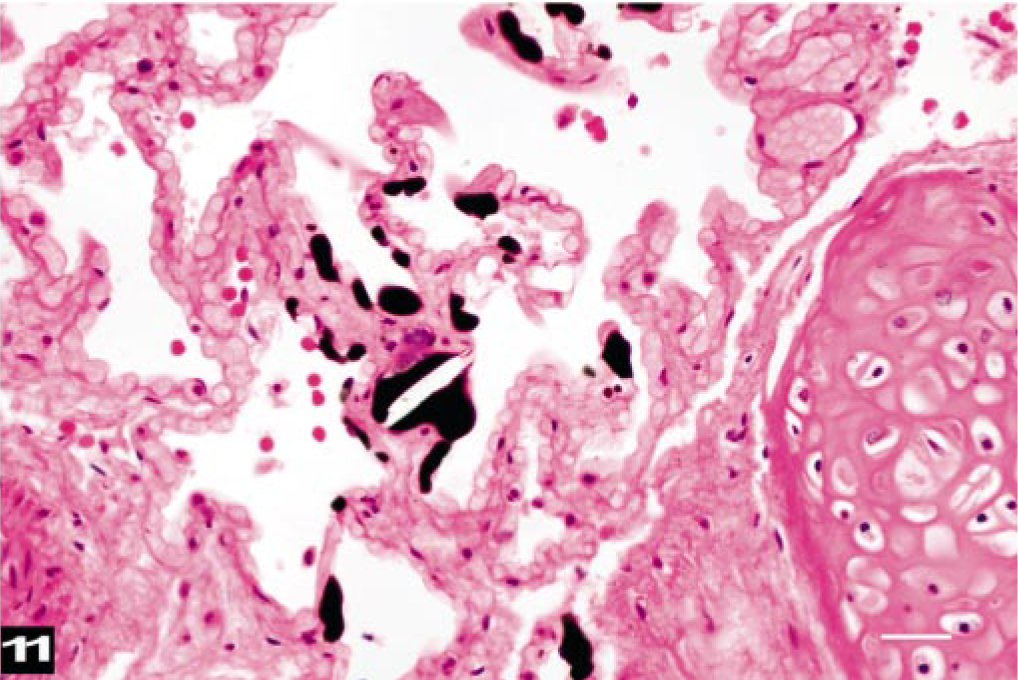
Lung; beaked whale (BW-9). Dark-staining fat emboli are scattered within midsize veins and capillaries. HE. Osmium tetroxide-postfixation. Bar = 25 μm.
For comparison, 86 archived samples of fresh lungs from different species of cetaceans that had been stranded on the Canary Islands were checked for fat emboli (data not shown). Only six showed intravascular fat emboli, and two of these were sperm whales that died after ship collision. The other four whales consisted of one stranded BW and an aged Globicephala macrorrhychus, for which the causes of death were not determined, and two pigmy sperm whales with skull fractures.
No bacterial pathogens were isolated on routine aerobic bacterial cultures of the brain, lung, and spleen.
Discussion
Atypical mass strandings occurred previously off the same coasts of the Canary Islands during naval exercises carried out in 1985, 1988, and 1989, 61, 67 but no postmortem studies were conducted. Similar strandings have been reported in the past two decades in Greece and in the Bahamas. 1, 17 Some common epidemiologic factors are notable among these strandings: (1) the oceanographic features were characteristic of steep-slope regions; (2) the species involved were predominantly BWs; and (3) these events were temporally associated with naval maneuvers that employed high-intensity, low- or midfrequency range sonar signals. 11, 17 These observations have been interpreted to suggest that BWs are found in association with particular oceanographic features and that they are either behaviorally or physiologically susceptible to the effects of sound exposure from certain types of anthropogenic sonar systems.
In the Greek stranding 17 and the stranding reported in this study, animals were in good body condition, without external lesions of collision trauma, and they had stomachs full of fresh ingesta. Conjunctival hemorrhages were common. In the Bahamas mass stranding of 2000, 3 of the 5 BWs were examined. These animals were in good body condition with food in their stomachs. The two freshest specimens had subcapsular renal hemorrhages, bilateral intracochlear hemorrhages, and unilateral temporal region and subarachnoid hemorrhages with blood clots observed bilaterally in the cerebral ventricles. It was concluded that these lesions were due to antemortem acoustic or pressure-related trauma caused by exposure to midfrequency sonar exposure. 11
The gross and histopathologic evaluations reported in this study indicate that the whales had no systemic, inflammatory, infectious, or neoplastic diseases. All the animals had similar pathologic findings of generalized congestion and acute, disseminated hemorrhaging that mainly affected the CNS, acoustic jaw fat, pharyngeal and laryngeal serosae, lungs, and kidneys. Impact trauma was initially considered a possible cause; however, with the exception of one whale with postmortem shark bites (BW-9), external trauma was not noted.
The clinicopathologic findings most similar to those of the animals of our study are those reported in acute DCS in human divers, 16, 20, 30, 46, 47 animals experimentally exposed to compression and decompression, 18, 37, 38, 59, 63 or animals exposed to underwater explosions. 32, 39, 64 According to navy sources, no explosions occurred during the naval exercise; thus, another energy source is thought to be the primary trigger of the pathogenic process involved in this event.
Detection of antemortem gas bubble accumulation within blood vessels and tissues can be difficult to demonstrate after death, because gases can dissolve back into surrounding tissues when the inciting injury ceases. 31, 38 Although many of the lesions noted in this study are those associated with the systemic gas bubble injuries that occur in acute cases of DCS in humans and laboratory animals, 3, 16, 18, 26, 33, 36, 37, 46, 58, 59 a diagnosis of DCS through identification of intravascular bubbles is made with caution.
The pathogenesis of fat emboli formation is still not fully understood, and it is likely multifactorial. 24 Fat embolism is infrequently diagnosed, but it has been described in cases of acute and subacute DCS. 26, 29, 31 It may arise from a number of different traumatic and nontraumatic processes. 24, 43, 49, 62 Pulmonary and systemic fat emboli that develop within hours of trauma are typically considered a consequence of injury to fat depots. 24, 62 Fat embolism also may occur by the coalescence of chylomicrons and intralipid liposomes in plasma during stress. 24 In both cases, intravascular lipid emboli can be demonstrated in several organs using histochemical methods on formalin-fixed tissues collected years earlier. 2, 7, 51 To our knowledge, this study provides the first description of fat emboli in cetaceans. 13, 25
Acute hemorrhaging in fat with necrosis of adipocytes, fat emboli in the lungs, epidural veins, and head and neck lymph nodes were identified in the freshest carcasses examined. These findings are similar to those observed with rapid fat emboli onset shortly after injury. 10, 24, 31, 49 Embolus distribution suggests that injured adipose tissue is the primary source. The large number of fat droplets and erythrocytes within the draining lymph nodes of the head and neck indicates this area was primarily affected by the pathologic process. No fat emboli were found within the vessels of the CNS or associated with hemorrhages in the cerebral sub-arachnoid spaces. It is presumed that the large vascular plexuses (including epidural and thoracic retia mirabilia) could protect the CNS from accumulating gas and fat emboli. 41 This hypothesis is supported by the observation that in BWs, disrupted adipocytes were often observed in association with hemorrhages, and fat emboli were observed within the epidural veins around the cervical spinal cord. The disruption of interstitial fat depots adjacent to veins could result from nitrogen bubble formation and growth and from tissue rupture that introduces both fat and gas into the venous blood stream, processes that are suspected of contributing to bubble formation in human DCS and in experimental animals after decompression. 18, 26, 29, 30
The low numbers of fat emboli found in the kidney were discovered in the medullary capillaries. Neither fat nor gas emboli were observed in glomeruli, implying that they were not circulating in the arterial blood delivered to the kidneys or that the renal circulation was shut down as in a dive mode. 21 Fat emboli were not associated with hemorrhages beneath the kidney capsule, where distended veins and disrupted capsular and interlobular connective tissue with large, nonfat-staining vacuoles (gas bubbles) were noted. It is speculated that the nitrogen bubble development observed in this study was merely nitrogen bubble escape from nitrogen-saturated tissues into free spaces and veins either by static or rectified diffusion. 6, 23
From the lack of published reports of stranded cetaceans (several hundreds of 15 different species) and on the basis of the experience of personnel in the Canary Islands and the UK (more than 2,000 cetaceans of several species), the incidence of fat emboli is presumed to be low in most of the species studied. A whale's agonal activity while stranded on the beach, a common scenario, does not commonly induce fat emboli. The distribution of fat emboli presented in this study, relative to other stranded cetaceans, and the conditions required for fat emboli development suggest that the generation of fat emboli in this study likely occurred before the animals became stranded. Further evidence that supports this conclusion is the observation that three whales with fat emboli were not stranded on the beach but were recovered offshore. Also, observations performed by the personnel present at the strandings indicated that animals were severely shocked and dying rapidly. The first dead animals were found 4 hours after the use of sonar began, so there was little time for the moribund whales to have developed the antemortem lesions while on the beach or rocks. These observations suggest the animals were severely injured before beaching and that gas and fat emboli were related to the injury and not secondary to the stranding event.
Although DCS is not always lethal, if severe, it can be rapidly fatal, or it can result in a more protracted syndrome leading to death. 16, 29, 31 Gas and fat emboli can cause nervous and cardiovascular dysfunctions, respiratory distress, pain, and disorientation. These signs common to human and experimental animal DCS 16, 18, 24, 27, 30, 36 may have been the main pathophysiologic process leading to the death and/or stranding of these BWs.
The mechanism of development of emboli remains to be determined. Three pathways were previously proposed for the development of the lesions observed in BWs that became stranded in the Bahamas: resonance, tissue diathesis, and acoustically mediated bubble formation. 11, 23, 44 Resonance and tissue diathesis could account for hemorrhagic trauma and fat emboli evolution, but they do not explain the presence of gas emboli or the distribution of hemorrhage and fat emboli within multiple tissues and organs. Acoustically mediated bubble formation provides a possible explanation for the observed lesions. 6, 14, 23, 25 Bubbles may grow via rectified diffusion, which is the growth or generation of bubbles driven by exposure to an acoustic field. 6, 23 Rectified diffusion includes the growth of bubbles through the diffusion of gas in the absence of a continuous acoustic exposure. According to this theory, the acoustic source destabilizes existing bubble nuclei, which grow due to the outflow of gas from gas-saturated tissues. 6, 23 Another proposed mechanism is that BWs are startled by an intense acoustic exposure and modify their surfacing behavior. In this way, supersaturation and the probability of bubble formation are increased due to changes from their normal dive profiles. 14, 23, 25, 45
Because nitrogen is more soluble in lipids than in other biologic tissue, there would be a propensity for bubble growth in lipid-rich tissue. 13, 18, 23, 24, 26, 29, 30, 44 The postprandial and presumably hyperlipidemic state of these whales may have favored lipid aggregation in blood. A greater degree of supersaturation should decrease the threshold for these putative processes.
Dolphins and human divers are known to develop relatively high levels of nitrogen supersaturation resulting from cycles of compression and decompression during repetitive, breath-hold diving. 6, 23, 52, 56 However, dolphins (and presumably other cetaceans) have evolved adaptations that mitigate the accumulation of excess nitrogen in tissues. 21, 34, 50, 54, 56, 57 The most notable adaptation is lung or alveolar collapse, which stops gas exchange at the depth when alveoli collapse. 21, 56, 57 In the presence of an intense energy field, the degree of tissue saturation required for bubble formation may decrease. 6, 23 It has been hypothesized that BWs should have a greater degree of tissue–nitrogen saturation than most cetaceans because of their slow and deep-diving behavior. 22, 23, 65
Damage to the acoustic fat of the head may provide clues to understanding the etiology of the observed lesions. Sound impulses are thought to be directed to the ear via the specialized acoustic fat channels of the lower jaw. 21 Hemorrhages were observed in the fatty acoustic channels of the jaw of all atypically mass-stranded BWs. Strong acoustic signals will affect these well-vascularized lipid-rich channels and could result in rapid bubble activation. Such a situation could result in rapid bubble activation driven by both tissue nitrogen off-gassing and continued acoustic exposure. 6, 23
The development of gas bubbles within tissues could have produced hemorrhages and necrosis, acute spongiotic changes in the CNS, and gas/fat venous embolism. Some of these lesions occur during the initial phases of acute DCS and DON lesions in humans and experimental animals and in these mass-stranded BWs. 18, 26, 29, 30, 59, 63
Gas and fat emboli released into the venous system and deposited in the pulmonary capillary beds may travel through arteriovenous shunts into the systemic circulation. 16 Respiratory and cardiovascular dysfunctions may occur with a biphasic response at the brain–spinal cord level: an initial venous embolic obstruction and vasoconstriction, followed by a secondary vasodilatation and prolonged (reactive) hyperemia. 60 This hemodynamic process may explain the widespread cerebral congestion and edema with spongiosis, intracranial perivascular hemorrhages, and subarachnoid and intraventricular hemorrhages that have been described in the BW mass strandings.
Unfortunately, evaluation of portomesenteric vasculature was not performed; however, the gas bubbles observed in liver sinusoids are similar to those reported in portomesenteric vessels following decompression in humans 4, 48 and in experimental DCS in dogs. 3 Subsequently, these acute lesions have been found by our group in two recently single-stranded BWs (M. densirostris) (unpublished observation) and have been observed in stranded cetaceans in the UK. 25
Gas embolism in the inner ear has been described in DCS. 27, 36 Emboli may induce otic hemorrhages and contribute to disorientation and/or vestibular dysfunction. Similar lesions have been described in other, sonar-associated mass strandings of whales. 11 Investigators should carefully dissect and sample ear structures to more completely document the potential contribution of fat and gas emboli in otic tissues to this stranding syndrome.
In summary, this study presents lesions in cetaceans that are similar to those of acute decompression-like disease in humans and laboratory animals. Evidence of gas bubbles, presumably nitrogen, is presented, and vascular and tissue changes consistent with those caused by gas bubble injuries and fat emboli were demonstrated in the vital organs of BWs from an atypical mass stranding. The stranding was temporally and spatially coincident with a naval exercise employing midfrequency sonar. The animals were presumed to have died or to have been stranded with a DCS-like disease and died shortly after with severe cardiovascular failure brought on by “stranding stress syndrome.” 9, 11 Researchers aware of the syndrome of gas bubbles and fat emboli should be better prepared to document the lesions in the event of similar mass strandings.
Gas and fat emboli should be considered in stranded cetaceans suspected of having been exposed to sonar. Comparative data should be collected from unexposed controls to gather evidence about the conditions and mechanisms by which acoustic exposure induces lesions. Further investigation of the behavioral 19, 45, 65 and physiologic effects of sonar on cetaceans is important to elucidating the pathogenesis of this syndrome.
Footnotes
Acknowledgements
This research was funded by the Ministry of Science and Research (Ren-2002-04162-C02-01/MAR), GD Fisheries (EU), and Ministry of Defense and Canary Islands Regional Government. We are also grateful to Dr. A. Gómez, A. Fernandez Jr., Dr. P. Jepson, Dr. D. Hauser, Dr. S. Rommel, J. Rico, and Dr. P. Tyack for constructive comments on and support for this work, as well as to J. A. Santiago, E. Degollada, E. Curbelo, A. Sosa, A. M. Afonso, E. Sierra, M. Mendez, H. Lorenzo, A. Castro, P. Calabuig, M. Carrillo, E. Urkiola, T. Gallardo, Dirección General Medio Natural, Cabildo de Gran Canaria, Cabildo de Fuerteventura, Cabildo de Lanzarote, and other anonymous contributors for providing assistance and support for some parts of the present work.
