Abstract
Stejneger’s beaked whales (Mesoplodon stejnegeri) are one of the lesser known species of mammals, with little information available on their population status or incidence of diseases. Recent pathologic investigations on stranded and bycaught wild cetaceans around Hokkaido, Japan, revealed an unusually high incidence of systemic amyloidosis in this species, warranting further investigation. The objective of this study was to further characterize the systemic amyloidosis of Stejneger’s beaked whales by retrospective histopathologic analyses of tissues from animals that stranded in Japan between 1994 and 2018. Various tissues from 35 individuals were examined histologically with hematoxylin and eosin, Congo red, and immunohistochemistry for amyloid A (AA), in which 12 (34%) were diagnosed with systemic amyloidosis. The organs with the highest severity of amyloid deposition were the stomach and intestine. The type of amyloid was confirmed as AA of approximately 9 kDa by 2-dimensional gel electrophoresis with extracted amyloid from the liver and subsequent Western blotting with an antiserum against AA peptide. There were no statistically significant associations between amyloidosis and sex, body condition of the whales, or the presence of chronic inflammation. The high prevalence of this disease might be of concern for overall population numbers, and continued pathologic monitoring of stranded animals is necessary throughout its distributional range.
Stejneger’s beaked whales (Mesoplodon stejnegeri) belong to the genus Mesoplodon, one of the lesser known members of mammals. 5 This species is known to inhabit temperate to subpolar, deep waters of the North Pacific basin 20 and has been rarely sighted at sea. Due to the inadequate records of live sightings, information on their population status or trends in abundance is lacking, and hence the Stejneger’s beaked whale is listed as Data Deficient in the International Union for Conservation (IUCN) Red List. 31 Likewise, disease accounts for this species are limited with 3 sporadic cases of amyloidosis 27,30 and 2 reports limited primarily to gross findings. 29,35 However, recent pathologic investigations on 11 species of stranded and bycaught wild cetaceans around Hokkaido, Japan, revealed an unusually high incidence of amyloidosis, presumably of the amyloid A (AA) type, specifically in Stejneger’s beaked whales (2/3 animals identified with amyloidosis), 23 warranting further investigation.
AA amyloidosis, the systemic form of the protein misfolding disease characterized by AA deposition in various organs, is the predominant type of amyloidosis in animals. 8 Chronic inflammation, infectious disease, or neoplasia is usually the prerequisite for disease onset, which results in persistently elevated concentrations of the serum amyloid A (SAA) in blood. 28 However, the exact mechanism by which soluble circulating SAA converts into insoluble aggregates of AA fibrils is not yet fully understood. 21 SAA is a highly conserved protein ranging in size from 104 to 112 amino acids, 33 and the N-terminal domain of these various SAA isoforms is the key in determining the amyloidogenic capacity of a particular isoform. 16
In free-ranging wild mammals, AA amyloidosis was suspected on the basis of immunohistochemistry (IHC) in 2 Stejneger’s beaked whales as mentioned above, 23 26 California sea lions (Zalophus californianus), 2 13 black-footed cats (Felis nigripes), 32 2 hares (Lepus europaeus), 12 a European pine marten (Martes martes), 19 and numerous beech martens (Martes foina). 11 Furthermore, AA amyloidosis is confirmed by amino acid sequencing in 69 island foxes (Urocyon littoralis) 9 and 2 red foxes (Vulpes vulpes). 25 While many of the wildlife cases are considered sporadic occurrences, the island foxes and beech martens are especially prone to developing this condition at the species/subpopulation level, with a prevalence of about 34% and 10%, respectively, posing a potential risk to recovery and survival in some populations. 9,19
The purpose of this study was to document the features of systemic amyloidosis in Stejneger’s beaked whales through a retrospective analysis of an extensive case series by investigating the prevalence of this disease and its organ distribution, analyzing the responsible protein, and identifying possible risk factors.
Materials and Methods
Animals
The study population consisted of 35 Stejneger’s beaked whales of all age groups and both sexes that stranded along the coast of Japan between 1994 and 2018. The total known number of stranded Stejneger’s beaked whales in Japan during this period was at least 218 (Marine Mammals Stranding DataBase, National Museum of Nature and Science, accessible at www.kahaku.go.jp/research/db/zoology/marmam). Cases selected for evaluation in this study were in a fresh or only moderately autolyzed condition (decomposition codes of 2 or 3 14 ) and had the majority of organs available for retrospective histopathologic examination. All animals used in this study were free-ranging in origin, either stranded dead or stranded alive but died shortly after without any human interventions. Previously published cases of amyloidosis 23,27,29,30 included 5 animals that were further investigated in this study.
Age groups of the animals were classified into the following 4 categories according to their body size (measured from the tip of maxilla to center of fluke), also taking into account factors such as body coloration, size of erupted teeth, skull morphology, and fetal creases: 191 to 230 cm as newborns, 231 to 300 cm as calves, 301 to 440 cm as juveniles, and 441 to 550 cm as adults. In addition, body conditions of poor, fair, and good were specified based on the degrees of (1) concavity ventrolateral to the dorsal fin (epaxial muscle mass) and (2) depression posterior to the blowhole through meticulous examinations of photographic records. The details of the 35 animals are presented in Supplemental Table S1.
Histopathologic Analysis
Histopathologic slides were prepared from archived formalin-fixed tissues and processed routinely. Available tissues from the liver, spleen, kidney, heart, lung, stomach, intestine, thyroid gland, pancreas, adrenal gland, gonad, uterus, mammary gland, urinary bladder, lymph node, skin, and brain were examined. Sections were stained with hematoxylin and eosin (HE) and Congo red (CR), in which the CR-stained sections were viewed under polarized light for confirmation of the green birefringence of amyloid. The tissue distribution of amyloid deposits in each animal was noted. A semiquantitative grading scale was applied to 7 selected organs: liver, spleen, kidney, heart, lung, stomach, and intestine. For the liver, spleen, and kidney, the following grading criteria were applied: − = no amyloid detected; liver, + = deposition primarily confined to the vascular walls but not apparent within the space of Disse, ++ = deposition extending into the space of Disse but with only mild changes to hepatocytes, +++ = severe deposition in the space of Disse with compression and reduced number of hepatocytes leading to distortion in the tissue architecture; spleen, + = deposition confined to vascular walls, ++ = deposition mainly in the marginal zones with no severe changes in the follicular structure, +++ = severe and extensive deposition in the follicles that displace the existing follicular structure and reduce the number of follicular lymphocytes; and kidney, + = minimal deposition limited to the interstitium of the medulla, ++ = mild to moderate, multifocal deposition in the interstitium of the medulla and mild segmental deposition in <25% of glomeruli, +++ = moderate to severe, multifocal to extensive deposition in the interstitium of the medulla and moderate to severe segmental to global deposition in ≥25% of the glomeruli. Representative histologic images of the applied grading scheme are shown in Supplemental Figures S2 to S10. The remaining heart, lung, stomach, and intestine were graded following previously reported criteria, with the same aforementioned 4 categories: −, +, ++, and +++ (− = no amyloid detected, + = minimal to mild deposition, ++ = moderate deposition with mild disruption of normal architecture, +++ = severe deposition with disruption of normal architecture and/or evidence of secondary tissue damage). 32
To determine the organs with the highest severity of amyloid deposition, scores of 0 to 3 were applied to correspond to −, +, ++, and +++, respectively (− = 0, + = 1, ++ = 2, +++ = 3). These scores were applied to the 7 organs in every amyloid-positive animal, and the mean scores for each organ were calculated.
All additional concurrent lesions were also documented to investigate for any associations between amyloidosis and other diseases.
Immunohistochemical Characterization
Immunohistochemistry for AA was performed on all available tissues of amyloid-positive individuals and on representative tissues of all amyloid-negative individuals using a mouse monoclonal antibody against AA (1:600, clone KM268; Kyowa Medex, Tokyo, Japan) as described previously. 23 A liver section of a Holstein cow (Bos taurus) with AA deposits served as a positive control, while tissue sections in which the primary antibody was replaced by normal mouse serum served as negative controls.
The validity of IHC was confirmed with the use of another antibody, rabbit antiserum raised against a synthetic peptide of mouse SAA1 (GHEDTMADQEAN; corresponding to positions 90–101 of mice SAA). This was applied to representative organs of each amyloid-positive whale with the same labeling methods. Normal rabbit serum in place of the primary antibody served as a negative control.
Statistical Analyses
The significance of sex, body condition, and chronic inflammation (renal crassicaudiasis) as factors for the occurrence of amyloidosis were evaluated using Fisher’s exact test or χ2 test, with P < .05 considered significant. Correlation of amyloidosis with age classes was not statistically determined due to the limited sample sizes of newborns, calves, and juveniles.
Amyloid Fibril Extraction
Frozen liver tissues of both amyloid-positive (n = 2) and amyloid-negative (n = 1) Stejneger’s beaked whales, confirmed by histopathology, were used for the extraction. The procedure was carried out according to Pras et al 24 with some modifications as follows.
Liver tissue weighing 1.5 g and stored at –25°C was thawed and subjected to initial homogenization (Multi Beads Shocker; Yasui Kikai, Osaka, Japan) under 40 000 × g twice, each for 1 minute. The homogenates were further processed by adding 15 ml saline and homogenized with a homogenizer (VP-050 N; TAITEC, Saitama, Japan) twice, each for 1 minute on ice. The suspension solution was centrifuged with an ultracentrifuge (Optima L-100XP; Beckman Coulter, Brea, CA) at 4°C, 35 000 × g for 50 minutes, and the supernatant was discarded. Subsequently, 15 ml saline was added to the remaining material, and homogenization and centrifugation were carried out as above. These operations were repeated 10 times. The final residue was homogenized with 15 ml distilled water, centrifuged at 4°C, 35 000 × g for 45 minutes, and the supernatant was discarded. The residue was homogenized again with 15 ml distilled water and left at 4°C overnight. The homogenate was then centrifuged at 4°C, 35 000 × g for 45 minutes, and the supernatant was collected. This collection procedure was repeated 3 times to end up with 45 ml supernatant. The supernatant was centrifuged at 4°C, 125 000 × g for 60 minutes, and the residue, suspended with a small amount of distilled water, was collected for analysis. To confirm the presence of amyloid fibrils, the collected solution was subjected to CR staining. The extracted material of both the amyloid-positive and amyloid-negative livers was stored at –80°C until use.
Two-Dimensional Gel Electrophoresis and Immunoblotting
Prior to the 2-dimensional (2-D) gel electrophoresis of the extract, the materials were purified with the ReadyPrep 2-D cleanup kit (Bio-Rad, Hercules, CA) according to the manufacturer’s instructions. About 90 µg protein was resolved by isoelectric focusing (IEF) on 7 cm Immobiline Dry-Strips (pH 4–7; GE Healthcare Bio-Sciences, Pittsburgh, PA) using a PROTEAN IEF Cell system (Bio-Rad) at 20°C for 12 hours for passive rehydration, then separated for 15 minutes at 250 V, 1 hour at 4000 V, and 10 000 V-h at 4000 V, with a maximum current of 50 μA. After the IEF, each strip was equilibrated in 6 M urea, 1 M Tris HCl (pH 8.8), 20% glycerol, 2% sodium dodecyl sulfate (SDS), and 2% dithiothreitol (DTT) for 20 minutes, followed by a further 10 minutes of equilibration in the same solution containing 2.5% iodoacetamide instead of DTT. Resolved proteins were further separated by SDS–polyacrylamide gel electrophoresis on 15% acrylamide gels in Tris/glycine/SDS buffer under standard conditions. Protein spots were detected by staining with a Coomassie brilliant blue R250 (CBB) stain. The extract from an amyloid-negative liver was used as a negative control.
To detect the AA protein by Western blot and to determine its molecular weight, the resolved proteins on acrylamide gels were transferred to polyvinylidene difluoride membranes (Immobilon P; Millipore, Billerica, MA) at a constant voltage of 20 V for 40 minutes using a Trans-Blot SD Semi-Dry Transfer Cell (Bio-Rad). The blots were washed with phosphate-buffered saline with 0.1% Tween 20 (PBST) and then blocked for 1 hour at room temperature with 1% skim milk in PBST. Subsequently, the membrane was incubated overnight with the same rabbit antiserum against mouse SAA1 synthetic peptide used for IHC at 4°C. The membrane was incubated with a horseradish peroxidase–conjugated anti-rabbit IgG secondary antibody (1:5000; GE Healthcare Life Sciences, Pittsburgh, PA) for 1 hour at room temperature. A chemiluminescence substrate (Clarity Western ECL Substrate; Bio-Rad) was used to detect positive spots using the ImageQuant LAS 500 system (GE Healthcare Life Sciences). As negative control, the primary antibody was omitted and replaced with normal rabbit serum.
Results
Prevalence
The 35 examined Stejneger’s beaked whales comprised 16 females and 19 males that ranged in body length from 191 to 525 cm, including a newborn, a calf, 3 juveniles, and 30 adults (Fig. 1). Most of these animals stranded along the Sea of Japan, while 1 was found along the Pacific Ocean, ranging from latitudes of 33°N to 45°N (Suppl. Fig. S11).
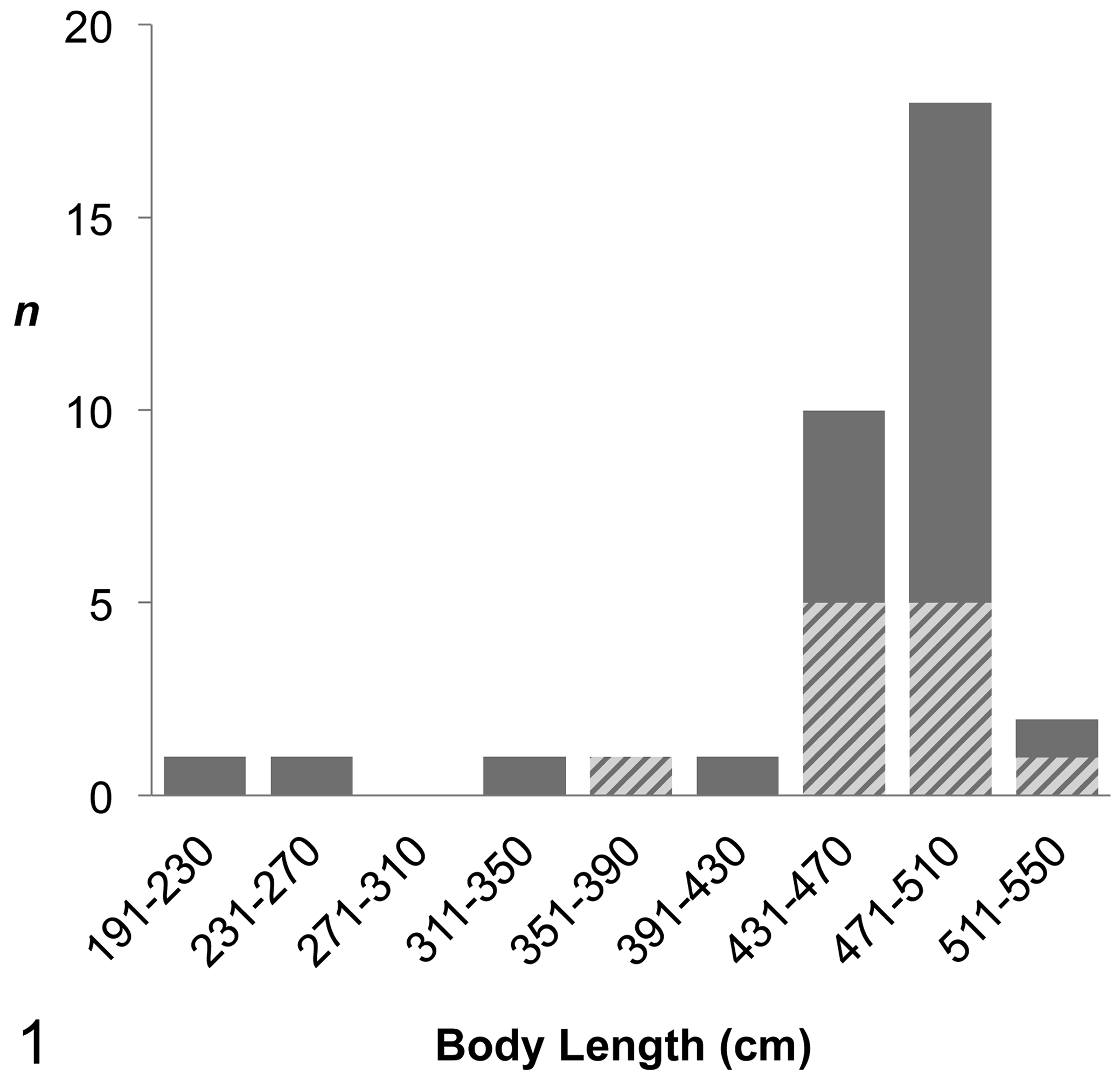
Number of Stejneger’s beaked whales examined by body length (n). Cross-hatched bars indicate the number of animals diagnosed with systemic amyloid A amyloidosis.
Of the 35 whales, 12 were diagnosed with systemic amyloidosis through examinations of HE- and CR-stained sections, indicating a prevalence of 34% in the examined population. The 12 whales included 7 females and 5 males; there was no significant sex difference (P = .311). The body length of these amyloid-positive individuals ranged from 389 to 520 cm (Fig. 1), where 11 of the 12 whales were in the adult class. The only nonadult animal (case No. 4) was a male estimated to be approximately 2 years old based on size, coloration, and lack of tooth eruption. The body conditions of animals with amyloidosis varied (3 “poor,” 4 “fair,” and 5 “good”), and statistically significant correlations were not obtained (P = .342). The strandings of whales with amyloidosis occurred north of 36°N in latitude, but there was no apparent geographic pattern.
Histopathology and Organ Distribution
Evaluation of HE- and CR-stained sections revealed a widespread distribution of amyloid deposits that were always immunohistochemically positive with an anti-AA antibody. Cases diagnosed as amyloid-negative on HE sections were never positive on IHC. Deposits were consistently detected in the liver, spleen, kidney, and gastrointestinal tracts of all whales with amyloidosis (Table 1). The organ with the highest mean deposition score was the intestine (2.67), followed by the stomach (2.56), while the liver (2.33), spleen (2.33), and kidney (2.27) also had high scores.
Organ Distribution and Severity of Amyloid Deposition in 12 Stejneger’s Beaked Whales With Systemic Amyloid A Amyloidosis.

Abbreviations: BL, body length (cm); F, female; M, male; NA, not available. Grading criteria: −, no amyloid detected; +, mild; ++, moderate; +++, severe.
In the gastrointestinal tract, amyloid deposits were predominantly found in the lamina propria and less in the muscularis mucosa, interstitium of muscularis, and tunica media of arteries and arterioles (Figs. 2, 3). The liver of animals with severe amyloid deposition had hepatocytes that were severely compressed and reduced in number and smaller in size due to substantial deposits in the space of Disse (Fig. 4). There were no considerable differences in the amount of amyloid deposition between different liver lobes in these individuals, except for that of the youngest amyloid-positive whale (case No. 4), which varied from no deposits to marked deposition between lobes, with only about half the hepatocytes remaining in some of the more severely affected areas. Deposits in the spleen were always associated with the tunica media of arterioles. The splenic follicles were also commonly affected, either by deposits surrounding the follicles or almost completely effacing the germinal centers. In the kidney, multifocal to coalescing amyloid deposits were observed in the peritubular interstitium of the medulla. Massive deposits were also common in the papilla, and the tubules in the severely affected areas were atrophic as well as reduced in number (Fig. 5). Amyloid deposits were detected in the glomeruli in 82% (9/11) of amyloid-positive animals with kidneys examined, which all had medullary amyloidosis. The affected glomeruli had segmental amyloid deposition that mildly expanded the mesangium. Two animals (case Nos. 9 and 30) had more prominent amyloid deposition in the glomeruli, leading to a global expansion of the mesangium. The heart and lung in the majority of animals were either negative or only mildly affected by amyloid deposits. These deposits were found in the myocardial interstitium or tunica media of various arteries and arterioles, with mean deposition scores of 1.13 and 0.33, respectively. The severities of deposition in the selected 7 organs are summarized in Table 1.
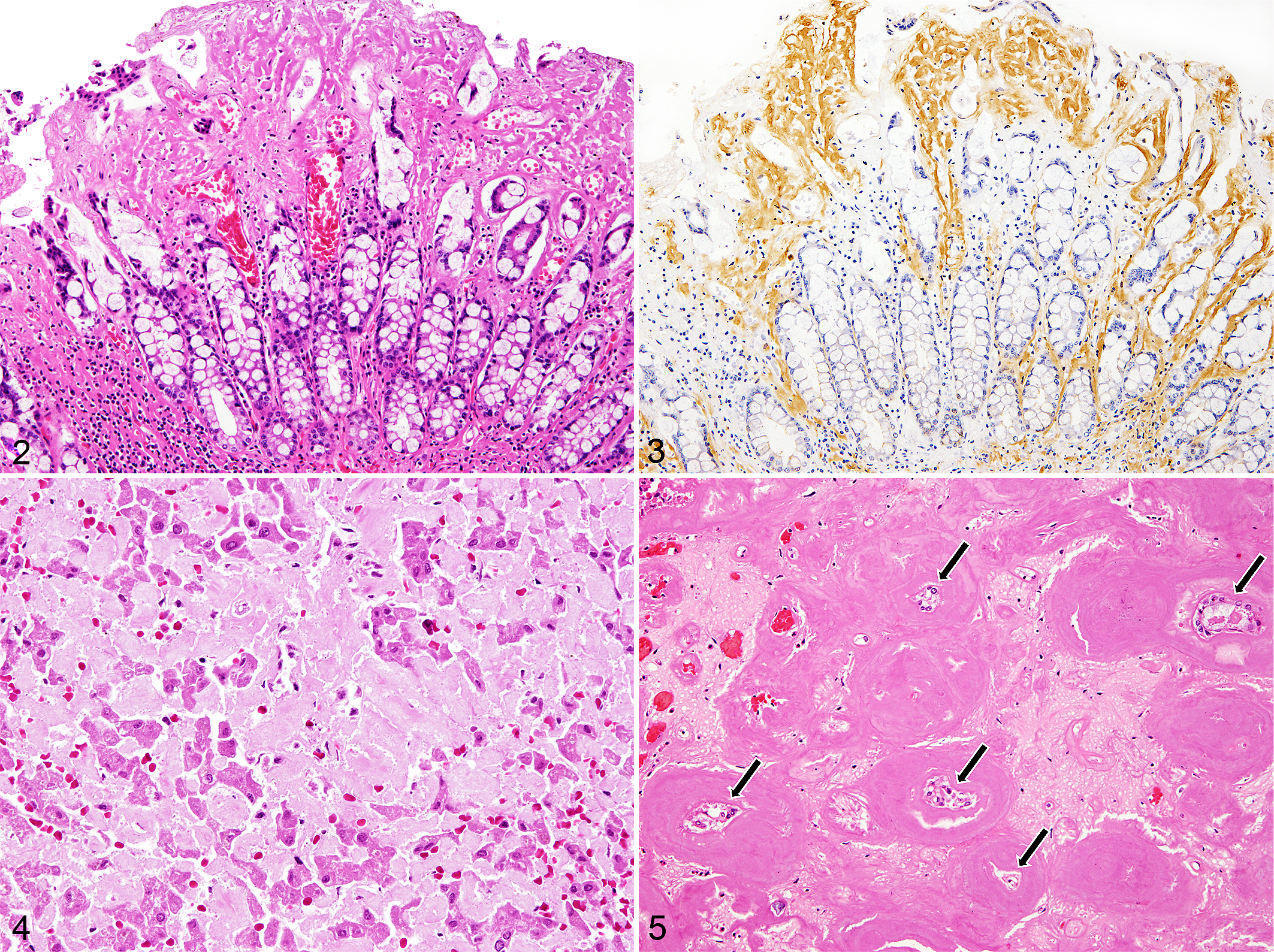
The thyroid gland (4/6, 67%), adrenal gland (6/7, 86%), pancreas (2/4, 50%), uterus (1/2, 50%), choroid plexus (2/2, 100%), thymus (1/1, 100%), and lymph node (3/4, 75%) were also sites with deposits of varying consistency, primarily in the interstitium and vascular walls. There were no amyloid deposits in the urinary bladder (0/4).
Concurrent Lesions
Renal crassicaudiasis, a cause of chronic inflammation, was the most frequently recorded condition in the 35 whales, with the 3 smallest individuals being the only animals without the parasite (91%; Suppl. Fig. S12). Histologically, the lesions were composed of mature granulomas containing the Crassicauda nematode or mineralized foci, effacing the renal parenchyma with areas of marked, extensive fibrosis (Suppl. Fig. S13). There was no significant correlation between this renal parasitism (and associated inflammation) and amyloidosis (P = .536), and no additional diseases were commonly shared among whales with amyloidosis.
Other diseases identified included pulmonary mycosis, bronchopneumonia, adrenal hematoma, hepatic biliary proliferation, balanitis, and polyglucosan body deposition in the heart and cerebellum (Suppl. Table S1). Cardiac lipofuscinosis was found in 4 whales, of which 2 were those with systemic amyloidosis.
AA Protein Analysis
The CBB-stained gel of both the amyloid-positive and amyloid-negative liver extracts revealed numerous spots, while the Western blot using an antibody against mouse SAA1 showed reactivity with 2 dominant spots at a molecular weight of approximately 9 kDa and 2 additional spots at higher molecular weights of approximately 18 and 36 kDa, suggestive of dimer and trimer structures (Fig. 6). The spots with Western blot reactivity were observed on the cathode end of the gel, indicating a positively charged protein. These spots were absent in the amyloid-negative extract.
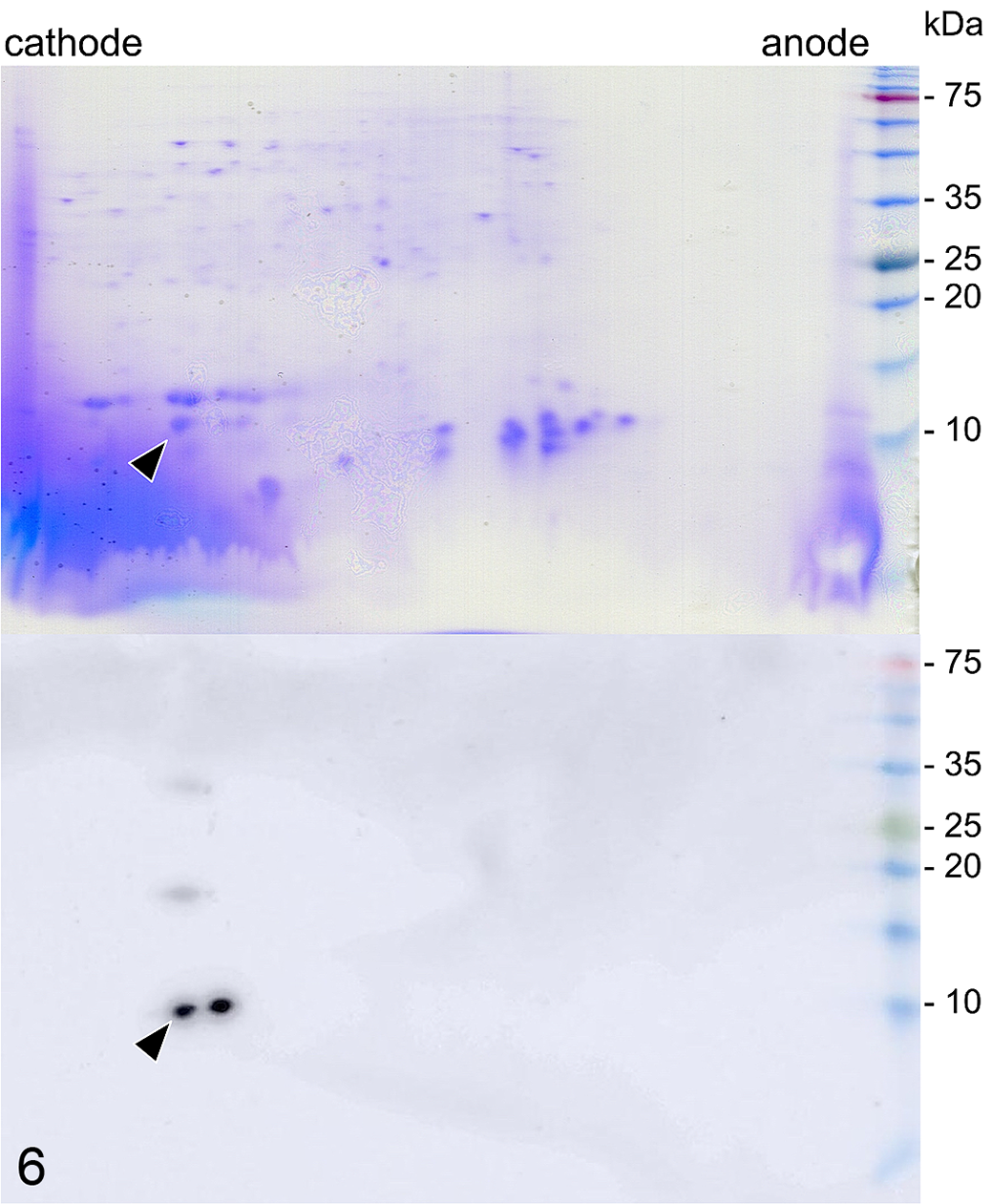
Two-dimensional gel electrophoresis (top) and Western blot (bottom) of extracted proteins from amyloid-laden liver, Stejneger’s beaked whale. Coomassie blue stain shows numerous spots (top). The immunoblot using an antibody against a mouse SAA1 peptide (bottom) shows 2 dominant spots near the cathode end, at a molecular weight of approximately 9 kDa, and 2 additional spots at molecular weights of approximately 18 and 36 kDa. Arrowheads indicate the target dominant amyloid protein.
Discussion
The current study identified that systemic AA amyloidosis is a highly prevalent disease in stranded Stejneger’s beaked whales around Japan, at a prevalence of 34%. The distribution of amyloid deposits was widespread in all the whales with this condition. The amyloid deposition was most severe in the gastrointestinal tract, liver, spleen, and kidney, which were considered sufficient to cause disease in the majority of animals. Although no particular risk factor was determined, the unusually high prevalence of AA amyloidosis in the population, which also affected a juvenile whale, may suggest a species predilection to developing systemic AA amyloidosis.
Of the 10 types of amyloidosis known in animals, 6 (AA, AL, AApoAI, AApoAII, ATTR, AFib) are considered to be systemic in distribution, and the AA type is by far the most common. 28 In cetaceans, despite the vast numbers of pathologic descriptions to date, reports of amyloidosis are extremely limited, of which systemic amyloidosis has been reported only in the Stejneger’s beaked whales 23,27,30 and bottlenose dolphins (Tursiops truncatus). 3 Moreover, the type of deposited amyloid has been determined by IHC in 2 species, as AA in the Stejneger’s beaked whale 23 and as Aβ in the striped dolphin (Stenella coeruleoalba). 15 The current study further supports the previous results of IHC by Western blot reactivity to an anti-SAA antibody and confirms the type of deposits in the systemic amyloidosis of Stejneger’s beaked whales as AA.
AA amyloidosis is a disease often secondary to cyclic or prolonged elevation of circulating SAA levels in response to chronic inflammation. 28 Persistent parasitic infections such as leishmaniasis 13 and filariasis 4 have been recognized as one of the causes of amyloidosis, presumably due to the same pathogenesis through chronic inflammation. 1 In the case of Stejneger’s beaked whales, renal crassicaudiasis was once suggested to be a contributing factor for the onset of amyloidosis. 30 While the parasite was identified in all animals with amyloidosis, the majority of animals without amyloidosis also had this condition, resulting in an overall infection rate of 91% in the sample set. This fact indicates that the influence of Crassicauda parasitism alone was not a significant risk factor in the development of amyloidosis in this species, and additional mechanisms were considered to take part. Furthermore, crassicaudiasis is a common parasitic infection in beaked whales (Ziphiidae), 6,18,29 but amyloidosis has never been reported in other ziphiids, indicating further support to the idea of a species predisposition.
Other commonly proposed contributors of AA amyloidosis include polymorphisms in the SAA gene and mutations in the genetic components outside the SAA genes such as the promoter, enhancing elements and transcription factors. 7,8 In the island foxes, an amyloidogenic SAA has been described and their high prevalence for AA amyloidosis is proposed to be due to a lack in genetic heterozygosity. 10,26 Although not much is known on the genetics of Stejneger’s beaked whales, their genetic pleomorphism seems quite limited, 17 which suggests a similar genetic predisposition in these whales. Moreover, one of the juveniles developing this condition early in life further supports the idea of an inherited predilection, similar to familial amyloidosis in humans. 28 The exact age of the examined whales was undetermined, but the fact that age-related histologic changes such as cardiac lipofuscinosis being rarely noted suggest that these animals were not senescent.
It is noteworthy that there were no significant correlations between body condition and amyloidosis, even though the severity of deposition was considered sufficient to cause malabsorption and/or protein loss through the gastrointestinal tract 34 as well as hepatic and renal failure, which might all potentially contribute to the stranding and death. This seeming inconsistency may be explained by the anatomic characteristics of cetaceans. The cetacean body is surrounded by a thick layer of blubber that can be as thick as 6 cm in Stejneger’s beaked whales, allowing them to survive in a temperate aquatic environment. Since the decrease in blubber can lead to hypothermia sooner in temperate waters than in the tropics, whales living in higher latitudes may die before becoming visually emaciated. 14 To support this theory, whales that stranded with systemic amyloidosis in good body condition were all found in latitudes higher than 41°N. Accurate assessment of the clinical manifestations of amyloidosis in dead stranded marine mammals ranging over wide water temperatures proved to be challenging.
Interestingly, the status of Stejneger’s beaked whales around Japan has changed drastically over the recent years. They were historically thought to be rare around Japan, having a primary distribution in the colder waters around Alaska. 22 However, this view started to change in the late 1980s when more than 20 whales stranded along the Sea of Japan in a period of 4 years, which included pregnant females with full-term fetuses. 36 Thereafter, strandings of this species in Japan had been stable in the high end, with the number of strandings during the first 12 years of this study period (1994–2005) being 145 (Marine Mammals Stranding DataBase, National Museum of Nature and Science). However, in the latter 13 years (2006–2018), the number of strandings decreased to 73 (Suppl. Fig. S14). Since data on the abundance of Stejneger’s beaked whales are unavailable on a global scale, we are uncertain if these stranding data reflect a declining population trend or merely a change in its distribution. Nonetheless, the unusually high prevalence of AA amyloidosis in the population is alarming.
The high incidence identified in this species might suggest an underlying genetic involvement in the development of systemic AA amyloidosis in Stejneger’s beaked whales, which requires further investigations into the molecular pathogenesis. Pronounced systemic amyloid deposition in the majority of diseased animals in this species is of concern to their health status. We speculate that the disease might contribute to the seemingly declining population, at least around Japan.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Nakagun_et_al - Systemic Amyloid A Amyloidosis in Stejneger’s Beaked Whales (Mesoplodon stejnegeri)
Supplemental Material, Combined_supplemental_materials-Nakagun_et_al for Systemic Amyloid A Amyloidosis in Stejneger’s Beaked Whales (Mesoplodon stejnegeri) by Shotaro Nakagun, Kenichi Watanabe, Yuko Tajima, Tadasu K. Yamada and Yoshiyasu Kobayashi in Veterinary Pathology
Footnotes
Acknowledgements
We thank all individuals, personnel, agencies, aquaria, respective municipal offices, and fishery cooperatives involved in the recovery and on-site examination of the beaked whale carcasses over the years, including Prof. Takashi Matsuishi from Stranding Network Hokkaido and its members. Likewise, we thank the Cetology Study Group of Japan for its continuing collaboration. We also thank Karin Uesaka for her assistance in protein analysis and Akiko Tomikawa for her excellent technical assistance throughout the research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grants-in-Aid for Scientific Research (KAKENHI) from the Japan Society for the Promotion of Science (#19J10574) and Sasakawa Scientific Research Grant from The Japan Science Society (#28-736 and #2018-7012).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
