Abstract
The major drawback of the current common bile duct ligation (CBDL)-induced hepatopulmonary syndrome (HPS) animal model is the extremely high mortality rate that hinders experimental studies. The purpose of this study was to investigate an improved method of CBDL with the goal of developing a simple and reproducible rat HPS model after a single CBDL treatment. Two groups of male Sprague–Dawley rats underwent separate methods of CBDL: (1) the upper common bile duct ligation (UCBDL) group (n = 40), in which the first ligature was made near the junction of the hepatic ducts, and the second ligature was made above the entrance of the pancreatic duct; (2) the middle of the common bile duct ligation (MCBDL) group (n = 40), in which the first ligature was made in the middle of the common bile duct, and the second ligature was made above the entrance of the pancreatic duct. The CBDL-induced HPS rats were evaluated by pulse oximeter, arterial blood analysis, histopathology, and cerebral uptake of intravenous technetium-99m-labeled albumin macroaggregates (which reflects intrapulmonary vascular dilation). The mortality rates of the UCBDL group and the MCBDL group were 42.5% and 77.5%, respectively (P < 0.05). These results suggest that the UCBDL, a single improved procedure, provides a better method compared to the established HPS model, because of the relatively high success rate and the decreased risk of complications.
Keywords
Hepatopulmonary syndrome (HPS) is characterized by a desaturation of oxygen in the arterial blood of patients with chronic liver disease.1,2 Three important components of HPS are hypoxemia with an increased alveolar–arterial oxygen partial pressure difference (AaPO2), increased intrapulmonary shunts, and chronic liver disease typically comprised of cases of portal hypertension and liver cirrhosis. 3 The results from liver transplantation centers indicate that the prevalence of HPS in cirrhotic patients ranges from 5 to 32%. 4 In the past, treatment was unsatisfactory, with the exception of liver transplantation, which has been effective.5,6 The pathophysiology of HPS continues to be investigated. 7
At present, the treatment of HPS is a challenging problem in clinical practice. Experimental studies are ongoing to develop new treatment modalities to prevent pulmonary vascular changes (such as medicines) and surgical techniques. Experimental induction of liver disease in animal models can accurately replicate the histological, biochemical, hemodynamic, renal and neurohumoral alterations seen in human liver dysfunction.8–11 Handling risks to the researcher have been eliminated, and the use of such models improves our understanding of the physiopathology of liver disease and of other involved syndromes. We can follow the physiopathological progress of cirrhosis and its complications through experimental studies in animal models of liver cirrhosis, secondary biliary cirrhosis and portal hypertension. An ideal experimental animal model of HPS should include hypoxemia with an increased AaPO2 and elevated degrees of intrapulmonary shunting. Many animal models have been studied for HPS, including the partial portal vein ligation (PPVL) model, thioacetamide-induced fulminant hepatic failure, and common bile duct ligation (CBDL)-induced biliary cirrhosis.12,13 Among these models, chronic CBDL in the rat is the only successfully established experimental model of human HPS. 2 Although this model mimics the characteristics of human HPS, the major disadvantage of the currently described CBDL-induced HPS animal models is an extremely high mortality rate that hinders the experimental studies or a relatively low recurrence rate of HPS requiring re-treatment. Due to bile duct obstruction, intraluminal pressure rises markedly, and numbers of bile duct cells also increase. 14 In our previous study significant cysts of common bile duct after ligation were found following an increase in CBDL duration, as has been reported elsewhere in the literature,7,15,16 and autopsy of dead CBDL rats has also found many ruptured bile ducts. In addition, dilated common bile duct rupture after ligation tends to result in a series of serious complications, and this may be an important factor in the death of CBDL rats.
To develop a simple and reproducible rat HPS model with CBDL after a single treatment, we used two methods of CBDL: middle of the common bile duct ligation (MCBDL) and upper common bile duct ligation (UCBDL). This approach has not previously been reported for rats. This study first proposed UCBDL and MCBDL, compared the results of the methods, and evaluated their reliability, practicality and application in parallel experiments. Defining well-characterized and easily accessible animal models that mimic human diseases is critical for exploring their pathogenic features and mechanisms and for developing effective therapeutic strategies for such diseases including HPS.
Materials and methods
Surgical procedures
All the procedures performed on the rats were conducted according to National Institutes of Health guidelines. The study protocol was approved by the committee on Animal Research of Southwest Hospital. Male Sprague–Dawley rats (220–250 g, Laboratory Animal Center, The Third Military University, Chongqing, PRC) were randomized into two groups in all the experiments. Core body temperature was maintained at 37 ± 1℃ throughout all the surgical procedures using a homeothermic heating system. Food and water were provided ad libitum post-operatively. CBDL-induced liver cirrhosis was used as the HPS experimental model. The rats were housed in plastic cages and allowed free access to food and water. All rats were fasted for 12 h before the operation. The CBDL operation was performed after anesthetizing the rats with 2% isoflurane delivered in oxygen. The common bile duct was exposed and doubly ligated with 3-0 silk. In the UCBDL group (n = 40), the first ligature was made near the junction of the hepatic ducts, and the second ligature was made above the entrance of the pancreatic duct (Figure 1a). In the MCBDL group (n = 40), the first ligature was made in the middle of the common bile duct, and the second ligature was made above the entrance of the pancreatic duct (Figure 1b). The ligatures were tightened, and the common bile duct was transected between the ligatures. The sham-operated group (n = 10) had the common bile ducts isolated without ligation. The incision was closed, and the animals were allowed to recover. All animals were sacrificed by inhalation of increasing CO2 after the completion of the experiment.
Effects of two methods of common bile duct ligations: middle of the common bile duct ligation (MCBDL) and upper common bile duct ligation (UCBDL). (a) In the MCBDL group, the first ligature was made in the middle of the common bile duct, and the second ligature was made above the entrance of the pancreatic duct. (b) In the UCBDL group, the first ligature was made close to the junction of the hepatic ducts, and the second ligature made above the entrance of the pancreatic duct. (c) The bile lake volume of the MCBDL group (5 ± 2.3 mL). (d) The bile lake volume of the UCBDL group (0.8 ± 0.3 mL). *P < 0.05 versus the UCBDL group.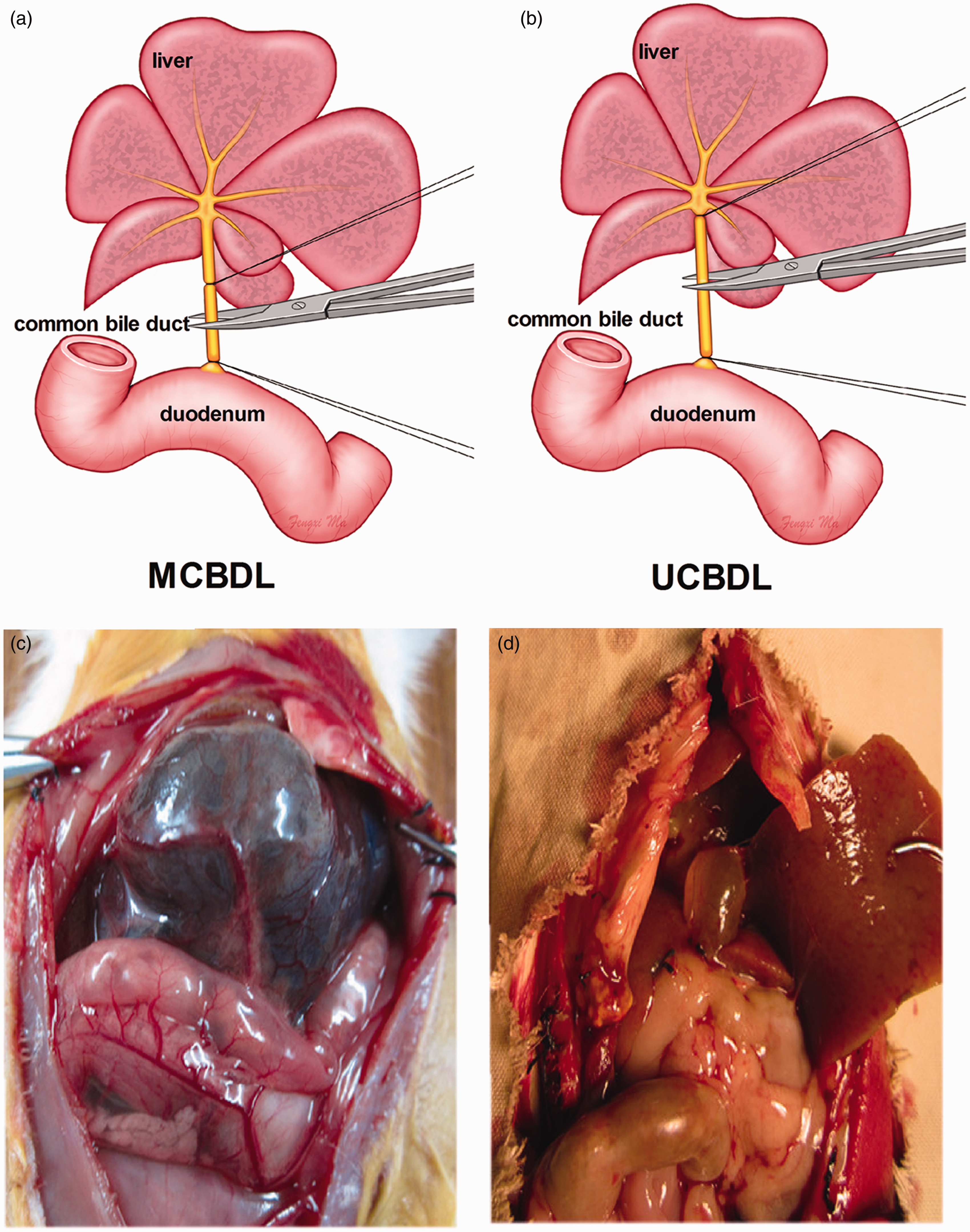
Pulse oximeter monitoring
In each rat, a clip probe (TDR-43W) from the stand alone pulse oximeter (CANL-425SV-A; Med Associates Inc, St Alban, VT, USA) was placed on the tail and connected to the pulse oximeter. The rats were allowed to stabilize for 30 min under room air; and the output of the pulse oximeter was recorded when the LED light was green, indicating the presence of a good signal and a regular wave pattern detected by the analysis software (SOF-425; MED Associates Inc). Eight rats were randomly selected for study in each group.
Arterial blood gas analysis
All the catheters were inserted into the carotid artery, tunneled subcutaneously and exteriorized at the back of the neck with the rats under identical anesthesia as described earlier. The animals were allowed to recover from the surgical procedure for 24 h with overnight fasting followed by free access to food and water; they were studied when conscious and unrestrained in room air. Before the experiments, the animals were left undisturbed for a minimum of 30 min after installation of the experimental apparatus until they reached a physiologically stable condition that was confirmed by minimal fluctuations in oxygen saturation, stable heart rates, and/or minimum body motion. The arterial blood sampling was performed in a quiet and air-conditioned room maintained at 24℃, and the arterial blood gas analysis was performed on an ABL 700 radiometer (Radiometer, Copenhagen, Denmark) in the Clinical Laboratory at Southwest Hospital. The AaPO2 was calculated using the modified alveolar gas equation 150 – (PCO2/0.8) – PO2. 10 Eight rats were randomly selected for study in each group.
Histopathological study
Samples from the lungs and liver were collected into 100 g/L phosphate buffered formaldehyde and fixed overnight. Serial 4 µm thick sections were prepared after the samples were dehydrated in graded ethanol solutions, cleared in chloroform, and embedded in Paraplast (Guangzhou Xiuwei Commerce Co., Ltd., GuangDong, China). Staining was performed with hematoxylin and eosin (HE), and the samples were studied under light microscopy in a blinded fashion. Eight rats were randomly selected for study in each group.
Detection of intrapulmonary shunt volumes
The intrapulmonary shunt volume was quantified by injecting technetium-99 m-labeled albumin macroaggregates (200 μCi, mean size = 20 µm, range 15 to 50 µm) into the tail vein and sacrificing the animal 30 min later by an overdose of isoflurane. The radioactivity of the resected lung and brain was measured by a gamma counter (GC-911; Zonkia, Hefei, China) at random. The volume of albumin macroaggregates passing through the lungs, which was defined as the ‘brain/lung radioactivity ratio’, was calculated using the following formula: brain/lung radioactivity ratio (%) = (radioactivity of the brain/radioactivity of the lungs) × 100. 17 Eight rats were randomly selected for study in each group.
Statistical analysis
The data were analysed statistically using the SPSS statistical program (SPSS Inc, Chicago, IL, USA). The values are presented as mean ± SEM. The results were evaluated using analysis of variance followed by Fisher’s post hoc test. A P-value of less than 0.05 was considered statistically significant.
Results
Mortality rates in the UCBDL, MCBDL and sham-operated rats
No deaths were observed in the sham-operated rats. A mortality rate of 42.5% (17 of 40 rats) was observed in the UCBDL group. Thirty-one of 40 rats (77.5%) died after five weeks of ligation in the MCBDL group (Figure 2a). The vast majority of deaths occurred in the first two weeks after ligation, and most deaths were the result of bile duct rupture or bile leakage, infection, and subsequent sepsis. During the development of fibrosis or cirrhosis, macroscopic signs of cholestasis were observed in the majority of the rats, namely weight loss, lethargy, limb and ear jaundice, yellow urine, and massive ascites. The physical activity levels of the CBDL rats decreased progressively, and there were decreased responses to external stimuli.
Comparison of two common bile duct ligations and the induced mortality and volume of bile lakes in the sham-operated, middle of the common bile duct ligation (MCBDL), and upper common bile duct ligation (UCBDL) groups. (a) The sham-operated group had no deaths, and the mortality of the UCBDL group was significantly decreased compared to that of the MCBDL group. (b) In the sham-operated group, the formation of bile lakes was not observed, and the volume of the bile lake in the MCBDL group was markedly increased compared to the UCBDL. Data are expressed as the mean ± SEM. *P < 0.05 versus the UCBDL group.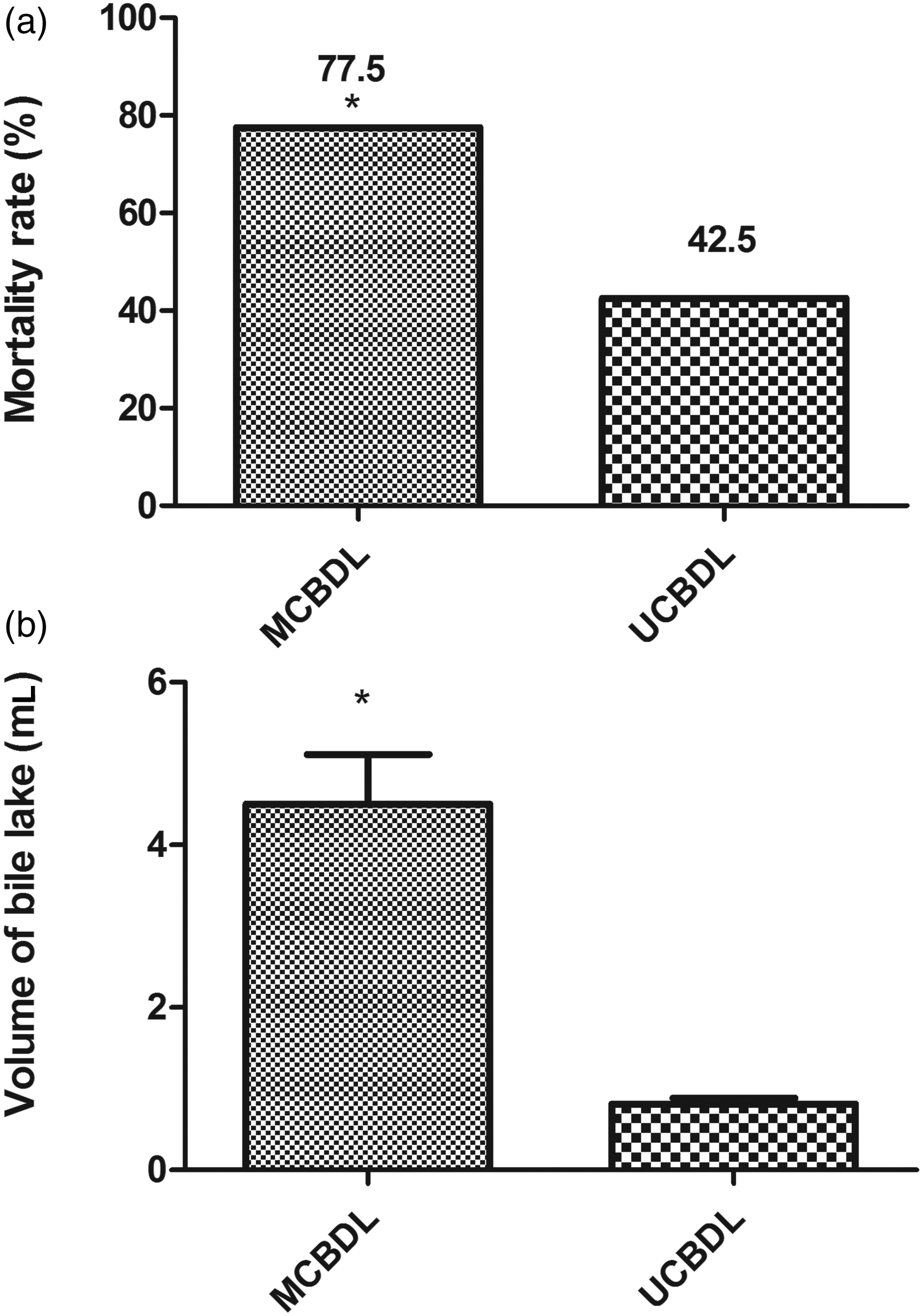
Common bile duct morphological changes induced by CBDL
Body weight, liver biochemistry and histology in the middle of the common bile duct ligation (MCBDL), upper common bile buct ligation (UCBDL) and sham-operated rats.
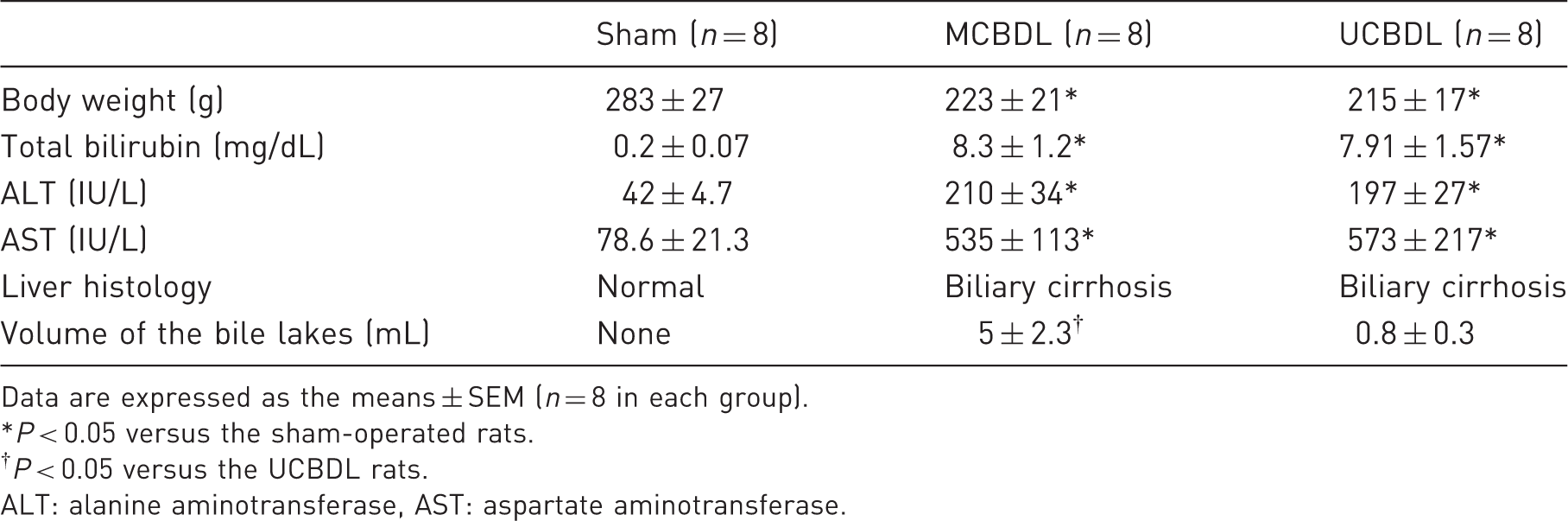
Data are expressed as the means ± SEM (n = 8 in each group).
*P < 0.05 versus the sham-operated rats.
†P < 0.05 versus the UCBDL rats.
ALT: alanine aminotransferase, AST: aspartate aminotransferase.
Development of hepatic injury after CBDL
To characterize the development of hepatic damage after CBDL, we assessed hepatic biochemical and histological features of injury in the sham-operated and five-week CBDL animals. Table 2 shows the results of these studies. Blood aspartate aminotransferase (AST) and alanine aminotransferase (ALT) concentrations as well as total bilirubin levels were similar in the UCBDL and MCBDL-induced cirrhotic groups and were significantly higher than in the sham-operated group (Table 1). Histological analysis confirmed progressive hepatic injury and the development of biliary cirrhosis, which was diagnosed as macroscopic nodular formation and microscopic secondary biliary cirrhosis resulting from the two methods of ligation. Although both methods of CBDL were effective in inducing cirrhosis, the UCBDL-induced cirrhosis was more serious than that in the MCBDL group (Figures 3c and 3e).
Morphological analysis of the liver and lung. (a) Liver histology was normal in the sham-operated group. (b) Lung histology was normal in the sham-operated group. (c) The five-week middle of the common bile duct ligation (MCBDL) animals had progressive ductular proliferation and fibrosis, leading to the development of biliary cirrhosis. (e) The five-week upper common bile duct ligation (UCBDL) animals developed progressive fibrosis and nodule formation, indicating the development of cirrhosis. (d, f) Five weeks after MCBDL and UCBDL separately, there was massive inflammatory cell filtration, and the capillary density and diameters were significantly increased. The alveolar space was shrunken down in some areas, and the alveolar septa were wider compared with the findings in a sham-operated group. Hematoxylin and eosin (HE) staining, original magnification ×100. Prevalence of hypoxemia and intrapulmonary shunt volumes in the two common bile duct ligation (CBDL) models and sham-operated rats. Results are expressed as the means ± SEM (n = 8 in each group). *P < 0.05 versus the sham-operated rats. MCBDL: middle of the common bile duct ligation, UCBDL: upper common bile duct ligation, SpO2: pulse O2 saturation; PaO2: arterial oxygen tension; PaCO2: arterial carbon dioxide tension; AaPO2: alveolar–arterial oxygen partial pressure difference.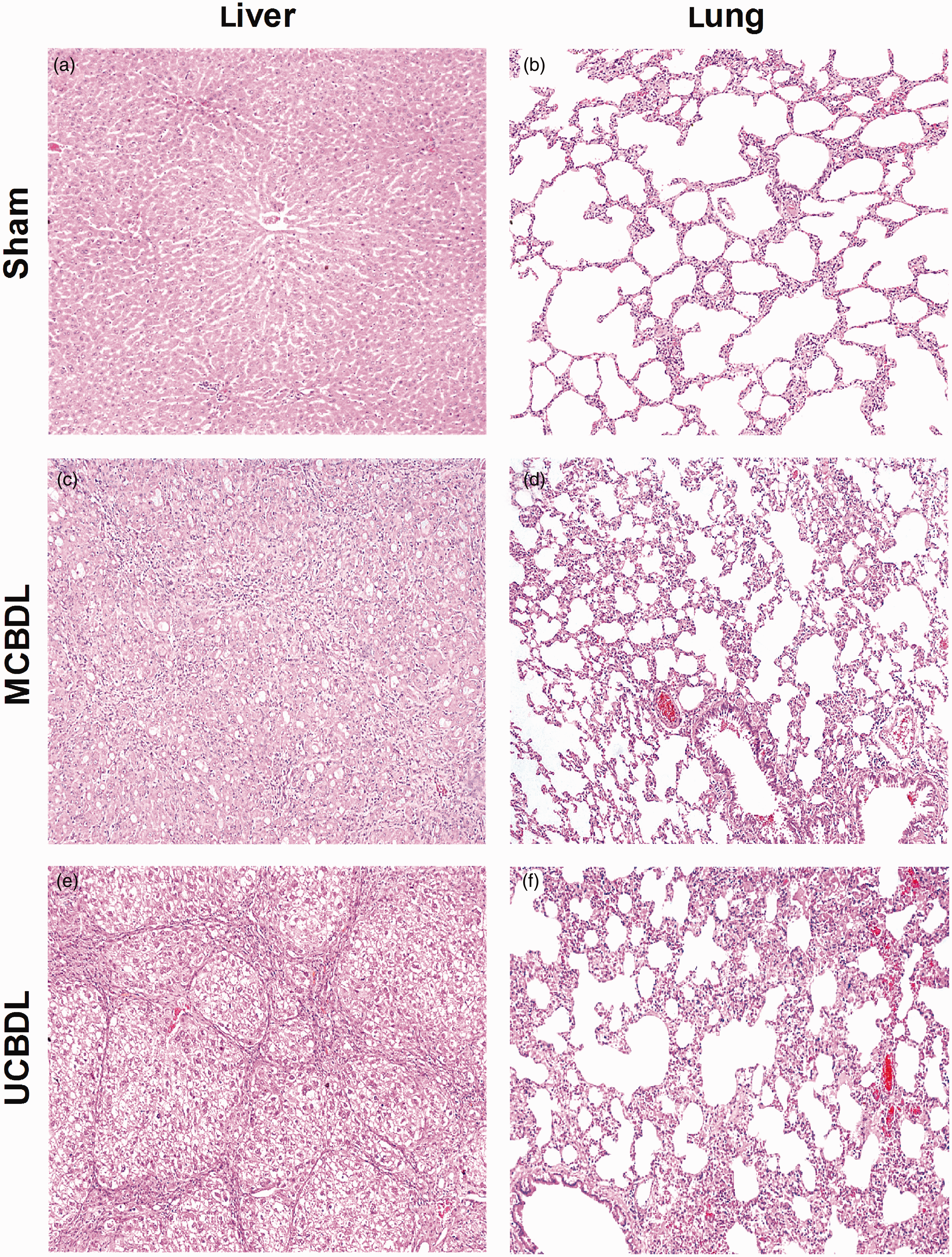
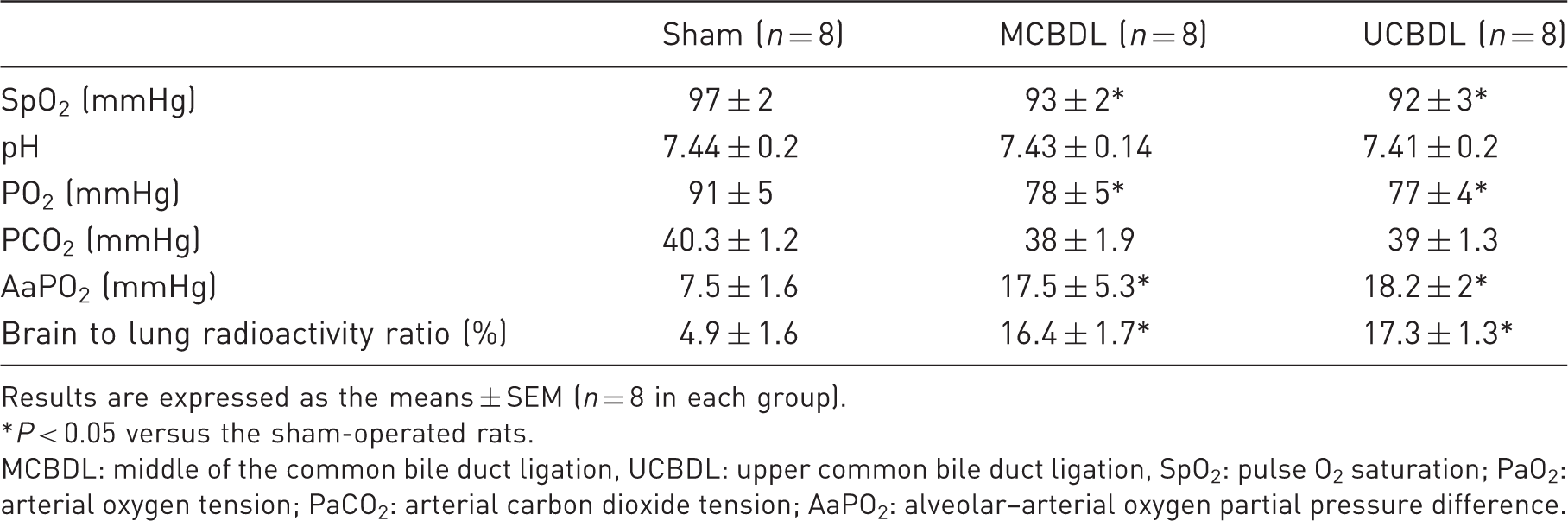
Oxygen saturation, arterial oxygenation and pulmonary alteration after CBDL
To assess the development of gas-exchange abnormalities after CBDL, the levels of oxygen saturation and arterial blood gases were measured in the animals after the five-week CBDL. To evaluate the lung tissue, the histology was analyzed. There were lower levels of oxygen saturation and PaO2 as well as higher AaPO2 levels in both groups of CBDL-induced cirrhotic rats than in the sham-operated rats (P < 0.05). There was little difference between the UCBDL and MCBDL groups (P > 0.05). Arterial blood pHs and PaCO2 were similar in all the groups (P > 0.05) (Table 2). In the five-week CBDL animals, the abnormalities were increased, and the increase was reflected by the development of mild hypoxemia and a widened AaPO2 (18.5 ± 5.3 and 18.2 ± 2.0 mmHg of the sham values, P < 0.05). Figure 2 shows representative low-power images of hematoxylin-stained histological sections from the sham and five-week CBDL animals (Figures 3b, 3d and 3f). There was a massive amount of inflammatory cell filtration, and capillary density was significantly increased in the five-week CBDL animals. The alveolar space was diminished in some areas, and the alveolar septa were wider compared with the sham-operated group. These results demonstrated the progressive development of gas-exchange abnormalities after CBDL that could not be explained by lung edema or architectural abnormalities.
HPS in the CBDL rats
The assessment of HPS in the CBDL-induced cirrhotic rats was based on a combination of gas exchange abnormalities and intrapulmonary vascular dilation (IPVD). In the CBDL-induced cirrhotic rats, the AaPO2 and the brain/lung radioactivity ratio were markedly higher than in the sham-operated rats (16.4 ± 1.7 and 17.3 ± 1.3% of sham values, respectively; P < 0.05), reflecting the presence of IPVD (Figure 4, Table 2). The UCBDL and MCBDL-induced cirrhotic rats exhibited similar AaPO2 and brain/lung radioactivity ratios, indicating that both methods could establish a model of HPS.
Individual measurements of the brain/lung radioactivity ratio after intravenous injection of 200 μCi (technetium-99m-labeled albumin macroaggregates) in the sham-operated rats (n = 8), middle of the common bile duct ligation (MCBDL) rats (n = 8), upper common bile duct ligation (UCBDL) rats (n = 8). *P < 0.05 versus the sham-operated group.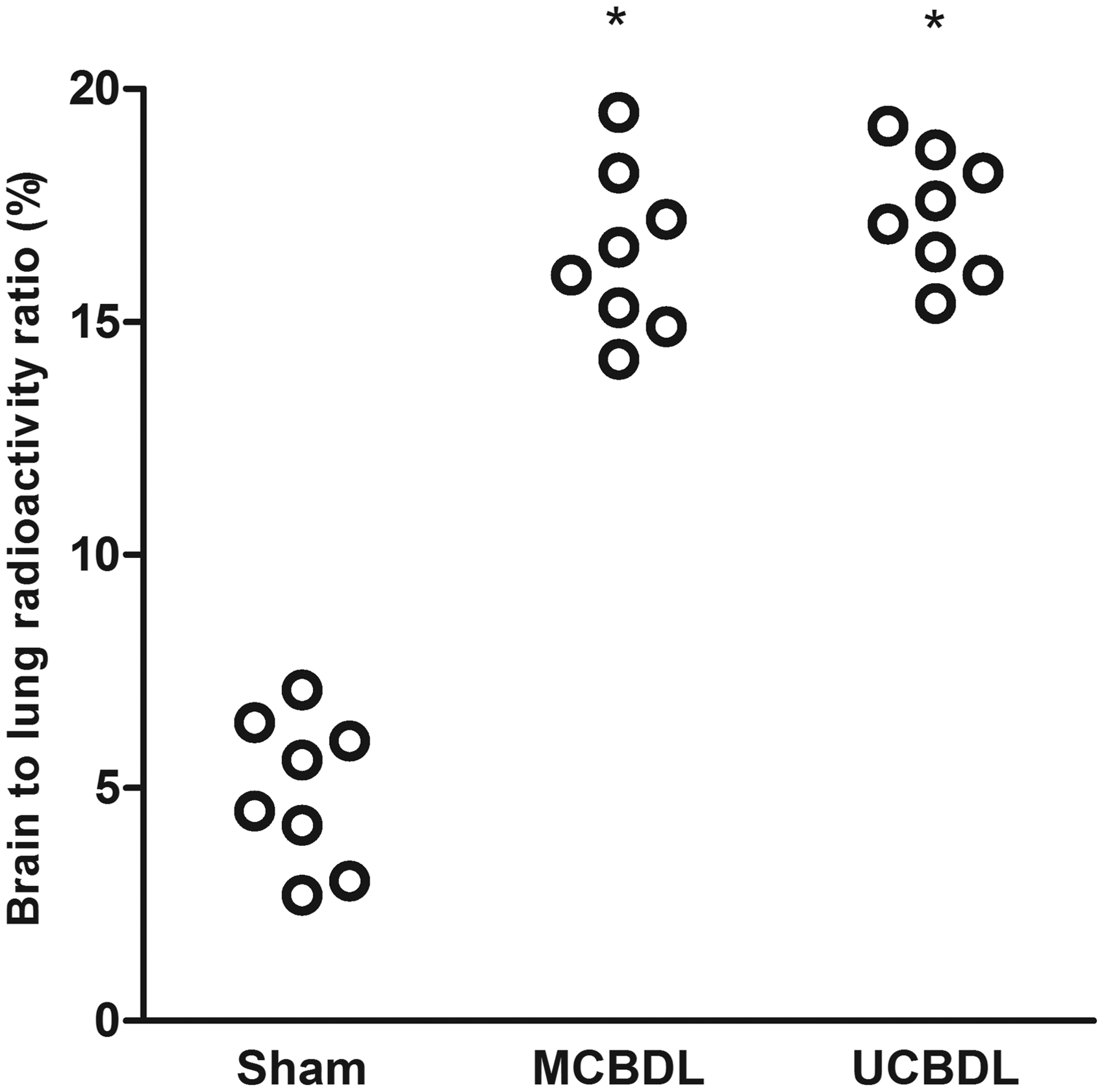
Discussion
Defining well-characterized and easily accessible animal models that mimic human disease is critical for exploring the pathogenic features and mechanisms of disease and, in this case, for developing effective therapeutic strategies for HPS. HPS occurs in 5–32% of patients with cirrhosis. Hepatic fibrosis in rats can be effectively induced by various chemical or surgical hepatic injuries such as the hepatotoxins alpha-naphthyl-isothiocyanate (ANIT), carbon tetrachloride (CCl4), 3,5-diethoxycarbonyl-1,4-dihydrocollidine (DDC), or by the injuries produced in rats subjected to CBDL and PPVL.10,12,13,18 Chronic CBDL in rats is the only well-established experimental model of human HPS.18–20 For this reason, this model of hepatic cirrhosis was used to study HPS. The mortality and morbidity of CBDL rats is high,21,22 which greatly limits experimental studies.
There is no explicit detail in the literature describing which portion of the common bile duct should be ligated. To our knowledge, this study is the first to show that the use of UCBDL can greatly reduce the mortality rate in CBDL-induced HPS rats. This finding suggests that the UCBDL method can mimic HPS better than the MCBDL method. The current MCBDL approach leads to high mortality and morbidity. The deaths associated with CBDL were typically observed within two weeks of the ligation. We found that these rats experienced symptoms including weight loss (Table 1), poor spirit, limb and ear jaundice, massive ascites, and yellow urine. The autopsies showed that all of the CBDL rats had different sized bile lakes (which reflects different degrees of common bile duct dilation); and many bile duct ruptures were found in the MCBDL rats, but no ruptures occurred in the UCBDL rats. The common bile duct normally ranges from 12 to 16 mm in length and from 0.6 to 1 mm in diameter, but it can be up to 45 mm long. 23 After ligation and excision of the common bile duct, the rats developed extrahepatic cholestasis characterized by transient growth retardation and marked hyperbilirubinemia (Table 2). Four to five weeks later, the histological changes of biliary cirrhosis became evident. 14 These changes were accompanied by portal hypertension with hepatosplenomegaly, ascites (in some rats), and liver dysfunction, 17 which ultimately resulted in liver failure and death. 18 We postulated that after MCBDL the increased pressure dilated the bile duct and the diaphragm was oppressed and rose, and this in turn seriously affected respiratory and digestive functions. This ultimately resulted in bile lake rupture, thereby increasing mortality in these rats. In the UCBDL rats, the first ligature was made close to the junction of the hepatic ducts, which greatly reduced bile duct dilation, accelerated the formation of secondary cirrhosis, and decreased the chance of bile leakage (Figure 1).
Furthermore, mortality in the UCBDL rats was decreased 1.8-fold compared to that in the MCBDL rats (Figure 2). Whether there is a significant correlation between the number of bile duct ruptures and the mortality of CBDL rats remains questionable. However, the rupture of dilated bile ducts definitely increases the mortality of CBDL rats. To some extent, this result suggests that the death of the MCBDL rats was at least partly related to bile duct rupture. Moreover, factors other than bile leakage may contribute to the death of the CBDL rats; these factors could include infection and intestinal endotoxin-induced sepsis and portal hypertension leading to gastrointestinal bleeding,24,25 and they are irreversible. In particular, these changes allow improved access to the portal vessels for hemodynamic work, which can be very difficult in the presence of a large bile lake. By using UCBDL we improved the method of CBDL, which reduced the degree of dilation of the duct, decreasing the chance of rupture of the duct and increasing the survival rate and the replication rate of HPS rats.
HPS is defined by the presence of liver disease and/or portal hypertension and IPVD that causes an abnormal age-corrected AaPO2.26,27 After five weeks of CBDL, we found that the impacts of the two types of ligation on liver function were not very different, but the severity of liver cirrhosis in the UCBDL group was more obvious than in the MCBDL group (Figures 3c and 3e). Compared to the sham group, the hepatic damage mentioned above was obviously different in both the MCBDL and UCBDL groups. Both of these CBDL methods led to progressive liver injury, which is a prerequisite for the formation of HPS.
Although we found that all types of CBDL could lead to liver cirrhosis, not all CBDL rats presented as hypoxic, as was similar to patients with chronic liver disease. 28 To date, no publications validating the heart rate feature of the pulse oximeter have been published for CBDL rats. We used a pulse oximeter to screen for hypoxemia, which coexisted with the pulmonary complications in the cirrhotic rats. In agreement with our findings, although several rats showed obvious liver cirrhosis after five weeks of CBDL, for unknown reasons, there was no hypoxemia. Moreover, arterial blood gas analysis was used to show that there were significant hypoxemia and AaPO2, more than 15 mmHg, which was consistent with our pulse oximeter findings. This result demonstrated that the pulse oximeter could be used to find early CBDL-induced HPS in rats, as is similar to the method of screening for HPS in patients.29,30
The normal pulmonary capillary diameter is approximately 8–15 µm. 31 IPVD is considered to exist when the pulmonary capillary diameter increases (15–90 µm), which is the major structural derangement in HPS.16,32 In this study, we found that these vessels were markedly expanded and the interalveolar spaces were widened in the CBDL-induced cirrhotic rats when compared to the sham-operated rats. Further evaluation of the extent of IPVD, using an intravenous injection of radiolabeled albumin macroaggregates with a diameter of 15–50 µm, allowed Schraufnagel and co-workers to detect an increase in the alveolar capillary diameter in cirrhotic rats that was still too small to allow the passage of macroaggregates. 33 Alternatively, the passage of macroaggregates may occur through the pulmonary artery to pulmonary vein anastomoses, which suggests the presence of IPVD.32,34 We calculated the shunting of microcirculation based on the brain/lung radioactivity ratio. This finding was in line with that in a study conducted by Fallon and co-workers using a similar technique in cirrhotic rats. 18 In our study, IPVD was markedly increased in the CBDL-induced cirrhotic rats, along with an elevated AaPO2 as compared with the sham-operated group. These characteristics were in accordance with manifestations of patients with HPS, suggesting that both methods of CBDL could successfully establish a model of HPS.
The results of this study have clearly demonstrated that the UCBDL-induced cirrhosis is more similar to HPS in humans than that induced by MCBDL, and the UCBDL model and humans showed hepatic dysfunction associated with IPVD and arterial hypoxemia. Compared to MCBDL, UCBDL has a lower mortality rate, making it a more reliable and reproducible model of liver cirrhosis in HPS. This model should aid understanding of the pathological mechanisms deduced from genetic and molecular approaches.
Footnotes
Acknowledgements
The authors greatly appreciate the technical assistance of Mrs Ma Xifeng with drawing.
YY and BC conceived of and designed the experiments. YY, BC, YC and BLZ performed the experiments. YY and BC analyzed the data. YY, BC, BY and KZL contributed reagents, materials or analysis tools. YY and BC wrote the paper. All authors read and approved the final manuscript.
Funding
The authors acknowledge this work was supported by grant Nos. 81170053 and 81170414 from the National Science Foundation of China (NSFC).
Declaration of conflicting interests
The authors declare that they have no competing interests.
