Abstract
The transgenic adenocarcinoma mouse prostate (TRAMP) model, designed for researching human prostatic cancer, was genetically engineered to harbor a transgene composed of the simian virus 40 Large-T/small-t antigen promoted by the rat probasin gene. In addition to prostatic neoplasms, the TRAMP mouse develops tumors in the seminal vesicles. This study was conducted to evaluate the pathology and histogenesis of TRAMP seminal vesicle neoplasms. Tissues of accessory sex organs harvested from 72 TRAMP mice of various ages (11-40 weeks of age) were fixed in neutral buffered formalin and stained with hematoxylin and eosin, desmin, 5-bromo-2′-deoxyuridine (BrdU, treated animals only), and SV40 Large-T antigen (SV40-Tag). In the seminal vesicles, we found neoplastic stromal cells that emerged multicentrically just beneath the epithelium, densely packed between the epithelium and the smooth muscle layer These stromal cells frequently exhibited mitotic figures and showed BrdU incorporation and SV40-Tag protein expression in the nuclei and immunopositivity for desmin. The proliferative mesenchymal cells were lined by cuboidal to columnar epithelium. Some of the larger papillary, polypoid lesions exhibited a phyllodes pattern resembling that seen in mixed epithelial-stromal tumors of the breast, prostate, and seminal vesicles of humans. Although the epithelium was negative for SV40-Tag and showed only occasional incorporation of BrdU, it clearly participated in the biphasic proliferation, forming papillary, cystic, and tubuloglandular structures. No conclusive evidence of malignancy (invasion or metastasis) was identified. Our recommended diagnosis of this lesion in the seminal vesicles is epithelial-stromal tumor.
Keywords
The transgenic adenocarcinoma mouse prostate (TRAMP, or C57BL/6-Tg(TRAMP)8247Ng) mouse was developed to enhance understanding of prostatic carcinogenesis. 11 TRAMP mice harbor a transgene composed of the rat probasin promoter region to drive expression of the simian virus 40 early genes (T/t antigen; SV40-Tag)–coding region. 11 The probasin gene is expressed only in prostatic epithelium under the regulation, both in vitro 31 and in vivo, 8 of androgens and zinc; thus, the probasin/SV40-Tag transgene was designed to be expressed only in the prostate. 11, 21 The TRAMP prostatic tumors are of epithelial origin; their morphologic characteristics and grading system have been described by our group. 34
In addition to being a model of prostatic cancer, the TRAMP mouse has been reported to develop other tumors, including neuroendocrine tumors in the prostate 14 and neoplasms in the seminal vesicles. 12, 19, 20, 29 However, the histopathologic features of seminal vesicular tumors have not been described fully in previous articles. 12, 19
The purpose of this study was to characterize the histopathologic features and assign an appropriate diagnosis for the TRAMP seminal vesicle tumors and to compare them with prostatic tumors of the same model by performing histopathologic and immunohistochemical examinations. Histogenesis was also investigated.
Materials and Methods
Animals and histopathology
TRAMP mice from a C57BL/6 background were kindly supplied by Dr. N. Greenberg (Fred Hutchinson Cancer Research Center, Seattle, WA) to establish our in-house TRAMP colony at Integrated Laboratory Systems Inc. (ILS, Research Triangle Park, NC). 26 In our study, male TRAMP mice of various ages from the ILS colony were used. Ages in weeks and numbers of mice at euthanasia were as follows: 11 weeks (10 mice), 14 weeks (5), 20 weeks (19), 24 weeks (6), 28 weeks (12), 38 weeks (8), and 40 weeks (12). Animals were housed in the ILS AAALAC-accredited Specific Pathogen-Free facility and handled in accordance with the guidelines provided in the Institute for Laboratory Animal Research Guide for the Care and Use of Laboratory Animals 13 as described elsewhere. 26 All the animals were fed pelleted NTP-2000 diet (Zeigler Brothers Inc., Gardeners, PA) ad libitum after weaning and given free access to Durham city tap water supplied in water bottles with stainless steel sipper tubes until euthanasia. The 28- and 40-week-old mice were treated with 5-bromo-2′-deoxyuridine (BrdU) in drinking water at a concentration of 200 ppm (200 mg/L) for 3 days before euthanasia. Animals were euthanatized at various ages by CO2 asphyxiation and subsequent exsanguination. The accessory sex glands including all lobes of prostate and seminal vesicles were processed for histopathologic examinations. 34, 35
Immunohistochemistry
Localization of SV40-Tag and desmin protein expression and BrdU incorporation were investigated using, respectively, a monoclonal antibody (clone PAb 101, BD Biosciences Pharmingen, San Diego, CA), a polyclonal antibody (Accurate Chemical & Scientific Corp., Westbury, NY), and a monoclonal antibody (clone B44, Pharmingen). Sections were deparaffinized in xylene and hydrated through a series of ethanols to 1× Automation Buffer (Biomeda Corp., Foster City, CA). Endogenous peroxidase was blocked using 3% H2O2 for 10–15 minutes at room temperature. Antigen unmasking was accomplished by heating sections in 200 ml of citrate buffer (pH 6.0) (BioCare Medical, LLC, Walnut Creek, CA) in a steamer for 30 minutes (SV40-Tag) or a microwave (Panasonic, Secaucus, NJ) at level-3 for 10 minutes (desmin), or by placing the sections in 0.01% trypsin at 37° C for 3–5 minutes (BrdU). Tissues were blocked with diluted normal horse or goat serum (Jackson Immuno-Research Laboratories Inc., West Grove, PA), depending on the species in which the secondary antibodies were raised. The primary antibodies, appropriately diluted, were applied at room temperature for 30 minutes (BrdU, 1:400) or 1 hour (SV40-Tag, 1:100; desmin, 1:500). Nonimmune immunoglobulin (IgG) (Jackson ImmunoResearch Laboratories Inc.) raised in the same species as the primary antibodies was used as the negative control at equivalent conditions in place of the primary antibody. Localization of the primary antibody was detected using a biotinylated horse anti-mouse IgG (SV40-Tag and BrdU) or a biotinylated goat anti-rabbit IgG (desmin) and a streptavidin complex conjugated to horseradish peroxidase (Vector Laboratories Inc., Burlingame, CA). Observation of the antibody complex was completed using liquid diaminobenzidine (DakoCytomation Inc., Carpinteria, CA). Finally, slides were rinsed in running tap water, counterstained with Harleco® Harris' hematoxylin (EMD Chemicals Inc., Gibbstown, NJ), dehydrated through a series of ethanols to xylene, and coverslipped with Permount® (Fisher Scientific International Inc., Fairlawn, NJ).
Results
Normal seminal vesicles
Seminal vesicles are saccular glands consisting of a central cavity and peripheral pouches. 36 The normal luminal surface of seminal vesicles is a system of anastomosing glandular architecture oriented in various directions and lined by cuboidal to tall columnar epithelium that forms an intricate arrangement of mucosal folds that ramify into secondary and tertiary folds (Fig. 1). 28, 36 Secretion filling the lumen usually expands the seminal vesicles, resulting in an extended vesicular wall lined by short epithelial papillae, a scanty sub-mucosal layer, and a thin smooth muscle layer.
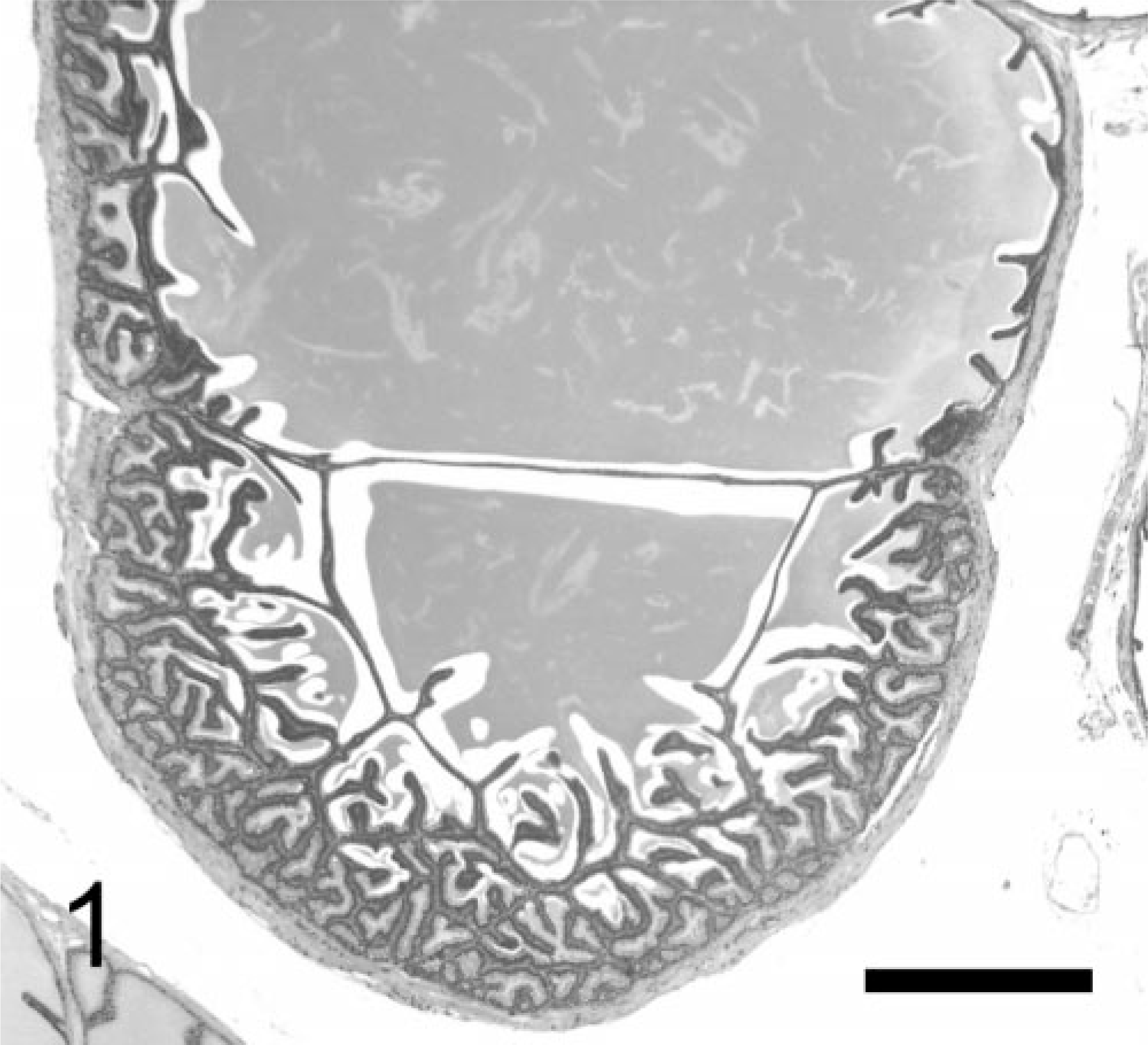
Normal seminal vesicle; TRAMP mouse (No. AJM-1, 24 wk old). Seminal vesicles are saccular glands composed of central cavity and peripheral pouches; luminal surface is system of anastomosing glandular structures oriented in various directions and lined by cuboidal to tall columnar epithelium. HE. Bar = 500 μm.
TRAMP seminal vesicular tumors
When compared macroscopically with normal seminal vesicles of the C57BL/6 parental strain, those of TRAMP mice became enlarged after 20 weeks of age (Figs 2, 3). Tumor masses projected into the luminal cavities and could not be seen from the outside of the seminal vesicles. Tumors were white and bulging on the cut surface (Fig. 4). Clefts and polypoidal structures were frequently seen. Masses were exclusively localized in the neck of the seminal vesicles. The seminal vesicles, especially in the tail, were distended secondarily to accumulation of secretion.
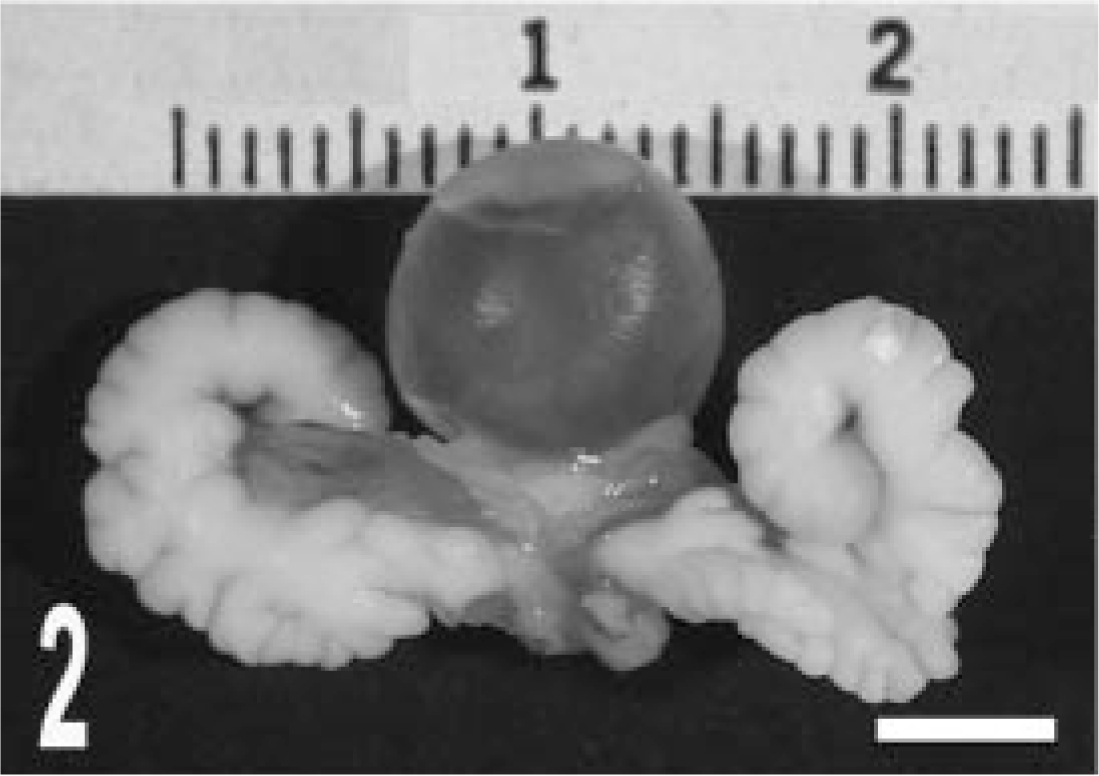
Gross appearance of seminal vesicles. Compared with seminal vesicles of a wildtype (C57BL/6) mouse (
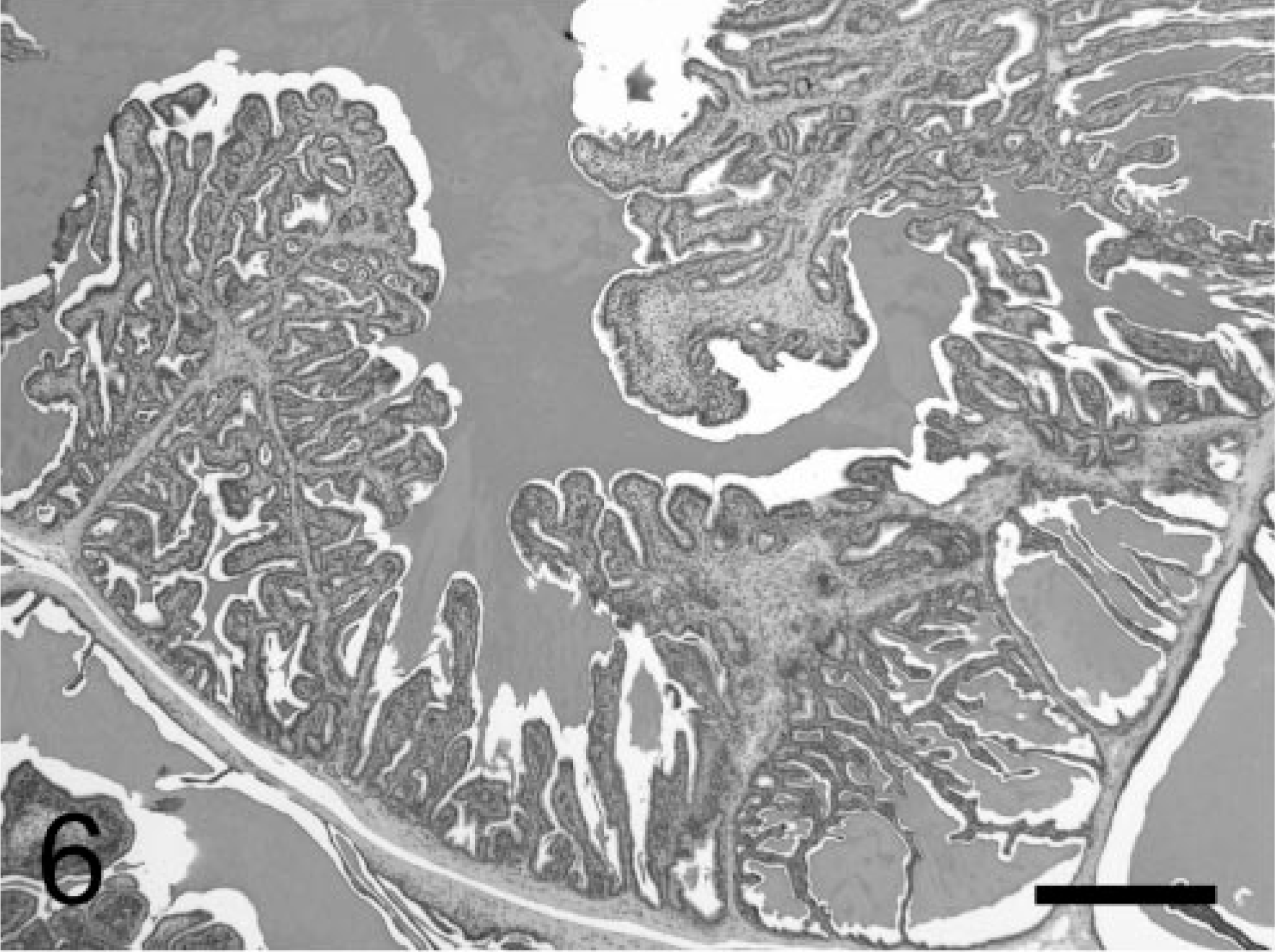
Tumors consisted of two main components: epithelial cells and stromal cells. Tumors initially exhibited a short papillary pattern, in which only submucosal stromal cells proliferated between the epithelium and smooth muscle layer (Fig. 5). Such lesions progressed and formed, at approximately 20 weeks, multiple papillary neoplasms with a prominent stromal component, based on the normal complex glandular structures, with an expanding mesenchyme (Fig. 6). Complex papillary growths into the acinar lumen were supported by a prominent proliferation of stromal cells and other mesenchymal components, including smooth muscle and connective tissues. Some of these papillary tumors further developed into larger lesions, with stromal expansion by myofibrous or myxomatous (or both) mesenchymal components (Fig. 7), resembling a phyllodes pattern often observed in the epithelial-stromal tumors in the human breast, prostate, and seminal vesicles. Throughout the developmental course of the neoplasms, there were always some areas in the seminal vesicles lined with relatively normal epithelium.
Seminal vesicle tumor; TRAMP mouse (No. 5, 28 wk old). Tumors are composed of proliferating stromal cells beneath the monolayered normal-appearing glandular epithelium. Note more mitotic figures in proliferating stroma (arrows) than in epithelium. HE. Bar = 75 μm.
Large papillary tumor of seminal vesicles; TRAMP mouse (No. 2, 28 wk old). Tumor exhibits a papillary pattern, composed of anastomosing glandular structures, similar to that shown in Fig. 1, supported by increased amount of stroma. HE. Bar = 500 μm.
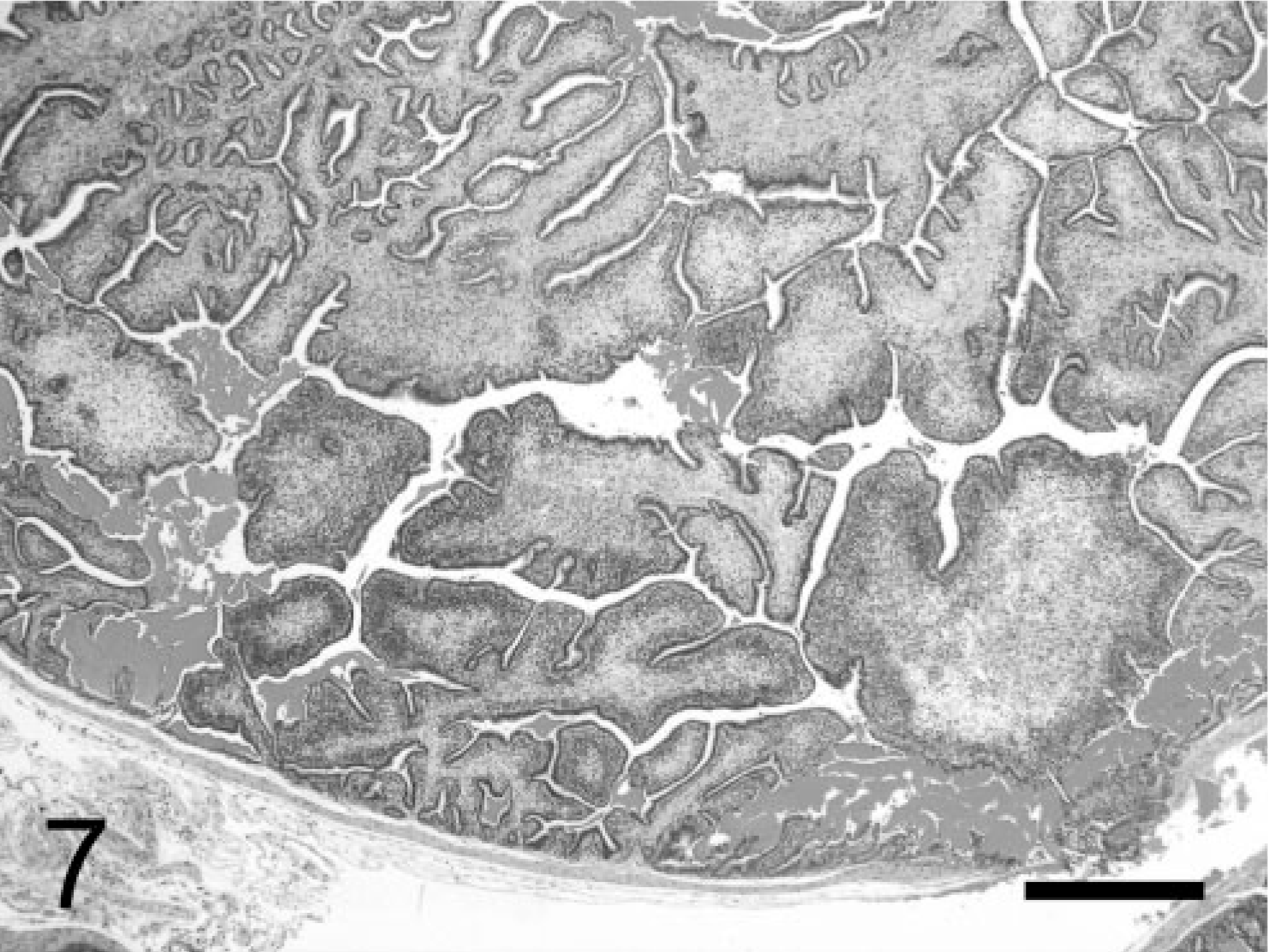
Large phyllodes-like tumor of seminal vesicles; TRAMP mouse (No. 8, 28 wk old). Subepithelial tissue is greatly expanded by large amount of myofibrous or myxomatous stroma (or both). HE. Bar = 500 μm.
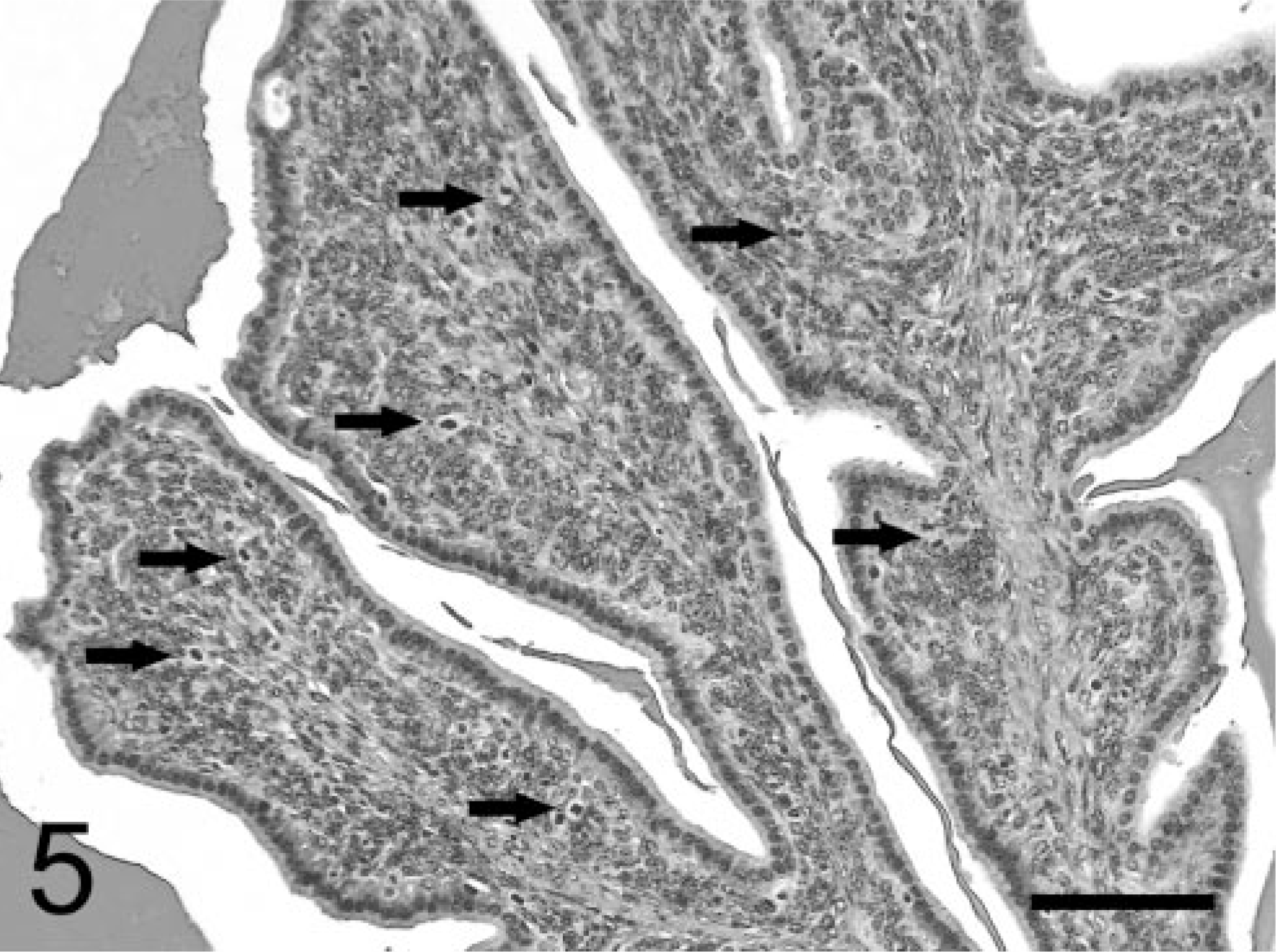
Neoplastic stromal cells (Fig. 5) arose multicentrically between the epithelium and smooth muscle layer, were spindle-shaped, and displayed round to oval nuclei. Cells were densely packed just beneath the lining epithelium, sometimes forming a cuff around the glandular structures. No basement membrane was evident using light microscopy. Mitoses were frequently observed (Fig. 5), and BrdU was incorporated by almost half of the neoplastic stromal cells (Fig. 8). Immuno-histochemistry revealed that these cells expressed SV40-Tag oncoprotein (Fig. 9), suggesting that this neoplastic stromal component was genetically induced. In addition, stromal tumor cells were immunolabeled with anti-desmin antibody, indicative of a myocytic origin or differentiation (Fig. 10).
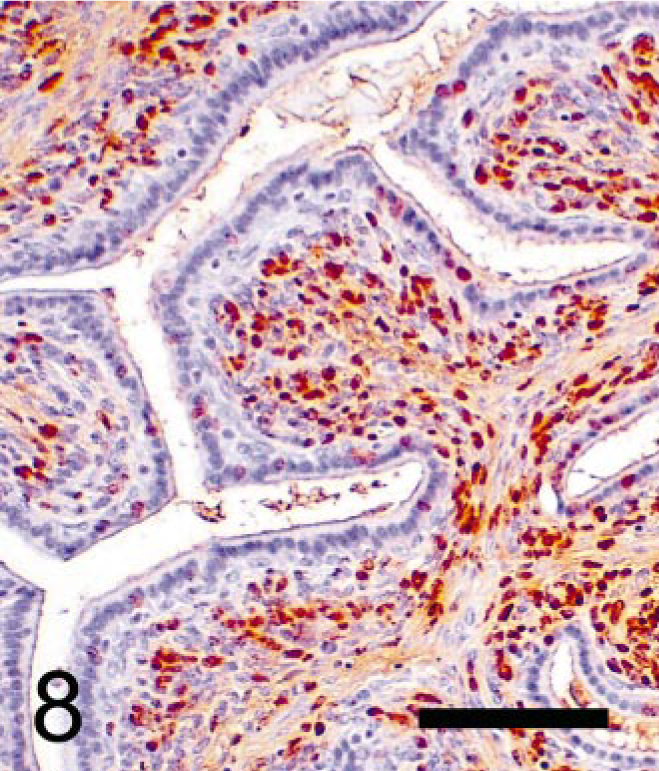
Seminal vesicular tumor; TRAMP mouse (No. 4, 28 wk old). Immunohistochemical labeling of BrdU using monoclonal antibody. Numerous stromal cells, but few epithelial nuclei, are labeled with BrdU. StreptABC/HRP method for BrdU with hematoxylin counterstaining. Bar = 100 μm.
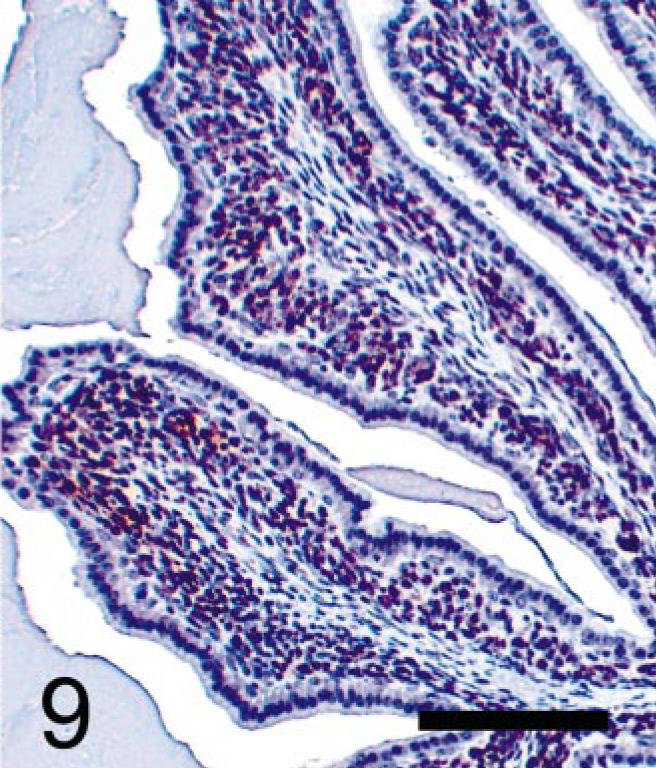
Seminal vesicular tumor; TRAMP mouse (No. 5, 28 wk old). Immunohistochemical labeling of SV40-T antigen (Tag) protein using monoclonal antibody. Stromal tumor cells are immunopositive, but epithelium is completely negative for SV40-Tag antibody. StreptABC/HRP method for SV40-Tag with hematoxylin counterstaining. Bar = 100 μm.
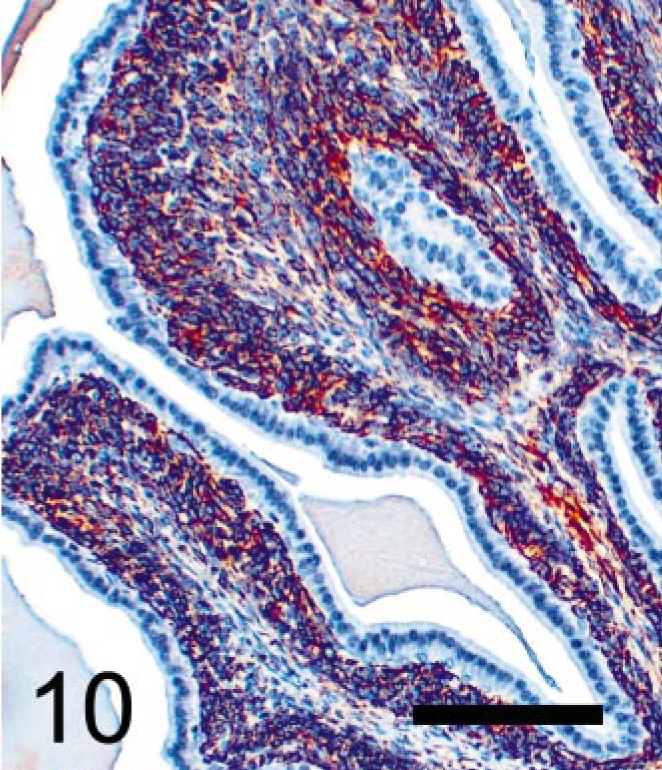
Seminal vesicular tumor; TRAMP mouse (No. 5, 28 wk old). Immunohistochemical labeling of desmin protein using polyclonal antibody. Stromal tumor cells are labeled with anti-desmin, suggesting myocytic origin or differentiation. StreptABC/HRP method for desmin with hematoxylin counterstaining. Bar = 100 μm.
The epithelium that lined the tumors (Fig. 5) was composed of bland-appearing, cuboidal to tall columnar epithelial cells with maintenance of nuclear polarity and sometimes with basophilic cytoplasm. Mitotic figures and the intranuclear immunoreaction localizing BrdU (Fig. 8) were occasionally observed in this population. However, their nuclei were consistently negative for SV40-Tag expression (Fig. 9). In large papillary and phyllodes-like tumors, cystic and tubulo-glandular structures were frequently observed (Fig. 11), lined by monolayered bland epithelium and usually cuffed by stromal cells.
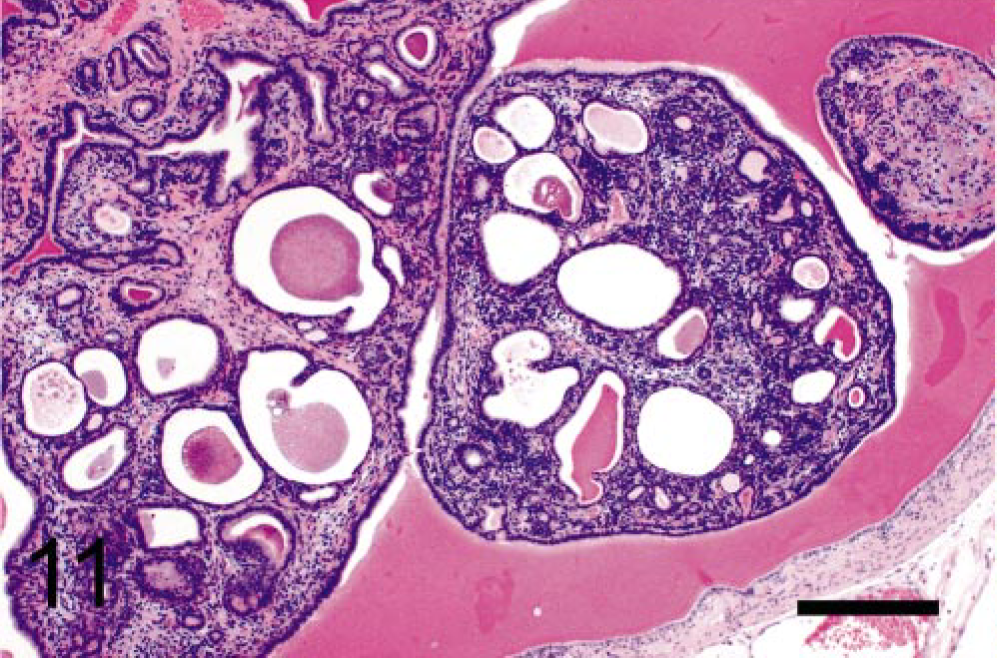
Seminal vesicular tumor; TRAMP mouse (No. 15, 40 wk old). Cystic and tubuloglandular structures are prominent. HE. Bar = 500 μm.
Up to 40 weeks, although tumors became quite large, there was no unequivocal evidence of malignancy of seminal vesicle tumors, such as invasion of the tumors into the smooth muscle layer or metastasis to other organs. Large papillary and phyllodes-like tumors were occasionally found to be infarcted.
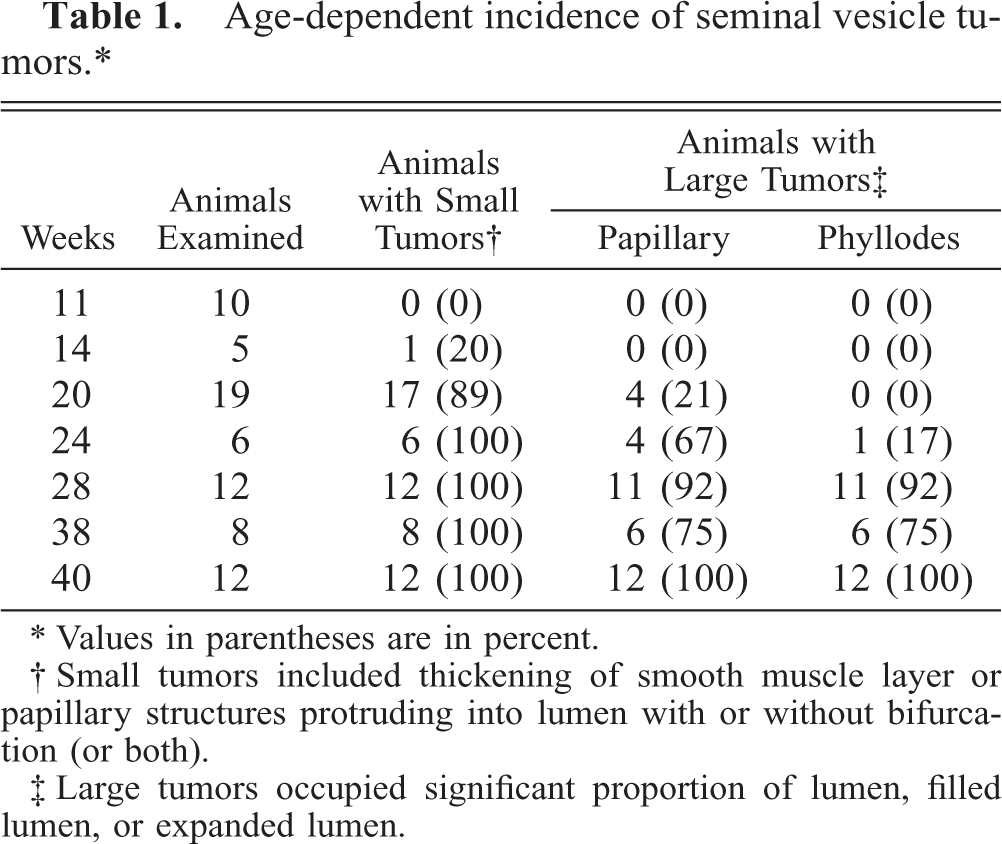
The chronologic incidences are summarized in Table 1. Small tumors included thickening of the smooth muscle layer or papillary structures protruding into the lumen with or without bifurcation (or both). Large papillary and phyllodes-like tumors occupied a considerable portion of the lumen, often filling or expanding it. The onset of seminal vesicular tumors occurred as early as 14–20 weeks. The incidence of seminal vesicle tumors was 100% at 24 weeks or later. Large papillary tumors were frequently observed at 24 weeks and phyllodes-like tumors at 28 weeks or later.
Age-dependent incidence of seminal vesicle tumors.∗
∗ Values in parentheses are in percent.
† Small tumors included thickening of smooth muscle layer or papillary structures protruding into lumen with or without bifurcation (or both).
‡ Large tumors occupied significant proportion of lumen, filled lumen, or expanded lumen.
TRAMP prostatic tumors
We observed that the epithelial component of the prostatic tumors grew in papillary, glandular, or cribriform patterns. Mitotic figures were frequently seen in the nuclei of epithelial tumor cells but occurred rarely in the mesenchyme (Fig. 12). BrdU was prominently incorporated into the nuclei of the epithelial component (Fig. 13) but rarely into those of mesenchymal cells. Only nuclei of neoplastic epithelial cells expressed SV40-Tag protein in the prostate (Fig. 14).
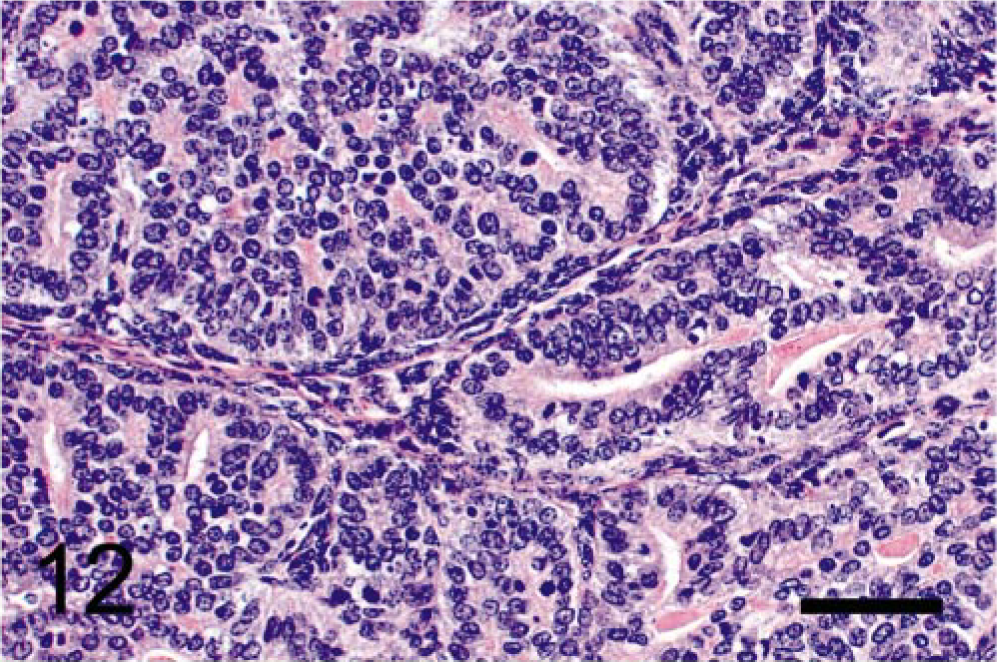
Prostatic tumor; TRAMP mouse (No. 12, 28 wk old). Note mitotic figures frequently seen in neoplastic epithelium but not in mesenchyme. HE. Bar = 50 μm.
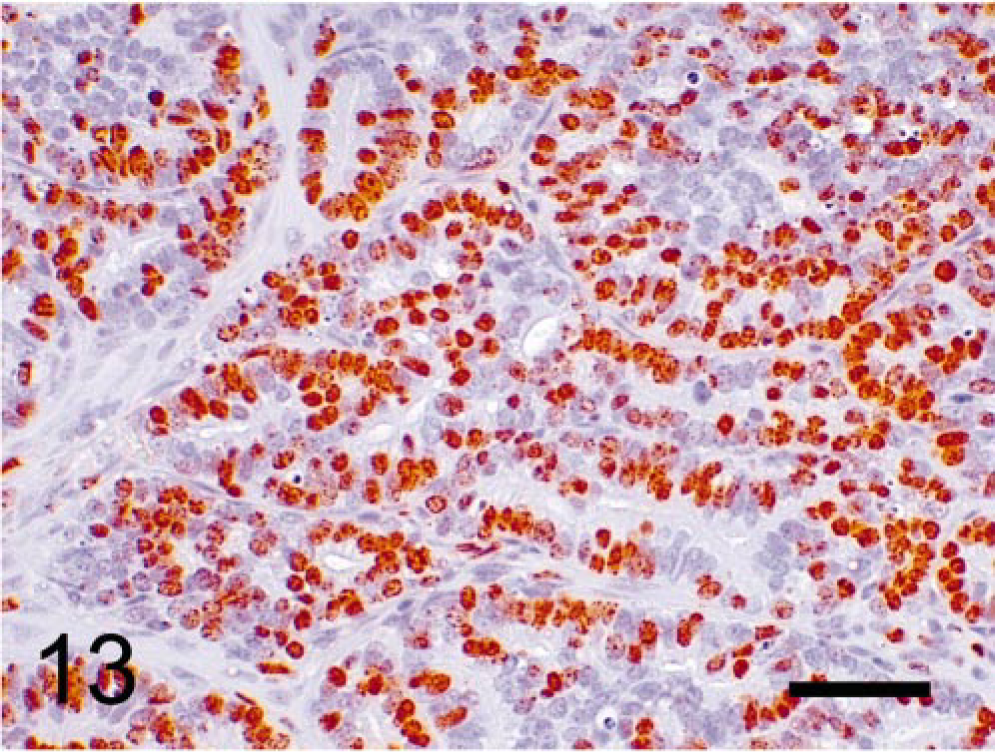
Prostatic tumor; TRAMP mouse (No. 9, 28 wk old). Immunohistochemical labeling of BrdU. Epithelial tumor cells show prominent immunopositivity, whereas mesenchymal cells are occasionally labeled. StreptABC/HRP method for BrdU with hematoxylin counterstaining. Bar = 50 μm.
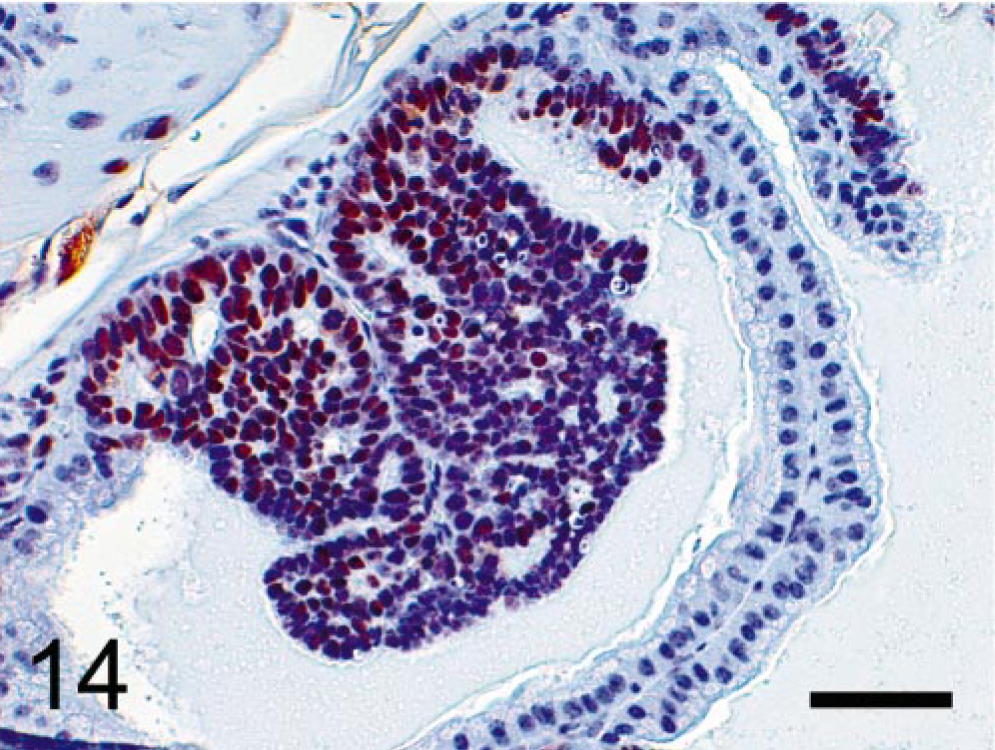
Prostatic tumor; TRAMP mouse (No. 5, 28 wk old). Immunohistochemical labeling of SV40-Tag protein. Only nuclei of hyperplastic and neoplastic epithelial cells express SV40-Tag protein; no expression in normal epithelial cells and stromal components. StreptABC/HRP method for SV40-Tag with hematoxylin counterstaining. Bar = 50 μm.
Discussion
Spontaneous seminal vesicular tumors occur extremely rarely in mice. In carcinogenicity studies conducted by the National Toxicology Program (NTP), only eight primary spontaneous seminal vesicular neoplasms among 46,752 B6C3F1 mice have been found, 24 including three adenomas, two adenocarcinomas, one squamous cell carcinoma, one sarcoma, and one undifferentiated carcinoma. Additional chemical-induced undifferentiated carcinomas in B6C3F1 mice treated for 2 years with reserpine were reported by the NTP. 25 These tumors were characterized by considerable cellular pleomorphism and appeared to involve the glandular epithelium and muscular wall of the seminal vesicles, although their origin was uncertain. Tumors were composed predominantly of spindle cells with multiple discrete areas of anaplastic epithelial-like cells. The spindle cells exhibited oval or elongated nuclei with variable amounts of streaming cytoplasm and appeared to be derived from the anaplastic cells that were pleomorphic with round, oval, or angular nuclei appearing often vesicular with a prominent nucleolus. Cytoplasm was usually abundant but the borders were indistinct. In some areas of two tumors, the cells were arranged in a trabecular pattern. Some tumor cells also differentiated toward squamous epithelium. Because no paraffin blocks were available in the reserpine study, we could not perform immunohistochemistry to reveal the nature of induced tumor cells, but their morphologic features suggested that these tumors were definitely of epithelial origin, not biphasic. Other investigators have described primary seminal vesicle tumors including adenocarcinoma, cavernous hemangioma, fibroadenoma, and papillary adenoma 6, 27, 33, 37 in a variety of strains of mice. Spontaneous lesions consisting of mesenchymal proliferation with decidual-like morphology have been found in seminal vesicles of aging mice; 15 these exhibited unique morphologic features including the presence of granules in the cytoplasm and granular metrial gland–like cells. We believe that the seminal vesicular tumors in the TRAMP model cannot be placed in any of these mouse-tumor categories.
The proliferating pattern of the seminal vesicle tumors in the TRAMP mice exhibited features of biphasic organization. Although stromal cells proliferated rapidly, they were always lined by a single-layered epithelial component and never formed solid stromal masses such as those seen in simple uterine stromal tumors, such as endometrial stromal nodules and endometrial stromal sarcoma 2 or leiomyoma/leiomyosarcoma. 3, 7
Morphologic observations as well as immunohistochemistry performed to localize SV40-Tag and BrdU clearly revealed differences in histologic characteristics and histogenesis between prostatic and seminal vesicular tumors of TRAMP mice. Histologically, seminal vesicular tumors showed prominent proliferation of stromal cells with frequent mitotic figures lined by bland epithelium, whereas prostatic tumors displayed epithelial tumor cell proliferation with scant stromal components. Immunohistochemistry for BrdU further supported these histologic findings. In seminal vesicular tumors, BrdU was incorporated into nuclei of stromal cells more frequently than those of epithelial cells, whereas in prostatic tumors, BrdU incorporation occurred more frequently in nuclei of proliferating epithelium than in those of stromal cells. SV40-Tag immunohistochemistry provided additional information concerning the differences in histogenesis of these two entities. Only hyperplastic and neoplastic prostatic epithelium expressed SV40-Tag protein, and stromal cells manifested no expression of the protein in the prostatic tumors, whereas only stromal tumor cells exhibited the protein expression in the seminal vesicular tumors. These results indicate that, in TRAMP mice, the seminal vesicular tumors arise from a completely different origin compared with the prostatic tumors. Furthermore, the tumors in the seminal vesicles did not develop until 14–20 weeks of age, compared with the onset of the prostatic hyperplasia or prostatic intraepithelial neoplasias that reportedly occurred in the same model within 10 weeks of age. 10, 12, 34
From the information on the TRAMP mouse seminal vesicular tumors that we have demonstrated in this study, we may conjecture as to their histogenesis. Although the origin of these tumors is still not known, we suspect that they arise multicentrically from stromal cells that express desmin protein, existing in the submucosal tissue between seminal vesicular epithelium and the smooth muscle layer. First, the stromal cells grow beneath and along the epithelium and then continue their proliferation toward the epithelium, associated with thickening of the stroma. As the stromal tumor cells grow, the lesions develop a prominent papillary pattern. In large papillary tumors, centrally located stromal neoplastic tissues or supporting mesenchymal tissues (or both) become myxoid and edematous, forming the phyllodes-like tumors. The tumor cells located beneath the epithelium exhibit the most prominent proliferative activity, regardless of the size of the tumors. Epithelial cells also proliferate, but generally form only a single cell layer lining the proliferating stromal tumors. Although we believe the desmin-positive spindeloid cells to be smooth muscle cells because of the uniqueness of the proliferative lesions in the TRAMP seminal vesicles, the possibility that these cells may represent a myoepithelial phenotype cannot be ruled out conclusively.
Several kinds of human neoplasms display characteristics similar to those of the TRAMP mouse seminal vesicle tumors. Epithelial-stromal tumors (phyllodes tumor or cystosarcoma phyllodes) in the human breast are more or less circumscribed, have a foliated structure, and are composed of connective tissue and epithelial elements resembling fibroadenoma in the breast. 5 In human breast epithelial-stromal tumors, polypoidal structures are lined by an elongated ductal epithelial component. Use of immunohistochemistry and ultrastructural analysis has revealed that the majority of the stromal tumor cells are fibroblasts, although occasional cells exhibit characteristics of myofibroblasts. 4 Although the seminal vesicular tumors of TRAMP mice are not as fibrotic or fibrogenic as the epithelial-stromal tumors of human breast, the fundamental architecture of both TRAMP seminal vesicle tumors and human breast epithelial-stromal tumors appears similar. Epithelial-stromal tumors (phyllodes tumors) of the prostate resemble human breast epithelial-stromal tumors and are biphasic. 40 Epithelial components, including cysts and glands, of human prostatic epithelial-stromal tumors are invaginated by an unusually cellular stroma that frequently forms papillary to polypoid projections. The stromal cells are spindle shaped, often condense around the glands, and occasionally lie in a myxoid matrix. 18, 30, 39 The stromal component is positive for muscle-specific actin and desmin. 40 These descriptions, including the immunopositivity for desmin, seem to be similar to those of the TRAMP mouse seminal vesicular tumors presented in this study. In addition, to date, four cases of epithelial-stromal tumors of the seminal vesicles have been reported in humans; 1, 9, 16, 22, 41 most of them recurred or metastasized (or both), indicative of malignancy.
Occasionally, in the seminal vesicles of TRAMP mice, large infarcted lesions were found. Generally, these were often pedunculated and appeared to have undergone torsion with subsequent infarction. We, therefore, do not necessarily consider the infarction of large lesions indicative of malignancy in the seminal vesicle tumors because there was no evidence of invasion or distant metastases. At this time, insufficient evidence exists to designate these tumors malignant. On the basis of findings in our study and case reports in humans, we propose “epithelial-stromal tumor” as an appropriate diagnosis for this seminal vesicular tumor of the TRAMP mouse.
Several articles have mentioned seminal vesicle tumors of the TRAMP mouse. Lin et al. 17 reported that the enlargement of seminal vesicles was caused by invasive prostatic malignant neoplasms. However, immunohistochemical analysis in this study clearly showed that the neoplasms observed in the seminal vesicles were completely different from the prostatic tumors. Several investigators also reported that seminal vesicles of TRAMP mice were swollen at 15 weeks of age or thereafter, but they described no histopathologic alterations. 29, 38 Hsu et al. 12 reported that seminal vesicles were often not visible because they had been destroyed by large prostatic carcinomas; however, when the prostatic tumors were small, they were seen to be associated with hyperplastic epithelium showing protrusion of papillary fronds into the seminal vesicle lumen. They reported that these papillary fronds showed markedly cellular stroma, representing papillary fibroadenoma. However, as shown in this study, the stromal component was strongly positive for desmin, suggesting a muscular origin or differentiation, even though a fibrous component was observed. Seminal vesicular tumors observed by Martiniello-Wilks et al. 19 were generally enlarged and often dilated. The monolayered epithelium consisted of cuboidal to columnar cells with large oval nuclei, with coarse chromatin, and increased mitotic activity. The subepithelial stroma displayed marked proliferation of atypical cells with variable sizes and shapes. The authors designated this type of tumor “epithelial-stromal tumor with a phyllodes appearance.” We agree with the term “epithelial-stromal tumor” but would not add the qualifier “with a phyllodes appearance,” because phyllodes-like lesions occurred at 28 weeks of age or later. We suggest that some researchers using TRAMP mice younger than 28 weeks of age might not encounter phyllodes-like lesions. In addition, in human pathology literature, the term “epithelial-stromal tumor” includes phyllodes patterns in the uterus, prostate, and seminal vesicles. 5, 23, 32, 40, 41
Although the tumors of TRAMP seminal vesicles resemble epithelial-stromal tumors found in the breast, prostate, and seminal vesicles in humans, no reports of this type of tumor in any other rodents exist, and the tumors reported in this study are believed to be TRAMP mouse–specific lesions.
Footnotes
Acknowledgements
We thank Dr. N. Greenberg of Fred Hutchinson Cancer Research Center, Seattle, WA, for supplying the mice to establish our TRAMP colony. We thank Dr. John Peckham from Experimental Pathology Laboratories Inc., Research Triangle Park (RTP), NC, and Drs. Darlene Dixon, Georgette Hill, and Hiroshi Satoh from the NIEHS, Research Triangle Park, NC, for reviewing histopathologic slides of TRAMP mice. We thank Ms. Natasha Clayton and Julie Foley, NIEHS, for their technical assistance in immunohistochemical staining. We gratefully recognize, from the NIEHS, Ms. JoAnne Johnson and Drs. Gail Pearse and Darlene Dixon for their critical review of the manuscript.
