Abstract
A mixed epithelial and stromal tumor of the seminal vesicle gland is an uncommon neoplasm characterized by a dual population of epithelial and stromal cells. In this case report, we present a 59-year-old male patient who presented with a large, 10 cm mass in the left seminal vesicle, which was preliminarily suspected to be a malignant tumor of the seminal vesicle based on magnetic resonance imaging findings. Histopathological evaluation, however, revealed a tumor with biphasic epithelial–mesenchymal differentiation, predominantly displaying mesenchymal characteristics. The epithelium and stroma displayed papillary and foliar structures, respectively. The epithelial cells were bland, arranged in either single or multi-layered cuboidal patterns, and the stromal cells were spindle shaped with a sparse distribution. The patient experienced a favorable postoperative outcome. The diagnosis of mixed epithelial and stromal tumor is challenging based on clinical and imaging findings alone, and definitive diagnosis relies on pathological examination. This case report addresses a rare presentation of low-grade mixed epithelial and stromal tumor in the seminal vesicle gland and aims to expand the understanding of this entity by reviewing the relevant literature.
Introduction
The seminal vesicle glands are a pair of intricate, coiled tubular structures situated posterior-laterally at the base of the bladder, deriving from the mesonephric duct system during embryogenesis. In clinical practice, tumors originating from the seminal vesicle are rare. seminal vesicles can be involved by tumors from adjacent organs, with prostate cancer being a significant consideration.
Primary neoplasms of the seminal vesicle primarily include adenocarcinoma, squamous cell carcinoma, and mixed epithelial and stromal tumor (MEST), adenocarcinoma was the most common type. MEST is a unique and rare pathological entity characterized by biphasic epithelial and mesenchymal components. The clinical manifestations and biological behaviors of MEST vary depending on the tumor location, size, and pathological morphology. The report received approval from the Research Ethics Committee of Hangzhou First People’s Hospital (Ethics approval number: ZN-20240117-0014-01).
Case report
A 59-year-old male patient was admitted to the hospital after a routine physical examination revealed a mass in the seminal vesicle that had been present for over 10 days. The patient remained asymptomatic. A digital rectal examination revealed a large, soft mass with indistinct margins at the base of the prostate. Serum prostate-specific antigen (PSA) levels were within the normal range at 0.993 μg/L (reference: 0.000–4.000 μg/L), and carcinoembryonic antigen (CEA) levels were 2.89 μg/L (reference: 0.00–5.00 μg/L). Computed tomography (CT) urography identified two well-demarcated, round, low-density lesions in the left seminal vesicle region, in close proximity to each other. The larger lesion, measuring approximately 5.4 cm × 5.8 cm, exhibited uniform density and did not show significant enhancement. The smaller lesion, measuring approximately 4.3 cm × 3.6 cm, had heterogeneous density and demonstrated mild to moderate uneven enhancement. Bladder magnetic resonance imaging (MRI) with both plain and contrast-enhanced sequences (3.0T) revealed two large, abnormal signal shadows in the left seminal vesicle gland area, measuring approximately 5.8 cm × 5.3 cm and 4.2 cm × 3.3 cm, which were closely associated. The cystic components appeared hyperintense on T1 and T2 sequences, whereas the solid components presented as isohypointense on T1 with small nodular slightly hyperintense areas and mixed signal on T2. Notably, the solid components demonstrated marked heterogeneous enhancement on the contrast-enhanced images, suggesting a suspected malignant tumor (Figure 1(a)).

(a) On magnetic resonance imaging, the cystic part of the tumor showed a high signal, measuring approximately 5.8 cm × 5.3cm, solid part displayed a low signal, with a small nodular slightly high signal within a size of about 4.2 cm × 3.3cm. (b) The tumor was cystic and solid, the solid part was 4 cm × 3 cm × 2.8 cm in size, soft and lobulated texture (the circle).
The patient underwent an ultrasound-guided core needle biopsy of the mass in the left seminal vesicle and prostate under local anesthesia. Histopathological analysis revealed a fibrous connective tissue matrix replete with muscular blood vessels, lined with cuboidal epithelial cells. The epithelial cells and the subepithelial stromal cells showed no atypia. The prostate exhibited hyperplastic changes. Although the biopsy pathology did not reveal evidence of malignancy, considering the large size and cystic-solid nature of the seminal vesicle tumor, the limited specimen from the biopsy may not truly represent the nature of the mass, and the possibility of a borderline or malignant tumor cannot be excluded. In addition, there was a clinical risk of torsion or incarceration of the mass. If the tumor in the seminal vesicle is malignant, it is often necessary to first rule out involvement from a malignant tumor of the prostate. Therefore, a surgical resection of the prostate and seminal vesicle tumor is indicated. After thorough preoperative preparation, the patient underwent robot-assisted laparoscopic resection of the prostate and seminal vesicle tumor. Macroscopic evaluation disclosed a left seminal vesicle tumor approximately 10 cm × 6 cm × 3 cm in size, presenting with both cystic and solid sections on the cut surface. The solid portion was lobulated and measured around 4 cm × 3 cm × 2.8 cm (Figure 1(b)). Microscopic inspection revealed a biphasic tumor composed of epithelial and mesenchymal elements. The epithelial and mesenchymal components exhibited a polypoid or leaf-like growth pattern, with focal areas of epithelial cyst formation. The epithelial cells were characterized by single or multi-layered cuboidal epithelium, while the stromal spindle cells were arrayed loosely with no evidence of atypia or mitotic activity (Figure 2). The result of an immunohistochemical marker is as follows: the tumor epithelial cells are positively stained for pan-cytokeratin and CK7 and negatively stained for CK20. The stromal cells are positive for both ER and PR (Figure 3). In addition, the stromal cells show partial expression of Calponin, SMA, and CD34, and negative expression of S100. Immunohistochemical staining for PSA reveals positive staining in both epithelial and stromal cells. The high sensitivity of PSA leads us to consider this marker less reliable. The diffuse positivity of stromal cells for ER and PR can effectively differentiate prostate stromal tumors. The Ki-67 index indicated a low proliferation rate of 1%. The pathological diagnosis was a low-grade MEST of the left seminal vesicle gland, associated with prostatic hyperplasia. Postoperatively, the patient was followed up for 16 months, during which time he experienced no significant discomfort, no abnormalities in blood tests, and no evidence of recurrence or metastasis on abdominal contrast-enhanced CT scans.
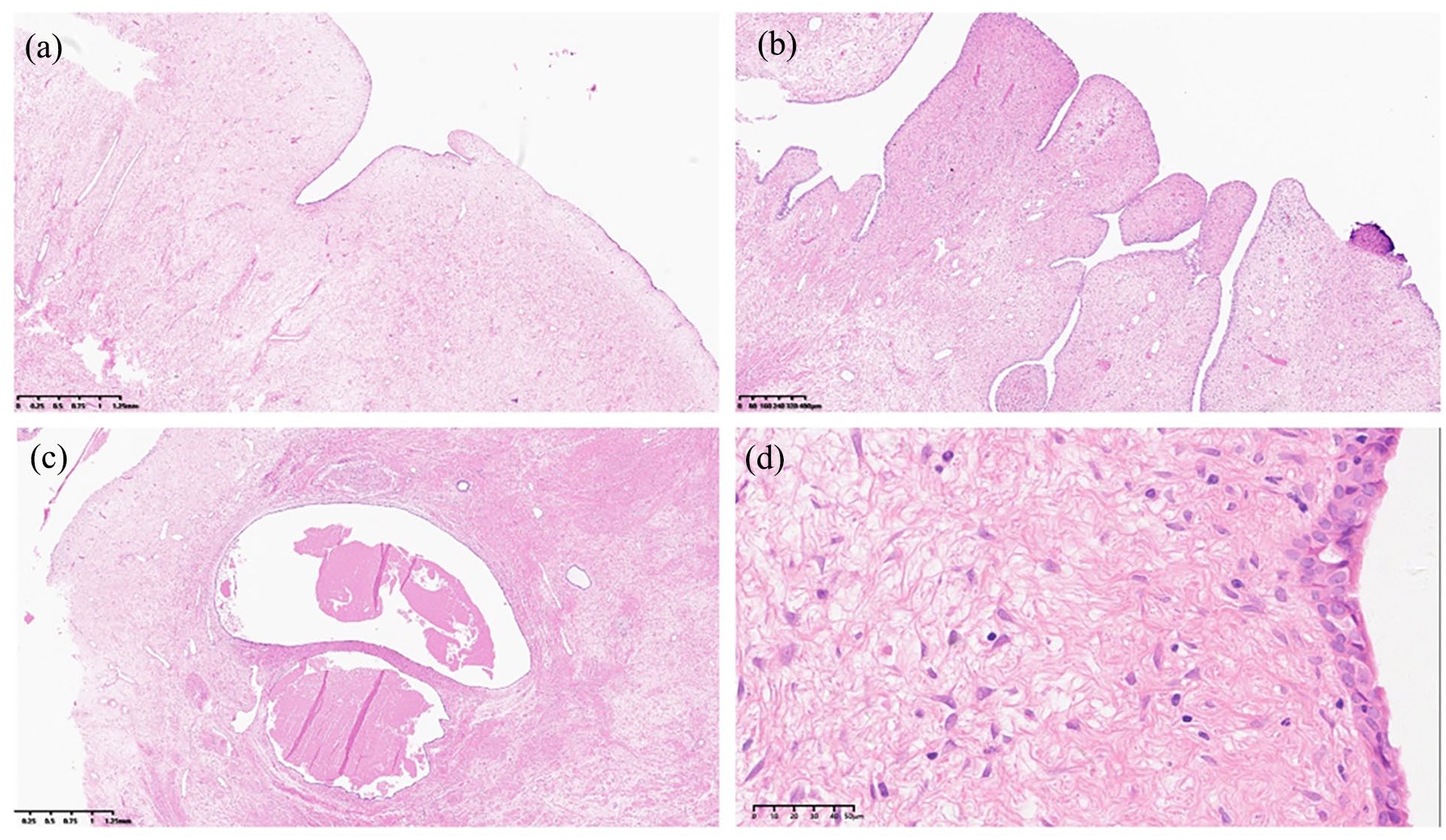
Histological morphology of the mixed epithelial and stromal tumor of the seminal vesicle: (a) The tumor was mainly composed of mesenchymal elements, and the surface was covered by epithelial cells (H&E 20×). (b) The epithelial layer and stroma predominantly exhibit a polypoid or leaf-like growth pattern (H&E 40×). (c) Focal epithelium forms cystic cavities within the stroma (H&E 20×). (d) The epithelium was composed of multilayered cuboidal cells with mild morphology and loose distribution of stromal cells without atypia or mitosis (H&E 400×).

Immunohistochemical examination: (a) and (b), respectively, revealed that the stromal cells are positive for both ER and PR (EnVision 100×).
Discussion
MEST are infrequent neoplasms that predominantly arise in the kidney and urogenital tract, comprising both epithelial and mesenchymal components. MEST primarily affects women in the perimenopausal stage and tends to involve the renal parenchyma. However, it is exceedingly rare in men. 1 The pathogenesis of MEST remains unclear: Renal MEST is thought to originate from remnants of the Müllerian duct such as Müllerian duct-like stromal cells, 2 whereas seminal vesicle MEST is believed to be related to congenital seminal vesicle cysts. 3 Both tumors share structural similarities, characterized by the proliferation of stroma and epithelium, with renal MEST commonly featuring an ovarian-like stroma, whereas seminal vesicle MEST typically exhibits a mild spindle cell morphology with a lower cell density. Both types may present with high-grade morphology in rare cases. Immunophenotypically, the epithelial and stromal markers are largely similar, with seminal vesicle MEST frequently showing stromal positivity for CD34, whereas renal MEST is often negative for CD34. A search on PubMed with the term “Seminal vesicle mixed epithelial stromal tumor” yielded 43 published cases, encompassing both benign and malignant tumors. The age of onset spanned from 31 to 70 years, with a median age of 49 years. The most frequent clinical manifestations observed included lower urinary tract symptoms in 23 cases (51.1%), hematospermia in 10 cases (22.2%), suprapubic, perineal, or groin pain in 5 cases (11.1%), abdominal pain in 4 cases (8.9%), hematuria in 4 cases (8.89%), and asymptomatic presentation in 7 cases (15.6%). These clinical variations may be attributed to the size and location of the seminal vesicle tumors. In the present case, the patient was asymptomatic, and the tumor was incidentally discovered during a physical examination.
Digital rectal examination can suggest prostatic hyperplasia, and the serum PSA level is typically within the normal range. Imaging examinations often indicate the presence of seminal vesicles or cystic solid lesions within the prostate. However, these signs, laboratory tests, and imaging examinations lack specificity and can lead to misdiagnosis. Therefore, pathological examination plays a crucial role in the diagnosis of this disease. Grossly, the tumors varied in size from 3 cm to 16 cm and displayed a cystic and solid appearance. The cut surface appeared multilocular and cavernous. Microscopically, the tumor exhibited a combination of epithelial and stromal components. The epithelium often formed irregular slit-like, polypoid, or foliar structures, and could also form round or oval cysts. The stroma exhibited varying degrees of proliferation, cell density, and mitotic figures. Based on the histological characteristics of the tumor stromal components, including cell density, atypia, mitotic count, and necrosis, Reikie et al. 4 proposed three grades (low/intermediate/high) of MEST. Low-grade MEST is characterized by the absence of stromal atypia and mitotic activity, with no nuclear pleomorphism or necrosis observed. Intermediate-grade MEST features increased stromal cellularity, low mitotic activity, nuclear pleomorphism, and atypia, yet lacks necrosis. Conversely, high-grade MEST, which portends a poor prognosis, is identified by invasive borders, marked nuclear pleomorphism and atypia, stromal overgrowth, significantly increased mitotic activity, hemorrhage, and necrosis. In this particular case, the stromal cells exhibited bland and loosely distributed characteristics, with no mitotic figures or necrosis, which is consistent with low-grade MEST. Previous case reports have summarized three cases of intermediate-grade MEST3,5,6 and only two cases of high-grade MEST,7,8 while the remaining cases were all low-grade MEST.
Due to the anatomical proximity of the seminal vesicles to the prostate, bladder, and rectum, it is essential to exclude invasion and metastasis from malignant tumors in adjacent tissues. PSA, CEA, and carbohydrate antigen 125 (CA125) are valuable in the differential diagnosis of seminal vesicle MEST. Generally, these three markers are not elevated in seminal vesicle MEST. In this particular case, the patient’s serum PSA and CEA levels were normal. CA125 was not measured. These findings do not provide evidence for the presence of malignant tumor metastasis and invasion from the prostate or intestinal tract. The primary tumor of the seminal vesicle was considered the first possibility. CT and MRI can determine the extent of lesion involvement, but they are challenged in differentiating between benign and malignant lesions due to their similar radiological presentations. When abnormal imaging findings are detected, a CT or ultrasound-guided core needle biopsy is necessary. 9 A notable characteristic of microscopic examination of MEST of the seminal vesicle is the presence of a cystic area lined by a single or multi-layered cuboidal epithelium surrounded by fibrous stroma. It is crucial to differentiate between benign and malignant MEST in the seminal vesicle. In this case, the imaging examination suggested a mass in the left seminal vesicle, which could not definitively exclude the possibility of malignancy. Therefore, a bilateral prostate and seminal vesicle tumor biopsy was performed under ultrasound guidance. The biopsy results of the left seminal vesicle showed no atypical epithelial and stromal fibrous components, consistent with the microscopic appearance of MEST of the seminal vesicle. However, due to the large size and cystic solid, the nature of the tumor cannot be fully ascertained by the core needle biopsy specimen, and the possibility of malignancy cannot be completely ruled out.
There are no established guidelines for the treatment of seminal vesicle MEST. The primary treatment approach is complete surgical resection of the tumor-bearing organs. Surgical intervention is recommended due to the absence of data supporting conservative treatment, even for asymptomatic cases. Previous cases have employed various surgical techniques, including tumor resection, seminal vesiculectomy, cystectomy, and prostatectomy, depending on the histological grade, size, and degree of tumor invasion. 10 For low or intermediate-grade MEST, tumor resection alone is typically performed. However, high-grade MEST may necessitate more aggressive surgical approaches, such as prostatectomy with seminal vesiculectomy. 11 In this case, we performed a robot-assisted radical prostatectomy and resection of the seminal vesicle solid tumor. The postoperative pathology confirmed a diagnosis of low-grade MEST, with no tumor component found in the surrounding prostate tissue.
Approximately 95% of previously reported cases of MEST are low- or intermediate-grade MEST, with only two cases demonstrating high-grade MEST. Fain et al. 7 previously documented a highly malignant case of MEST in a patient who presented with lower urinary tract symptoms, underwent cystoprostatectomy, received chemotherapy, and subsequently developed lung metastases after a 6-month follow-up period. Another case of highly malignant MEST was reported by Abe et al., 8 where the patient also presented with lower urinary tract symptoms and unfortunately succumbed to lung metastasis 11 months after seminal vesiculectomy. It is worth noting that MEST with a low or intermediate grade generally has a favorable prognosis and rarely recurs. However, the potential for malignant transformation in MEST cannot be definitively determined due to the limited follow-up time in the reported cases, as no instances of distant metastasis have been observed thus far.
Conclusion
This case report documents a large, clinically and radiologically suspected malignant lesion of the seminal vesicle, which was confirmed to be a low-grade MEST following radical surgical pathology. Despite the lack of long-term follow-up data, current evidence suggests a favorable prognosis for low-grade MEST with no risk of recurrence or metastasis. This report contributes to the expanding body of literature on MEST, fostering a deeper understanding of its clinical presentation and histopathological characteristics. Further research is warranted to elucidate the pathogenesis and long-term outcomes of MEST, ultimately refining diagnosis and treatment protocols for this enigmatic neoplasm.
Footnotes
Acknowledgements
None.
Author contributions
C.W. and Y.W.: conceived and designed the study; J.X.: administrative support; Y.W.: provided clinical treatment and management of patient; C.W.: collection and assembly of data; J.X.: performed pathological diagnosis; and C.W. and Y.W.: drafted the manuscript. All authors interpreted the data.
Data availability statement
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Medical and Health Science and Technology Program of Zhejiang Province under Grant Number 2024KY177.
Ethical approval
Ethical approval for this study was obtained from Research Ethics Committee of Hangzhou First People’s Hospital (Ethics approval number: ZN-20240117-0014-01)
Informed consent
Written informed consent was obtained from the patient for his anonymized information to be published in this article.
