Abstract
In November 2002, a 2-year-old, spayed Maltese Terrier in central Mississippi was presented for an acute illness characterized by uncontrolled hyperactivity that rapidly progressed to generalized tremors, ataxia, and intermittent hyperthermia. Postmortem examination after a 2-week course revealed mild, multifocal, nonsuppurative meningo encephalitis, with focal necrosis in the medulla. Reverse transcriptase-nested-polymerase chain reaction for West Nile virus (WNV) was positive on brain and negative on other tissues. Immunohistochemistry was negative on all tissues. The clinical, postmortem, and laboratory findings are consistent with acute encephalitis due to WNV infection. WNV infection should be considered in dogs showing signs of encephalitis when and where WNV and mosquito vectors occur.
After the introduction of a particularly neuroinvasive and virulent strain of West Nile virus (WNV) into the northeastern United States, there was a dramatic outbreak of fatal infections in birds, accompanied by much smaller but alarming numbers of fatal infections in horses and humans. 1, 16, 17, 20, 22 Within 3 years, the newly introduced Flavi-virus had swept across the North American continent, leaving in its trail untold thousands of dead birds and thousands of confirmed cases of illness including hundreds of fatalities in horses and humans. Reports of morbidity and mortality in other domestic animals have been less numerous. Avian and equine cases peaked in Mississippi during 2002.
At the beginning of November 2002, a 2-year-old, spayed Maltese Terrier was presented to the referring veterinarian with an acute onset of episodic, uncontrolled rolling, which progressed rapidly to whole-body tremors and ataxia. Initial treatment with oral phenobarbital and a short course of prednisone gave minimal improvement. After 1 week, the case was referred to the Animal Health Center, College of Veterinary Medicine, Mississippi State University, MS. The neurologic examination demonstrated signs of chorea consisting of involuntary, irregular, jerky adventitious movements involving the entire body. The ocular globes also demonstrated similar erratic movements. Conscious proprioception and hopping reactions were also mildly decreased in the front limbs. Diseases causing involuntary adventitious movements usually affect areas of the cerebral cortex, or specific extrapyramidal nuclei of the telencephalon, or cranial brainstem. 10 The differential diagnosis included multifocal inflammatory diseases—infectious, noninfectious, and immune mediated. Complete blood count and serum chemistries were unremarkable, and cerebrospinal fluid (CSF) analysis revealed no significant abnormalities. An electroencephalogram revealed changes suggestive of encephalitis. On the basis of clinical signs and electroencephalogram results, the dog was treated for infectious causes of encephalitis. During hospitalization in the intensive care unit, the dog became very excitable. Treatment with doxycycline, clindamycin, diazepam, diphenhydramine, and fluids failed to prevent increasing severity of involuntary movements and intermittent periods of hyperthermia (up to 108 F/42.2 C).
Serology for Toxoplasma gondii, Neospora caninum (ProtaTek, Chandler, AZ), Ehrlichia canis, and Rickettsia rickettsii (Antech, Southaven, MO) were negative, and paired serum and CSF titers for canine distemper virus and parvovirus (Colorado Veterinary Diagnostic Laboratory, Ft. Collins, CO) indicated protective immunity but failed to reveal intrathecal antibody production. After 6 days of hospitalization and treatment, approximately 2 weeks after the onset of clinical signs, the dog was euthanatized because of its deteriorating condition.
Necropsy examination revealed no gross abnormalities. Microscopic examination of the brain revealed a mild, multifocal, nonsuppurative meningoencephalitis, which involved gray matter or mixed gray/white matter areas such as the brainstem. This was characterized by small perivascular cuffs of lymphocytes within the parenchyma and sometimes the adjacent leptomeninges. Mild microgliosis of the neuropil often accompanied the perivascular cuffing (Fig. 1). Foci of inflammation, which were sometimes bilateral, were present in the pyriform lobe and associated parahippocampal gyrus and hippocampus, the pons and medulla, and the cerebellar cortex. A unique lesion close to the midline raphe of the medulla at the level of the olivary nuclei consisted of an area of necrosis with fibrin effusion and hemorrhage, clusters of macrophages (gitter cells), and many swollen axons (spheroids) with a couple of small lymphocytic cuffs nearby (Fig. 2). Spheroids stained positively by the Bielschowsky's method for axons. 19 The only other significant findings were limited to the liver, which showed patchy individualization of hepatocytes, cytoplasmic eosinophilia, nuclear pyknosis, and lysis of hepatocytes (acute diffuse moderate hepatic necrosis), with numerous binucleate hepatocytes and without significant inflammatory cell infiltration. Numerous budding yeast with pseudohyphae (Candida sp.) were present in the gastric mucus without evidence of mucosal invasion or inflammation.
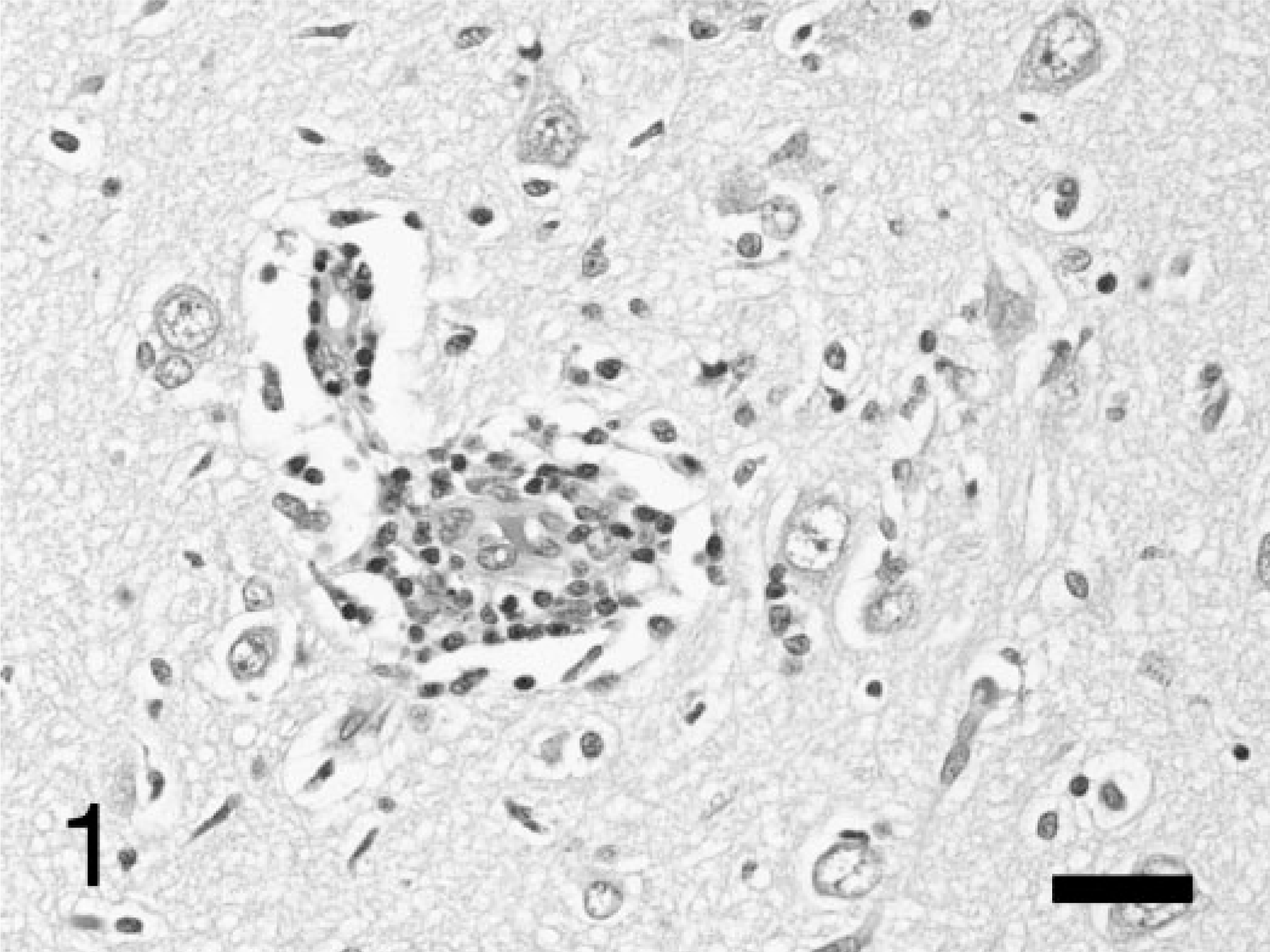
Cerebrum; dog. Scattered small perivascular cuffs of mononuclear inflammatory cells and mild microgliosis in the pyriform lobe. HE stain. Bar = 35 μm.
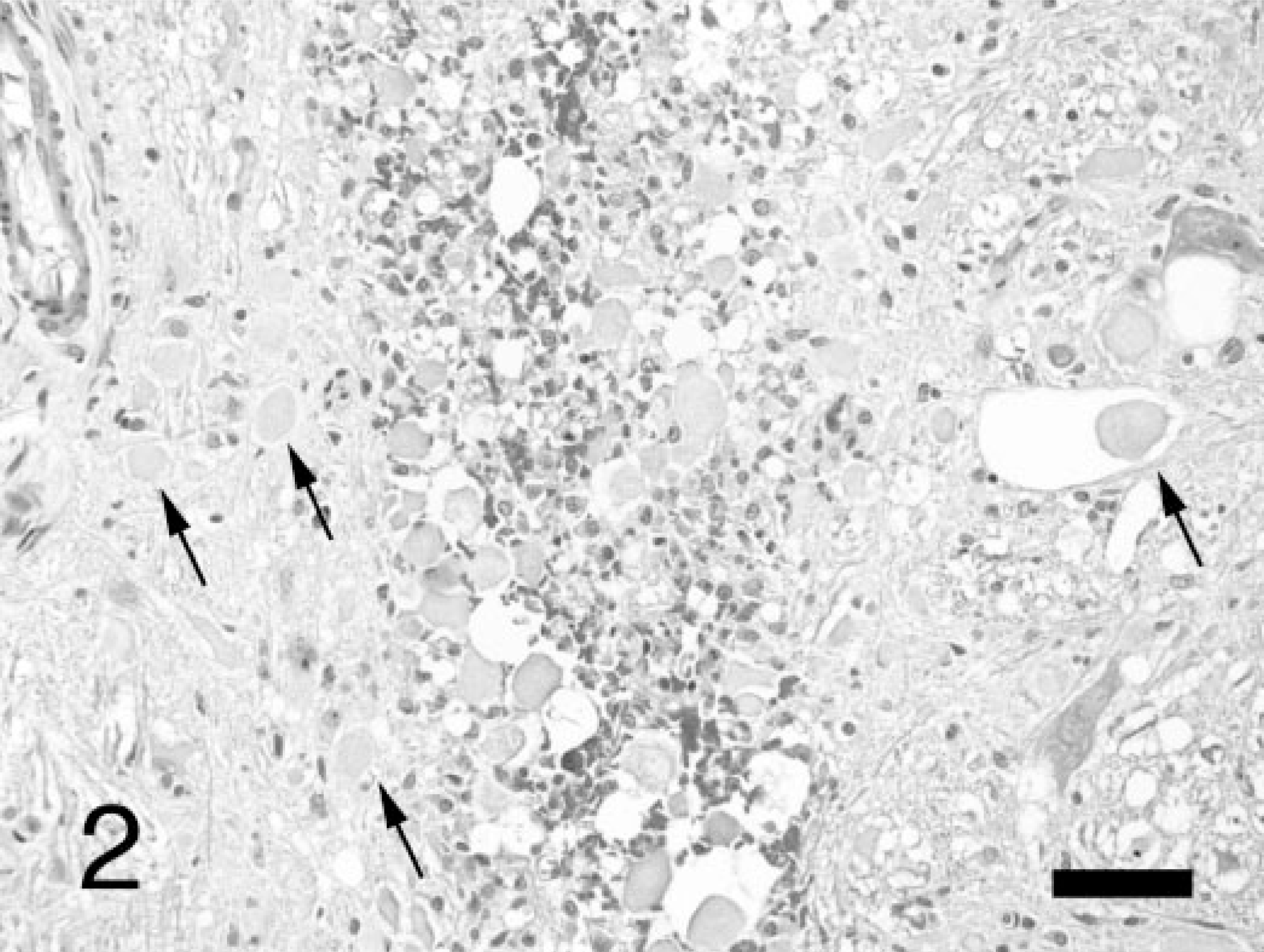
Medulla oblongata; dog. An area at the midline raphe of the medulla showing many swollen axons (spheroids) indicated by arrows, surrounding an area of necrosis with hemorrhage and macrophages. HE stain. Bar = 70 μm.
Polymerase chain reaction (PCR) for WNV was performed on fixed brain and unfixed spinal cord, liver, and kidney. Pieces of formalin-fixed brain weighing 30–100 mg were removed from the medulla oblongata near the lesioned site and various other sites in the brain stem, cerebral cortex, and cerebellum, rinsed with water, and placed in RNAlater™ reagent (QIAgen, Valencia, CA) for at least 1 hour before RNA extraction. Samples of frozen unfixed spinal cord, kidney, and liver were extracted without preprocessing. Tissue samples were disrupted using a motor-driven pestle (PelletPestle, Kontes, Vineland, NJ), and RNA was extracted using Trizol™ reagent (Invitrogen, Carlsbad, CA) or RNea-sy™ mini spin columns (QIAgen), according to manufacturer's instructions. Reverse transcriptase-nested-polymerase chain reaction (rtnPCR) was performed using the nested primers and a modification of the method described by Johnson et al., 12 with commercial reagent kits (OneStep™ RT PCR, Qiagen, Valencia, CA, and Taq SuperPak™, Sigma, St. Louis, MO). Positive control was a 1 : 1,000 dilution of pooled RNA extracted from unfixed WNV-positive avian tissues (predominantly kidney and brain). DNA bands were detected after agarose gel electrophoresis using GelStar (Cambrex, East Rutherford, NJ). Three of five pieces of formalin-fixed medulla were positive on rtnPCR, whereas all other specimens of fixed brain (cerebral cortex, cerebellum, and brain stem) and unfixed (frozen) spinal cord, kidney, and liver were negative.
Immunohistochemistry (IHC) was performed in an attempt to demonstrate WNV antigen. Sections of formalin-fixed, paraffin-embedded (FFPE) tissues were cut at 5 μm and stained using an autostainer (Dako, Carpinteria, CA) and prepackaged streptavidin–biotin–horseradish peroxidase detection reagents (LSAB2, Dako), according to manufacturer's instructions. Sections of brain, kidney, pancreas, liver, heart, spleen, adrenal gland, and tongue were stained using murine polyclonal antibody for WNV (VR-1267-AF, ATCC, Manassas, VA) after pretreatment with proteinase K (Dako). This polyclonal antibody showed moderate diffuse background staining in all tissues, complicating interpretation. No specific staining was seen except possibly in the brain where neurons diffusely showed uniform light cytoplasmic staining compared with control slides stained with irrelevant monoclonal IgG1. The results were considered to be equivocal because of the high background staining and diffuse nature of neuronal staining, with little variation in intensity. These results suggested a paucity of antigen in the dog's brain, when compared with infected avian tissues where robust cytoplasmic staining was seen.
Brain sections were also stained using monoclonal antibody to canine distemper virus nucleoprotein (CDV-NP, VMRD, Pullman, WA), with negative results. IHC on FFPE brain for rabies virus was performed in another laboratory (Prairie Diagnostic Service, Saskatoon, SK, Canada), with negative results.
Although fatal encephalitis due to WNV is well documented in many avian species, 22 horses, 8 and humans, 20 clinical signs of disease caused by this virus have rarely been documented in dogs. In areas where WNV is endemic, a high prevalence of specific neutralizing antibody indicates that infection in dogs is common, even if clinical disease is not. One serosurvey from South Africa, where WNV is endemic, showed 37% of dogs had neutralizing antibodies to WNV. 5 A serosurvey in New York City, immediately after the first identification of WNV in the western hemisphere, showed that a higher percentage of dogs (10%) than horses (3%) or humans (2.5%) had specific neutralizing antibody near the epicenter of the outbreak (Queens). 15 A previous report from South Africa described the isolation of a virus, later identified as WNV, from the brain of a dog with severe clinical signs of encephalitis. 7, 21 In one experiment, infection of three dogs with WNV produced asymptomatic seroconversion in all the three but measurable viremia in only one of these animals, which was later found to suffer from hyperadrenocorticism. 5 A more recent study using the US strain demonstrated low-level viremia in all the four dogs and eight cats infected, 2 with no clinical signs in dogs and only mild, transient febrile disease in cats.
Two contemporary reports of severe WNV infection in US dogs have been published. Buckweitz et al. described postmortem evidence of WNV replication in many tissues of an 11-year-old, male, mixed-breed dog with renal and central nervous system (CNS) disease; quantitative reverse transcriptase–polymerase chain reaction (RT-PCR) found that viral sequences were most abundant in kidney, followed by heart, brain, lung, and spleen. 6 Lichtensteiger et al. describe postmortem findings after euthanatization of an 8-year-old, mixed-breed dog with severe somatic disease that included myocarditis, as well as fatal WNV encephalitis in a 3-month-old wolf pup. 18 Both these reports documented antigen and genetic sequences of WNV, using IHC and RT-PCR, respectively, in multiple extraneural tissues associated with lesions of necrosis and inflammation.
This report documents a case of severe neurologic disease in a young adult dog with a 2-week duration of CNS signs, where evidence of WNV infection was limited to the brain. Histologic lesions in the brain were nonspecific but compatible with findings of WNV encephalitis reported in horses, 8 humans, 20 and the two reports in dogs, 6, 18 both with respect to location (brain stem and medulla) and type (non-suppurative encephalitis with areas of necrosis). Clinical signs of balance loss (rolling) and ataxia would be compatible with cerebellomedullary disease, whereas whole-body tremors suggest a more diffuse disturbance of the brain. Evidence of meningoencephalitis was found in the pyriform lobe and hippocampus, brain stem, and cerebellum/medulla but spared much of the prosencephalon. Hepatic necrosis in this case without evidence of local viral replication (PCR and IHC negative) was possibly because of the severe recurrent hyperthermia. Hepatic damage due to previous viral replication that was cleared during the illness cannot be excluded although no inflammation was apparent. Numerous binucleate hepatocytes suggested subacute damage with regeneration. The presence of Candida yeast in the stomach was considered an incidental finding attributable to treatment with antibiotics. Contrasting with the findings of Buckweitz et al. 6 and Lichtensteiger et al., 18 no lesions, viral nucleic acids, or viral antigen were detected in any extraneural tissues including frozen and fixed kidney and liver from this dog.
The area of necrosis found in the brain stem of this dog was associated with a localized macrophage response (gitter cells) but not overt local inflammatory infiltrate suggesting, as described in horses, 8 that the lesion may have a vascular or possibly immune-mediated etiology rather than a direct cytolytic or inflammatory etiology. IHC for WNV on brain was unable to convincingly demonstrate viral antigen in this case and in the case reported by Buckweitz et al., 6 indicating lower levels of viral replication than seen in avian tissues, again similar to findings in horses. 12 Lichtensteiger et al. 18 did not report IHC of the brain.
The comparatively higher rate of neurologic disease seen in equine infections with the US strain of WNV compared with those in Italy 8 is supported by experimental studies in mice, which indicate that the US strain is highly neuroinvasive compared with other strains. 3 Additional studies in mice have demonstrated that exposure to gaseous anesthetics and CO2 increase WNV neuroinvasion in a concentration-and time-dependent manner, 4, 13 suggesting that other environmental or host factors may influence the development of neurologic disease in animals. The low incidence of clinical disease seen in dogs in the face of high seroprevalence suggests that dogs are more resistant to neuroinvasion than either humans or horses.
Evidence of extraneural infection was not found in horses with WNV encephalomyelitis 8 as in this dog. Perhaps, in this Maltese Terrier the immune response had cleared the virus from extraneural tissues. The presence of abundant viral replication in extraneural tissues in the two other reports of US canine infections suggests that some viral replication occurs in extraneural tissues before neuroinvasion and that the kidney and heart are likely target tissues in the dog. Buckweitz et al. 6 suggest renal biopsy for the diagnosis of WNV infection in dogs, but Lichtensteiger et al. 18 found no viral antigen in the kidney of the dog they examined. The clinical value of renal biopsy 18 or even urine testing for WNV, presumably using RT-PCR, has not yet been critically evaluated.
Extraneural replication of WNV in humans may be extrapolated from reports of fatal encephalitis in recipients of kidneys from infected donors that began 13–18 days after transplantation and from severe encephalitis in a heart recipient, 9, 11 indicating that the disease can be transmitted through blood or organ transplantation. Further evidence of robust WNV replication in extraneural tissues of mammals is presented in a recent report documenting fatal WNV infection in three Eastern Fox squirrels, with evidence of renal, cerebral, cardiac, hepatic, and pulmonary viral replication. 14 Additional studies are needed to elucidate the details of the biology and pathophysiology of WNV infections in mammals.
The continued significance of WNV to animal and human health in this country will probably involve the virus becoming endemic with sporadic outbreaks. Public health agencies often provide timely information regarding outbreaks of human disease. It is possible that analysis for antibodies to WNV in canine and feline sera could provide valuable epidemiologic information on the spread and incidence of WNV.
Footnotes
Acknowledgements
We thank Stephanie Mays and Chelsea Miller for technical assistance and the Mississippi Veterinary Diagnostic Laboratory System for financial support.
