Abstract
Tissues were obtained at necropsy from the nasal vestibule, turbinates, nasopharynx, trachea, tracheobronchial bifurcation, and lung from each of 10 clinically healthy calves persistently infected (PI) with bovine viral diarrhea virus (BVDV) serotype 2a. Tissues from the nasal vestibule were obtained by biopsy from five additional PI calves. Formalin-fixed tissues were processed for immunohistochemistry to localize the distribution of BVDV throughout the respiratory tract. Antigen distribution and intensity were subjectively evaluated. Throughout the respiratory tract, mononuclear leukocytes, vascular smooth muscle, and endoneural and perineural cells had BVDV immunoreactivity (BVDV-IR). Multifocally, squamous and ciliated columnar epithelium throughout the respiratory tract contained weak to moderate BVDV antigen. Viral antigen was not seen in goblet cells. BVDV-IR in mixed tubuloalveolar glands of the nasal cavity was weak to strong in serous secretory cells and ductular epithelium. Chondrocytes of the concha often contained BVDV antigen diffusely. Nasal mucus-secreting and tracheobronchial glands multifocally contained weak viral signal. In all cases, alveolar macrophages had moderate to strong BVDV-IR, whereas BVDV-IR in alveolar epithelial cells was weak to moderate. BVDV was present in interalveolar leukocytes and mesenchymal cells. Results indicate that serous secretions of the nasal cavity, productive viral infection of epithelium, and infected leukocytes in respiratory secretions are likely major sources of infectious BVDV from PI calves. The presence of BVDV antigen in respiratory epithelium is, at least, indirect support for the notion that this virus predisposes PI cattle to secondary microbial infections.
Keywords
Bovine viral diarrhea virus (BVDV), a pestivirus, infects cattle worldwide. BVDV isolates are classified into two biotypes on the basis of their cytopathic (CP) or non-CP (NCP) effects in cell culture. 5 Genetic and antigenic differences further separate BVDV isolates into types 1a, 1b, 2a, and 2b. 10, 13, 33, 34 BVDV causes several clinical syndromes including acute or chronic infections with manifestations of disease in several organ systems and immunosuppression. 4, 5, 15 Fetal infections with a NCP strain at less than 150 days of gestation can result in persistent infections of the fetus with immunotolerance to the infecting BVDV strain. 5 Upon birth, persistently infected (PI) calves are permanently infected and viremic with BVDV while being more susceptible to other infectious diseases than are non-PI cattle. Mucosal disease can develop in PI calves if they are infected with a CP BVDV isolate that is similar to the NCP strain. Finally, PI calves may not respond adequately to immunization for other infectious agents and, most importantly, are sources of infectious virus for other cattle within a herd. 5, 22, 31 Demonstration of persistent viremia with NCP BVDV has been the gold standard for diagnosis of PI cattle. The commonly used test to detect PI cattle is the so-called ear notch test in which formalin-fixed skin from the ear is examined with immunohistochemistry (IHC) for BVDV antigen in keratinocytes, hair follicle epithelium, and hair bulbs. 27
BVDV is considered a major factor in precipitating many outbreaks of severe bovine respiratory disease in transported beef cattle.
12,
14,
15,
30,
37
In those situations, BVDV infection predisposes cattle to infections with bacterial pathogens such as
PI cattle readily shed BVDV to other cattle when in feedlot pens, and the presence of a PI calf can increase health costs and reduce weight gains among other cattle within the pen. 22, 31, 37 The sources of infective BVDV shed by PI cattle are alimentary, reproductive, and respiratory tract secretions as well as milk. 22 BVDV has been demonstrated in epithelial cells of the skin, digestive tract, pancreas, mammary glands, adrenal gland, liver, lung, kidney, reproductive tracts, and central nervous systems of PI cattle. 2, 9, 11, 20, 27, 35, 36, 39 Viral antigen is also found in lymphocytes, macrophages, and blood vessels. 24, 35, 40 In addition, BVDV antigen is distributed in the skin in keratinocytes, hair follicle epithelium, and hair bulb in PI cattle. 27 This study was undertaken with two objectives. The first objective was to determine the distribution of BVDV antigens in cells from the respiratory tract of PI cattle to elucidate the potential for the respiratory tract to be a source of viral shedding. The second objective was to determine the extent of respiratory epithelial infection as a possible mechanism by which PI cattle are more susceptible to other respiratory pathogens.
Materials and Methods
Animals
Ten PI calves, 4–6 months of age, were obtained from two different beef herds. Five were from herd A, and five were from herd B. All calves were clinically normal and diagnosed as PI by the IHC test on ear notches at least 2–4 weeks before the collection of other samples. All calves were euthanatized by barbiturate overdose. At the time of sample collection or necropsy, another ear notch was collected for IHC evaluation, and all samples were positive for BVDV antigen. Before euthanasia by barbiturate overdose, blood samples were obtained from calves in ethylenedi-aminetetraacetic acid–treated tubes for viral isolation. A complete necropsy was done, and tissues from the nasal vestibule, turbinates, nasopharynx, trachea, tracheobronchial bifurcation, and lung were collected immediately after euthanasia and fixed in 10% neutral buffered formalin solution.
In addition to the respiratory samples obtained at necropsy, biopsy samples were obtained from the nasal vestibules from five PI cattle. One PI calf was from herd A and was not necropsied. Four other cattle examined (herd C) were approximately 18-month-old steers that had been diagnosed as PI when 6 months of age and maintained at an experimental feedlot for use in transmission studies of BVDV. Persistent infection with BVDV had been repeatedly established through viral isolation from peripheral blood leukocytes. Tissue samples were similarly fixed in formalin solution.
Virus isolation and identification
Virus isolation from blood samples was done in Madin-Darby bovine kidney and in bovine turbinate cells. 15 BVDV isolates were subtyped by differential polymerase chain reaction testing as described previously. 10, 33 Sequencing of the 5′ UTR and subsequent analysis indicated that these BVDV isolates were subgenotype BVDV2a. 10
Immunohistochemistry
IHC was used to identify BVDV antigen in formalin-fixed tissues. Tissues were kept in formalin for 7 days for herd A and 3 days for herds B and C before processing. Primary antibody was an immunoglobulin-G1 (IgG1) monoclonal antibody (designated 3.12F1) that is directed against the GP48 BVDV protein (developed by Dr. J. T. Saliki, Oklahoma State University, Stillwater, OK). The antibody in ascitic fluid was diluted 1 : 75,000 in phosphate-buffered saline solution containing Tween 20. Secondary and tertiary antibodies were rabbit anti-Mouse IgG1 antibody diluted 1 : 500 and biotinylated goat anti-rabbit IgG (H & L), respectively (Zymed Laboratories Inc., San Francisco, CA). Streptavidin-horseradish peroxidase and Nova Red Substrate (Vector Laboratories, Burlingame, CA) were used to develop the color reaction. All reactions were conducted in a DAKO Model LV Auto Immunostainer (Dako, Carpinteria, CA). Six-microgram sections of paraffin-embedded, formalin-fixed tissue were cut, pronase treated, and incubated with the primary antibody solution. Positive control sections included BVDV-positive skin sections. For negative controls, duplicate sections of tissues from the PI calves were treated with normal mouse IgG before treatment with secondary and tertiary antibodies, and respiratory tissues from a noninfected calf from herd B were processed with the primary, secondary, and tertiary antibodies. Negative control tissues were treated for color development and examined microscopically as described above.
The distribution of antigen in respiratory epithelium, glands, lamina proprial structures, and leukocytes was documented throughout the respiratory tract. Antigen distribution was designated as multifocal or diffuse. Intensity of BVDV immunoreactivity (BVDV-IR) was subjectively scored using the following system: 0 = no detectable antigen; + = weak, antigen faintly detected uniformly within the cytoplasm or in a stippled pattern; ++ = moderate, antigen readily detected uniformly within the cytoplasm; +++ = strong, antigen staining is intense within the cytoplasm.
Results
NCP BVDV was isolated from each PI calf from herds A, B, and C, and each isolate was typed as BVDV2a. Pneumonia was not present in any of the calves examined. Histologically, one calf from herd A and three calves from herd B had mild suppurative rhinitis characterized by neutrophilic infiltrates within the superficial epithelial layers of the vestibule and respiratory passages along with mild increases in lymphocytes and plasma cells in the lamina propria.
Using IHC, BVDV-IR was demonstrated in the cytoplasm of infected cells. Mononuclear leukocytes contained BVDV antigen within tissue throughout the respiratory tract (Tables 1, 2). Morphologically these cells were primarily macrophages, although in tonsillar tissue of the nasopharynx, cells that appeared to be lymphoblasts and were within germinal centers contained BVDV antigen. Macrophages containing BVDV antigen were usually within the connective tissues of the lamina propria; however, in calves with mild rhinitis, those cells were also readily visible within the mucosal epithelial layers (Figs. 1, 2). Throughout all sections, smooth muscle of arteries and veins contained BVDV antigen; however, immunoreactivity of endothelium was rarely and only weakly detected (Fig. 3). In addition, in most cases, endoneural and perineural cells within peripheral nerves contained BVDV antigen (Fig. 4).
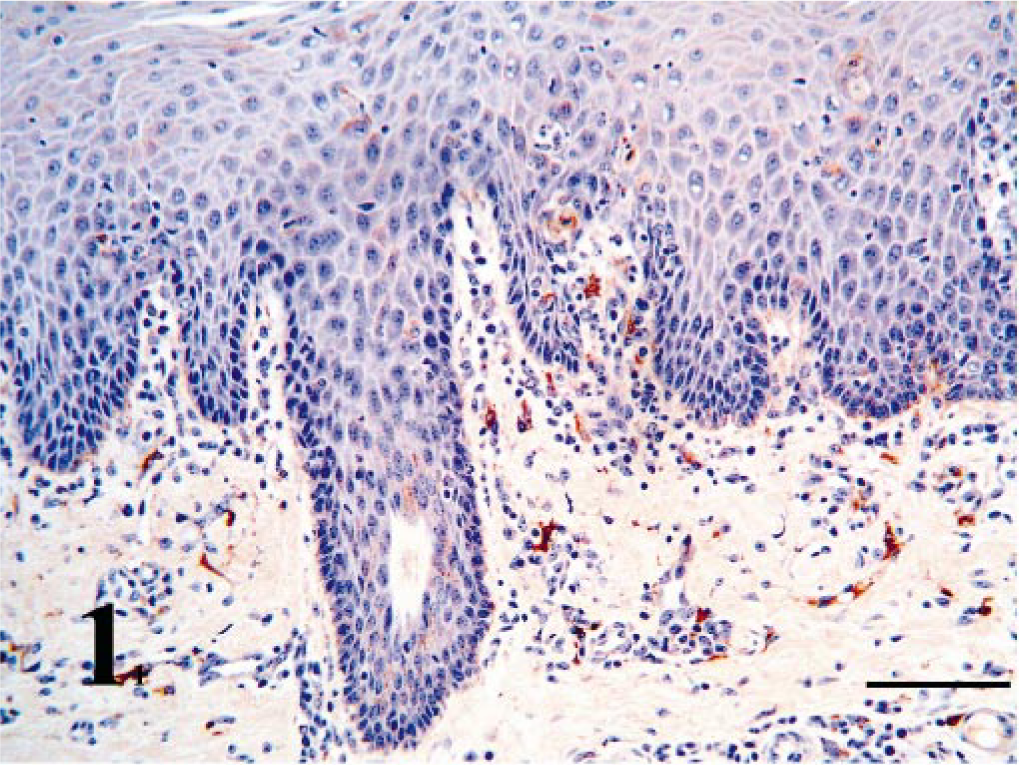
Nasal vestibule; PI calf without rhinitis. BVDV antigen is present in epithelium and in macrophages in the lamina propria. Bar = 90 μm.
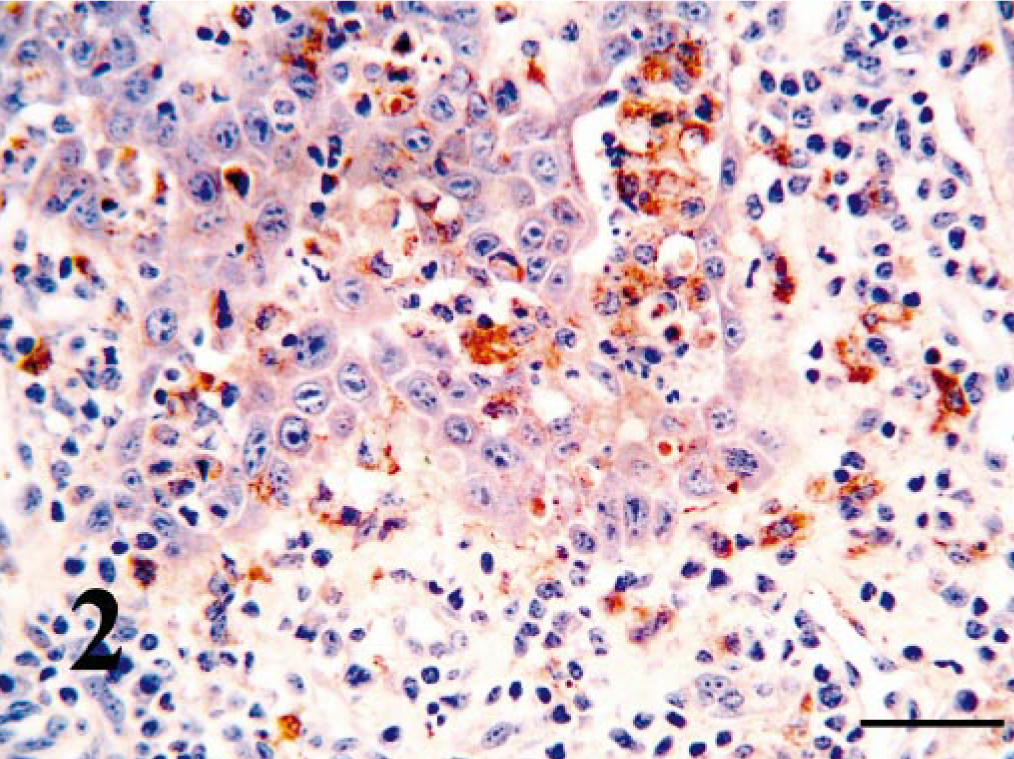
Nasal vestibule; PI calf with rhinitis. BVDV-positive leukocytes have infiltrated the hyperplastic epithelial layers and are present in the lamina propria. Bar = 25 μm.
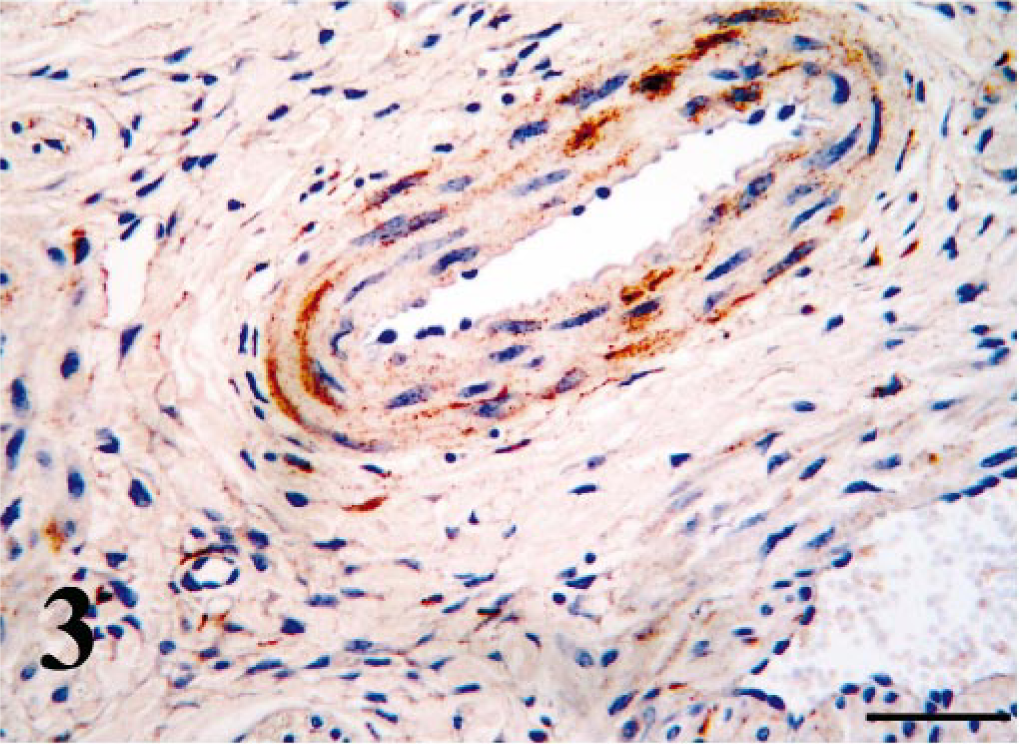
Arterial wall, nasal vestibule; PI calf. Strong antigen signal is present in smooth muscle. Bar = 50 μm.
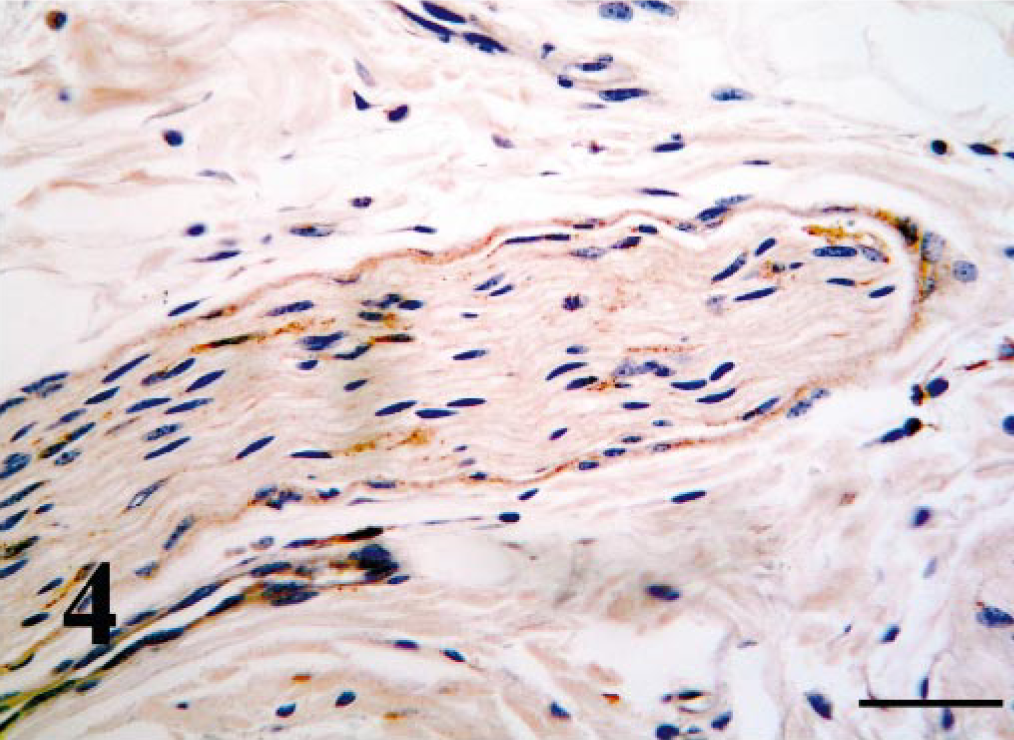
Peripheral nerve, nasopharynx; PI calf. Endoneural and perineural cells contain a weak antigen signal. Bar = 50 μm.
Distribution of BVDV antigens in the respiratory tract of necropsied PI calves.∗
∗ 0 = no detectable antigen; + = weak, antigen faintly detected uniformly within the cytoplasm or in a stippled pattern; ++ = moderate, antigen readily detected uniformly within the cytoplasm; +++ = strong, antigen staining is intense within the cytoplasm.
BVDV antigen distribution in biopsies from the nasal vestibule from PI calves.∗.

∗ 0 = no detectable antigen; + = weak, antigen faintly detected uniformly within the cytoplasm or in a stippled pattern; ++ = moderate, antigen readily detected uniformly within the cytoplasm; +++ = strong, antigen staining is intense within the cytoplasm.
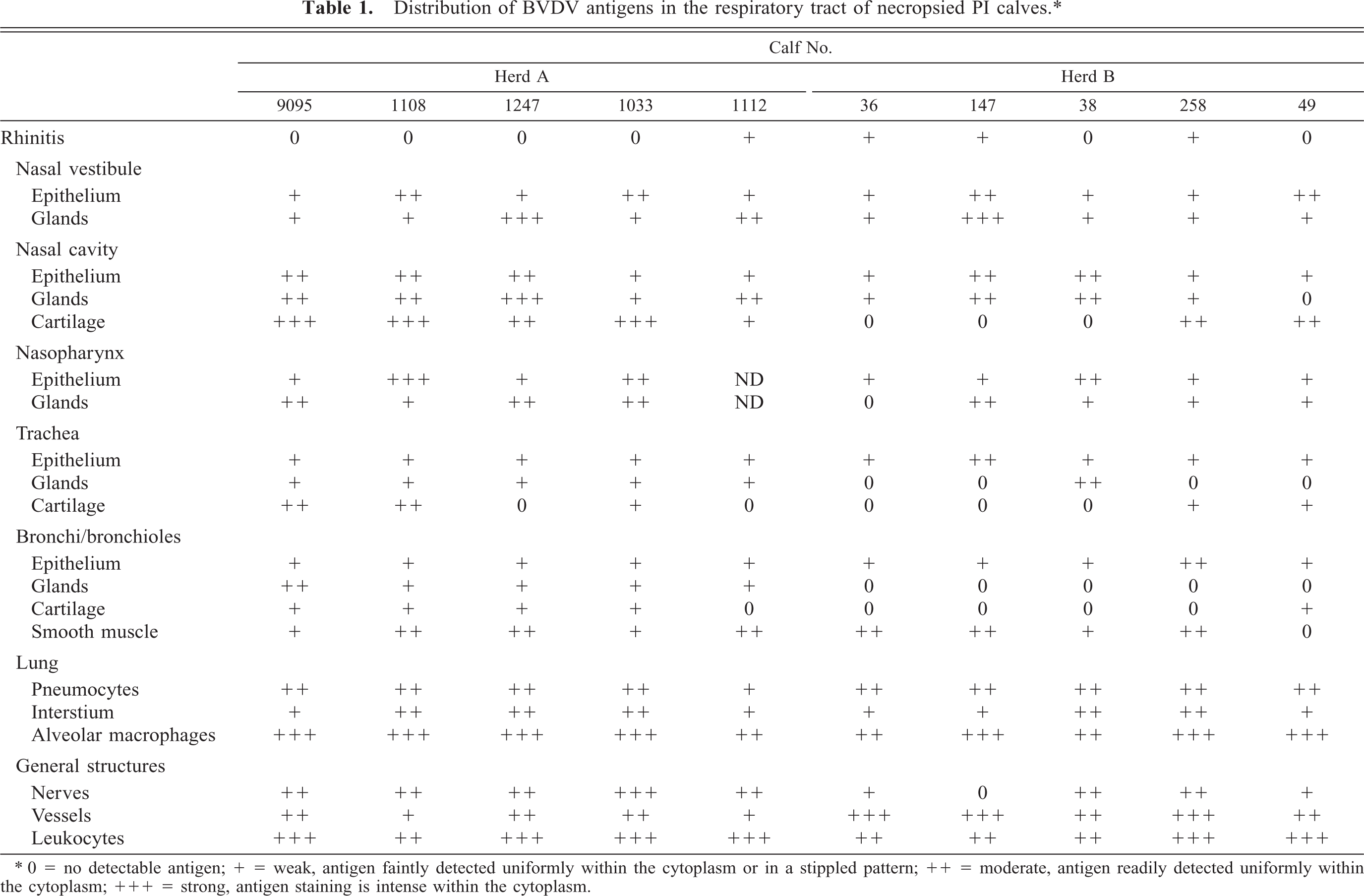
Within the nasal vestibule, multifocal areas of nasal epithelium contained weak to moderate BVDV-IR in all cattle necropsied or biopsied (Tables 1, 2). Multifocally, both basal cells and keratinocytes contained BVDV antigen as fine stippled cytoplasmic granules (Fig. 1). Mixed mucosal tubuloalveolar glands ranged from no immunoreactivity in two biopsied cases to strong immunoreactivity in one calf each from herds A and B. Antigen signal was rarely found in mucus-secreting glandular cells but was found primarily in serous secretory cells and ductular epithelium (Fig. 5).
Nasal vestibule; PI calf. Mucus glands ducts contain strong antigen signal, whereas mucus-secreting cells are mostly antigen negative. Bar = 50 μm.
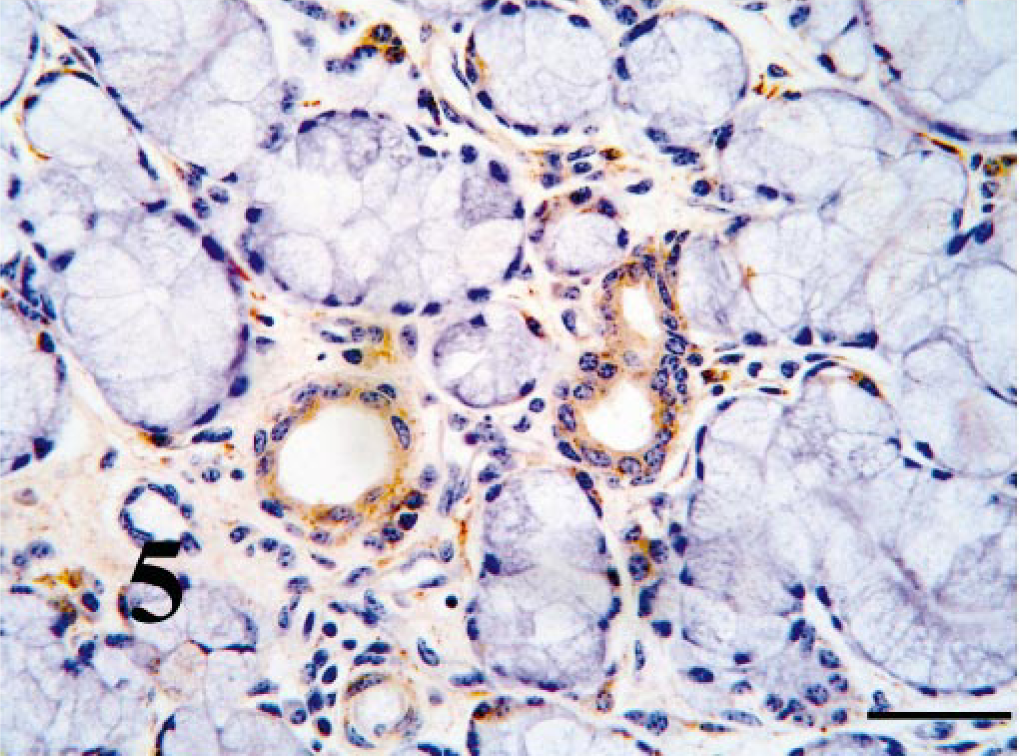
Within the turbinate region of the nasal cavity, ciliated epithelium BVDV-IR ranged from weak to moderate (Fig. 6). Viral antigen was not seen in goblet cells. BVDV-IR in mixed tubuloalveolar glands ranged from weak to strong with strong reactivity being concentrated in serous secretory cells and ductular epithelium (Fig. 7). Chondrocytes of the concha often had diffuse weak to strong BVDV-IR (Fig. 8). In those cases, cells of the perichondrium had similar BVDV-IR. Nasopharyngeal epithelium and tubuloalveolar glands were usually multifocally BVDV positive.
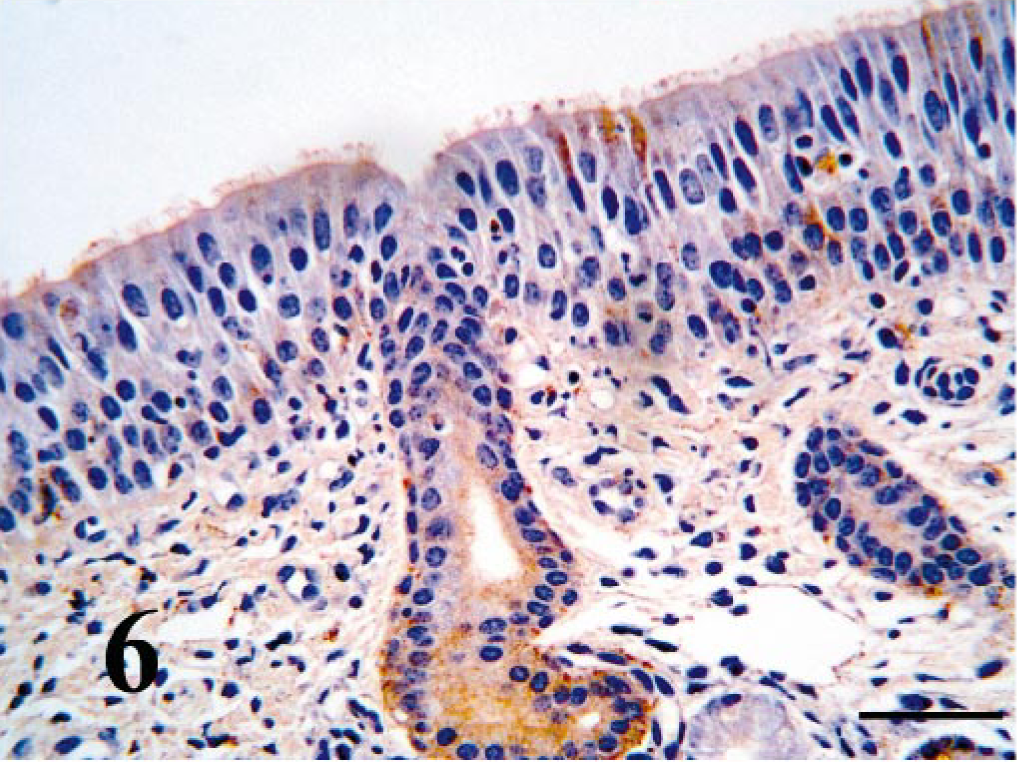
Nasal turbinate mucosa; PI calf. Surface epithelium and serous gland duct contain strong antigen signal. Bar = 25 μm.
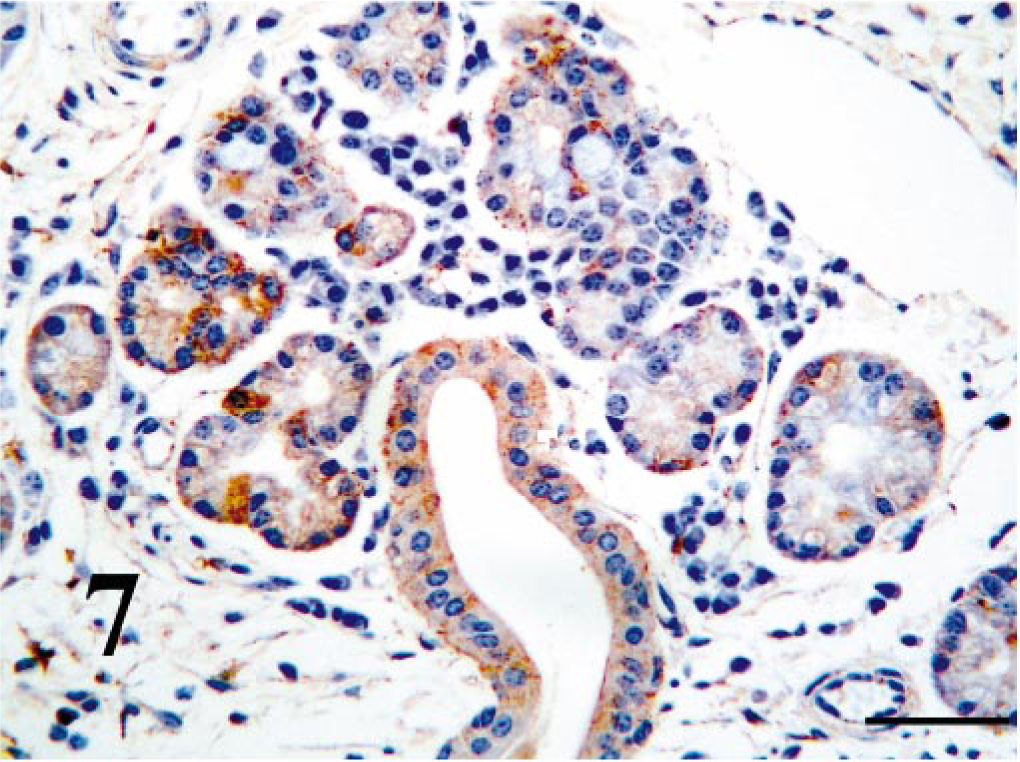
Nasal turbinate mucosa; PI calf. Serous glands and duct contain strong antigen signal. Bar = 25 μm.
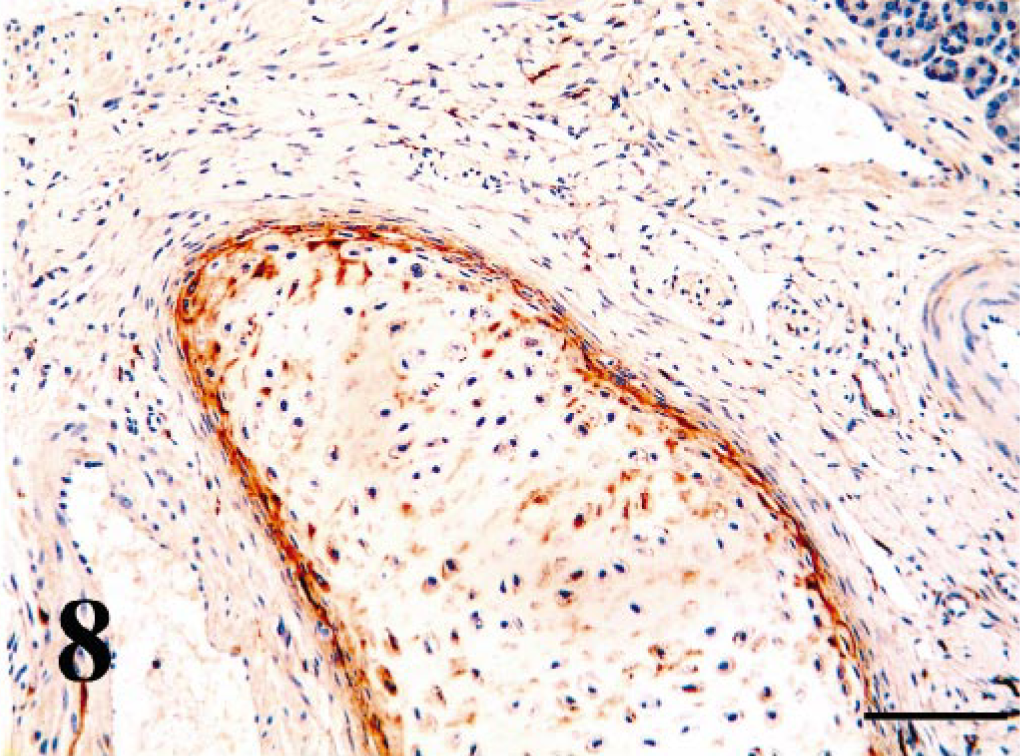
Nasal turbinate concha; PI calf. Chondrocytes and pericondrial cells contain strong antigen signal. Bar = 90 μm.
Overall, BVDV-IR in tracheal and bronchial columnar epithelium was usually diffusely weak with sparing of the goblet cells (Fig. 9). Multifocally, BVDV-IR in tracheal and bronchiolar glands ranged from negative to weak. Chondrocytes and perichondrial mesenchymal cells contained BVDV antigen in six of the calves. Peribronchial smooth muscle cells contained weak to moderate BVDV-IR in nine of the calves.
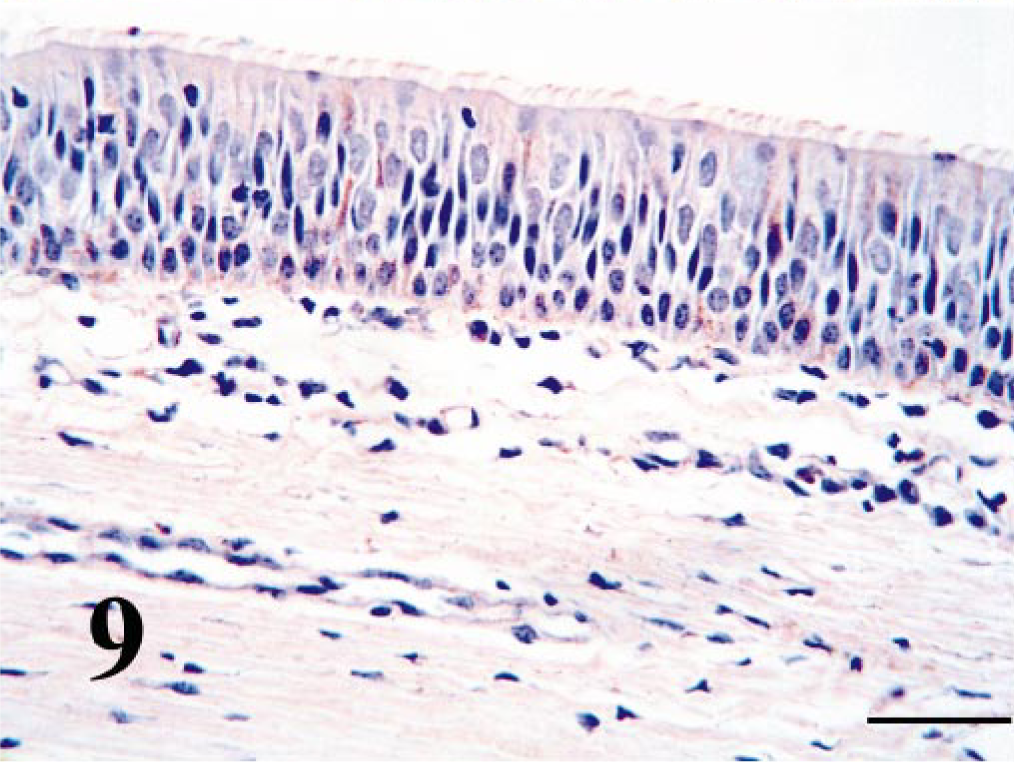
Trachea; PI calf. Epithelium contains weak, multifocal antigen signal. Bar = 25 μm.
Within the lung, BVDV-IR in alveolar macrophages was diffusely moderate to strong in all calves. BVDV-IR in alveolar epithelial cells was diffusely weak to moderate (Fig. 10). Multifocally cells within the inter-alveolar septa often contained BVDV antigen. Those cells appeared to be both leukocytes and mesenchymal cells.
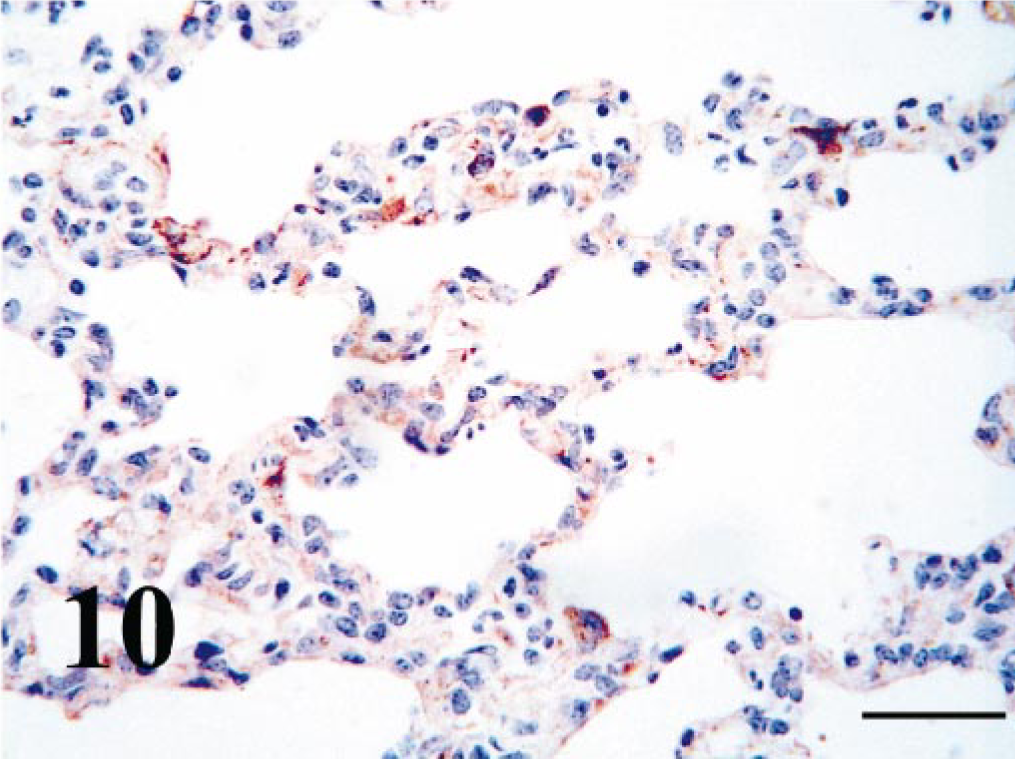
Lung; PI calf. Pneumocytes and cells within the interalveolar septa contain strong antigen signal. Bar = 25 μm.
Discussion
In previous studies, BVDV was demonstrated by IHC in epithelial cells of the skin, digestive tract, pancreas, mammary glands, adrenal gland, liver, lung, kidney, reproductive tracts, and central nervous systems of PI cattle. 2, 9, 11, 20, 27, 35, 36, 39 Macrophages/histiocytes were BVDV positive within all tissues examined. 35 In the lung, BVDV antigen was demonstrated as faint minimal staining within bronchial mucosa, alveolar macrophages, and blood vessels of one PI calf. 35 In contrast, in a study of cattle experimentally and acutely infected with BVDV, virus was isolated from the lung in high titer but BVDV antigen was not reported in lung tissue by IHC. 25 In this study, we focused entirely on the respiratory tract, and BVDV antigen was present within surface and ductal epithelium, glands, vessels, nerves, and leukocytes throughout the respiratory tract in most of the PI calves. In addition, chondrocytes and perichondrial cells were often infected. There were variations in intensity of the BVDV-IR among different animals and in some respects from different herds. For instance, BVDV-IR was not found in cartilage cells as commonly from cattle in herd B compared with those in herd A. This could represent a difference in viral tropism between BVDV isolates or might represent differences in maturity of cartilage. Tissues from herd B cattle were in formalin only 3 days before processing, whereas tissues from herd A were in fixative for 7 days. Therefore, loss of antigenicity because of prolonged exposure to formalin is most likely not a cause of these results.
During the development of bacterial pneumonias in beef cattle, the inciting bacteria are often part of the normal nasal flora, proliferate when the animal is stressed or viral infected, and are inhaled within the lungs where they colonize, proliferate and cause disease.
6,
19
Therefore, bacterial adhesion to mucosal surfaces plays a major role in colonization and disease production. Viral infection of epithelial cells in vitro has been associated with increased adherence of
BVDV also most likely enhances respiratory disease through immunosuppression. Potgieter et al.
32
demonstrated increased susceptibility of cattle experimentally acutely infected with to experimental challenge with
Houe 22 recently reviewed the epidemiology of BVDV infection and indicated that PI cattle are considered to be the most important source of BVDV infections. BVDV is secreted in nasal discharge, saliva, semen, urine, feces, tears, and milk, with direct and indirect contact probable when PI cattle are present in herds. In this study, we found mild to strong BVDV-IR in serous secretory cells and duct epithelium of the mixed tubuloalveolar glands of the nasal cavity. BVDV-IR was almost completely absent from mucus-secreting cells. Therefore, the serous secretions of the nasal cavity are likely a major source of infectious BVDV. In addition, productive viral infection of epithelium and the presence of infected leukocytes in respiratory secretions probably further enhance the infectivity of PI calves for other cattle.
Footnotes
Acknowledgements
This project was funded in part by a grant from the Noble Foundation, Ardmore, Oklahoma. The authors wish to thank Jeanenne Duffy, Lurinda Burge, and Marie Montelongo for technical support.
