Abstract
Phenobarbital (PB) therapy is frequently associated with elevated serum alanine aminotransferase (ALT) and alkaline phosphatase (AP) activities in dogs without clinical signs of liver disease. The goal of this study was to determine if increased serum ALT and AP activities in clinically healthy PB-treated epileptic dogs are due to hepatic enzyme induction or to subclinical liver injury. Liver biopsies were obtained from 12 PB-treated dogs without clinical signs of liver disease but with elevated serum ALT and/or AP activities or both. Liver biopsies were obtained from eight healthy control dogs not receiving PB. Biopsies were evaluated histopathologically (all dogs) and liver homogenates were assayed for ALT (all dogs) and AP (six treated dogs, all controls) activities. As a positive control, liver cytochrome P4502B, an enzyme known to be induced by PB, was measured by benzyloxyresorufin-O-dealkylase activity and immunoblotting (five treated dogs, all controls). Serum AP isoenzyme analyses were performed. Results showed that ALT and AP activities in liver homogenates were not increased in treated dogs compared with controls, whereas the positive control for induction, CYP2B, was dramatically increased in treated dogs. Histopathological examination of liver biopsies revealed more severe and frequent abnormalities in treated dogs compared to controls, but similar types of abnormalities were found in both groups. Serum AP isoenzyme analyses in treated dogs demonstrated increased corticosteroid-induced and liver isoenzyme activities compared to controls. Results do not support induction of ALT or AP in the liver as the cause of elevated serum activities of these enzymes due to PB.
Keywords
Phenobarbital (PB) is one of the most commonly used anticonvulsant drugs in dogs. 5, 40 Although PB is generally considered to be a fairly safe and effective drug, hepatotoxicity is an infrequent but potentially fatal adverse effect. 10 Dogs that develop clinical signs of liver disease associated with PB treatment usually have increased serum liver-related enzymes due to liver injury. 10 However, increased serum liver-associated enzymes are also commonly seen in PB-treated dogs with no clinical signs of liver disease. 7, 8, 16, 22, 33 This has raised the possibility that these enzymes might be induced by PB. Enzyme induction is the increased synthesis of the enzyme, resulting in increased amounts of the enzyme protein and its related activity. Phenobarbital has been shown to induce a number of hepatic enzymes, 4, 23, 29, 38, 39 including certain cytochrome P450s, glucuronosyl transferases, and glutathione-S-transferases. Whether or not liver enzymes commonly used as markers of liver injury in serum biochemical profiles can be induced by PB remains unclear. These enzymes include alanine aminotransferase (ALT) and alkaline phosphatase (AP) in particular, as well as others such as aspartate aminotransferase (AST), gamma-glutamyl transferase (GGT), and sorbital dehydrogenase (SDH).
The two serum liver-associated enzymes most commonly used as markers of hepatic disease in dogs are ALT and AP. ALT is relatively liver specific in dogs 14, 15 and is involved in intermediate metabolism and gluconeogenesis. In hepatocytes, ALT is primarily a cytosolic enzyme but is also found in small quantities in mitochondria. 18 Increased serum ALT activity is typically associated with hepatocellular membrane damage or distortion and leakage of the enzyme from the hepatocytes. Text books and journal articles often state that ALT is induced by PB. 1, 3, 5, 7, 12, 22, 33, 45, 54 However, the conclusion of induction of ALT has been based on observations that some PB-treated dogs have elevated serum ALT activities but are either clinically healthy or do not have histopathologic lesions in the liver. No studies have been done that evaluate ALT activities within liver tissue of PB-treated dogs to confirm induction.
AP in dogs is found in many organs, including liver, bone, intestine, and kidney. 2, 24, 26, 27, 44, 50, 51 Because of the very short half-lives of the intestinal and kidney isoenzymes, the only isoenzymes found in canine serum are the bone (B-AP) and liver (L-AP) isoenzymes and a unique isoenzyme called the corticosteroid-induced (C-AP) isoenzyme. 27, 51 The B-AP isoenzyme originates from osteoblasts, and increased activity of this isoenzyme in serum is associated with conditions involving increased osteoblastic activity. 13, 51 The L-AP and the C-AP isoenzymes are produced in the liver and are located on the hepatocyte plasma membranes lining the bile canaliculi and sinusoids. 41, 42, 46, 51 Increased activity of the L-AP isoenzyme in serum is typically associated with cholestasis. Increased serum C-AP activity is frequently associated with exogenous glucocorticoid treatment or endogenous corticosteroid excess such as occurs with hyperadrenocorticism. However, increases in this isoenzyme have also been shown to be associated with many chronic illnesses, 27, 50, 51 possibly as a result of stress and the resultant increase in endogenous glucocorticoid secretion. Serum total AP activity is commonly elevated in PB-treated dogs. The isoenzymes contributing to the increased total activity have been determined for only a small number of PB-treated dogs, 27, 49, 55 with no one specific isoenzyme consistently increased.
Several authors have suggested that AP is induced by PB in dogs. 3, 5, 7, 12, 22, 33, 45, 54 However, as discussed above for ALT, induction was inferred because either no histopathologic hepatic lesions were present in the dogs with elevated serum total AP or the dogs were clinically healthy at the time. Only two studies have investigated whether or not increased serum AP activity in PB-treated dogs is associated with increased amount and activity of AP in the liver. 30, 55 These two studies used small numbers of PB-treated dogs (n = three each) and PB doses nearly 10 times the typical maintenance dose of PB used to treat canine epilepsy. One of the studies 30 evaluated only the microsomal fraction of the tissue homogenates, thereby excluding evaluation of much of the plasma membrane where both the L-AP and C-AP isoenzymes are located. These studies found significantly increased AP activity (eight- to 12-fold increases) in liver tissue of treated dogs compared with controls, and the conclusion that PB induces AP became firmly ingrained in the literature.
Theoretically, induction of ALT and AP by PB would increase the amounts and activities of these enzymes in the liver and subsequently cause increased “spill-over” activities in the blood indistinguishable from that caused by hepatocellular damage and cholestasis. This poses a serious problem for veterinarians when presented with PB-treated dogs that do not have clinical signs of liver disease but have elevated serum liver-associated enzyme activities. At present, there is no noninvasive method to differentiate between elevations due to enzyme induction and those due to early liver injury. The dilemma faced by the veterinarian is deciding whether or not to discontinue the PB. If the serum liver enzymes are elevated because of induction, there would be no indication to alter the anticonvulsant regimen. However, if the elevations are due to early subclinical liver damage, discontinuation of PB and initiation of alternative anticonvulsant therapy would be warranted to prevent continued liver damage that might ultimately result in liver failure.
The purpose of this study was to determine if elevated serum ALT and AP activities in epileptic but otherwise clinically healthy PB-treated dogs are due to induction of these enzymes or due to subclinical liver damage. Specific aims were 1) to determine which isoenzymes of AP accounted for increased serum AP activities, 2) to examine liver samples histologically to look for evidence of hepatic injury, and 3) to measure ALT and AP activities in liver samples to determine if induction had occurred.
Materials and Methods
Study population
PB-treated dogs
Between September 1997 and July 1999, client-owned epileptic dogs were recruited for the study by veterinarians throughout the Atlantic provinces of Canada, as described previously. 19, 20 Our goal was to identify dogs receiving PB that had elevated serum AP and ALT activities. Dogs of various breeds, age, sex, and duration of PB treatment were eligible for enrollment in the study. Dogs had to be clinically healthy (other than having epilepsy) as determined by physical examination and history and could not be receiving any other major therapeutic drugs. Owners of all animals signed informed client consent documents for participation in the study. The study was approved by the University of Prince Edward Island Animal Care Committee in accordance with the Canadian Council of Animal Care.
Data collected for each dog included signalment, physical examination results, complete histories (including degree of seizure control, duration of treatment and PB dosage, exposure to other drugs or chemicals, and concurrent or previous medical conditions) and results of complete blood count (CBC) analyses, serum PB concentration determinations, and complete serum biochemical analyses (Hitachi 911, Boehringer Mannheim Corp., Indianapolis, IN) performed at the Atlantic Veterinary College Diagnostic Laboratory. Serum biochemical analyses included AP, ALT, GGT, AST, SDH, thyroxine, conjugated and free bilirubin, urea, total protein, albumin, cholesterol, and fasting bile acids.
Dogs with serum total AP and/or ALT activities greater than two times (or both) the upper limit of the normal Atlantic Veterinary College Diagnostic Laboratory reference range (greater than two times N) were eligible for additional diagnostic tests, including abdominal ultrasound examination, blood coagulation tests, pre- and postprandial bile acids tests, and liver biopsy. Of 128 PB-treated dogs enrolled in the study, 30 dogs (23%) met the criteria for additional diagnostics and liver biopsy. Of these 30 eligible dogs, liver biopsies were obtained from 15 dogs. Owners of the remaining 15 dogs declined to have biopsies performed. Three of the 15 dogs that had liver biopsies were excluded from analyses for the following reasons: subsequent history of exposure to another chemical in one dog; subsequent evidence of concurrent unrelated disease in one dog; and inadequate amount of liver tissue to allow enzyme activity determinations for one dog. Therefore, the final N for analysis was 12 PB-treated dogs.
The 12 PB-treated dogs consisted of 10 males (eight neutered) and two females (one spayed). Ages ranged from 1.5 to 8 years (median 4.5 years). Breeds represented included two Huskies, one each of Labrador Retriever, English Springer Spaniel, Collie, Poodle, Shetland Sheepdog, and Pomeranian breeds, and four mixed-breed dogs. Serum PB concentrations ranged from 42 to 192 μmol/liter (9.7 to 44.2 μg/ml), median 99 μmol/liter (22.8 μg/ml); and total duration of PB treatment ranged from 4 months to 6.5 years (median 1.7 years). Total daily dose of PB ranged from 2.1 to 12.9 mg/kg/day (median 5 mg/kg/day). Seizure control was classified as good for three dogs (no seizures in the previous three months), fair for three dogs (one to two seizure episodes in the previous three months), and poor for six dogs (one or more seizure episodes in the past month).
Seven of 12 PB-treated dogs (58%) had serum ALT activities greater than the upper limit of the normal range, and 3 of 12 (25%) had serum ALT activities greater than two times N. Eleven of 12 (92%) had serum AP activities greater than the upper limit of the normal range, and all 11 were greater than two times N. No dogs had increased AST, SDH, GGT, or conjugated bilirubin greater than two times N.
Control dogs
Liver biopsy samples were obtained from eight healthy mixed-breed dogs not receiving PB that were scheduled for euthanasia at a local humane society. Dogs were healthy on the basis of physical examination, normal biochemical profile and CBC results, and had no recent histories indicative of concurrent disease or medication. Three were males (all intact) and five were females (three spayed). Ages were unknown, but physical examinations suggested estimated ages ranging from 1 to 3 years (median 1.75 years).
Serum AP isoenzyme determination
Serum AP isoenzyme analysis for quantification of L-AP, C-AP, and B-AP isoenzymes were performed for 11 PB-treated dogs and eight control dogs. Inadequate amounts of serum were available for the remaining PB-treated dog to allow isoenzyme determination. Methods were those of Sanecki et al. and Hoffman et al. 25, 43 Cellulose acetate affinity electrophoresis as described by Kidney and Jackson 27 was performed on all samples to qualitatively evaluate serum AP isoenzymes.
Liver biopsies
Multiple liver samples were obtained through ultrasound-guided percutaneous needle biopsy from each of 11 PB-treated dogs, using a Biopty (C.R. Bard Inc., Covington, GA) spring-driven biopsy instrument with an 18-gauge needle. A wedge liver biopsy sample was obtained during exploratory laparotomy in the 12th PB-treated dog. Liver biopsies were obtained from the eight Humane Society control dogs under anesthesia immediately before euthanasia, using the Biopty instrument with a 14-gauge needle. One portion of each biopsy was fixed in 10% buffered formalin for histopathology, and another portion was flash frozen in liquid nitrogen and stored at −80 C.
Histopathology
Sections from formalin-fixed liver samples were stained for light microscopy by the Atlantic Veterinary College Diagnostic Laboratory using standard techniques. Sections were stained with hematoxylin and eosin, rhodanine (copper), prussian blue (iron), periodic acid–Schiff (glycogen), and reticulum stain (fibrosis). Histopathology slides were randomized and scored in a blinded fashion by a single pathologist (LMM). Histopathologic categories scored included those indicative of induction (hepatocyte size and cytoplasmic characteristics such as granularity representative of endoplasmic reticulum induction) and those indicative of injury (necrosis, inflammation, fibrosis, vascular characteristics, and an overall injury score based on the pathologist's impression). Degree of staining for copper, iron, and glycogen was scored. The scoring system used in all categories consisted of a scale of 1 through 4 (1 = no abnormalities, 2 = mildly abnormal, 3 = moderately abnormal, and 4 = markedly abnormal).
Liver homogenate enzyme assays
Frozen liver samples were thawed and manually homogenized in nine volumes of Tris–KCl buffer (0.05 M Tris base, 150 mM KCl; pH 7.4) using disposable pestle-and-microfuge homogenization sets (Kontes Glass Company, Vineland, NJ). Protein contents of homogenates were determined using the Biorad DC protein assay (Biorad Laboratories, Hercules, CA). Linear ranges of all assays were determined and quantities of liver homogenate were chosen to provide results within the linear ranges.
ALT activity
ALT activity in each liver homogenate was determined using a quantitative, colorimetric end-point assay (Procedure #104, Sigma Diagnostics, St. Louis, MO) that used α-ketoglutaric acid as the substrate and that detected production of pyruvic acid. Results are expressed as units per milligram protein.
AP activity
Total AP activities in liver homogenates were determined using a quantitative, colorimetric end-point assay kit (Procedure #505, Sigma Diagnostics) that used para-nitrophenyl phosphate as the substrate, 2-amino 2-methyl-1-propanol buffer and detected the end product para-nitrophenol. Results are reported in units per milligram protein.
Benzyloxyresorufin-O-dealkylase activity
Benzyloxyreso-rufin-O-dealkylase (BROD) activity, a specific assay for the major PB-induced cytochrome P450 in dogs (CYP2B11), 28 was measured in liver homogenate as a positive control for verification of induction of an enzyme known to be induced by PB. The assay was performed as a modification of previously published techniques. 6, 28, 31, 32, 34, 56 Briefly, a reaction mixture containing liver homogenate (75–100 μg protein), 5 μM benzyloxyresorufin substrate, physiologic-buffered saline, and 0.5 μM dicumarol (to minimize reduced nicotin-amide adenine dinucleotide phosphate [NADPH]–mediated reduction of resorufin 32, 35, 36, 56 ) was equilibrated at 37 C. The reaction was initiated by adding 0.25 mM NADPH and the rate of production of the resorufin end product was measuring fluorometrically. Reaction rate (nmol/minute/mg protein) was determined by comparison with a resorufin standard.
Immunoblotting of CYP2B protein
Amounts of CYP2B protein in liver homogenates were determined using standard western blotting techniques. 18, 53 Briefly, 100 μg of crude homogenate protein per sample was separated on a 10% sodium dodecyl sulfate-polyacrylamide gel. The proteins were transferred to a nitrocellulose membrane by a semidry transfer process, and immunodetection of transferred CYP2B proteins was performed using goat anti-rat CYP2B antibody (Daiichi Pure Chemicals Co. Ltd., Tokyo, Japan) and rabbit anti-goat immunoglobulin G peroxidase conjugate secondary antibody (Sigma Chemical Company, St. Louis, Mo). Bands were observed by enhanced chemiluminescence (ECL, Amersham Biosciences, Baie d'Urfe, Quebec, Canada) and exposure to radiographic film.
Statistical analyses
Because of small sample sizes and nonnormal data, non-parametric methods were used for all statistical analyses (Mann-Whitney test for comparisons, Spearman's correlation coefficient (r) test for correlations) (SPSS Inc. Software, Chicago, IL; GraphPad Software Inc., Prism2, San Diego, CA). One-tailed analyses were used in comparisons of serum ALT and serum total AP activities between PB-treated dogs and controls because increased serum ALT and AP activities were selected for and hence expected to be higher in treated dogs than controls. Two-tailed analyses were used for all other comparisons.
For serum ALT and AP activities, histopathologic scores, and liver homogenate ALT comparisons, 12 PB-treated dogs were compared with eight controls. For liver homogenate AP activity comparisons, six PB-treated dogs with adequate amounts of liver homogenate to perform the assay were compared with eight control dogs. For CYP2B comparisons, five PB-treated dogs with adequate amounts of liver homogenate were compared with eight controls. For serum AP isoenzyme activity comparisons, 11 PB-treated dogs were compared with eight controls. Significance was defined as P ≤ 0.05 throughout.
Results
Serum ALT and AP activities
Median serum ALT activity in 12 PB-treated dogs was significantly (P < 0.05) higher than controls (92.5 and 43.5 U/liter, respectively), and median serum total AP activity was significantly (P < 0.001) higher in treated dogs compared with controls (378.5 and 80.5 U/liter, respectively) (Fig. 1). There were no significant correlations between serum AP and ALT activities or between either serum AP or ALT activities and age, duration of therapy, total daily dose of PB, or serum PB concentration in PB-treated dogs.
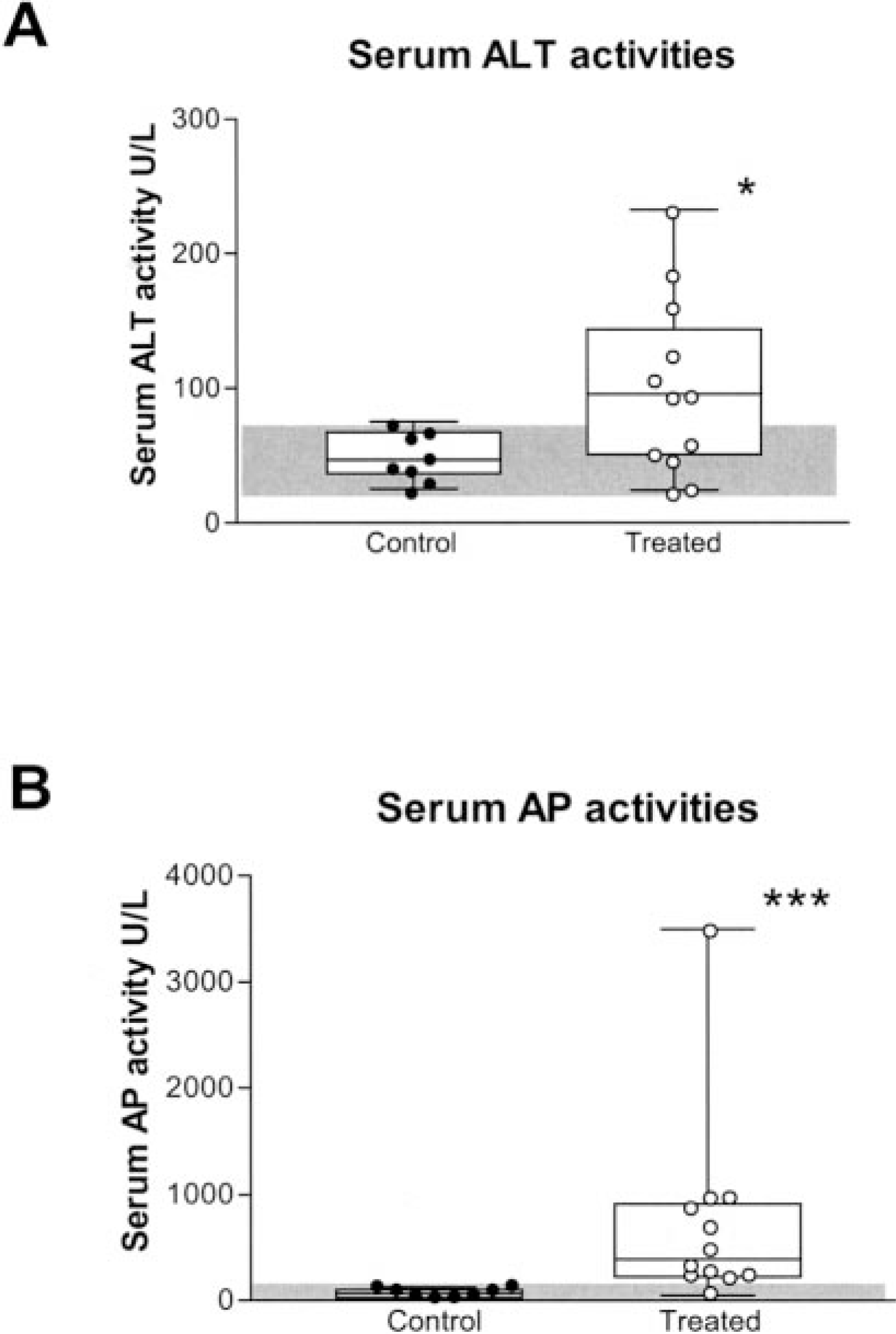
Serum AP isoenzyme analyses
Serum isoenzyme analyses were performed for 11 of 12 PB-treated dogs (Fig. 2). The C-AP isoenzyme was the predominant isoenzyme in six of the 11 dogs. Five of these six dogs had serum total AP activities greater than 500 U/liter (reference range: 23–87 U/liter). L-AP was the predominant isoenzyme for the remaining five of 11 dogs, all of whom had serum total AP activities less than 500 U/liter. Serum total AP activity was significantly correlated with C-AP activity (P < 0.001, r = 1.0).
Serum AP isoenzyme activities in 11 PB-treated epileptic dogs (ages ranging from 1.5 to 8 years) from whom liver biopsies were obtained.
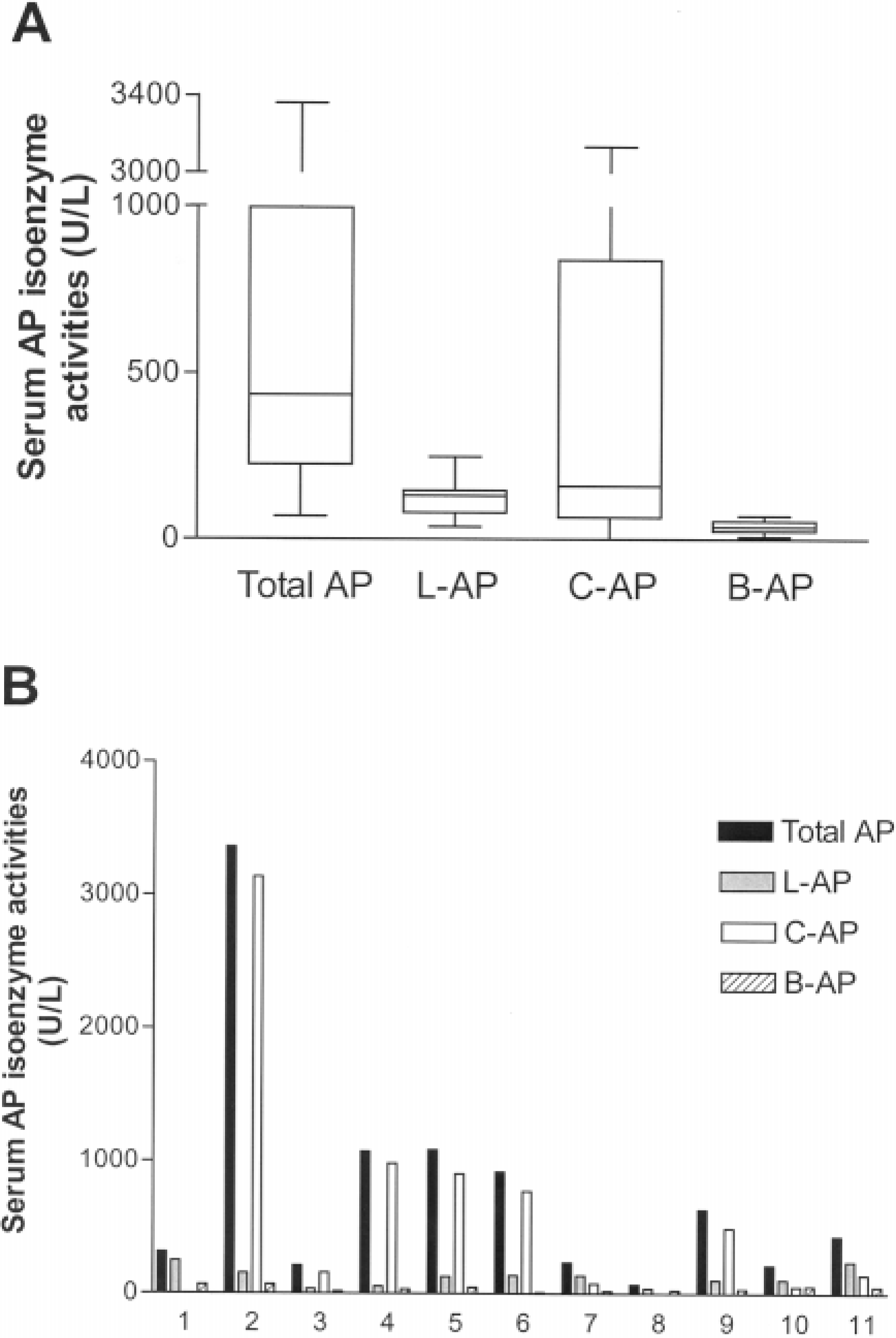
Serum AP isoenzyme analyses were performed for all eight control dogs (Fig. 3). L-AP and B-AP were the predominant isoenzymes. C-AP activity was present in serum from only one control dog. B-AP was significantly negatively correlated with age (P < 0.05, r = −0.83) and positively correlated with serum total AP (P < 0.01, r = 0.85).
Serum AP isoenzyme activities in eight healthy control dogs not receiving PB from whom liver biopsies were obtained.
Median serum C-AP and L-AP activities were significantly (P < 0.01) higher in PB-treated dogs compared with controls. B-AP activity did not differ significantly between the two groups.
Cellulose acetate affinity electrophoresis produced bands only in the previously identified locations for LAP, C-AP, and B-AP. No unusual or previously unidentified bands were observed for any PB-treated or control dogs.
Histopathology
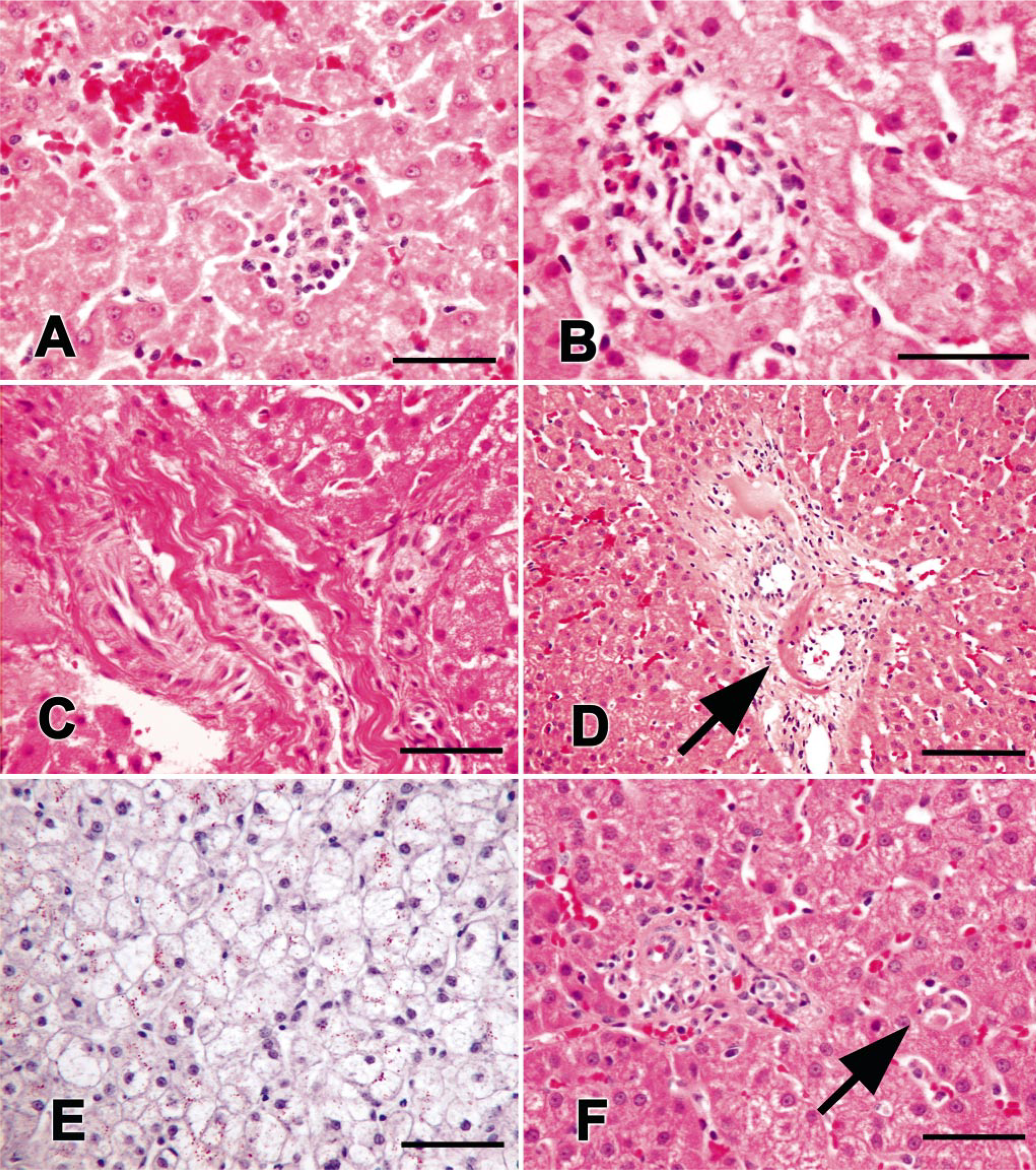
The 12 PB-treated dogs had enlarged, swollen hepatocytes with finely granular cytoplasmic appearance indicative of increased amounts of endoplasmic reticulum (Fig. 4A). Inflammatory cell infiltration, single cell and piecemeal necrosis, fibrosis, and vascular abnormalities resembling veno-occlusive changes were found in many treated dogs (10, 9, 8, and 7 dogs, respectively; Fig. 4B–D). Sections from 4 of 12 PB-treated dogs stained positively for copper (Fig. 4E) and nine of 12 stained positively for iron, which was found predominantly in macrophages and Kupffer cells but was also occasionally present within hepatocytes (Fig. 4F). No vacuoles were noted in hepatocytes, and there was no evidence of biliary stasis or biliary hyperplasia in any sample. A number of PB-treated dogs had scores ≥ 3 in the following categories: hepatocyte size (six dogs); cytoplasmic changes (three dogs); vascular changes (three dogs); inflammation, copper staining, iron staining, and overall injury score (two dogs each); and fibrosis (one dog).
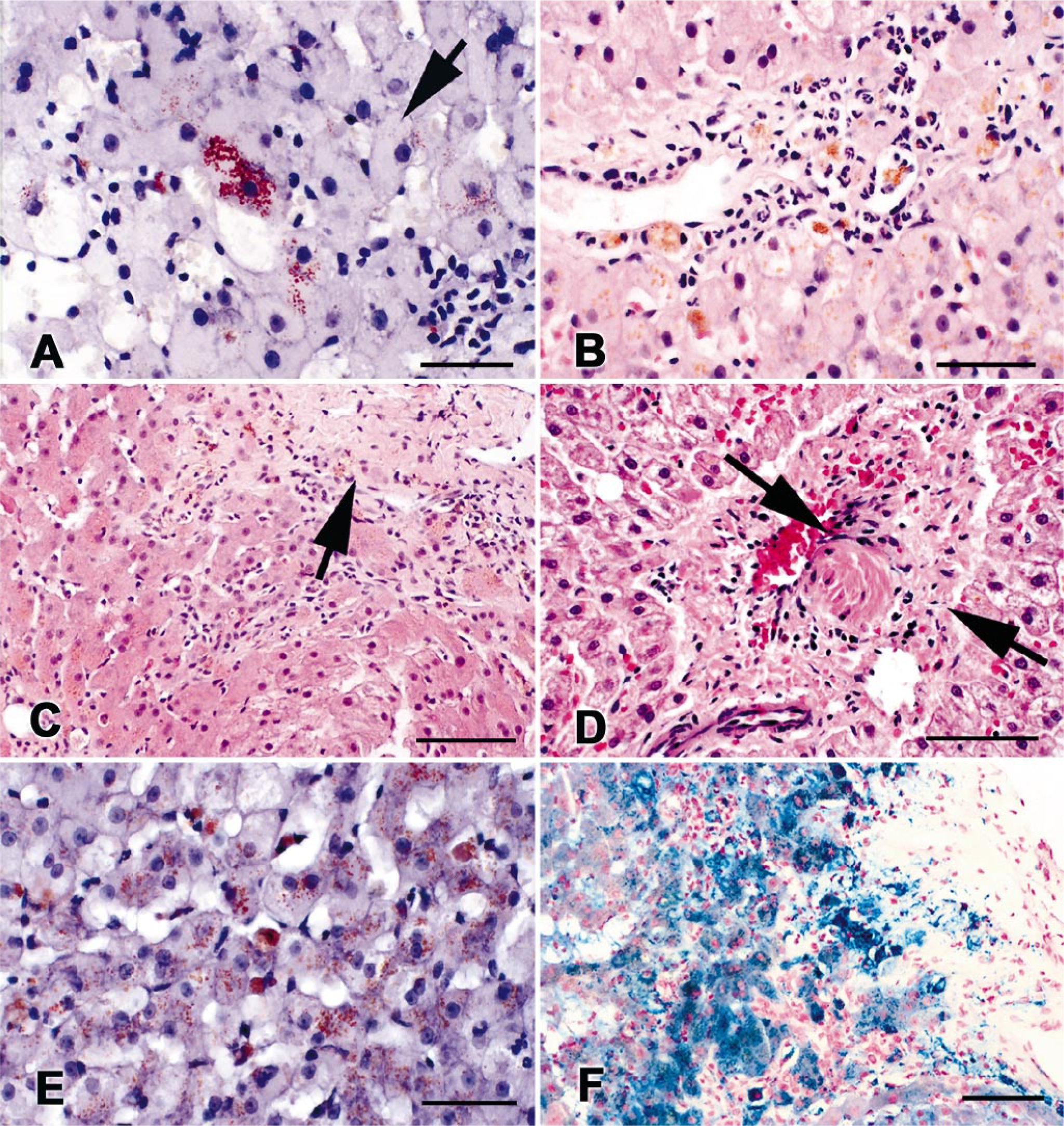
Photomicrographs of liver biopsies from PB-treated epileptic dogs.
Control biopsy samples had lesions similar to the PB-treated dogs, but with less frequency and to a lesser degree (i.e., lower scores) than treated dogs (Fig. 5). Median histopathologic scores and ranges of scores for treated and control dogs are shown in Table 1. Inflammatory cell infiltration, piecemeal necrosis, fibrosis, and vascular changes were found in three, four, two, and four of the control dogs, respectively. Sections from five of eight control dogs stained positively for copper and four of eight stained positively for iron. No control dog samples had scores ≥ 3 in any category except copper staining where three of eight had scores ≥ 3.
Photomicrographs of liver biopsies from healthy control dogs not receiving PB. Control dogs had hepatic lesions similar to PB-treated dogs, but with less frequency and to a lesser degree (i.e., lower scores) than treated dogs.
Histopathologic scores for liver biopsy sections from 12 phenobarbital-treated epileptic dogs and eight untreated control dogs.∗
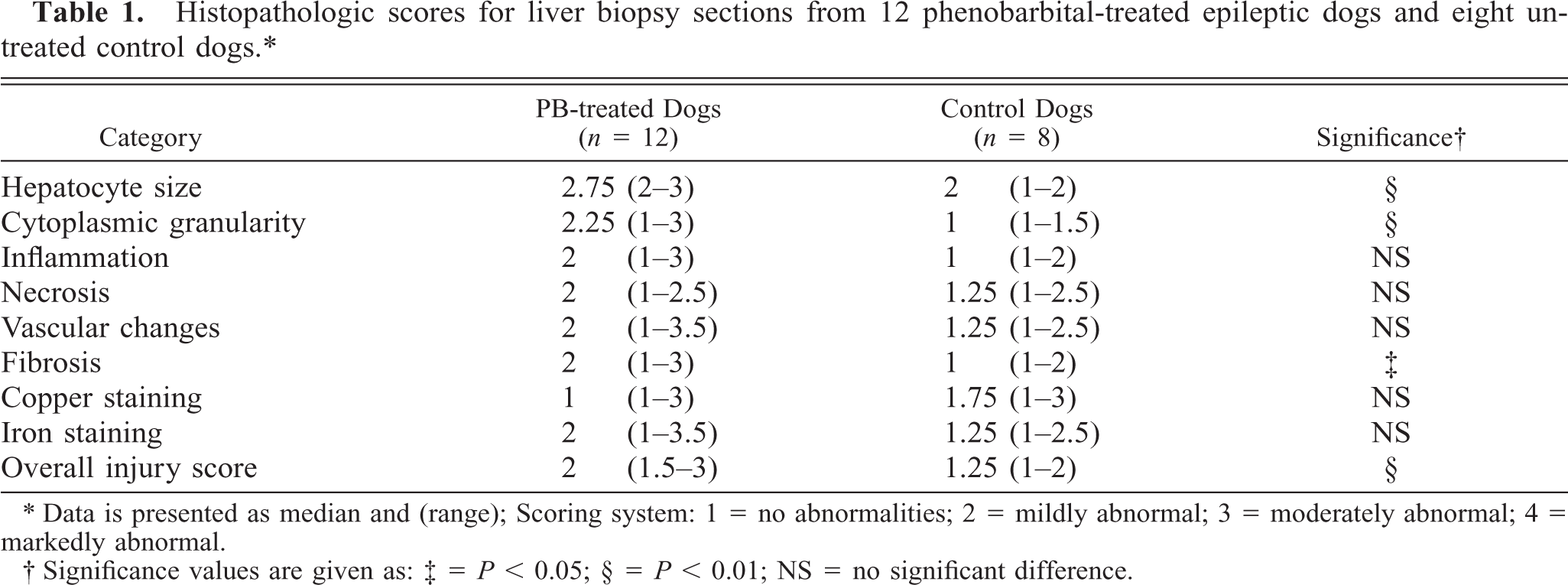
∗ Data is presented as median and (range); Scoring system: 1 = no abnormalities; 2 = mildly abnormal; 3 = moderately abnormal; 4 = markedly abnormal.
† Significance values are given as: ‡ = P < 0.05; § = P < 0.01; NS = no significant difference.
In PB-treated dogs, no associations were found between scores of any histopathologic categories and dose of PB, serum PB concentration, serum ALT or AP activities, or liver homogenate ALT or AP activities. However, duration of treatment was significantly associated with overall histopathologic score (Fig. 6) and with degree of iron staining (P < 0.01, r = 0.79). Age was also highly correlated with duration of therapy (P < 0.01, r = 0.83), so colinearity of these variables existed. Pre- and postprandial bile acids test results were not correlated with severity of histopathologic scores or with degree of elevation of serum AP or ALT activity in PB-treated dogs. There were no correlations between overall histopathologic score and serum or liver homogenate ALT or AP activities or age in controls.
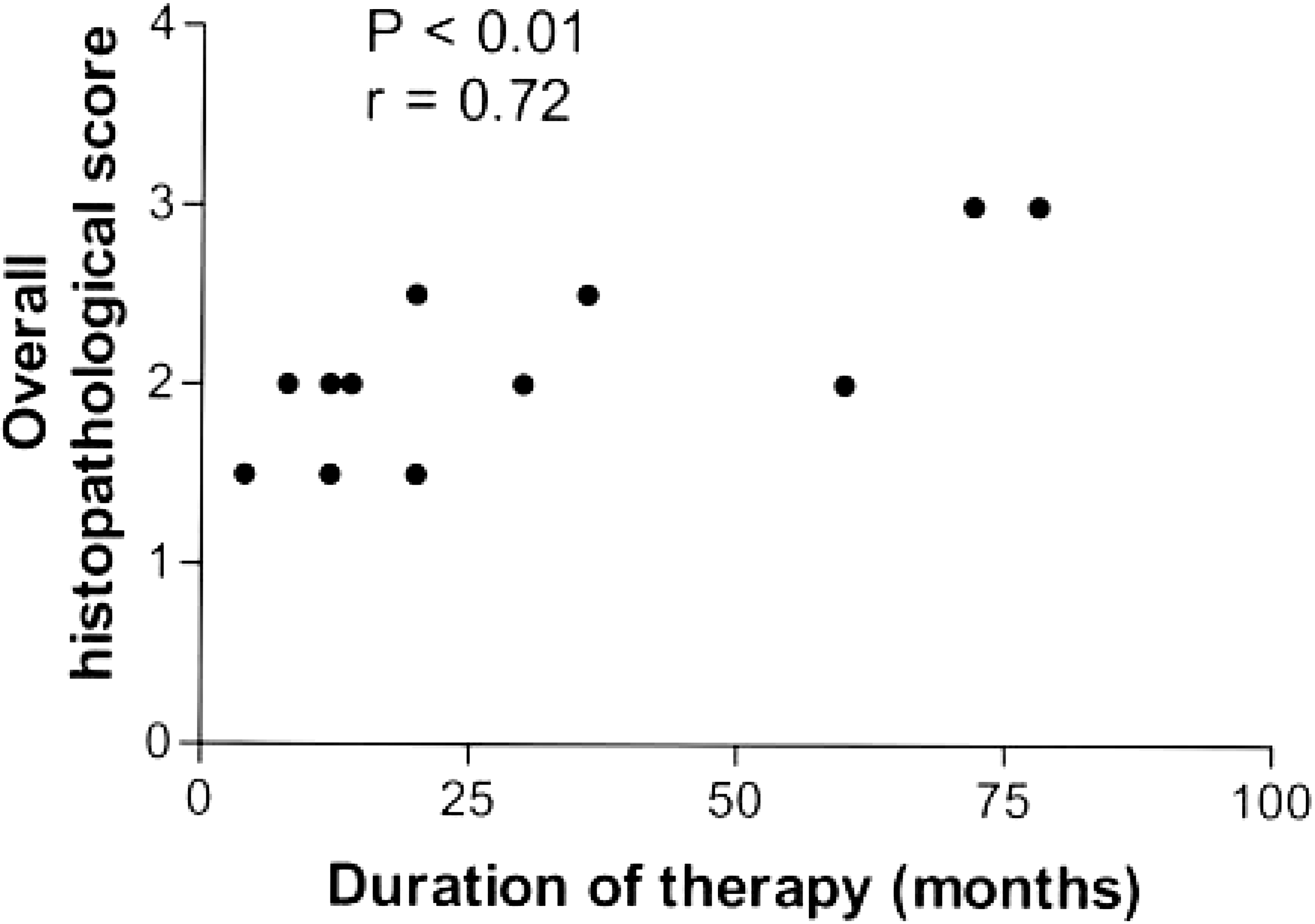
Scatterplot showing a significant association between duration of therapy and overall histopathologic score of liver biopsies from 12 phenobarbital-treated epileptic dogs.
Liver homogenate enzyme assays
ALT activities
Median ALT activity in liver homogenates from 12 PB-treated dogs was significantly lower than controls (0.069 and 0.115 U/mg protein, respectively) (Fig. 7). When only the seven PB-treated dogs with serum ALT activities above the upper limit of the reference range were compared with controls, median liver homogenate ALT activity was still significantly lower in treated dogs. Liver homogenate ALT activities in PB-treated dogs were significantly correlated with age and with duration of therapy (Fig. 8). Age and duration were also highly correlated, so colinearity existed between duration of therapy, age, and liver homogenate ALT activity. No significant correlations were found between liver homogenate ALT activities and serum PB concentration, total daily dose of PB, or serum ALT activity. In the control dogs, no significant correlations were found between liver homogenate ALT and serum ALT activities or age.
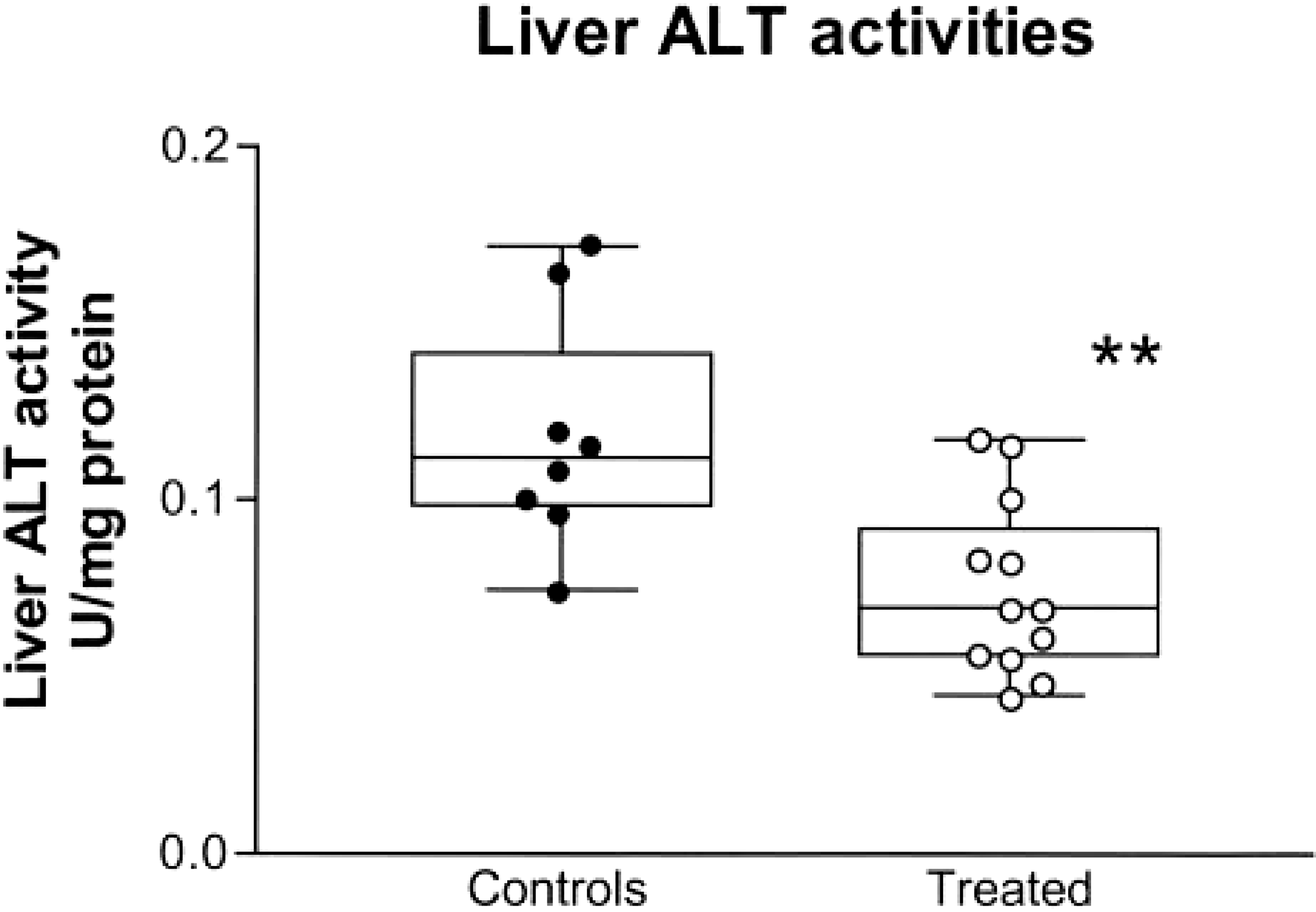
ALT activities in liver homogenates from 12 phenobarbital-treated (treated) epileptic dogs compared with eight healthy control dogs. Box extends from the 25th percentile to the 75th percentile, with a horizontal line at the median. Whiskers show the range of the data, extending from highest to lowest data points. Data points represent values for specific dogs. ∗∗Median value differs significantly (P < 0.01) from controls.
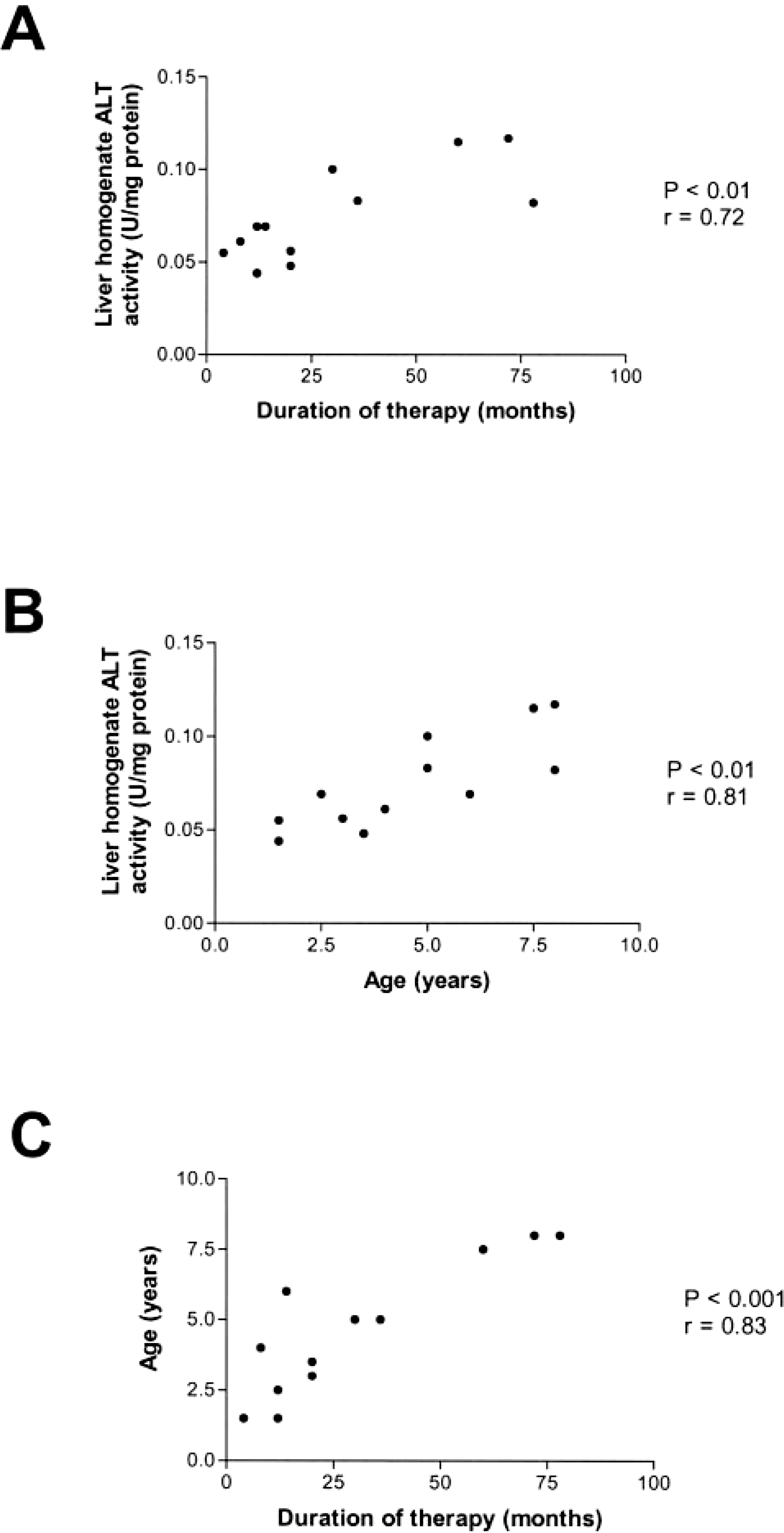
Scatterplots showing significant correlations between liver homogenate ALT activities in 12 phenobarbital-treated epileptic dogs and
AP activities
Six PB-treated dogs had adequate amounts of liver homogenate remaining to perform the AP assay. All had serum total AP activities greater than two times N. Median total AP activity in liver homogenates from PB-treated dogs was not significantly different than controls (10.9 and 14.7 U/mg protein, respectively) (Fig. 9). In the PB-treated dogs, significant correlations were found between liver homogenate AP activities and serum total AP and C-AP activities (Fig. 10). In the control dogs, liver homogenate AP activities were not correlated with serum total AP activity, C-AP, L-AP, B-AP, or age.
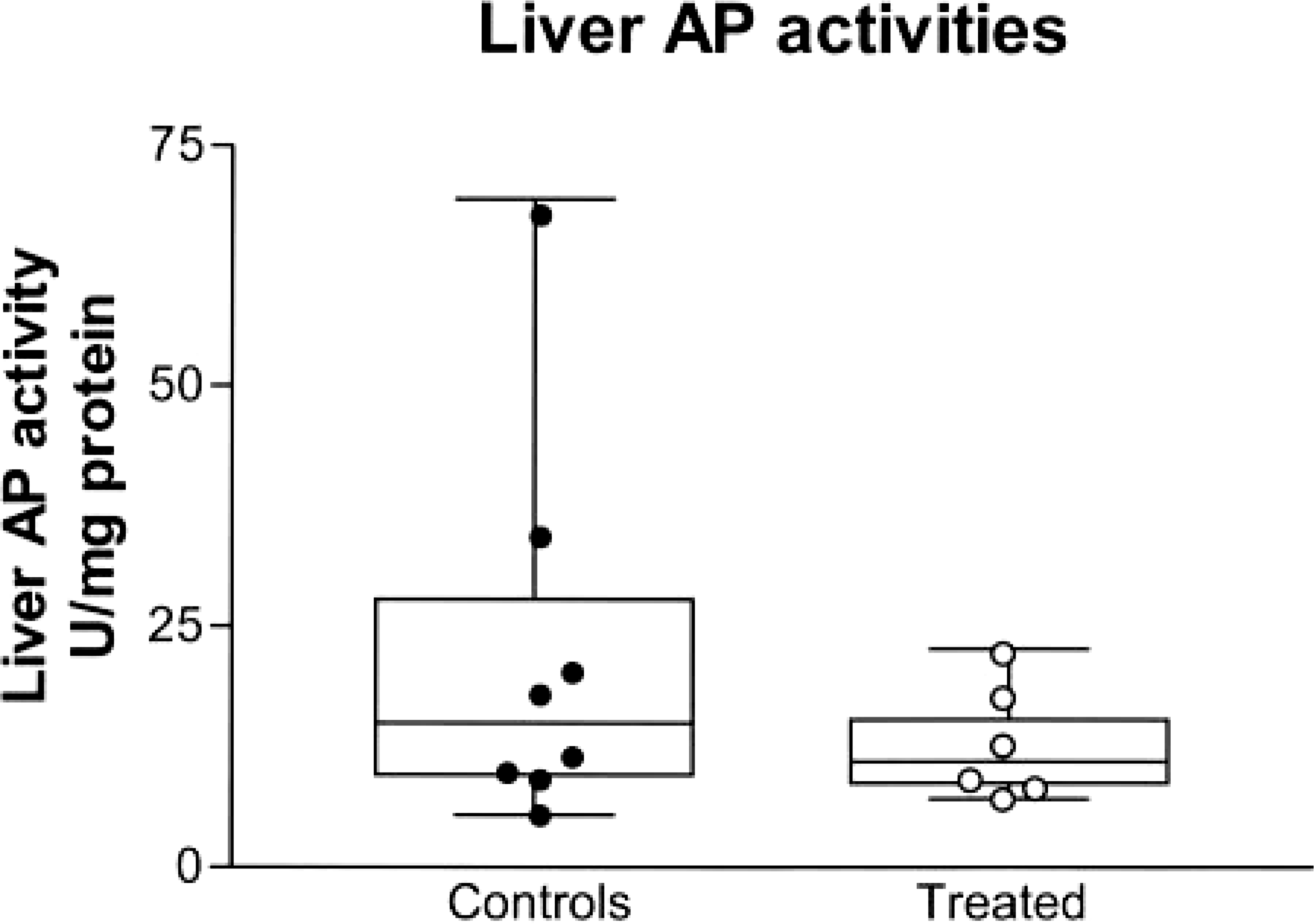
Total AP activities in liver homogenates from 6 phenobarbital-treated (treated) epileptic dogs compared with 8 healthy control dogs. Box extends from the 25th percentile to the 75th percentile, with a horizontal line at the median. Whiskers show the range of the data, extending from highest to lowest data points. Data points represent values for specific dogs.
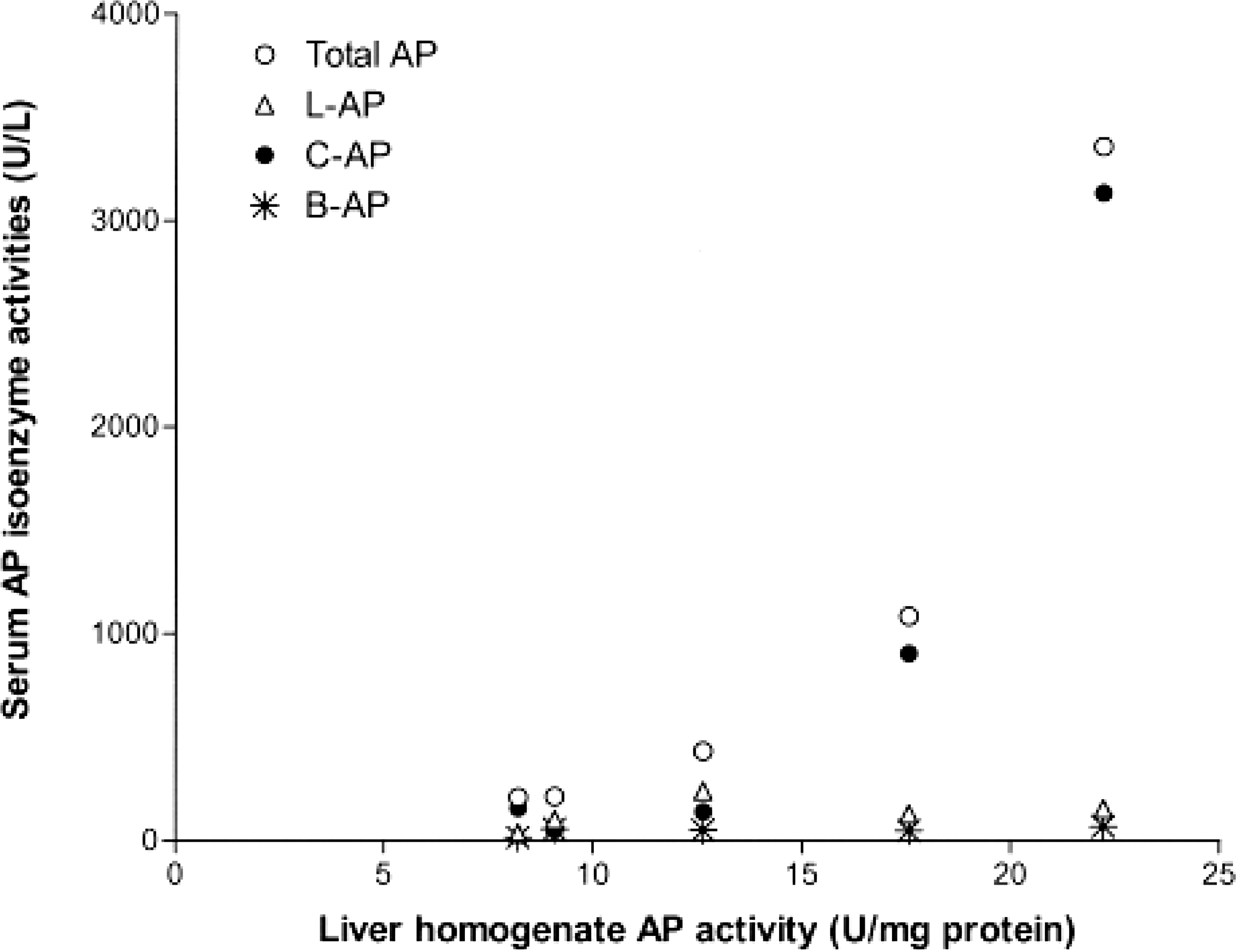
Scatterplot of liver homogenate AP activities versus serum AP isoenzyme activities from 5 of 6 phenobarbital-treated epileptic dogs. Liver homogenate AP activity was significantly correlated with serum total AP activity (P < 0.05, r = 1.0) and with serum C-AP activity (P < 0.001, r = 1.0) but not with serum L-AP or B-AP activities. (Isoenzyme determinations were not available for the sixth dog).
BROD activity and CYP2B immunoblotting
Metabolism of benzyloxyresorufin to resorufin (representative of CYP2B activity, a positive control for induction) was significantly greater in five PB-treated dogs than controls (median rates of 0.22 and 0.02 nmol/minute/mg protein, respectively) (Fig. 11A). Immunoblotting confirmed CYP2B protein induction (Fig. 11B).
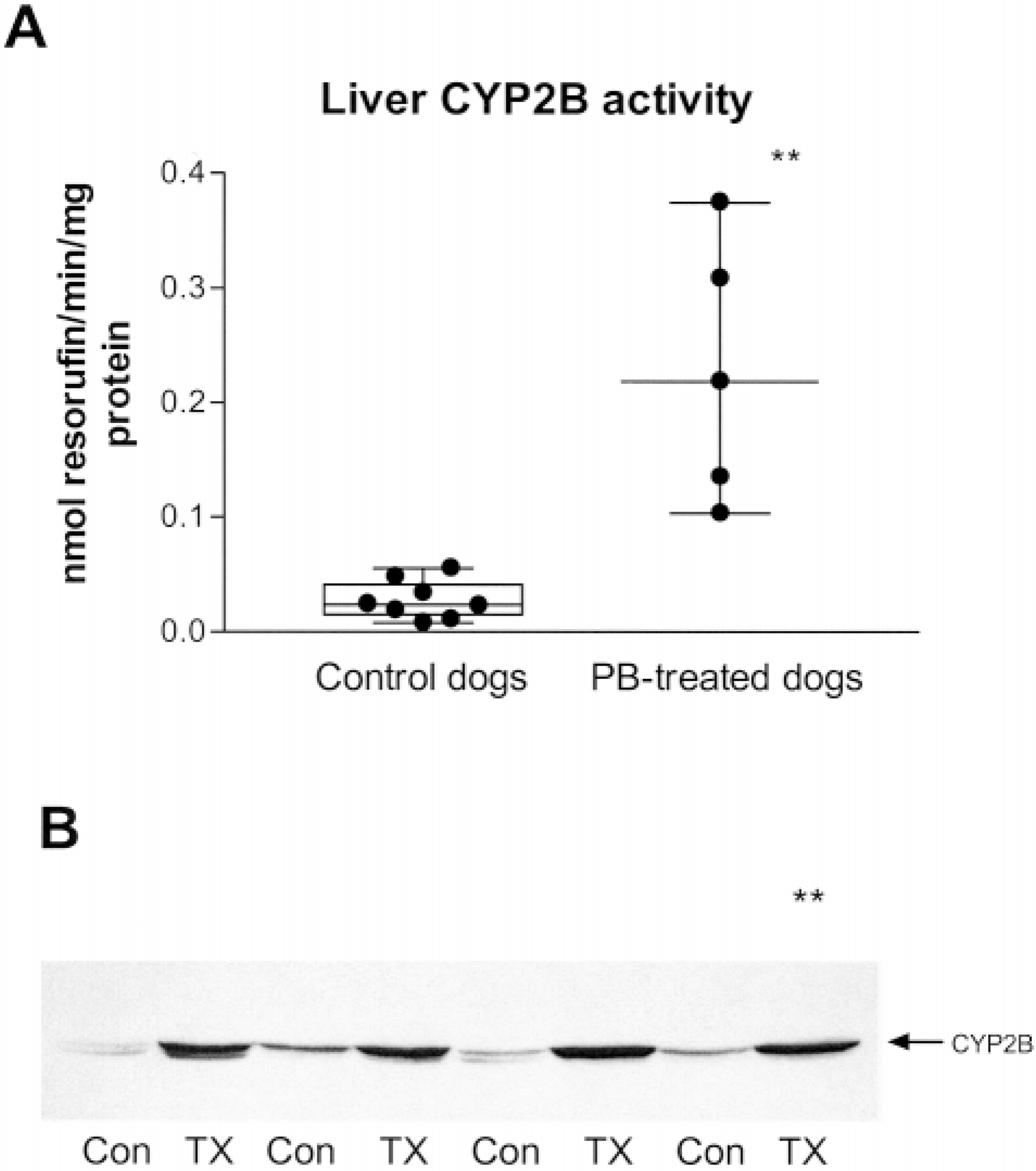
Induction of activities and protein quantities of CYP2B, a liver enzyme known to be induced by phenobarbital
Discussion
The primary goal of this study was to determine if increased serum ALT and AP activities in clinically healthy appearing PB-treated epileptic dogs could be attributed to induction of these enzyme activities in the liver. If induction of activities occurred, one would expect 1) increased liver homogenate ALT and AP enzyme activities in PB-treated dogs compared with controls and 2) greater histopathologic evidence of induction (i.e., enlarged hepatocytes and changes in cytoplasmic characteristics) but not of liver injury in PB-treated dogs. In this study, liver homogenate ALT and AP activities were not increased in PB-treated dogs compared with control, and histopathology revealed greater evidence of liver injury in PB-treated dogs.
Median liver homogenate ALT activity in PB-treated dogs was significantly lower than controls and thus does not support induction of hepatic ALT activity by PB. Treated dogs had a significantly greater degree of fibrosis than did the controls. This may have resulted in fewer functional hepatocytes per gram of liver tissue and therefore in fewer hepatocytes containing ALT. The possibility that induction of ALT did occur but was masked by an increased release of ALT from hepatocytes as a result of damage or other mechanisms cannot be ruled out. However, in PB-treated dogs, there were no correlations between serum ALT activity and liver homogenate ALT activity, dose of PB, duration of therapy, or serum PB concentration, and no correlations between liver homogenate ALT activity and serum PB concentration or dose of PB. The lack of correlations suggests that liver ALT was not induced in these animals. There was a significant correlation between liver homogenate ALT activity and duration of therapy, but liver homogenate ALT activity was also significantly correlated with age, and age was highly correlated with duration of therapy. Because of the colinearity of these three variables, interpretation is difficult.
Median liver homogenate total AP activity was also not increased in PB-treated dogs compared with controls. The lack of increased liver homogenate total AP activity in PB-treated dogs in this study differs from findings of the two previous studies investigating liver AP activity in PB-treated dogs 30, 55 where 8- to 12-fold increases in liver AP activities were observed. There are several possible reasons why our results differ. The previous studies used much higher doses of PB than are used therapeutically in dogs, with doses of 40–50 mg/kg/day as compared with a median dose of 5 mg/kg/day in our study. Smaller numbers of dogs (n = 3) in the previous studies also decreased the certainty of the conclusions. One study 30 evaluated only the microsomal fraction of liver homogenates for AP activity whereas we evaluated whole liver homogenates. This is potentially an important difference. The AP isoenzymes originating from liver tissue (L-AP and C-AP) are found predominantly on external hepatocyte plasma membranes, primarily in sinusoidal and canalicular regions. 41, 42, 46, 51 Therefore, the majority of AP activity would be in the cell fraction containing the hepatocyte cell membranes (i.e., the pellet from the first 9,000 × g spin) and not the microsomal fraction. Only a very small portion of the total hepatocyte AP would be found in the microsomal fraction. Enhanced synthesis of the AP enzyme would be expected to result in increased amounts of the enzyme in endoplasmic reticulum and golgi, both of which sediment in the microsomal fraction after differential centrifugation. This increased AP enzyme concentration in endoplasmic reticulum and golgi could result in a very large increase of AP enzyme concentration in the microsomal fraction, but the increase in overall AP activity in the entire hepatocyte might still be immeasurably small.
Results of recent studies 11, 46–48 investigating mechanisms of increased serum AP enzyme activities raise the possibility that induction of AP might occur but be masked by a simultaneous increase in release of the enzyme from hepatocyte cell membranes. These studies suggest that increased serum L-AP and C-AP activities are caused by increased cleavage and release of the enzymes from hepatocyte membranes through bile acid–enhanced activity of glycosylphosphatidylinositol phospholipase D (GPI-PLD), an enzyme found endogenously in plasma and on cell membranes including hepatocyte cell membranes. Because L-AP and C-AP isoenzymes accounted for the majority of the elevated total serum AP activity in PB-treated dogs in our study, it is possible the elevation was caused by an increased release of these isoenzymes from the hepatocyte membrane, perhaps due to either cholestasis or induction of GPI-PLD by PB. Although there was no overt biliary stasis in PB-treated dogs, there was significant enlargement and swelling of hepatocytes, which might contribute to intrahepatic cholestasis. Therefore, AP might have been induced by PB but a simultaneous increased rate of release of AP from plasma membranes could cause the total AP enzyme amount in liver tissue to remain unchanged or even decrease. The previous study 30 showing increased AP activity in the microsomal fraction from PB-treated dogs supports the hypothesis that induction of liver AP enzymes by PB occurs. The AP in the microsomal fraction would not yet have been subjected to GPI-PLD, so AP concentration in microsomes might be elevated compared with whole homogenate containing hepatocyte plasma membrane and GPI-PLD. Liver samples from our study were too small to allow preparation of microsomal fractions to test this hypothesis.
As additional support that induction of AP enzymes by PB might occur, a prospective study of PB-treated dogs showed that all three AP isoenzyme activities (CAP, L-AP, and B-AP) increased significantly in serum with time. 21 In that study, serum AP isoenzyme activities were determined for 23 epileptic dogs before the start of PB and at three weeks, six months, and 12 months after the start of PB. Median serum C-AP, LAP, and B-AP were all significantly increased at six months and 12 months compared with time 0. Median serum L-AP was also significantly increased at three weeks as well. The fact that all three isoenzyme activities significantly increased with time suggests either induction of the various isoenzymes in several tissues or induction of enzymes responsible for release of AP isoenzymes from cellular membranes. If increased serum total AP activities had been solely due to early or subclinical liver changes, we would not have expected B-AP to increase significantly with time. In our study, there was no significant difference in B-AP activity in the PB-treated dogs compared with controls. This is possibly because of the young age of the controls and the high B-AP activities due to bone growth in this young group. If the control dogs had been age-matched to the treated dogs, it is possible that a significant difference in B-AP activity between the two groups would have been found.
We did not find any unusual or previously unidentified AP bands with cellulose acetate affinity electrophoresis in PB-treated liver biopsy dogs. This suggests that PB does not induce a novel isoform of AP. This cannot be ruled out completely because a novel band might have been hidden within one of the known isoform bands. However, because affinity electrophoresis causes much more distinctive separation of isoform bands than traditional electrophoresis, the possibility is diminished.
Serum total AP activity was highly correlated with liver homogenate AP activity and with serum C-AP but not with serum L-AP in PB-treated dogs. This might suggest that C-AP is induced but that L-AP is not. If such were the case, measuring serum C-AP and L-AP activities might help differentiate between increased serum AP activity due to induction (increased C-AP) or due to hepatocellular injury (increased LAP). However, in this study, there were no correlations between serum C-AP or L-AP activities and histopathologic scores indicative of liver injury. That is, PB-treated dogs with the highest histopathology scores did not have the highest serum L-AP activities, and dogs with the least amount of liver abnormalities often had higher serum L-AP activity than C-AP activity. Therefore, measuring serum AP isoenzymes does not appear to be helpful at differentiating between induction and early liver injury.
Histopathologically, changes indicative of liver injury were more pronounced and found more frequently in treated dogs compared with controls. In particular, scores for fibrosis and overall injury were significantly higher in PB-treated dogs compared with controls. Varying degrees of inflammation, necrosis, and vascular changes were noted in both groups, but more treated dogs showed these changes and although not significantly different, median scores were higher in PB-treated dogs than controls. During the course of the study, postmortem liver samples were also evaluated from four additional PB-treated dogs not enrolled in the study that died for reasons unrelated to liver disease (unpublished data; Gaskill). Before death, all had moderately elevated serum AP activities, and two of four had moderately elevated serum ALT activities. When scores from these four postmortem liver samples are included in statistical analyses, PB-treated dogs had significantly higher scores for inflammation as well as for overall injury and fibrosis. Other statistical results remained the same. There were no unique histopathologic changes in PB-treated dogs other than the changes typically associated with increased amounts of endoplasmic reticulum (i.e., swollen hepatocytes and ground-glass appearance to the cytoplasm).
More control dog samples stained positive for copper than did treated dog samples. Liver copper accumulation has typically been thought to be either a consequence of or a cause of liver injury. 9, 17, 37, 52 However, dogs have a very wide and poorly defined range for normal hepatic copper concentrations. Studies of clinically normal dogs have found liver copper concentrations ranging from less than 100 up to 6,800 ppm dry weight and no histologic evidence of hepatitis in dogs with up to 2,000 ppm copper in the liver. 52 Rhodanine, the copper stain we used, provides a qualitative measure of copper and does not detect copper until tissue concentrations are approximately 400 ppm. 17 One investigator has proposed that high liver copper concentrations found in many normal dogs are because of a overabundance of copper in the typical canine diet. 52
We found a significant association between age and several histopathologic liver abnormalities in PB-treated dogs in this study. Median age of PB-treated dogs was significantly higher than controls (4.5 and 1.75 years, respectively; control ages estimated). Age matching of controls to PB-treated dogs was not possible in this study because of the clinical nature of the study and the lack of older dogs euthanized by the humane society.
Additional studies would help complete the investigation of PB induction of ALT and AP in dogs. Evaluation of ALT and AP messenger RNA as well as ALT and AP protein concentrations in liver homogenates of PB-treated and untreated dogs would determine if increased transcription and increased synthesis of these enzymes occur. Immunohistochemistry of liver sections to determine location and relative amounts of CAP and L-AP and determination of GPI-PLD quantity and activity would test the hypothesis of increased release of AP from hepatocyte cell membranes. Because of the clinical nature of this study and the small sizes of the needle biopsies, insufficient sample materials remain to pursue further studies with these samples.
The results of this study do not support the hypothesis that increased serum AP and ALT activities in PB-treated dogs are the result of induction of these enzyme activities in the liver. However, induction of AP activity in the liver might have been masked by a simultaneous increase in release of AP from hepatocyte membranes. Serum AP isoenzyme analysis does not appear to be helpful in differentiating hepatic enzyme induction from early hepatic injury. Although histopathologic evidence of hepatic injury was greater in PB-treated dogs compared with controls, there were no unique histopathologic lesions indicative of injury that were specific to PB therapy. Further investigation is required to fully address the question of whether or not ALT or AP is induced in livers of PB-treated dogs and to determine the best serum marker of early hepatic injury due to PB therapy.
Footnotes
Acknowledgements
We thank the Diagnostic Laboratory of the Atlantic Veterinary College, the veterinarians throughout Atlantic Canada who participated in the study, and Dr. David Sims for his assistance with photomicrographs.
1Present address: Department of Veterinary Clinical Sciences, College of Veterinary Medicine, Ohio State University, 601 Vernon L. Tharp Street, Columbus, OH.
