Abstract
Monkeypox with extensive lesions was diagnosed in a prairie dog that was involved in a recent human outbreak of monkeypox in the Midwestern United States. Gross lesions included oral ulcers, pulmonary consolidation, enlarged cervical and thoracic lymph nodes, and multifocal, small, white umbilicated plaques in the gastrointestinal wall. Microscopic lesions were extensive in the lungs and consisted of fibrinonecrotic bronchopneumonia with vasculitis and poorly defined eosinophilic intracytoplasmic inclusion bodies in cells thought to be alveolar epithelial cells, histiocytes, and fibroblasts. Multifocal necrotizing lesions, often accompanied by myxedema, were also present in most of the other examined organs. Aggregates of pox viral particles were observed within lesions by transmission electron microscopy. Monkeypox virus infection was confirmed by real-time polymerase chain reaction and virus culture at the Centers for Disease Control and Prevention. This report highlights the difficulties of rapid diagnosis of exotic or emerging diseases and further substantiates the prairie dog as an animal model of monkeypox.
Monkeypox, a member of the Orthopoxvirus genus, appears to be enzootic among wild mammals in the west and central African rainforest, where the principal reservoirs are thought to be squirrels and other rodents. 2 In humans in the Western Hemisphere, a monkeypox virus outbreak occurred for the first time during May and June 2003 among several residents of the Midwestern United States. 1,3,4 As of 8 July 2003, 35 (49%) of a total of 71 human cases of monkeypox had been confirmed at the Centers for Disease Control and Prevention (CDC). 1 Trace-back investigations determined that all confirmed human cases were associated with exposure to captive prairie dogs. These captive prairie dogs had been cohoused at one or more distributors with African rodent species that originated in Ghana, including Gambian giant pouched rats (Cricetomys sp.) and dormice (Graphiurus sp.). Laboratory tests demonstrated the presence of monkeypox virus in several rodents that died unexpectedly without exhibiting characteristic signs of monkeypox and that had originated from a shipment from Ghana on 9 April 2003. 1 This article provides a detailed description of gross and microscopic lesions in an affected prairie dog from the outbreak as well as results of diagnostic testing accomplished through cooperating local and federal agencies.
A 12-week-old female prairie dog was purchased at a Reptile and Exotic Trade Show in Indianapolis, IN, and 5 days later was presented to a local veterinarian with swollen eyes. The prairie dog became increasingly depressed and anorexic and developed ocular and nasal mucous discharge, respiratory distress, oral ulcers, and skin nodules over a period of 4 days. Because of unresponsiveness to antimicrobial therapy, the prairie dog was euthanatized 10 days after onset of the illness. Two days later, the owner developed fever, headache, cough, sore throat, and peripheral lymphadenopathy, followed the next day by a vesicular skin rash that was subsequently confirmed as monkeypox. At the same time, a family friend who had close contact with the prairie dog also developed flulike clinical disease and a skin rash that was confirmed as monkeypox (James Howell, personal communication).
The prairie dog was submitted to the Purdue Animal Disease Diagnostic Laboratory, where a complete necropsy was performed. Specimens of multiple tissues were immersion-fixed in 10% neutral buffered formalin and were routinely processed, paraffin embedded, sectioned at 5 µm, and stained with hematoxylin and eosin for microscopic examination. Specimens of formalin-fixed lung and small intestine were also processed by routine methods and stained with uranyl acetate and lead citrate for examination by transmission electron microscopy. Specimens of lung, liver, and spleen were collected for bacterial culture that included media for cultivation of Haemophilus spp., Yersinia spp., and Francisella tularensis.
A homogenized tissue pool of spleen, liver, intestine, lung, kidney, and brain was tested at the CDC in two real-time polymerase chain reaction (PCR) tests for the detection of monkeypox virus DNA. The generic orthopox real-time PCR (i.e., E9L nonvariola) used conserved orthopoxvirus gene sequence derived from comparison of various orthopoxvirus DNA polymerase gene sequences (E9L is the descriptor for the DNA polymerase gene of vaccinia, the type species for orthopoxviruses). The monkeypox-specific real-time PCR test used envelope gene sequence derived from the B6R gene of monkeypox virus. 3,4 Monkeypox virus was isolated at the CDC from the same original prairie dog tissue pool, using continuous African green monkey cells (BSC-40 cells), and viral DNA was similarly identified.
Necropsy examination revealed numerous variably sized (1–5 mm) discrete ulcers on the dorsal, lateral, and ventral surface of the tongue (Fig. 1) and on the hard palate. The cranial and middle lobes of the lungs were extensively dark red, slightly depressed, and firm. The remaining pulmonary parenchyma was pale tan and failed to collapse. Multiple 1-to 3-mm white plaques were present in the visceral pleura. The cervical and thoracic lymph nodes were markedly swollen, measuring up to 7 × 4 mm (Fig. 2). Small (2–3 mm) white, firm foci with umbilicated necrotic centers were sparsely distributed throughout the glandular portion of the stomach and the intestine. These foci were deeply embedded in the gastrointestinal wall (Fig. 3), were visible from the serosal surface, and were slightly raised on the mucosal surface.
Tongue; prairie dog (Cynomys sp.). The dorsal surface of the tongue has several variably sized ulcers.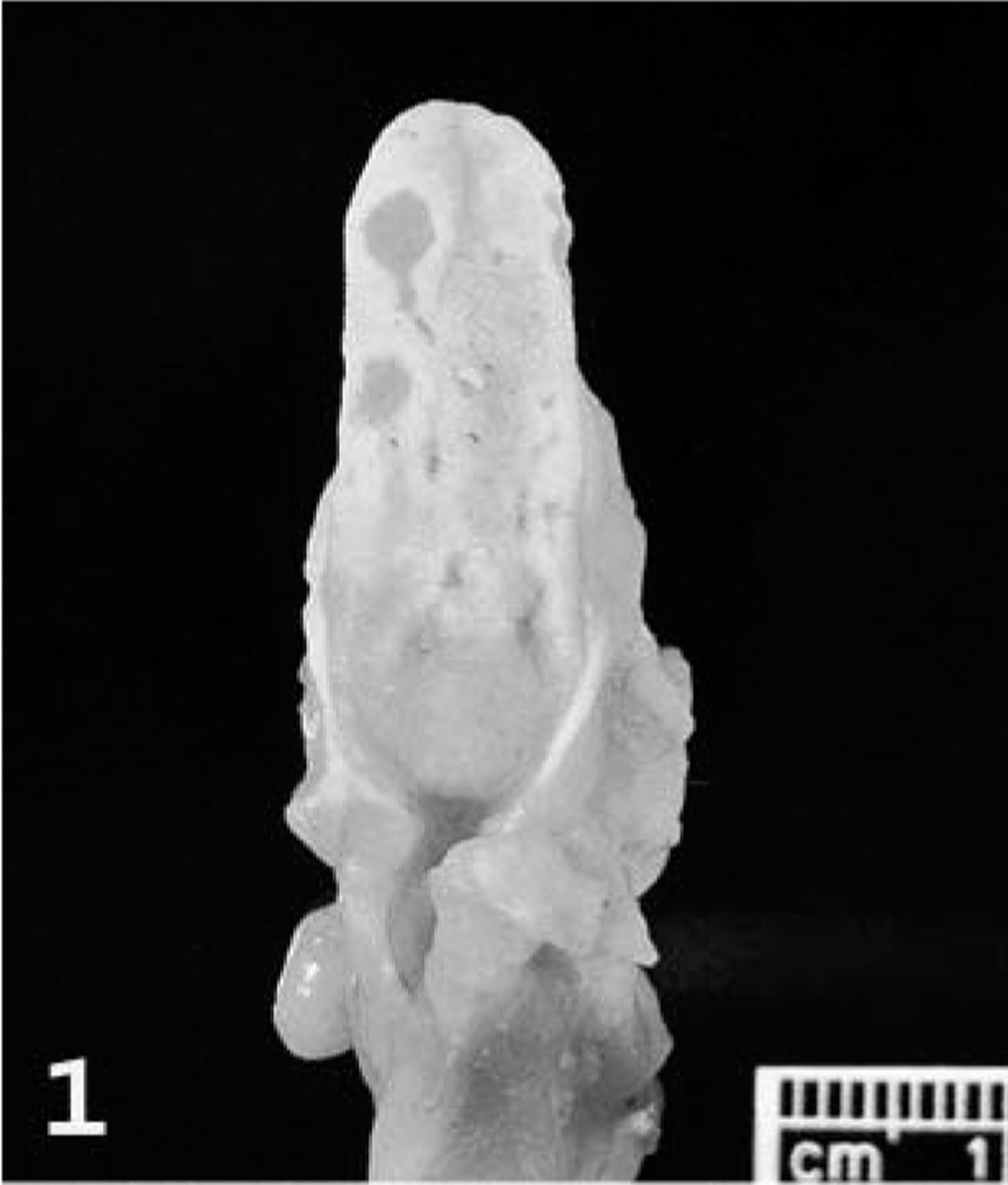
Lung; prairie dog (Cynomys sp.). The cranial and middle lobes are congested and slightly depressed. Multiple, small, white plaques thicken the visceral pleura. The bronchial lymph nodes are markedly swollen (arrows).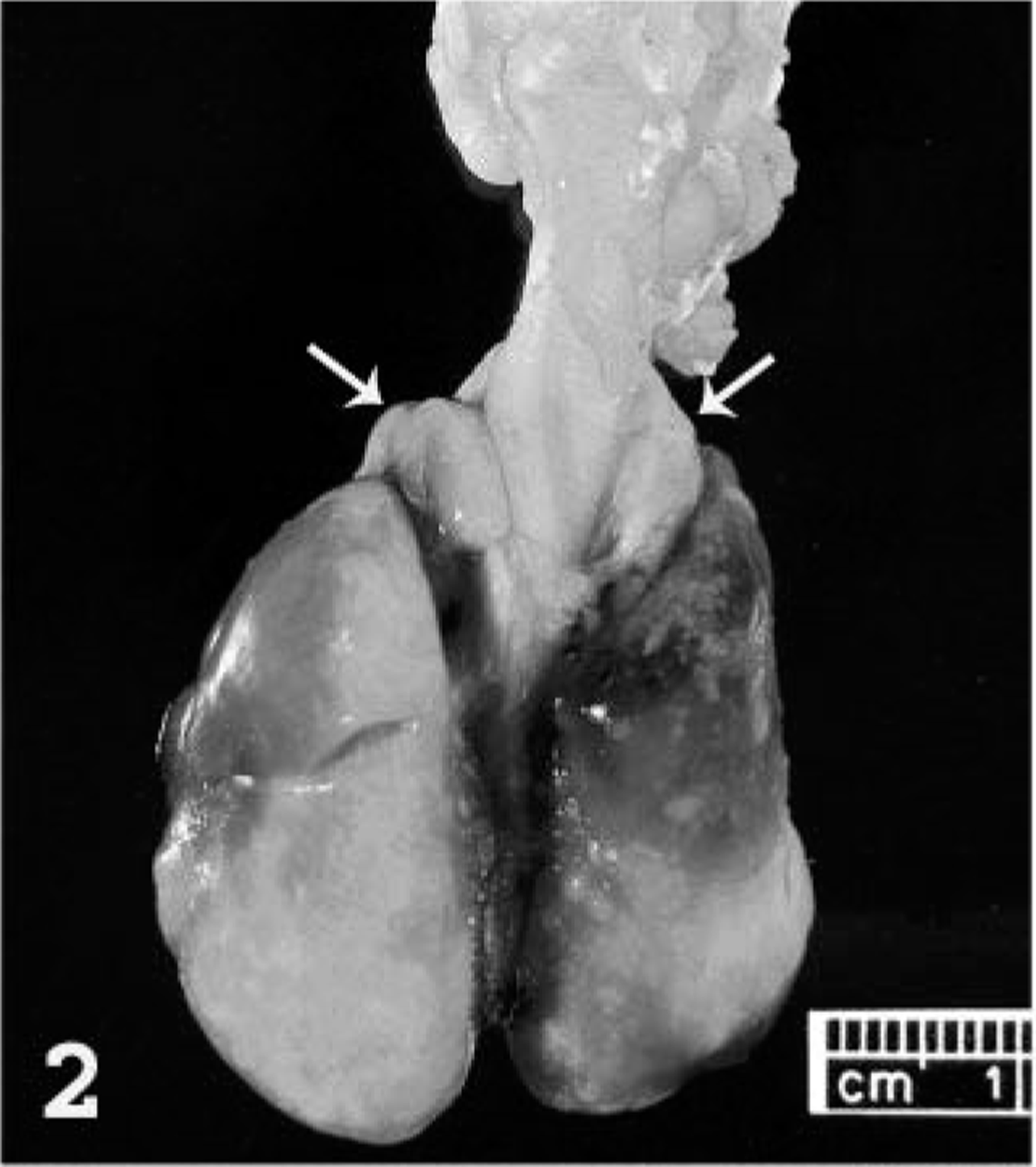
Intestinal tract; prairie dog (Cynomys sp.). Several white foci (arrowheads) in the intestinal wall are readily visible from the serosal surface.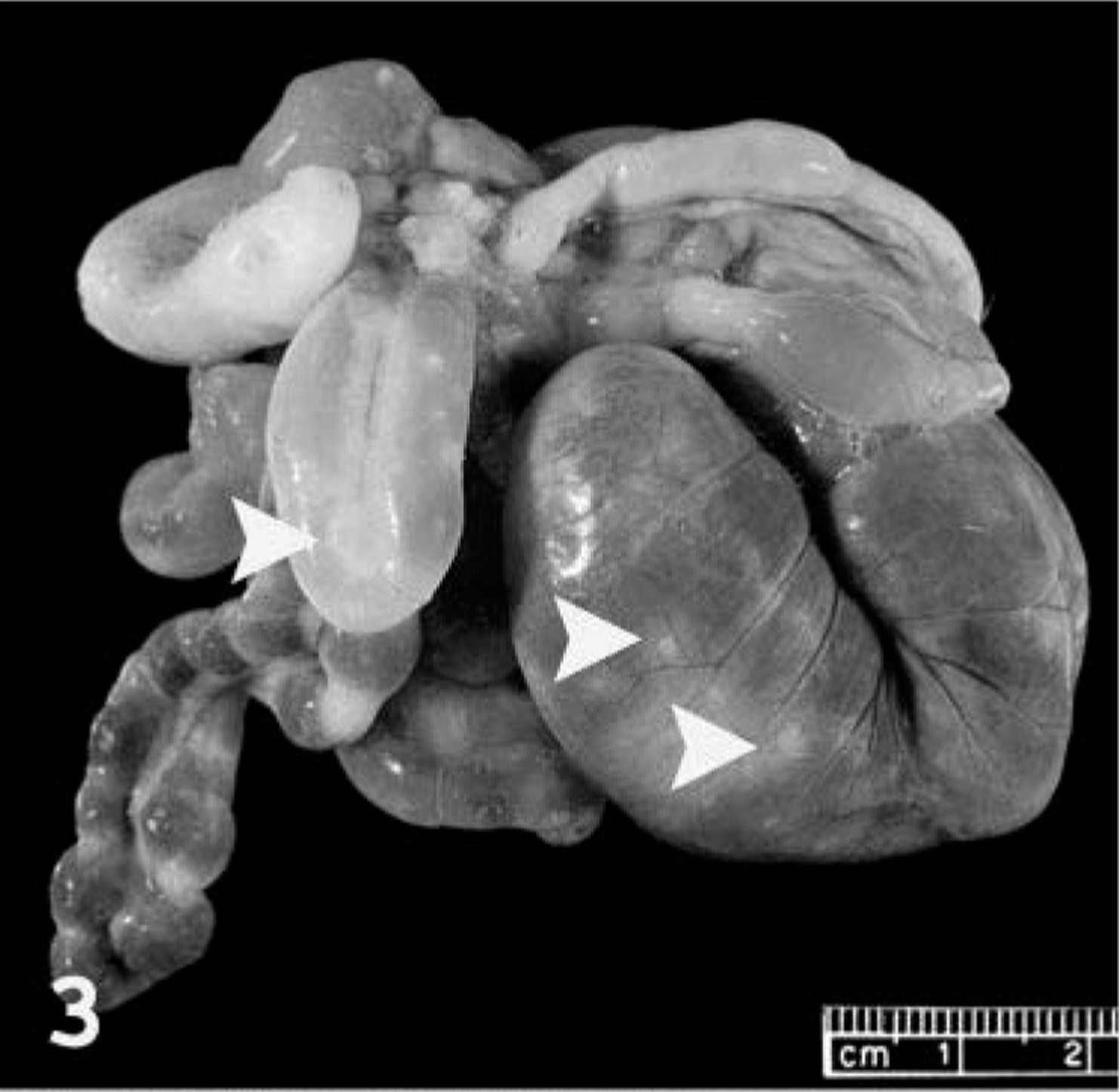
Microscopically, there was severe multifocal to coalescing fibrinonecrotic bronchopneumonia (Fig. 4) with vasculitis and poorly defined eosinophilic intracytoplasmic inclusions. The epithelium lining the bronchi and bronchioles was often necrotic and sloughed, occluding the lumen of the affected airways. The connective tissue surrounding the airways and blood vessels was markedly expanded by myxoid ground substance and inflammatory cells, many presumably of histiocytic origin, as well as syncytial cells and spindle cells (morphologically consistent with fibroblasts). Poorly defined eosinophilic inclusions measuring 3–7 µm in diameter were present either within the cytoplasm of alveolar epithelial cells, histiocytes, syncytial cells or fibroblasts, or were freely scattered throughout the necrotic debris. The walls of blood vessels were often segmentally or completely effaced by homogeneous eosinophilic material, neutrophils, and karyorrhectic debris (vasculitis). Dilated lymphatics in the interstitium contained fibrin, mixed inflammatory cells (primarily neutrophils and macrophages), and cellular debris. There were multifocal to diffuse areas of necrosis in the pulmonary parenchyma, containing eosinophilic debris, fibrin, erythrocytes, and degenerate leukocytes. The pleura was mildly thickened by fibrin and necrotic debris. Less affected areas of the pulmonary parenchyma were flooded with edema fluid and infiltrated by variable numbers of neutrophils and macrophages.
Lung; prairie dog (Cynomys sp.). Extensive necrosis of a bronchiole (B), blood vessel (V), and surrounding pulmonary parenchyma. The inset shows an inclusion body (arrowhead) within the cytoplasm of a degenerating cell presumed to be an alveolar macrophage. HE. Bar = 62.5 µm; inset: 15.5 µm.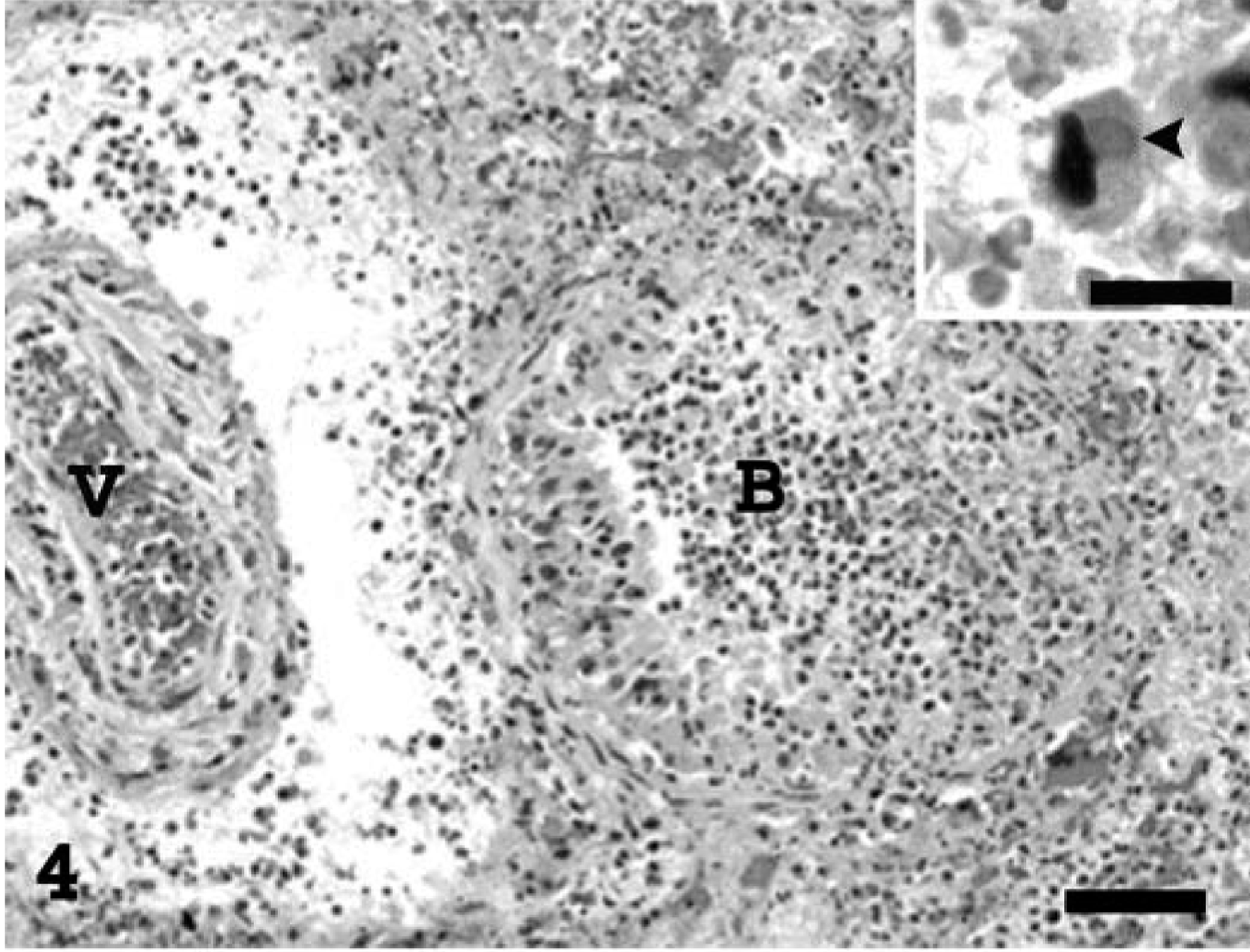
The severe necrotizing inflammatory process affecting the lungs extended into the adjacent mediastinal adipose and lymphoid tissue. Myxoid ground substance admixed with degenerate cells similar to those described in the pulmonary interstitium extensively expanded the subcapsular sinuses of the mediastinal lymph nodes, compressing the underlying lymphoid tissue (Fig. 5). The surrounding mediastinal fibroadipose tissue was necrotic.
Mediastinal lymph node; prairie dog (Cynomys sp.). Accumulation of abundant myxoid ground substance and degenerate cells, including several multinucleated syncytial cells, expand the subcortical sinus and compress the underlying lymphoid tissue. A high magnification of a multinucleated syncytial cell containing an intracytoplasmic inclusion body (arrowhead) is depicted in the inset. HE. Bar = 345 µm; inset: 36 µm.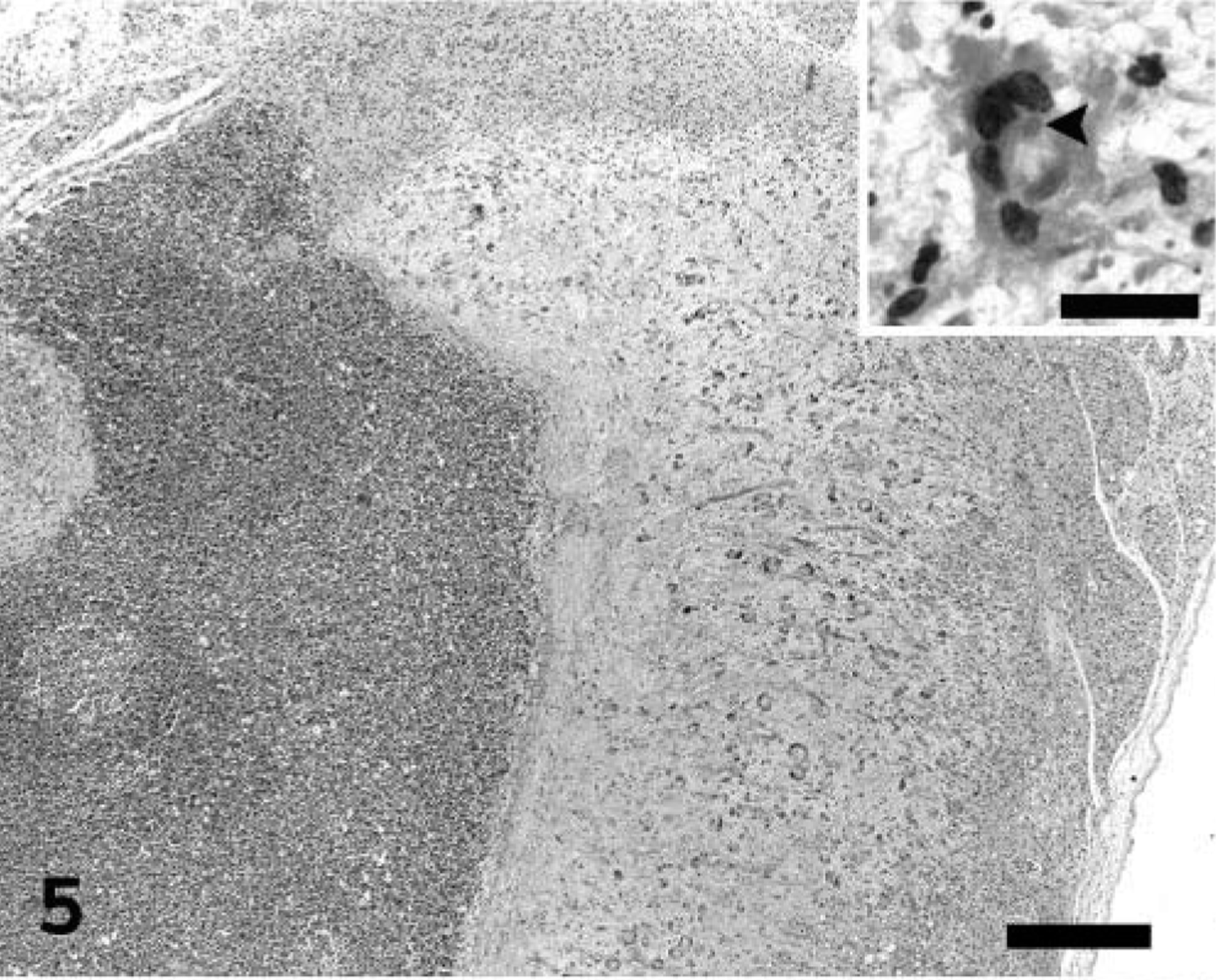
Multifocal necrotizing lesions, often accompanied by vasculitis, myxedema, and scattered poorly defined eosinophilic intracytoplasmic inclusion bodies in cells as described in the lungs, were also present in nasal turbinates, trachea, thymus and adjacent brown fat, lips, tongue, esophagus, stomach, jejunum, cecum, colon, liver, kidney, adrenal gland, broad ligament of the uterus, vagina, vestibule, female accessory genital glands, conjunctiva, and cornea. Mucus admixed with sloughed epithelial cells, cellular debris, fibrin, and extravasated erythrocytes filled the nasal cavity. In the trachea, the necrotic changes diffusely affected the mucosa and submucosa, extending into the smooth muscle and adventitia through the gaps in the cartilage rings. In the tongue, the necrotizing changes occasionally extended deep into the muscle and were associated with mild to moderate mixed inflammatory infiltrate. Multiple clusters of small (2–4 µm) yeasts morphologically consistent with Candida sp. were adherent to the mucosa at the ulcer margins. In the remaining digestive tract, multifocal necrosis involved predominantly the submucosa, with extension into the overlying mucosa, causing focal areas of ulceration (Fig. 6). Occasionally, the underlying muscularis externa was also involved, characterized by nuclear swelling and vacuolar degeneration of the smooth muscle cells. Sporadically, the necrosis affected the gut-associated lymphoid tissue. The liver had prominent Kupffer cells as well as rare necrotic foci in portal areas. The vaginal and vestibular mucosa had mild segmental acanthotic hyperplasia and ballooning degeneration of the stratum spinosum, with rare eosinophilic intracytoplasmic inclusion bodies (Fig. 7). In the vestibule, the affected squamous epithelium was partially ulcerated and mildly infiltrated with neutrophils. The adjacent skin had rare intracorneal neutrophilic aggregates (pustules). The follicular epithelium at the oronasal mucocutaneous junction had mild ballooning degeneration with occasional eosinophilic intracytoplasmic inclusion bodies and mild neutrophilic infiltrate.
Intestine; prairie dog (Cynomys sp.). The necrotizing lesions in the intestinal tract affect primarily the submucosa (here associated with the gut-associated lymphoid tissue), often followed by ulceration of the overlying mucosa. HE. Bar = 300 µm.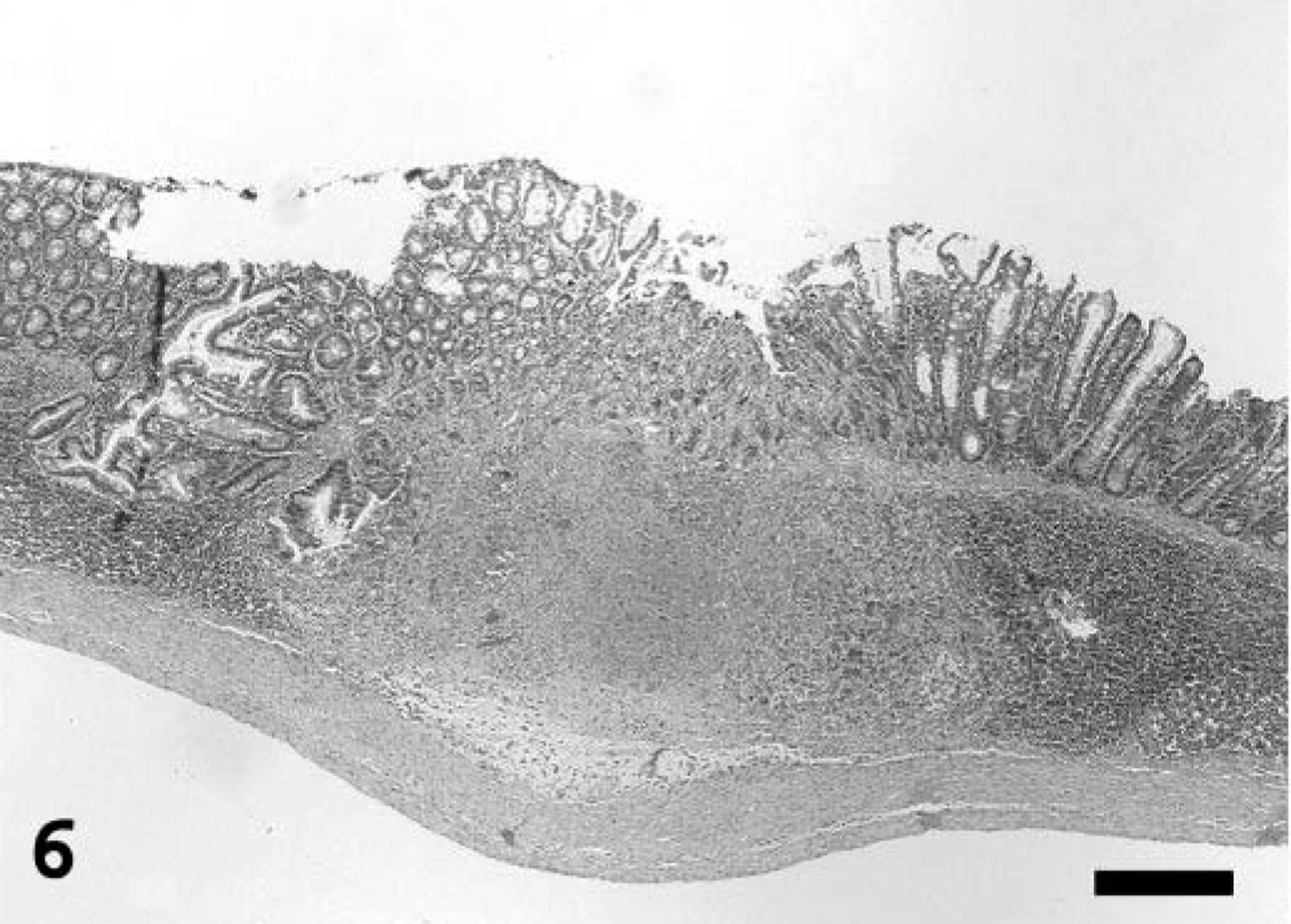
Vestibule; prairie dog (Cynomys sp.). The mucosa is mildly hyperplastic and vacuolated, with rare intracytoplasmic inclusion bodies (arrowheads). HE. Bar = 37.5 µm.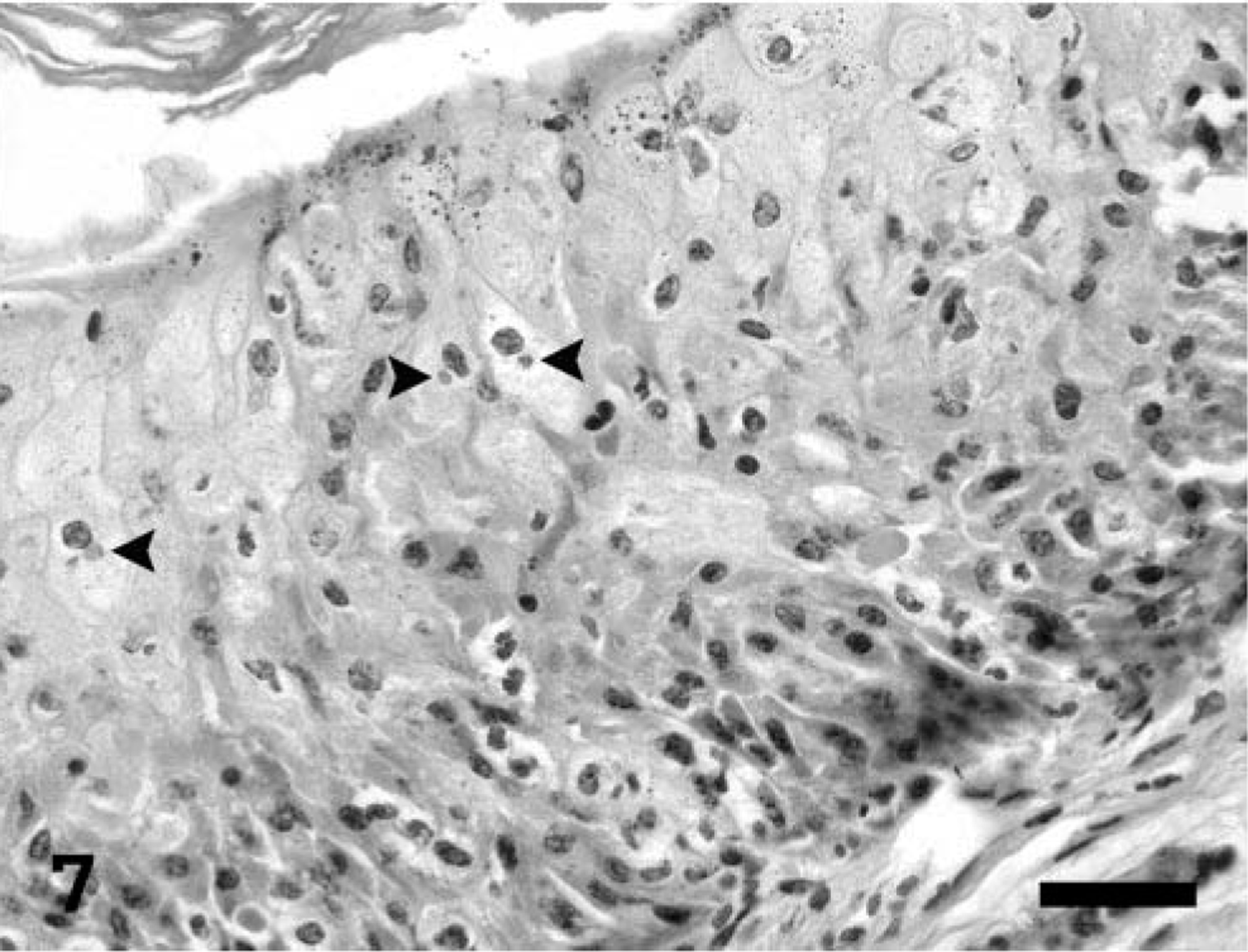
The femoral bone marrow was hypercellular, with increased numbers of myeloid precursor cells. No significant lesions were observed in the brain, Harderian and meibomian glands, heart, pancreas, major and minor salivary glands, epaxial, upper hindlimb and extraocular skeletal muscles, spleen, urinary bladder, or urethra.
Ultrastructural examination of lung and intestine revealed aggregates of mature, nonenveloped, up to 375 × 200 nm viral particles located free within the cytoplasm of scattered degenerating cells (Fig. 8). The virions had an outer membrane enclosing a biconcave electron-lucent core and two lateral bodies with the typical dumbbell-shaped appearance of poxviruses. The affected cells were thought to be type II pneumocytes and smooth muscle cells of the muscularis externa of the intestine, but the cell type could not be definitively determined because of necrosis.
Electron micrograph. Lung; prairie dog (Cynomys sp.). Aggregates of nonenveloped virions are located within the cytoplasm of a degenerate cell. Mature virions have the typical structure of poxviruses, with an outer membrane enclosing a biconcave core and two lateral bodies (inset). Uranyl acetate–lead citrate stain. Bar = 0.56 µm; inset: 0.188 µm.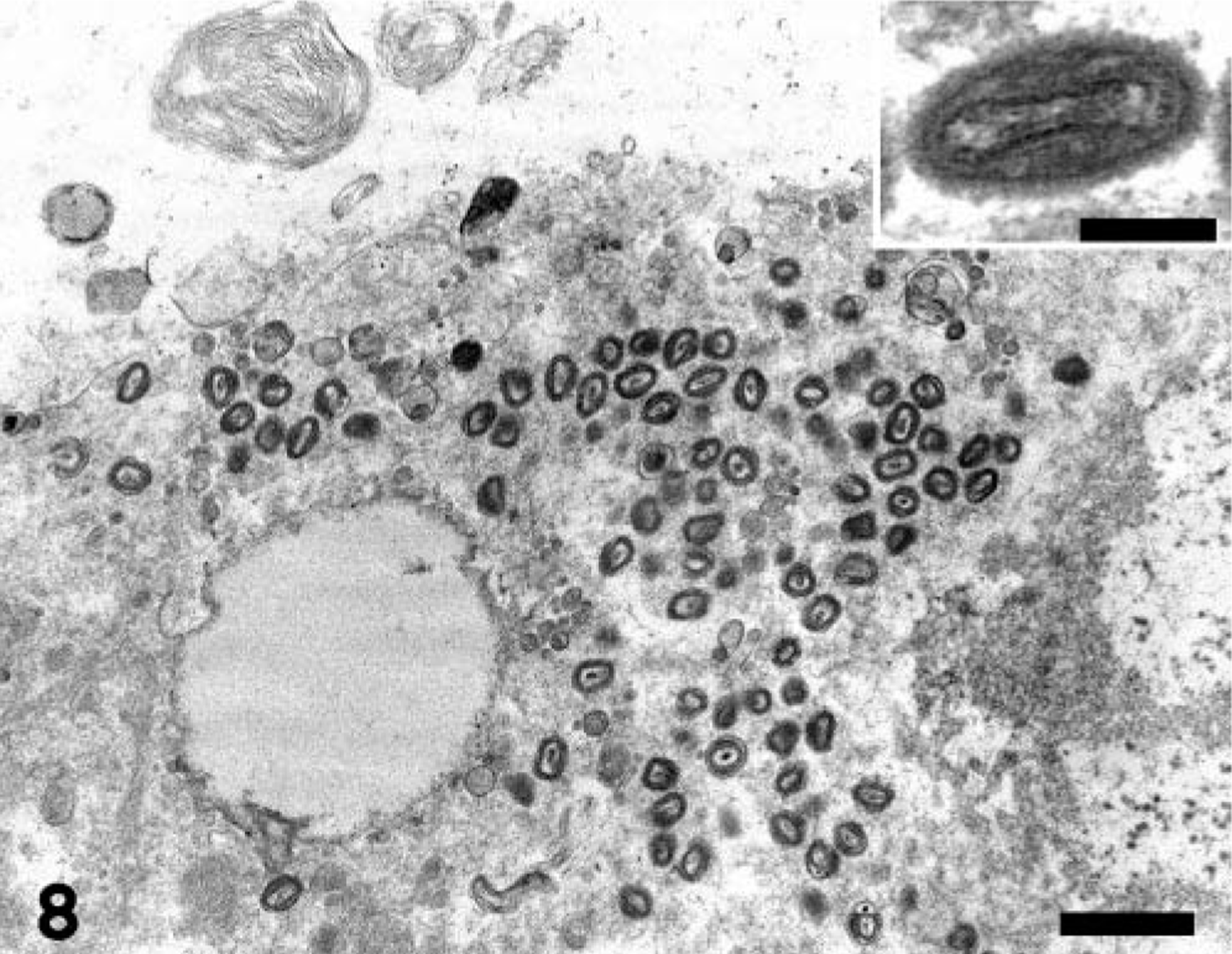
Bacillus circulans was cultured from the lung. Cultures for Haemophilus spp., Yersinia spp., and F. tularensis on the lung, and for Yersinia spp. and F. tularensis on the liver and spleen were negative.
The presence of orthopox generic DNA and monkeypox virus–specific DNA was confirmed in the homogenized tissue pool by the two real-time PCR tests. Cellular cytopathic effect consistent with monkeypox virus infection, characterized by focal syncytia formation, was observed in BSC-40 cells 4 days after inoculation with the phosphate buffered saline–tissue homogenate. This original culture was passaged one time, after which extensive macroscopically visible virus plaques were apparent by 2–3 days after infection.
A systemic monkeypox virus infection with extensive and severe lesions in numerous organs was diagnosed in this prairie dog. A presumptive diagnosis was made by ultra-structural observation of typical pox viral particles in affected tissues only after history was provided by the submitting veterinarian of suspected zoonotic disease in the owner as well as nearly simultaneous first reports of monkeypox in people associated with diseased prairie dogs. Early correct presumptive diagnosis was not made because of several factors. There was no previous report of monkeypox in North America. Cell degeneration and necrosis in a variety of organs were the most striking histologic lesions, resembling cases of acute septicemic bacterial diseases known to exist in North America, including tularemia and sylvatic plague. Although Bacillus circulans was isolated from the lung, it was considered to be an incidental finding because no report was found indicating B. circulans association with disease. Intracytoplasmic inclusion bodies, which are regarded as confirmatory for poxvirus infections, were not initially recognized in this prairie dog. The inclusion bodies that were later identified were poorly defined, did not stain, or stained poorly with eosin, and strongly resembled cell fragments or keratohyalin granules. Similarly, in some reported monkeypox cases in humans and monkeys, typical poxviral inclusion bodies were not numerous or readily identifiable. 6–8 At the time of submission, there was no history suggesting either skin eruptions during clinical examination of the prairie dog or illness in exposed humans. History of skin eruptions that were observed in the ill prairie dog at a later stage of the disease was provided by the submitting veterinarian only during discussions several days after necropsy examination. Skin lesions were inapparent during necropsy, so specimens of skin were not collected for microscopic examination. Skin lesions were likely obscured by the dense hair coat on the prairie dog. Humans did not become ill until several days after the prairie dog was euthanatized and submitted for diagnostic testing. Because the incubation period for monkeypox in humans is approximately 12 days (range 4–24), 4 it is likely that future cases of monkeypox in prairie dogs or other susceptible animal reservoirs will be submitted for clinical or postmortem diagnostic testing prior to known zoonotic disease. All these aspects clearly illustrate the challenge in making the diagnosis of monkeypox virus infection in non-usual hosts. These cases of monkeypox in prairie dogs and humans in the Midwestern United States also highlight the danger of emergence of a variety of diseases, previously considered to be geographically restricted, because of modern international travel and commerce.
The first clinical sign in the prairie dog was “swollen eyes,” most likely because of necrotizing blepharoconjunctivitis that was observed at necropsy. This sign has also been reported as the first sign in other infected prairie dogs involved in this outbreak, progressing to respiratory signs and nodular skin lesions in some cases. 3,4 Presence of conjunctivitis in prairie dogs or exotic rodents may be a discriminating clinical or postmortem finding justifying the inclusion of monkeypox in the list of preliminary differential diagnoses when it is accompanied by pyrexia or any of the described gross lesions.
The lesions in this prairie dog were severe and present in the majority of the examined organs, resembling in general those seen after experimental and natural infection in animals. 3,5,7,8 Alterations described recently in two other captive prairie dogs infected with monkeypox in the same outbreak of the disease in the United States were restricted to conjunctiva, tongue, and lung. 3 Euthanasia might have interfered with the full development of lesions associated with monkeypox virus infection in these animals. The lesions in the prairie dog in this study were also essentially identical in characteristics and distribution with that described in an aerosol model of monkeypox in cynomolgus monkeys, which reportedly resembled the features of monkeypox and smallpox in humans. 8 Thus, this report confirms the susceptibility of prairie dogs to severe monkeypox virus infection and further substantiates this animal species as a small, relatively inexpensive, and easily available animal model for monkeypox as suggested by Guarner et al. 3
Footnotes
Acknowledgements
We thank Ms. Phyllis Lockard (Indiana Animal Disease Diagnostic Laboratory, West Lafayette, IN) for electron microscopic preparations, the CDC monkeypox laboratory response team (including Drs. Victoria A. Olson, Yu Li, James V. Lange, Mr. Richard Kline, Mr. Edgardo J. Vergne, Ms. Tiara J. Harms, Linda Stempora, Elizabeth Sargent, Bette Jensen, and Dr. Inger K. Damon; Atlanta, GA) for confirmation of the presumptive diagnosis by real-time PCR and virus isolation, Dr. James Howell (Epidemiology Resource Center, Indiana State Department of Health, Indianapolis, IN) for contributions on epidemiologic and clinical data of the prairie dog and related human cases, and Dr. José A. Ramos-Vara (School of Veterinary Medicine, Purdue University, West Lafayette, IN) for critical review of this manuscript.
