Abstract
One-day-old gnotobiotic piglets were inoculated intranasally with in vitro passaged porcine circovirus 1 (PCV-1), PCV-2, and porcine parvovirus (PPV) alone or in combination (PCV-1/PCV-2, PCV-1/PPV, and PCV-2/PPV). Piglets were evaluated for 1) the development of porcine postweaning multisystemic wasting syndrome (PMWS), 2) distribution of viral antigens by immunochemistry, and 3) viremia and the presence of viral DNA in nasal and ocular secretions and feces. All single agent-infected piglets and piglets infected with PCV-1/PCV-2 or PCV-1/PPV were clinically asymptomatic. They were transiently viremic and seroconverted to homologous virus(es). At termination of the study on postinfection day (PID) 35, microscopic lesions were restricted to focal inflammatory cell infiltrates in livers and myocardia. One piglet given PCV-1/PPV was PPV viremic for 2 weeks after infection and had lymphangiectasia of the spiral and descending colon associated with granulomatous inflammation. All four PCV-2/PPV-inoculated piglets developed PMWS, characterized by sudden onset of depression and anorexia, icterus, and submucosal edema. One piglet became moribund on PID 27, and the remaining three piglets were euthanatized between PID 27 and PID 30 because of severe disease. Lymph nodes were small and the livers were mottled. Disseminated angiocentric granulomatous inflammation was present in all tissues examined except the brain. Multiple lightly basophilic intracytoplasmic inclusion bodies were identified in macrophages and histiocytes. PCV-2 antigen was widely distributed within macro-phages; PPV antigen was sparse. Hepatocellular necrosis and bile retention were prominent. PCV-2 DNA was identified in ocular, fecal, and nasal secretions. Terminal sera contained antibodies to PPV (4/4) and PCV-2 (3/4). Production of PMWS in gnotobiotic swine appears to require PCV-2 and additional infectious agents such as PPV for full disease expression in gnotobiotic piglets.
Porcine postweaning multisystemic wasting syndrome (PMWS) was first identified in western Canada 5 , 7 , 10 , 13 and more recently in Europe 5 , 6 , 24 and the far east (G. Allan, personal communication, 1998). In the United States, wasting disease has been reported in California and recently in the Midwest. 5 , 21 The disease is characterized by a constellation of clinical signs, including progressive weight loss, jaundice, and pneumonia. 7 , 13 , 25 Losses in affected herds are substantial, with up to 40% mortality in weanling pigs. Gross lesions consist of generalized lymphadenopathy, hepatitis, nephritis, and pneumonia. Histologically, the disease is characterized by widespread granulomatous inflammation, syncytial giant cells, and variable numbers of intracytoplasmic basophilic viral inclusions within infiltrating histiocytes and macrophages. 7 , 21 , 25 A novel viral agent, porcine circovirus 2 (PCV-2), has been consistently isolated from tissues, and both PCV-2 DNA and PCV-2 protein have been demonstrated within histologic lesions in pigs with PMWS. 5 , 6 , 10 , 12 , 19 , 21 , 25
The complete DNA sequence of PCV-2 is known; 12 , 18 , 21 it is less than 80% homologous to the nonpathogenic porcine circovirus 1 (PCV-1; PCV-CCL33). PCV-1 was recovered as a noncytopathic viral contaminant of primary and continuous porcine kidney cell lines 27 and appears to be widespread within swine populations. 9 Although PCV-1 has been recovered from mummified fetuses, 3 experimental infection of neonates with PCV-1 has not produced clinical disease. 3 , 31 PCV-1 and PCV-2 share at least one common antigen but can be distinguished from each other using virus type–specific primers in polymerase chain reactions (PCRs) and with monoclonal antibodies. 5 These porcine viruses, along with two avian agents and several plant viruses, belong to a new viral family, the Circoviridae. 17 , 30 , 32
The natural history of infection in susceptible pigs or within herds is not known. The pathogenesis of infection, cellular tropisms, and method(s) of spread have not yet been delineated, although it appears that the virus preferentially infects macrophages and is hematogenously disseminated in vivo. There is an urgent need to identify tissue tropisms, the extent and nature of lesions, and the patterns of viral persistence and shedding so that reliable diagnostic tools and control measures can be developed. Toward this end, experimental challenge studies in both gnotobiotic 11 and snatch-farrowed conventional 2 swine were recently conducted. Subclinical infection and histologic lesions compatible with early PMWS were obtained after inoculation of gnotobiotic piglets with clinical material or low-titer tissue culture origin virus; 11 moderate to severe PMWS was produced in conventional pigs 2 after inoculation of PCV-2 with or without added porcine parvovirus (PPV). In the gnotobiotic transmission study, 11 convalescent sera from virus-inoculated but not control piglets contained antibodies to PPV. Retrospective analysis of viral inocula revealed low levels of PPV antigen and DNA, indicating that PPV was also present in the organ homogenates prepared from some PMWS cases and in the isolated tissue culture virus stocks derived from them. PPV has been recovered from field cases of PMWS. 2
The mechanisms whereby minimally pathogenic PPV 2 , 8 potentiates PCV infection in gnotobiotic animals are not known. Moreover, it is not known if the apparent disease-potentiating effects of PPV is restricted to interaction with PCV-2 or if a similar pathogenic influence might also render PCV-1 virulent. The importance of these interactions is best determined in gnotobiotic swine, which are free of unrelated infectious disease and confounding differences in management and microflora. The objectives of this study were to 1) confirm the synergistic relationship between PCV-2 and PPV in producing severe PMWS in gnotobiotic swine, 2) determine if PCV-1 is rendered virulent by coinoculation of neonatal gnotobiotic swine with this virus and PPV, and 3) identify probable mode(s) of virus spread both within the pig and between pigs.
Materials and Methods
Piglets
Four date-mated pregnant sows were purchased from a closed confinement swine operation, and a total of 26 piglets were derived by Caesarian section 15 into sterile pentub isolation units, three or four piglets per unit. Each infection group was housed in separate self-contained isolation units to maintain biosecurity, and piglets were fed a commercial sow milk replacement diet as previously described. 15 Prior to and at the conclusion of the experiments, each isolation unit (food, feces, and cages) was cultured for aerobic and anaerobic bacterial growth to confirm gnotobiotic status; all cultures were negative for bacterial growth.
Viruses
A tissue-culture–propagated strain of PCV-2 5 was used as the source of viral inoculum. Virus was propagated in PCV-free PK15 cells 9 and characterized as PCV-2 by nucleotide sequence analysis 6 , 19 and reactivity with PCV-2–specific monoclonal antibody. 5 , 10 For challenge, a viral pool containing 1.0 × 105.5 TCID50/1.0 ml was prepared from the 24th in vitro passage of this virus and screened for selected porcine viruses (PPV, porcine reproductive and respiratory syndrome virus, porcine hepatitis E virus, and Aujesky's disease herpesvirus) as previously described. 5 , 11 , 20 PK-15 cell-origin PCV-1 30 (provided by Dr. G. Allan 3 , 4 ) was propagated as described, 29 and a viral pool (2.0 × 105.0 TCID50/1.0 ml) for inoculation was created as described for PCV-2 above, aliquoted, and stored at −70 C until use. PPV was recovered from tissues of a pig with PMWS by culture on primary porcine kidney cell monolayers. The virus was identified as PPV by nucleotide sequence analysis 2 and was purified by the limiting dilution method. A viral pool containing 2.0 × 105.0 TCID50/1.0 ml was prepared, aliquoted, and stored at −70 C for in vivo challenge.
Experimental design
The piglets were divided into infection groups as follows: group A (n = 3), cell lysate (PK-15) control; group B (n = 3), PCV-1 alone; group C (n = 3), PCV-2 alone; group D (n = 3), PPV alone; group E (n = 4), PCV-1 and PPV; group F (n = 4), PCV-2 and PPV; group G (n = 4), PCV-1 and PCV-2. Two uninfected piglets were housed with the PCV-1/PCV-2–infected piglets to assess horizonal transmission of PCVs.
At 1 day of age, preinoculation serum and an unclotted blood sample were collected, and each piglet was inoculated with 1.0 ml of viral material (0.5 ml/nostril). Blood samples were collected at weekly intervals thereafter until euthanasia on postinfection day (PID) 35 or until death. Heparinized blood was divided into aliquots, and one portion was used to isolate peripheral blood mononuclear cells (PBMC) by discontinuous gradient centrifugation. 16 In some instances, replicate cytospin preparations of PBMC were prepared for determination of the presence of viral protein(s) by indirect immunoperoxidase (IPO) stain. For isolation of total cellular DNA for determination of the presence of viral DNA by PCR, 0.2 ml PBMC suspension (5.0 × 106 cells/ml) was blotted onto Whatman filter paper, air dried, and stored at −70 C. Replicate 0.2-ml samples of whole blood were also blotted onto filter paper and processed as above. Fecal, nasal, and ocular swabs were collected at necropsy for determination of the pattern of virus excretion using PCR.
Histopathology and immunocytochemistry
Samples of myocardium, cerebellum, cerebrum, lung, liver, kidney, spleen, thymus, mandibular, bronchial, and mesenteric lymph nodes, stomach, pancreas, duodenum, jejunum, ileum, spiral colon, terminal colon, and all grossly evident lesions were collected into 10% (v/v) phosphate buffered formalin and processed for histopathologic examination. Six-micrometer sections were stained with hematoxylin and eosin for histopathologic evaluation. IPO staining for PCV-2 antigen was performed manually or with an automated staining apparatus (Fisher Biotech MicroProbe, Fisher Scientific) using a monoclonal antibody specific for PCV-2. 5 Deparaffinized sections were quenched for endogenous peroxidase with 3% H2O2 in phosphate-buffered saline (PBS), reacted with purified monoclonal antibody or cell culture supernatants containing monoclonal antibody blocked with 4% (v/v) heat-inactivated horse serum, reacted with biotinylated horse anti-mouse IgG, reacted with avidin-peroxidase, and visualized with diaminobenzidine (DAB) (Vectastain®).
For PPV, a second set of samples of lung, liver, kidney, spleen, thymus, lymph nodes, duodenum, jejunum, ileum, spiral colon, and terminal colon were collected, embedded in OCT medium, and frozen (−70 C) for determination of the distribution of PPV antigen by immunohistochemistry. Frozen sections were fixed in acetone, quenched with 0.3% H2O2 in PBS, and incubated with monoclonal antibody to PPV (VMRD, Pullman, WA) followed by biotin-conjugated horse anti-mouse IgG. Bound secondary antibody was detected with avidin-peroxidase developed with DAB as chromagen.
Virology, serology, and PCR for viral DNA
Serum antibody titers to PCV-1 and PCV-2 were determined on individual or pooled 10-fold dilutions of sera by indirect immunofluorescence using acetone-fixed monolayers of PK15 cells infected with PCV-1 or PCV-2 as described. 11 Antibody titers to PPV were determined by microtiter hemagglutination inhibition (HAI) assay. 11 Viral DNA was amplified from samples of total cellular DNA extracted from serial whole blood and PBMC by PCR amplification with primers specific for PCV-1, PCV-2, and PPV DNA as previously described. 5 , 6
Results
Piglets inoculated with single agents or uninfected cell lysates
All piglets inoculated with uninfected cell lysates or single virus inocula (PPV, PCV-1, or PCV-2) remained clinically normal. They were euthanatized on PID 35. The only gross evidence of viral infection was a 2–4-fold increase in the size of some lymph nodes. Tissue samples from uninfected control gnotobiotic piglets were histologically normal. Minimal histopathologic changes were seen in piglets inoculated with PPV alone. In this group, multifocal lymphoplasmacytic hepatitis and cholangitis were present in two piglets; one of these had similar cellular infiltrates in the myocardium. In lymphoid tissues, mild lymphoid hyperplasia and germinal center formation were observed. Gnotobiotic piglets inoculated with PCV-1 had very mild lymphoid hyperplasia and germinal center formation in lymph nodes. All piglets that received PCV-2 had mild lymphoplasmacytic hepatitis and cholangiohepatitis similar to those seen in PPV-infected piglets. Similar multifocal cellular infiltrates were seen in the myocardium of three piglets and in the lung and the pancreas of one piglet.
Piglets inoculated with PCV-1/PPV and PCV-1/PCV-2
All piglets that received PCV-1/PCV-2 were clinically asymptomatic. Lymphadenopathy similar to that seen in the single agent inoculation groups was observed postmortem. Three of four piglets inoculated with PCV-1/PPV were clinically and grossly indistinguishable from the single-agent pigs. The remaining piglet in this group exhibited thickening of serosal surfaces throughout the intestine, which was most evident in the spiral colon (Fig. 1). On cut section, numerous small (1–5 mm2) cystic spaces were observed in the subserosa. Piglets inoculated with PCV-1/PCV-2 had multifocal lymphoplasmacytic cholangiohepatitis quantitatively similar to that seen in piglets inoculated with PPV or PCV-2 alone. Moderate to severe lymphoplasmacytic myocarditis was observed in three of four animals (Fig. 2). Viral inclusion bodies were not seen.
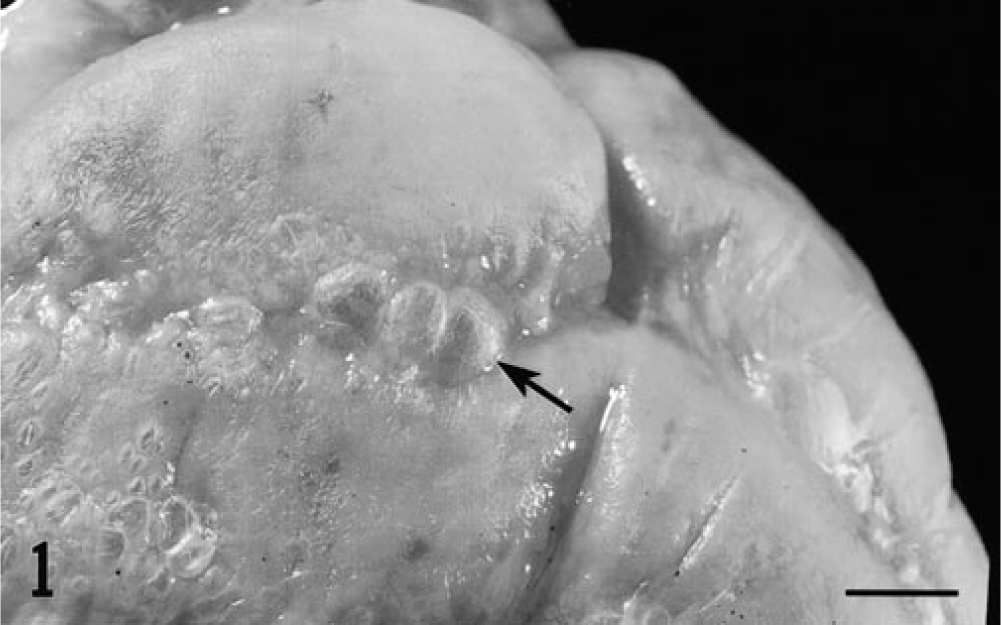
Serosal surface of spiral colon; piglet inoculated with PCV-1/PPV and terminated on postinfection day 35. Note the numerous small cystic spaces evident beneath the serosal surface of the colon. Bar = 0.5 cm.
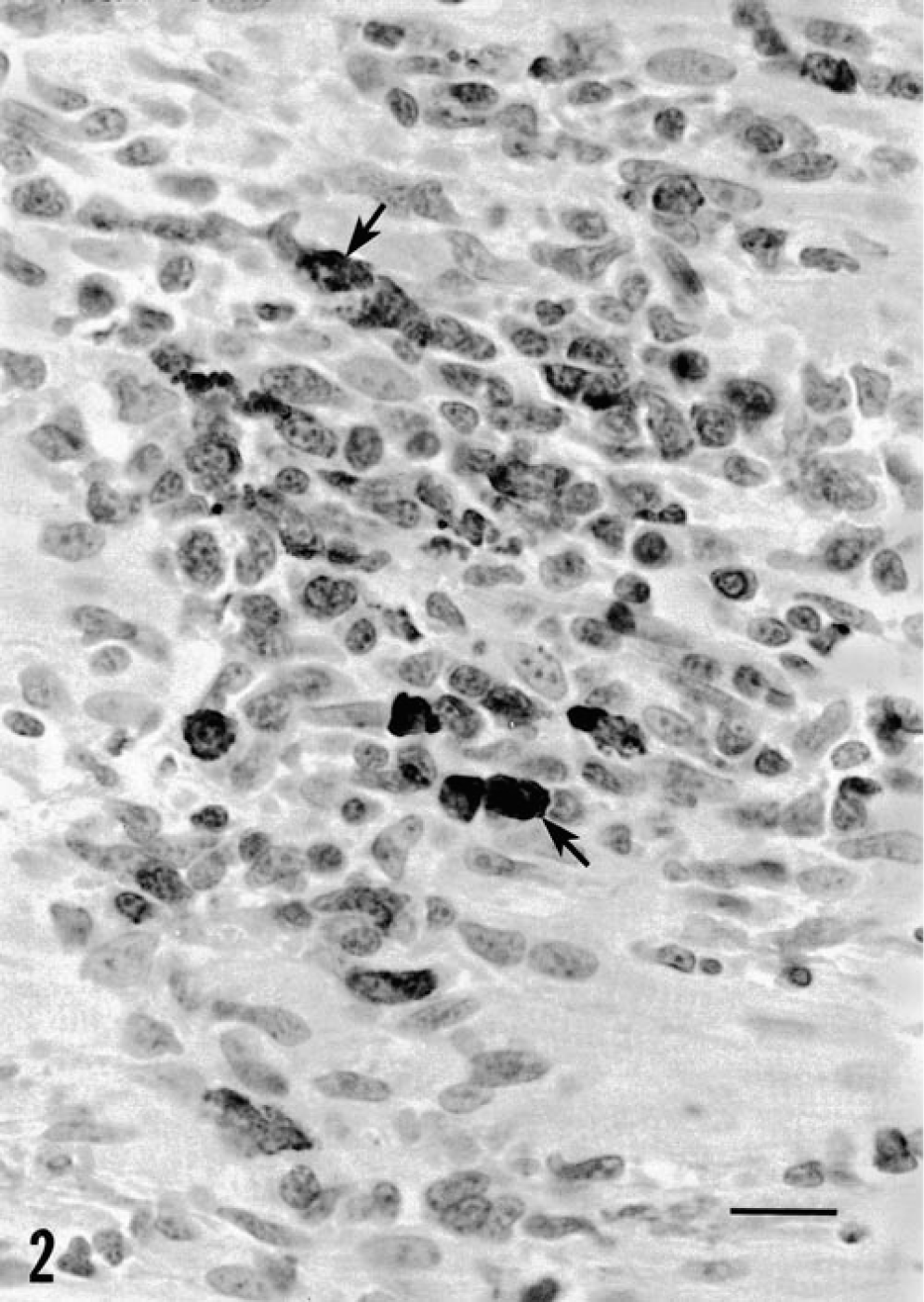
Myocardium; piglet inoculated with PCV-1/PCV-2 35 days previously. A serial section of this focal area of lymphoplasmacytic inflammation stained for PCV-2 (arrows) reveals the presence of numerous antigen-positive infiltrating macrophages and cardiac myocytes. HE. Bar = 25 µm.
PCV-1/PPV–inoculated piglets also exhibited variable lymphoid hyperplasia and germinal center formation in lymph nodes; three of four piglets had small accumulations of neutrophils and eosinophils within medullary sinuses. Multifocal lymphoplasmacytic cellular infiltrates were seen in the kidney (4/4 piglets), liver (4/4), myocardium (4/4), gastrointestinal tract (2/4), and brain (1/4). Histologic lesions in the piglet that had gross lesions in the intestine consisted of moderate to severe granulomatous inflammatory cell infiltrates closely associated with dilated cystic lymphatics. Infiltrates of macrophages and syncytial giant cells were prominent. Segmental lymphangiectasia was interpreted to be the result of blockage of regional draining lymphatics by inflammatory cell accumulations (Fig. 3). Viral inclusion bodies were not observed.
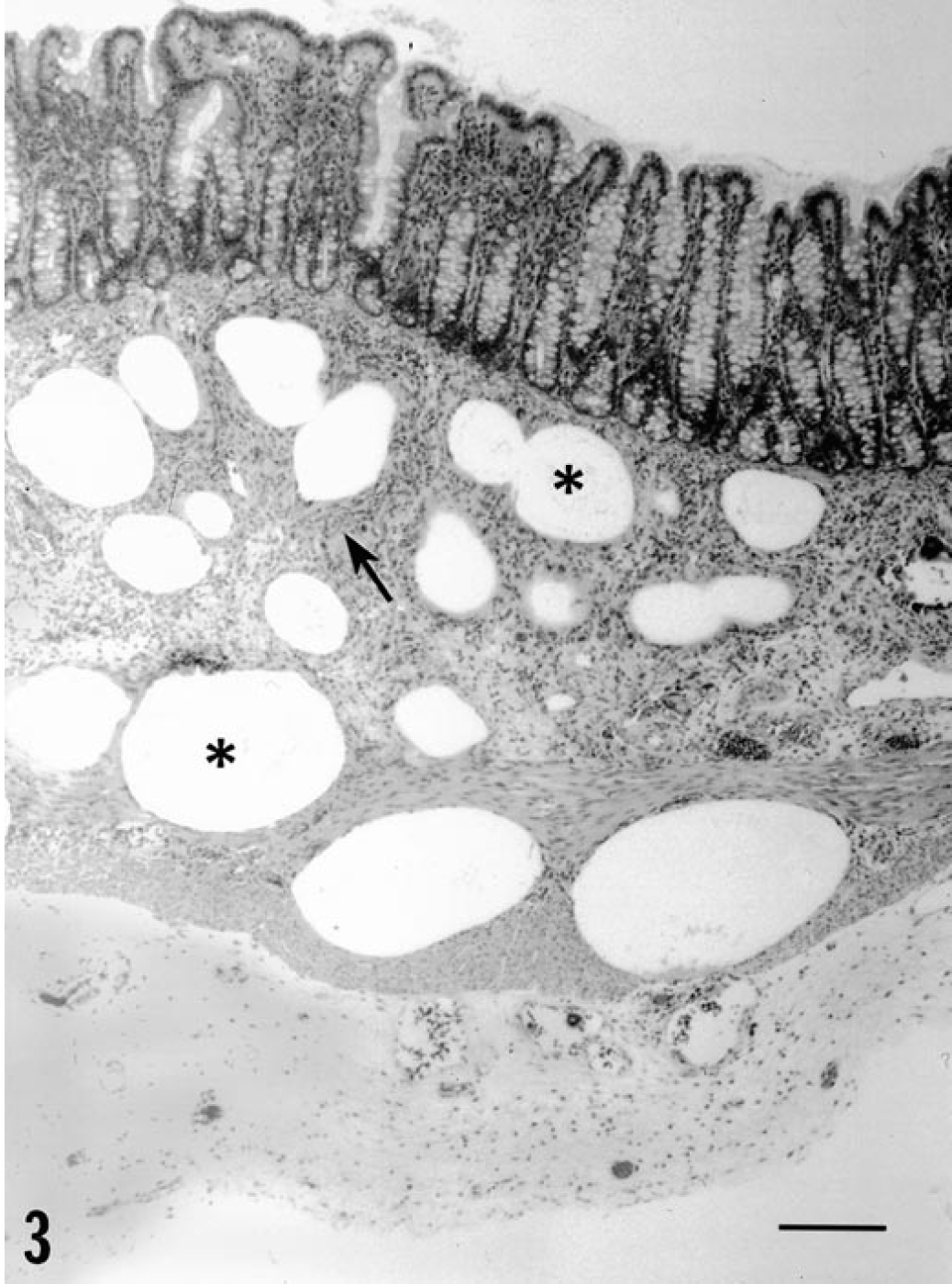
Serosal surface of spiral colon; piglet inoculated with PCV-1/PPV. Note the dilated submucosal lymphatics (∗) and the presence of syncytial giant cells and associated granulomatous inflammation (arrow) in the lamina propria. HE. Bar = 300 µm.
Piglets inoculated with PCV-2/PPV
Severe clinical disease and gross lesions of PMWS were observed in all piglets inoculated with PCV-2/PPV (Table 1). In this infection group, one piglet became anorectic and developed icterus on PID 24. The icterus increased in severity over the next several days and was accompanied by generalized subcutaneous edema. This piglet was euthanatized on PID 27. By PID 27, the three remaining piglets developed icterus and mild generalized subcutaneous edema and were euthanatized on PID 30. At necropsy, icterus, edema, and red to yellow mottling of the liver were seen. Thymuses were reduced in three of four piglets. Lymph nodes were comparable in size to those of uninfected controls. Ulcers of the nonglandular gastric cardia, 2–3 cm in diameter, were present in two piglets. Histologic lesions in piglets inoculated with PCV-2/PPV were severe and widespread in three of four piglets and moderate in the remaining animal. The primary lesion was widespread disseminated angiocentric granulomatous inflammation. A modest infiltrate of lymphocytes, neutrophils, and eosinophils accompanied macrophages. Lymph nodes were depleted of mature lymphocytes; germinal centers were reduced or absent. Syncytial giant cells were prominent and frequently contained mitotic figures. Many macrophages within the developing granulomas contained numerous lightly basophilic cytoplasmic viral inclusion bodies (Fig. 4). The granulomatous inflammatory infiltrate was most severe and widespread in the liver. Hepatic parenchyma was diffusely infiltrated with macrophages, lymphocytes, neutrophils, and eosinophils that displaced and replaced hepatocytes. Viral inclusion bodies similar to those identified in lymph nodes were detected within macrophages. Most hepatocytes exhibited nuclear pyknosis and karyorrhexis or were anucleate (Fig. 5); some were enlarged and vacuolated with centrally placed nuclei. Bile retention was prominent, and there was substantial fibroblastic proliferation.
Results ∗ in gnotobiotic piglets inoculated with virus(es) recovered from piglets with postweaning multisystemic wasting syndrome (PMWS).
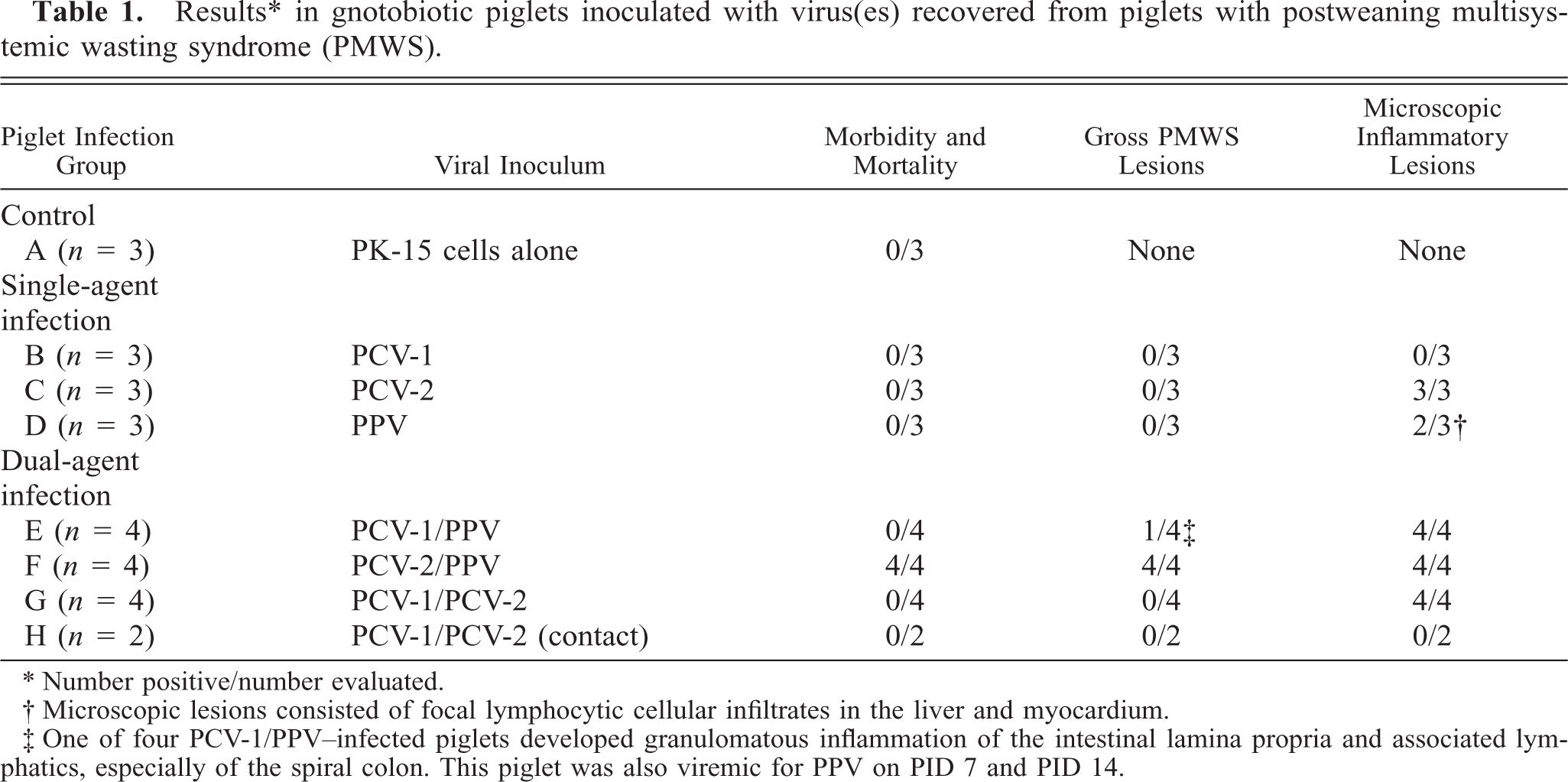
∗ Number positive/number evaluated.
† Microscopic lesions consisted of focal lymphocytic cellular infiltrates in the liver and myocardium.
‡ One of four PCV-1/PPV-infected piglets developed granulomatous inflammation of the intestinal lamina propria and associated lymphatics, especially of the spiral colon. This piglet was also viremic for PPV on PID 7 and PID 14.
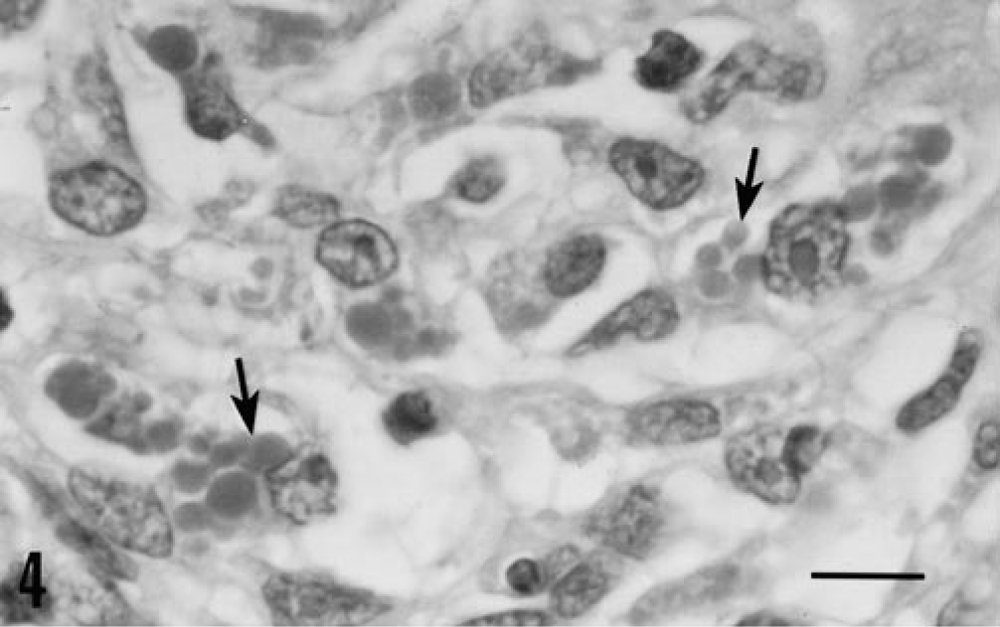
Lymph node; piglet inoculated with PCV-2/PPV 27 days previously. This node was PCV-2 positive. Note the presence of well-developed lightly basophilic intracytoplasmic inclusion bodies (arrows) within macrophages and histiocytes. HE. Bar = 1 mm.
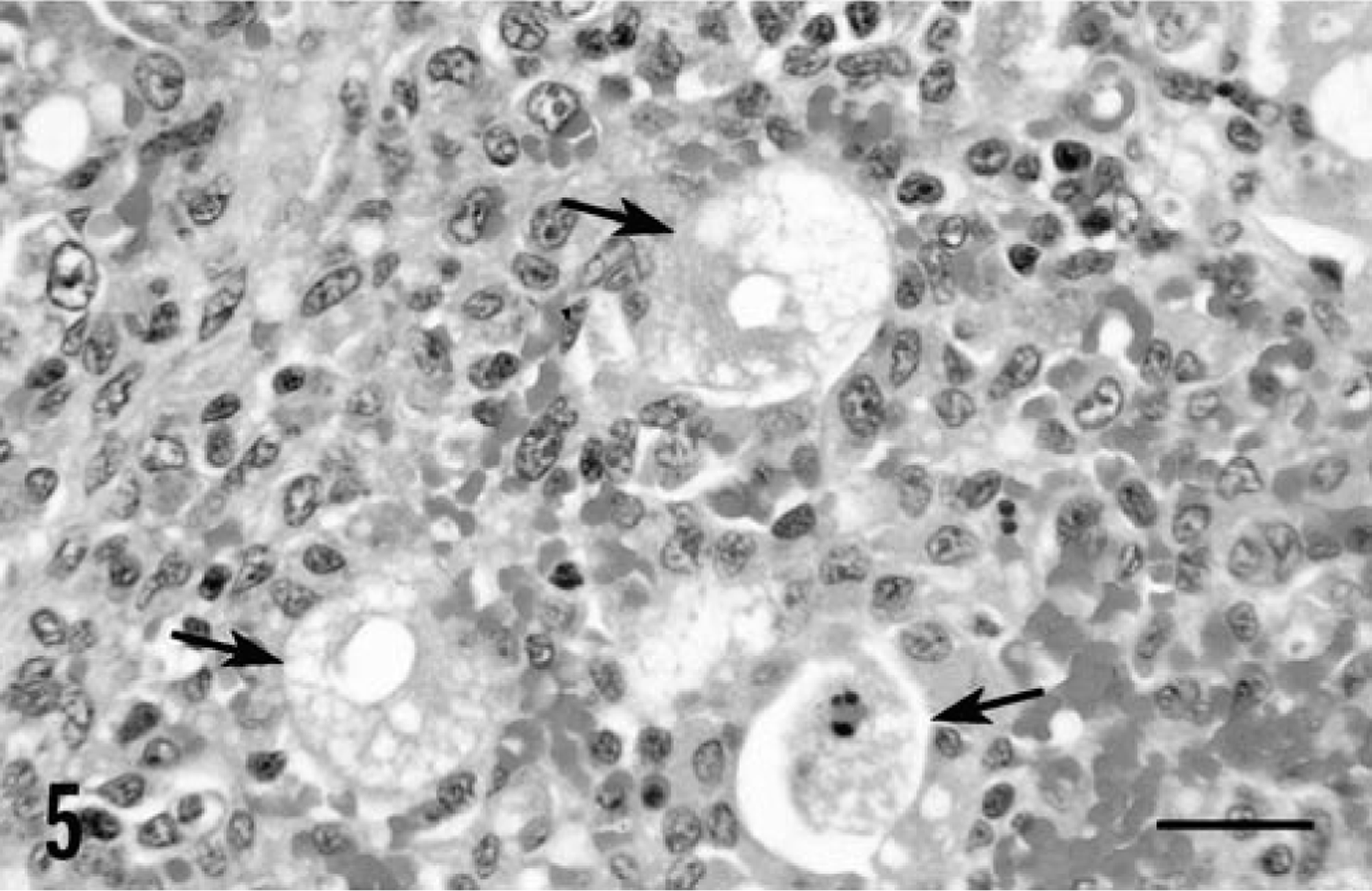
Liver; piglet inoculated with PCV-2/PPV 27 days previously. Note the necrotic hepatocytes (arrows) and the infiltration by histiocytes and macrophages. HE. Bar = 40 µm.
Multifocal angiocentric granulomatous inflammatory lesions were present in the kidneys. These lesions were most prominent at the corticomedullary junction. Degeneration and necrosis of adjacent renal parenchyma accompanied these lesions. Small multifocal interstitial granulomas were diffusely scattered throughout all lung lobes. Lymphocytic to granulomatous inflammatory cell infiltrates were identified in the lamina propria throughout the gut. Occasional gastric and duodenal mucosal ulcers accompanied these inflammatory cell infiltrates. Lymphoplasmacytic to granulomatous myocarditis was detected in all animals. Perivascular infiltrates of lymphocytes, plasma cells, and macrophages were identified in the brain of one piglet.
In the least severely affected piglet of the PCV-2/PPV infection group, moderate lymphocytic and plasmacytic cholangiohepatitis was observed. Mild angiocentric granulomatous inflammation was also present in the lung, liver, and myocardium. In the kidney, these vessel-oriented lesions were accompanied by occasional foci of thrombosis and perivascular fibrosis. Lymphoid and macrophage hyperplasia and germinal center formation were present in lymph nodes.
Immunocytochemistry for PCV
One or more tissues from all piglets infected with PCV-2, either alone or in combination with PPV, stained positively for PCV-2 antigen. Reactivity in piglets inoculated with PCV-2 alone or with PCV-1 was sparse and largely restricted to single cells or small foci of cells in lymph node germinal centers and within inflammatory cell infiltrates in other tissues. The tissues of piglets inoculated with PCV-2/PPV contained large amounts of PCV-2 antigen in macrophages. In lymph nodes, prominent accumulations of viral antigen were detected chiefly in follicular macrophages in collapsed germinal centers (Fig. 6). Residual lymphocytes in nodes were essentially PCV-2 negative. A similar distribution pattern of PCV-2 antigen was evident in the spleen, although many macrophages located between germinal centers were antigen negative. In the liver, virtually every macrophage in granulomatous inflammatory lesions was virus positive (Fig. 7). The amount of antigen in the liver far exceeded that suggested by the frequency of cytoplasmic inclusion bodies. Although viral antigen was occasionally seen in hepatocyte nuclei, virtually all (>95%) of the identifiable hepatocytes (Fig. 7) stained negatively for PCV-2 antigen; however, closely adjacent macrophages infiltrating hepatic parenchyma were strongly PCV-2 positive. In macrophages, antigen was primarily cytoplasmic, although intranuclear staining was occasionally observed. Endothelial cells within the liver rarely contained virus. In other parenchymal tissues, granulomas contained PCV-2 antigen.
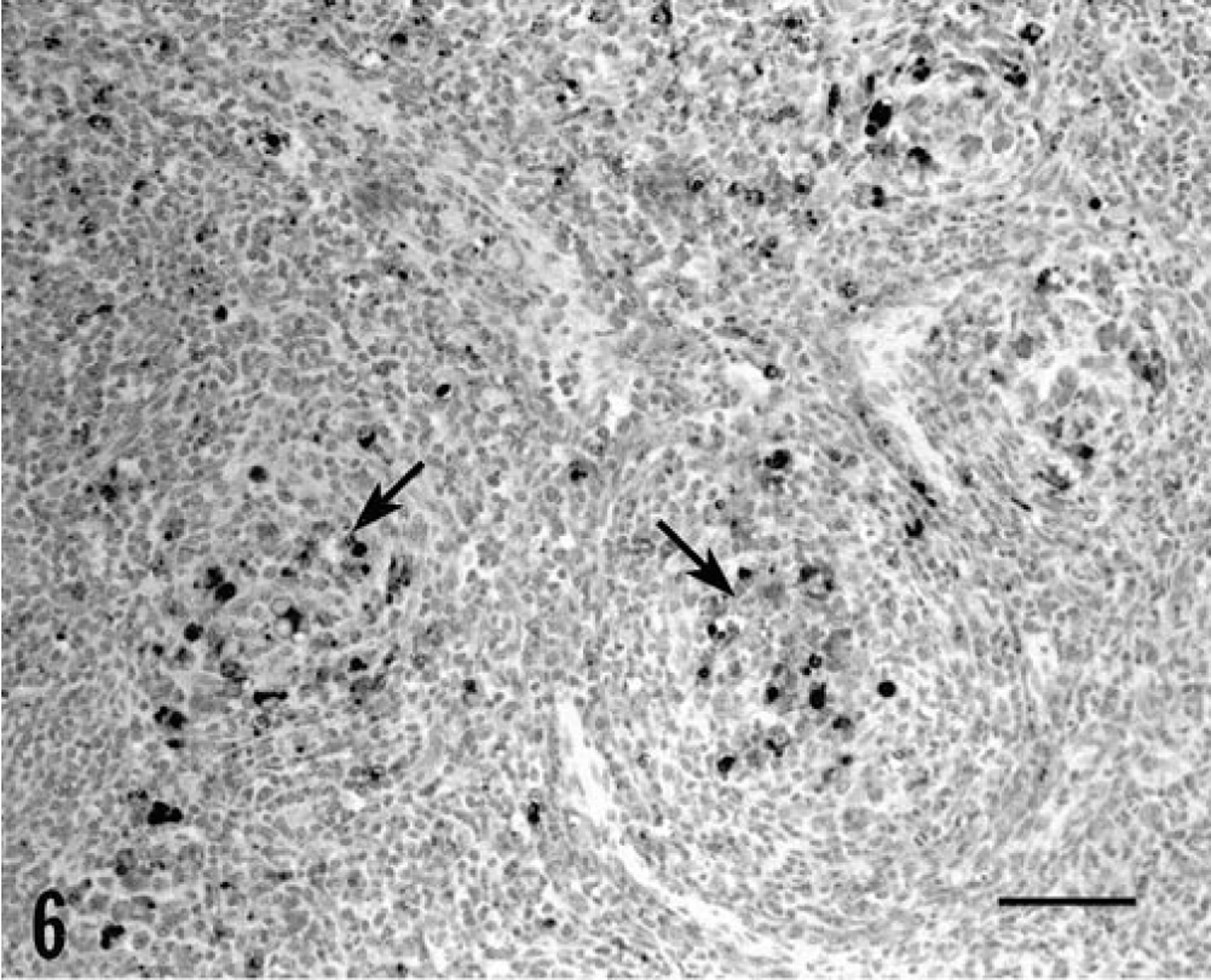
Lymph node; piglet inoculated with PCV-2/PPV 27 days previously. Note the presence of PCV-2 antigen–positive cells (arrows) within follicular macrophages located in the centers of collapsed germinal centers. Anti-PCV-2 monoclonal antibody stained with biotinylated equine anti-mouse IgG, reacted with avidin-peroxidase, developed with DAB, and counterstained with hematoxylin. Bar = 100 µm.
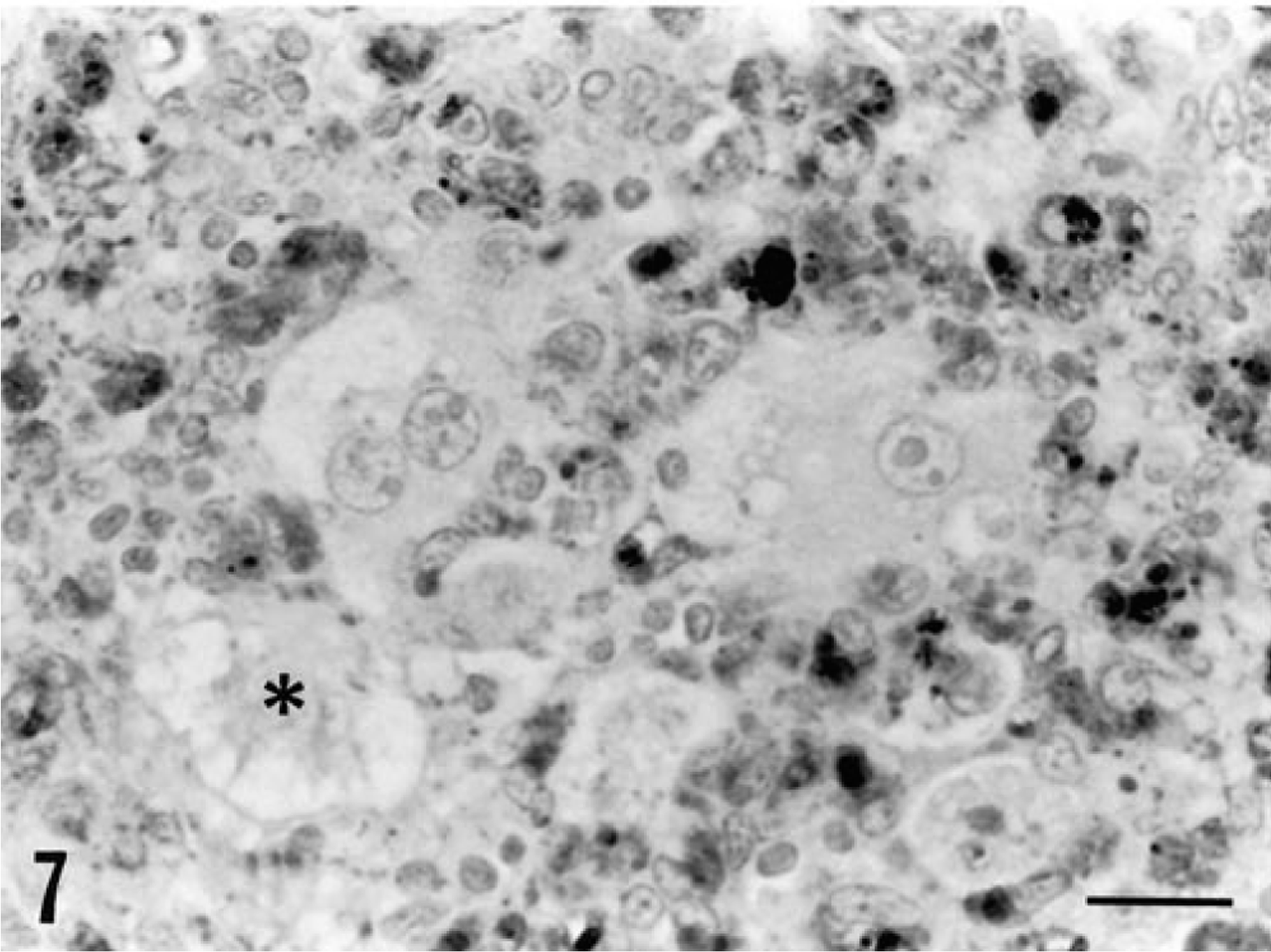
Liver; piglet inoculated with PCV-2/PPV 27 days previously. Note the distribution of viral antigen within infiltrating macrophages and histiocytes (DAB reaction product). Necrotic (∗) and degenerating hepatocytes do not contain viral antigen. Anti-PCV-2 monoclonal antibody stained with biotinylated equine anti-mouse IgG, reacted with avidin peroxidase, developed with DAB, and counterstained with hematoxylin. Bar = 25 µm.
Immunocytochemistry for PPV
Parvovirus antigen was not detected in the piglets given PPV alone or in combination with PCV-1. In contrast, modest numbers of PPV-positive cells were identified in piglets given PCV-2/PPV. PPV antigen was restricted to macrophages.
PCR for viremia and secretion of PCV-1, PCV-2, and PPV
Viremia was assessed by PCR using primers specific for each virus. Results are summarized in Table 2. Uninfected control piglets were negative for viral DNA throughout the study. With one exception, piglets given PPV, alone or in combination with another virus, never became viremic or were viremic only on PID 7. Five of ten piglets inoculated with PCV-1 were transiently viremic (PID 7 and PID 14). In piglets infected with PCV-2 alone, viral DNA was first detected in PBMC or whole blood in two of three animals on PID 14. Thereafter, viral DNA was detected in most PCV-2–infected piglets throughout the experiment. PCV-2 DNA was identified in dually infected (PCV-2/PPV) piglets earlier (1/4 on PID 7) and was present in all pigs from PID 14 onward. Although quantitation was not attempted, the signal intensity in the DNA from PCV-2/PPV–infected piglets was much stronger than in that of the PCV-2–infected group. Detection of viral antigen in PBMC cytospin preparations by immunochemistry was of limited value; only two of four piglets given PCV-2/PPV had detectible viral antigen.
Results ∗ obtained using PCR for viral DNA in porcine peripheral blood mononuclear cells and/or whole blood from gnotobiotic piglets infected with various combinations of PCV-1, PCV-2, and PPV.
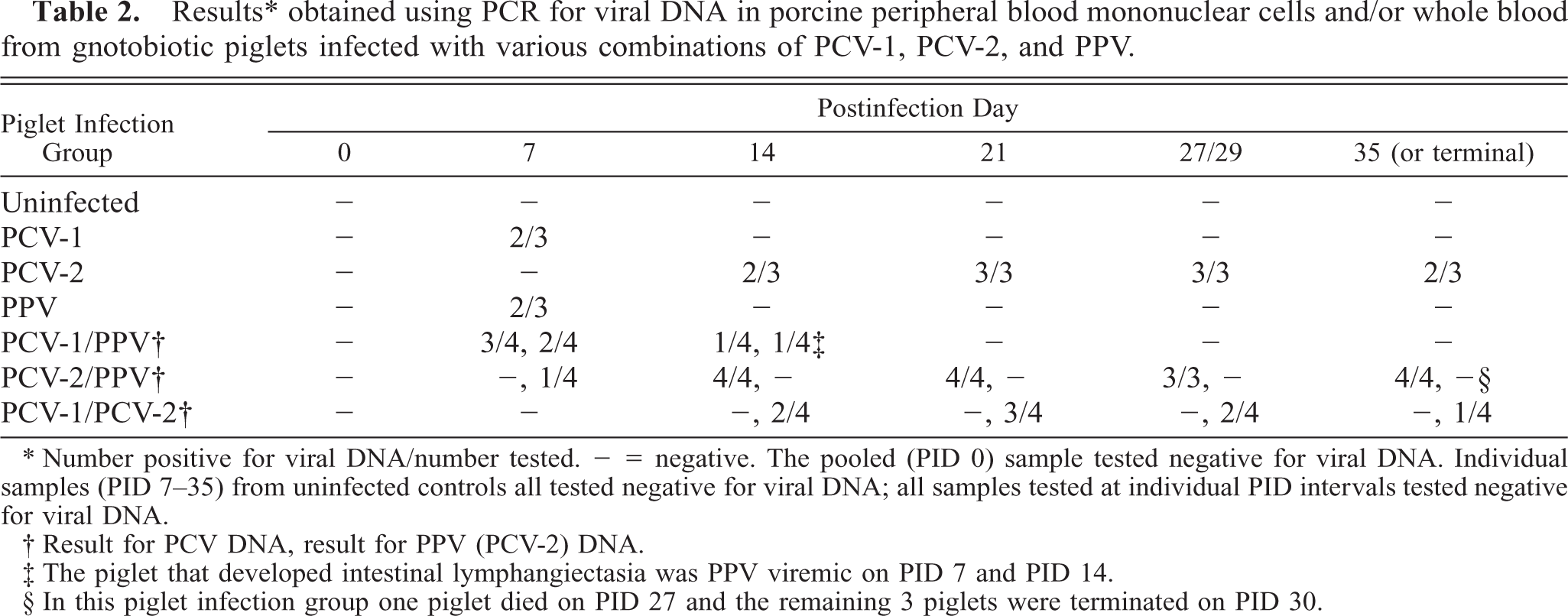
∗ Number positive for viral DNA/number tested. = negative. The pooled (PID 0) sample tested negative for viral DNA. Individual samples (PID 7–35) from uninfected controls all tested negative for viral DNA; all samples tested at individual PID intervals tested negative for viral DNA.
† Result for PCV DNA, result for PPV (PCV-2) DNA.
‡ The piglet that developed intestinal lymphangiectasia was PPV viremic on PID 7 and PID 14.
§ In this piglet infection group one piglet died on PID 27 and the remaining 3 piglets were terminated on PID 30.
Fecal, ocular and nasal swabs were collected at necropsy and assessed by PCR for viral DNA (Table 3). Viral DNA was not demonstrated in uninfected control piglets. PCV-2 DNA was recovered from secretions collected from piglets inoculated with PCV-2 alone or in combination with PPV. PPV DNA was present in the ocular and nasal secretions of one of three animals given PPV alone, all ocular/fecal and two of four nasal secretions of piglets inoculated with PCV-1 and PPV, and two of three nasal secretions of piglets inoculated with PCV-2 and PPV. Neither viral DNA nor viral antigen was detected in the two uninfected piglets housed together with the PCV-1/PCV-2–infected piglets.
Results ∗ obtained using PCR for viral DNA in various excretions and secretions collected at termination from gnotobiotic piglets infected with various combinations of PCV-1, PCV-2, and PPV.
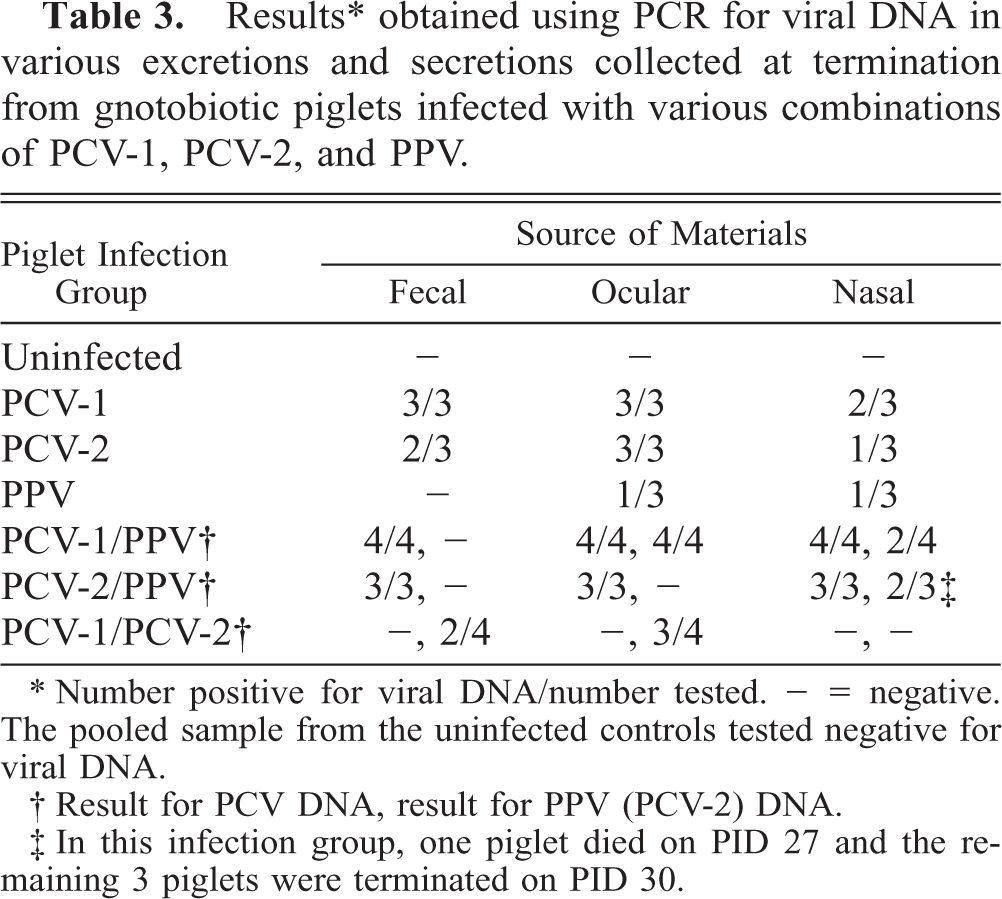
∗ Number positive for viral DNA/number tested. - = negative. The pooled sample from the uninfected controls tested negative for viral DNA.
† Result for PCV DNA, result for PPV (PCV-2) DNA.
‡ In this infection group, one piglet died on PID 27 and the remaining 3 piglets were terminated on PID 30.
Serology
Preinoculation sera from all piglets were negative for viral antibodies; uninfected control piglets remained seronegative to all three viral agents. All piglets given PPV promptly seroconverted to PPV, and HAI antibody was detected in convalescent sera from PID 7 onward. Similarly, piglets given PCV-1 or PCV-2 alone rapidly seroconverted to their homologous inocula only after infection. Group-specific antibodies, reactive with both PCV-1 and PCV-2, were detected in convalescent sera from most piglets inoculated with these two agents (3/4 seropositive to PCV-1, 4/4 seropositive for PCV-2). Because of the partial cross-reactivity between these two agents, no attempt was made to determine antibody titers to each circovirus. Three of four piglets given PCV-2/PPV had both PPV and PCV-2 antibodies in convalescent sera. The terminal sample from the piglet that died on PID 27 did not have detectible PCV-2 antibody but was PPV HAI positive. Terminal PBMC from this piglet stained strongly for PCV-2 antigen.
Discussion
PMWS has emerged as a new and potentially devastating infectious viral disease in major swine-producing areas of the world. The primary organ system affected (pulmonary, hepatic, or renal) and the mortality rate vary. The predominant clinical finding in affected piglets is a wasting syndrome. Even piglets identified as survivors fail to thrive, exhibit poor weight gains, and as a result represent economic losses to the producer. The bulk of available evidence both from naturally occurring disease and the experimental data generated here and elsewhere 2 , 5 , 6 , 7 , 10 , 11 , 21 , 25 indicate that PCV-2 is the primary infectious viral agent causing PMWS. The presence of viral inclusion bodies, viral protein, and viral DNA within the lesions of PMWS, the recovery of PCV-2 from affected animals, and the production of PMWS in conventional colostrum-deprived piglets with PCV-2 2 support this conclusion. In this study, we confirmed the results of previous transmission experiments 2 , 11 and have identified PCV-2 and not PCV-1 as a porcine viral pathogen. In gnotobiotic pigs, PCV-2 was essential but not sufficient by itself to produce PMWS.
PCV-1 or PCV-2, when inoculated into gnotobiotic piglets as single agents, caused no clinical disease and only modest histologic inflammatory lesions. Were it not for the fact that tissues from uninfected gnotobiotic control piglets were also available for comparison, the histologic changes associated with any of the single agent–infected pigs would likely be dismissed as nonspecific background changes. Unlike PCV-1–infected piglets, PCV-2–infected piglets had widespread but low level intracellular viral antigen at termination, and excretions and secretions from these piglets contained PCV-2 DNA. These data suggest that the infection in these animals was active, in spite of seroconversion. PMWS might eventually have developed in these animals; moderate PMWS has been produced in conventional colostrum-deprived piglets inoculated with PCV-2 alone. 2
The role of a second agent (PPV) in the genesis of severe PMWS in gnotobiotic pigs is problematic. PPV is not regarded as a significant cause of mortality in young swine, 8 , 31 although the virus has a tropism for lymphoid tissues 14 , 22 , 24 and causes reproductive failure and fetal deaths when piglets are infected in utero. Infection of neonatal swine with PPV is clinically asymptomatic and results in only minimal histologic evidence of infection in tissues; 2 , 8 these findings for PPV were replicated in this study. In contrast, in mid- and late-term PPV-infected porcine fetuses, the virus is widely distributed, especially in the liver, although viral-antigen–positive cells are not accompanied by inflammatory cell infiltrates. 22
Although PPV clearly potentiated PCV-2 disease in dually inoculated piglets, PPV was difficult to demonstrate in tissues at necropsy. The most reliable evidence for infection was PPV seroconversion after challenge. Moreover, it is clear from the PCR data that ocular and nasal secretions contained PPV DNA; thus, convalescent piglets may remain carriers after seroconversion and may serve as a source of virus transmission to other animals. There are precedents for the synergistic effects of other viruses in potentiating circovirus disease. Both Marek's disease 23 and infectious bursal disease 18 , 26 agents potentiate chicken anemia virus (CAV) infection in poultry. Severe anemia and full expression of disease occurs only in coinfected chickens. Both CAV and a related circovirus, psittacine beak and feather disease virus, replicate in myeloid precursor cells and are known to be immunosuppressive. 1 , 23 The experimental design employed in the present study did not elucidate the mechanism(s) of apparent synergy between PCV-2 and PPV. Synergy must be related to the virulence of PCV-2, because dual infection of swine with PCV-1 and PPV did not produce clinically significant disease. These data confirm previous reports of PCV-1 avirulence. 3 , 28 Thus, even though PCV-1 is widespread in swine populations, it is not the cause of PMWS.
Immunocytochemistry was used to determine the distribution of PCV-2 in infected tissues. The suspicion that PCV-2 exhibits preferential tropism for cells of the monocyte/macrophage lineage was confirmed by localization of viral antigen within this cell type and its derivatives. Other cell type(s) likely support virus replication in vivo; viral antigen has been demonstrated in lymphocytes, endothelia, and epithelial cells of the liver and kidney. 21 In PCV-2/PPV–infected piglet liver sections, rare hepatocytes stained positively from PCV-2. Liver failure as evidenced by icterus, edema, and widespread hepatocellular necrosis was the immediate cause of death in PCV-2/PPV–infected piglets. PPV also has an in vivo tropism for monocytes and lymphoblasts 14 , 22 and is hepatotropic in developing fetuses. 22 Both PPV and PCV-2 are nonenveloped DNA viruses and both are dependent upon host cell DNA synthesis for efficient viral replication. 31 Transient PPV infection of macrophages could promote high levels of replication of PCV-2 during the preclinical phase of dual infection by providing monocyte proliferation signals to this shared target cell. Alternatively, the mechanism of cooperation may be indirect, wherein transient PPV-associated immunosuppression promotes the spread of PCV-2 beyond this tissue compartment. Clearly, this intriguing and unique synergistic relationship must be explored further.
Demonstration of viral DNA in blood with the concurrent serum antibody response to PCV-2 suggest that PCV-2 infection may be persistent. Persistent DNA viremia after onset of specific antibody indicates that 1) developing humoral antibodies are ineffective in virus clearance for at least 7 weeks after infection and 2) direct detection of virus by immunochemistry and/or PCR in blood is a convenient method for diagnosis of PMWS. The implications of persistent virus as a constant or intermittent source of virus to other pigs and the environment are important to the producer in developing methods for control or eradication of this infection. Both PCV-2 and PPV are highly resistent to heating, freezing, ultraviolet light, and chemical disinfectants, suggesting that unusually stringent methods for handling contaminated premises must be a part of any control program for PMWS.
The experiments reported here complement previous transmission studies. 2 , 11 PCV-2 is essential but not sufficient by itself to cause PMWS in gnotobiotic pigs. The mechanism(s) that accounts for the synergistic relationship between PCV-2 and PPV infections and clinical expression of PMWS under gnotobiotic conditions remains elusive. It is not known if other pathogens of swine such as porcine respiratory and reproductive syndrome virus 22 , 27 are also involved in the wasting disease complex.
Footnotes
Acknowledgements
Portions of this research were supported by a research grant from The National Pork Producers Council, Ames, IA; Merial SAS, Lyon, France; and USDA formula funds, the Ohio State University. The excellent technical assistance of S. S. Ringler, J. Younger, K. Harrick, and S. Zanick is appreciated.
