Abstract
This report describes a unique distribution of cerebral cortical necrotic lesion, which was diagnosed as hepatic encephalopathy in a 2-year-old Maltese dog. The dog showed splenocaval shunt and small liver with marked hepatocellular fatty degeneration. Histopathologic examination revealed that diffuse laminar cortical necrosis composed of neuronal necrosis, marked infiltration of gitter macrophages, and astrogliosis were found bilaterally in the dorsolateral area of the cerebrum. No necrotic lesions were observed in the cerebral paleopallium and archipallium, the central gray matter, cerebellum, and brain stem. Astrocytes with large and pale nuclei (Alzheimer type II astrocytes) were apparent throughout the brain. Immunohistochemically, a decrease of immunostains for glutamine synthetase and glutamate transporter antibodies was seen in Alzheimer type II astrocytes and neuropil. This is, to our knowledge, the first report of extensive involvement of cerebral neopallidum in canine hepatic encephalopathy.
Hepatic encephalopathy refers to a complex neuropsychiatric syndrome with hyperammonemia resulting from severe liver failure and disturbance of ammonia metabolism such as intrahepatic or extrahepatic shunt in humans and animals. 5,17 Ammonia in the central nervous system is metabolized by astrocytes to glutamine, and evidence for a direct toxic effect of ammonia on astrocytes has been reported. 6,14 Alzheimer type II cell, which is morphologically characterized by large and pale nucleus, is the histopathologic hallmark of hepatic encephalopathy. 5,17 A disturbance in astrocytic function may lead to neuronal degeneration and necrosis because astrocytes have been considered to play an important role in the blood-brain barrier or neuronal metabolism. 11 In humans, it has been previously reported that in some cases degeneration and loss of neurons are associated with pronounced microcavitation in the cerebral cortex and basal ganglia (mainly in the putamen), 5 but, to our knowledge, none as severe as in animals. Furthermore, immunohistochemical studies on metabolic regulations of neurotransmitter glutamate (Glu) have demonstrated that glutamine synthetase (GS) and glutamate transporter (GLT-1) immunolabelings in astrocytes decrease in experiments of hepatic encephalopathy. 1,7 This report describes severe necrotic lesions in the cerebral neopallidum with preservation of other areas of the brain and discusses the vulnerability of the brain in hyperammonemia on the basis of distribution of lesions. In addition, immunohistochemistry using GS and GLT-1 antibodies was carried out to examine the condition of astrocytic function relating to Glu metabolism.
A 1.5-year-old Maltese dog was presented to a veterinary clinic for anorexia and weight loss of several months duration, and then a nutritious diet was given. The dog showed sudden seizure after nystagmus and circling 6 months later. The results of routine blood biochemistry and hematologic examination revealed high values of alanine aminotransferase (362 U/liter; reference range, 21–102 U/liter), ammonia (136 µg/dl; reference range, 20–110 µg/dl) and total biliary acid (60.2 µmol/liter; reference range, < 16 µmol/liter) and mildly low values of serum protein (5.0 g/dl; reference range, 6–7.3 g/dl). Mild regenerative anemia (packed cell volume 29.4%; reference range, 37–55%) was documented. The value of blood glucose was 74 mg/dl (reference range: 65–120 mg/dl) Radiographs and ultrasound revealed microhepatia. Seizure signs were not observed 1 day later. At 2 years of age, the dog developed anemia (packed cell volume 21.0%) and neurologic signs, recumbency, decreased consciousness, and coma, and died despite the guarded treatment. The value of ammonia, which was from the final hospital admission of the dog, was 162 µg/dl.
Necropsy findings included small liver with marked fatty change and a clear and yellow ascitic fluid (5 ml). A splenic vein connected the caudal vena cava directly, which was mildly enlarged (Fig. 1). The cerebrum showed laminar cortical necrosis, which often represents softening and microcavity formation in the dorsolateral area at the level of the caudate head to lateral geniculate nucleus (Fig. 2). Bilateral ventricles were moderately dilated. No gross findings were found in other areas of the brain.
Spleno-caval shunt; dog. A splenic vein (black arrows) connected directly the caudal vena cava (white arrow). The vena cava was mildly enlarged.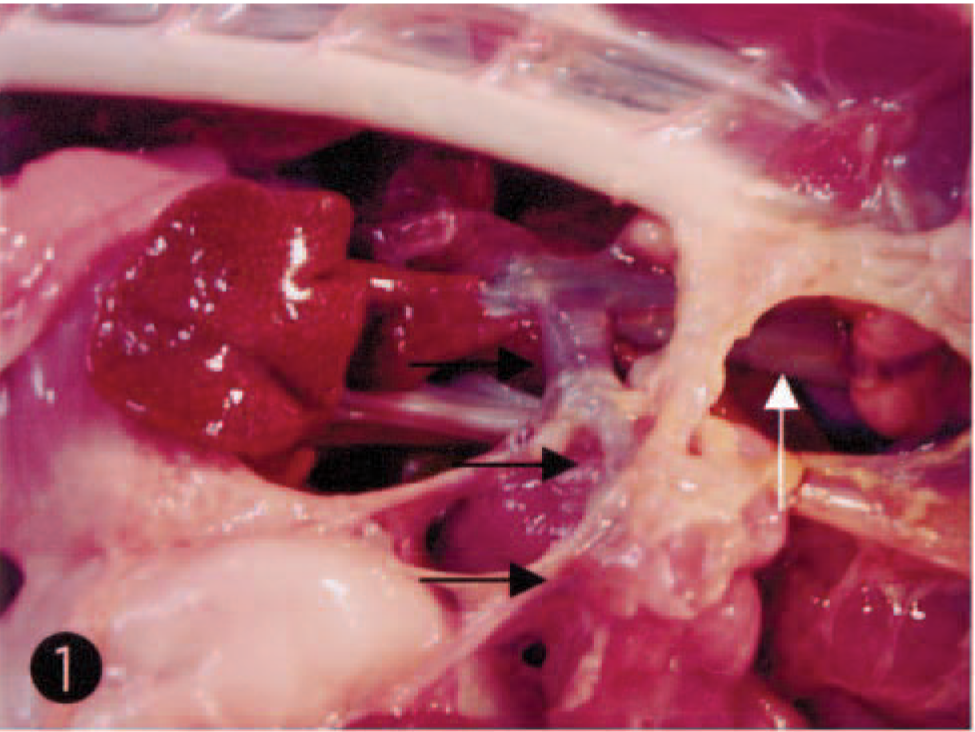
Cerebrum; dog. Note bilaterally cortical atrophy with whitish discoloration and microcavites formation (arrowheads) in the dorso-lateral area. Bilateral ventricles are moderately dilated.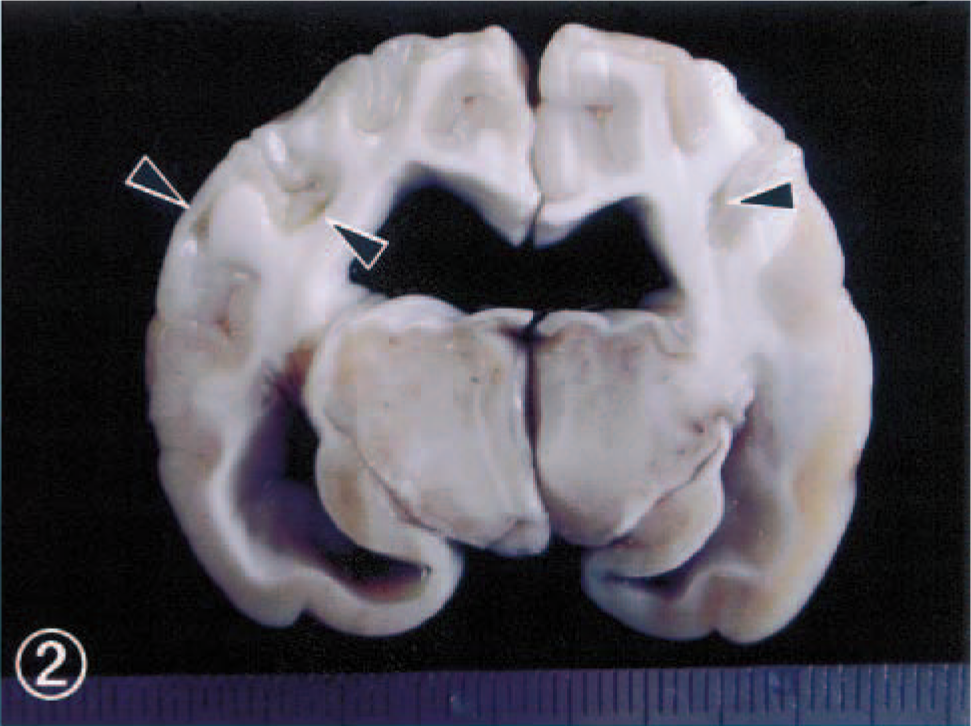
Samples of systemic organs, including the brain, were collected and fixed by immersion in 10% neutral-buffered formalin. They were then dehydrated, embedded in paraffin wax, sectioned at 4 µm, and stained with hematoxylin and eosin (HE). Selected brain sections were stained with the Klüver-Barrera and Bodian stains. In addition, selected brain sections were examined immunohistochemically with a streptavidin-biotin kit (DAKO, Glostrup, Denmark) and the following specific antisera: anti-human glial fibrillary acid protein (GFAP) (DAKO), anti-ovine GS (Chemicon, Temecula, CA), and anti-rat GLT-1 (Wako, Richmond, VA). Paraffin wax–embedded brain tissues of four normal Shetland sheepdogs and two mixed breed dogs, which were consistent with the dog in this study in postmortem interval and fixation, were used as controls.
On histopathologic examination of the brain, necrotic lesions were found to be confined to the cerebrum, where weak staining of HE, Klüver-Barrera, and Bodian stains was observed bilaterally in the dorsolateral area of the cerebral cortex and subcortical white matter at the level of the frontal tip to occipital pole. The lesions comprised laminar neuronal loss and necrosis, marked infiltration of gitter macrophages, and astrogliosis (Fig. 3). In the adjacent white matter in this area, there was demyelination, axonal loss, and marked astrogliosis. Cingulate and insular cortices were mildly affected. No necrotic lesions were seen in other areas of the brain, including cerebral paleopallium (olfactory bulbs and peduncles and pyriform lobe cortex) and archipallium (hippocampus and fornix), the central gray matter, cerebellum, and brain stem. Alzheimer type II astrocytes were frequently observed in the cerebral cortex (Fig. 4) and central gray matter, including putamen and globus pallidus. There was no thrombi formation in the lumens of the middle cerebral artery and its branches. Few perivascular cuffs of lymphocytes were seen. Immunohistochemically, GFAP-positive reactive astrocytes were diffusely detected in or around the necrotic lesions and their subcortical white matter, but Alzheimer type II astrocytes were frequently negative for GFAP immunostains. In six control dogs, fine granular immunostains for both GLT-1 and GS were diffusely observed with no differences in the intensity for their immunostains in the gray matters (cerebral cortex and central gray matter), whereas few immunopositive stains were seen in the white matter. GS immunohistochemistry demonstrated fine granular immunostains in neuropil of control brains (Fig. 5), whereas a weak or decrease in positive reactions was found in Alzheimer type II astrocytes and neuropil of nonnecrotic cortex adjacent to necrotic lesions (Fig. 6). As compared with control brain (Fig. 7), a decrease of immunolabeling by GLT-1 antibody was exclusively detected in the neuropil of this nonnecrotic cortex (Fig. 8). Both GS and GLT-1 immunostains reduced prominently in necrotic foci of the cerebral neopallidum. In the liver, severe fatty degeneration of hepatocytes was diffusely observed.
Cerebral cortex in the dorsal area at the level of the caudate head; dog. Note neuronal loss, marked infiltration of gitter macrophages and astrogliosis. HE. Bar = 43µm.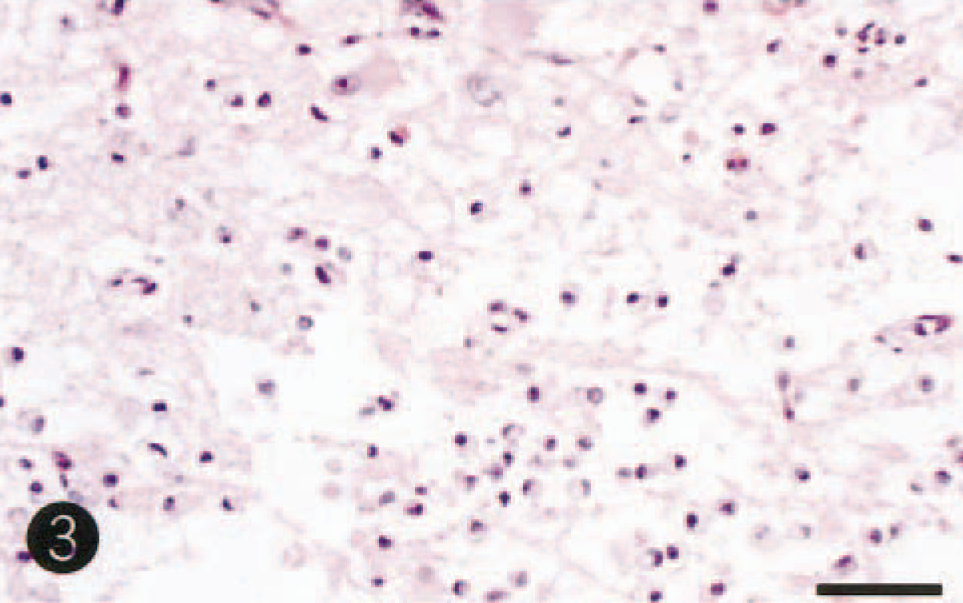
Cingulate cortex; dog. Alzheimer type II astrocytes with large and pale nuclei were observed in the nonnecrotic area adjacent to the necrotic lesion. HE. Bar = 45µm.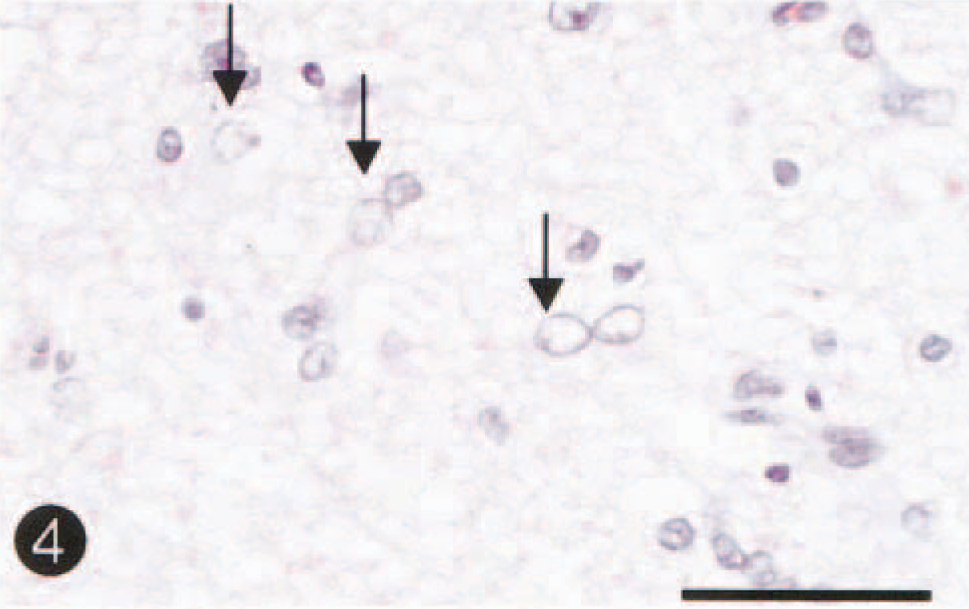
Cingulate cortex; control dog. Note fine-granule immunostains for GS antibody in neurophil. Astrocytic processes are also positive for GS antibody (arrows). Labeled streptavidin biotin, hematoxylin counterstain. Bar = 35µm.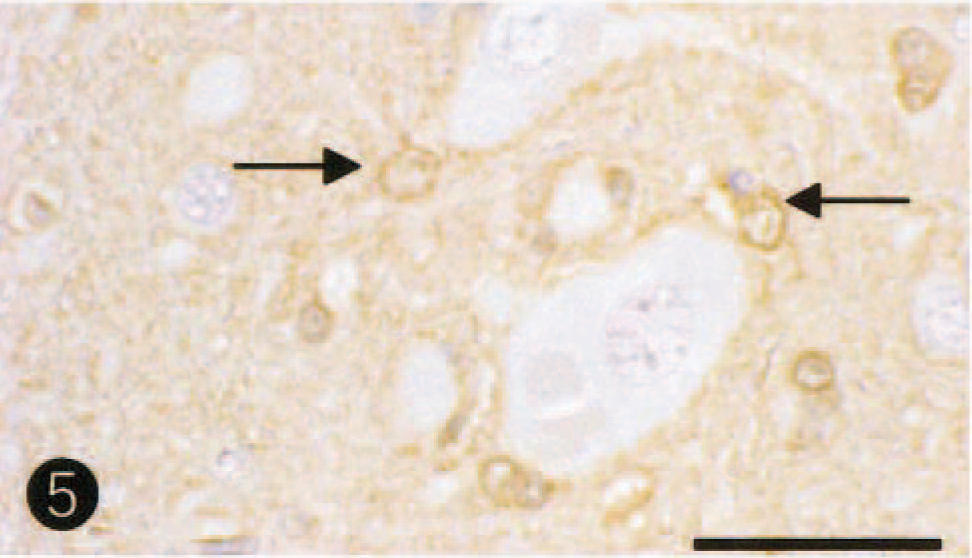
Cingulate cortex; dog. Note a decrease of immunostains for GS antibody in astrocytic (Alzheimer type II cells) processes (arrows) and neurophil. Labeled streptavidin biotin, hematoxylin counterstain. Bar = 45µm.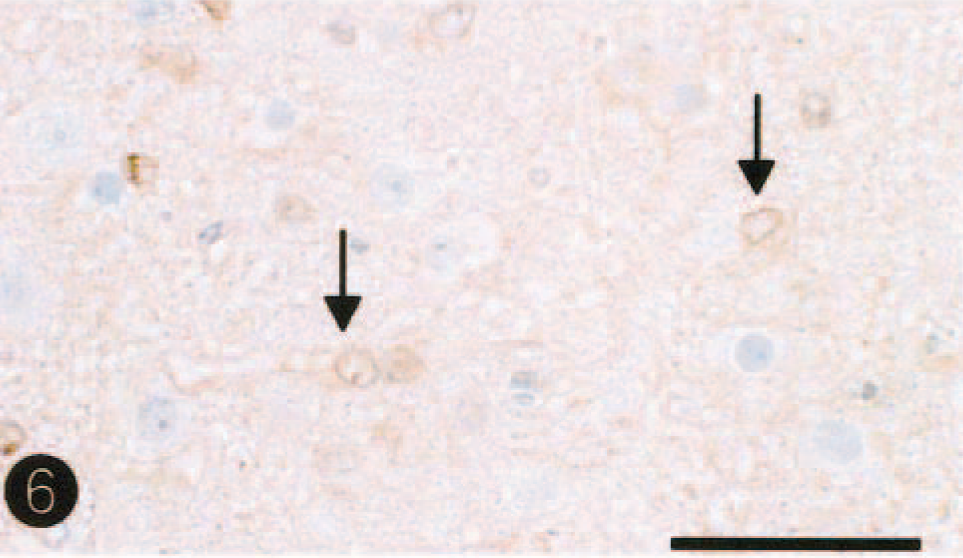
Cingulate cortex; control dog. Note fine-granule immunostains for GLT-1 antibody in neurophil. Labeled streptavidin biotin, hematoxylin counterstain. Bar = 45µm.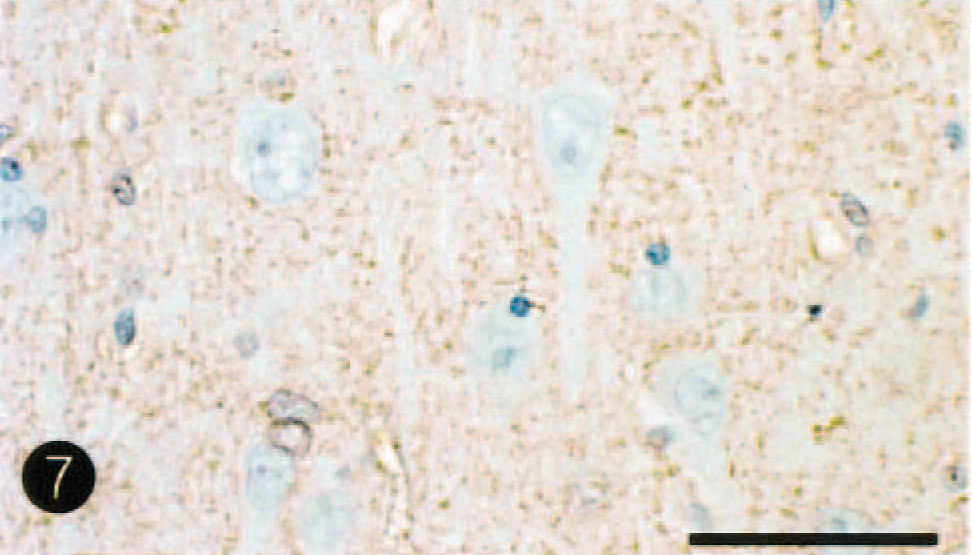
Cingulate cortex; dog. Note a decrease of GLT-1 immunostains in astrocytic cytoplasm and neurophil. Labeled streptavidin biotin, hematoxylin counterstain. Bar = 45µm.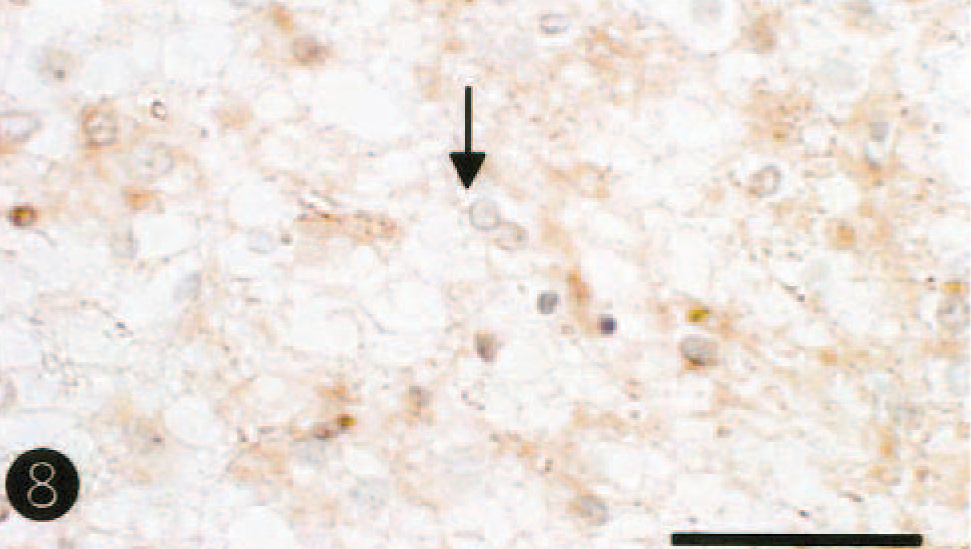
Clinical and pathologic findings of the dog in this study can be summarized as follows: 1) the dog presented neurologic signs and hyperammonia, which had been ongoing for at least 6 months, 2) the dog had a splenocaval shunt and small liver with marked fatty degeneration, 3) Alzheimer type II astrocytes were frequently demonstrated in the cerebrum. On the basis of these findings, this dog was diagnosed as hepatic encephalopathy with extrahepatic shunt. Splenocaval shunt has been reported in humans, 10,18 and a human case had no accompanying advanced liver disease or episodes of hepatic encephalopathy. 10 In this dog, it was considered possible that hyperammonia was evoked by both splenocaval shunt and severe hepatocellular fatty degeneration. Causes of splenocaval shunt and hepatocellular change remain to be determined.
The pathogenesis of hepatic encephalopathy has not been completely understood; however, this syndrome has been recently interpreted as an ammonia-induced gliopathy followed by neuronal necrosis. 6,12,13,16 A weak or decreased GFAP, GLT-1, and GS immunolabeling in Alzheimer type II astrocytes and neurophil was apparent in this dog. Similar immunohistochemical stains have been reported in experimental cases. 1,7 GS is a ubiquitous enzyme that catalyzes the adenosine triphosphate–dependent conversion of Glu to glutamine using ammonia as the nitrogen source, 9,16 and therefore, it plays a central role in the detoxification of brain ammonia and in the metabolic regulation of Glu. In addition, GLT-1 maintains low resting levels of extracellular Glu, thereby preventing Glu-mediated neurotoxicty. 2,3 The findings of low expression of GS and GLT-1 in this dog may reflect astroglial dysfunction of Glu metabolism, leading to high levels of Glu in synaptic cleft and thereby to neuronal necrosis because of its neurotoxicity. 2,3
The differential diagnosis for the dog in this study is necrotizing meningoencephalitis, which has been reported in young small breed dogs including Maltese dogs, because the lesions of necrotizing meningoencephalitis are associated with severe necrosis in the brain parenchyma. 15 In necrotizing meningoencephalitis of Maltese dogs, there was extensive necrosis and nonsuppurative inflammation (perivascular cuffs of lymphocytes) of the cerebral gray matter and white matter, overlying meninges, and adjacent thalamus and hippocampus. The dog with a long clinical history (20 weeks) showed extensive atrophy and astrocytic sclerosis, but perivascular cuffs of lymphocytes were also present in these sclerotic areas. In the dog in this study, there was little perivascular lymphocytic infiltration in the cerebrum. Furthermore, in the present study, cerebral lesions were distributed bilaterally, which have not been characterized by ‘necrotizing meningoencephalitis of Maltese dogs’. 15 Therefore, the dog in this study could not be diagnosed as necrotizing meningoencephalitis. Because this dog had a history of one seizure, there may be a possibility that the cortical lesion was caused by seizure-related changes. However, in general, predilection sites of necrotic lesions associated with seizure have been reported to be hippocampus, cingulated gyrus, amygdala, dorsomedaial nucleus of thalamus, and pyriform lobe. 17 This dog showed no neuronal necrotic changes in these areas.
Of interest in this dog is that there is severe involvement (complete necrosis) in the dorsolateral cortex of the cerebrum, which is anatomically categorized as neopallidum, 4 whereas cerebral paleopallium and archipallium as well as central gray matter were well preserved. There were no findings suggesting circulatory disturbances such as vascular thrombosis and hemorrhage in this dog. Furthermore, neuronal necrosis was not apparent in the piriform gyrus or hippocampus, which is considered to be secondary change due to seizure. 17 In humans with hepatic encephalopathy, it has been described that a variable degree of neuronal loss is observed in the basal ganglia, together with microcavitation (mainly in the putamen) when cortical cavitation is present. 5,8 However, the predilection of necrotic changes in the cerebral neopallidum has not been reported. This is the first case report suggesting that cerebral neopallidum may be most vulnerable site in hyperammonia. Although Alzheimer type II astrocytes were frequently present in the cerebral paleopallium and archipallium as well as central gray matter of this dog, there were few evidences for neuronal necrotic changes in those areas. The differences of pathology may reflect the degree of astroglial dysfunction between neopallidum and others as a result of hyperammonia.
