Abstract
The DNA mismatch repair (MMR) system preserves genomic stability by identifying and repairing mismatched nucleotides in the DNA replication process. The dysfunction of the MMR system, also known as mismatch repair deficiency (dMMR), is implicated as a predictive biomarker for the efficacy of immune checkpoint blockade therapy regardless of the tumor type in humans. This study aimed to evaluate the immunolabeling of MMR proteins in canine tumors and to identify the types of tumors having dMMR. First, we performed immunohistochemistry in 8 different canine tumors (oral malignant melanoma, high-to-intermediate grade lymphoma, mast cell tumor, malignant mammary gland tumor, urothelial carcinoma, hepatocellular carcinoma, osteosarcoma, and hemangiosarcoma) with 15 samples each to analyze the immunolabeling of canine mismatch repair proteins (MSH2, MSH6, and MLH1) using anti-human monoclonal antibodies. We found that more than half of canine oral malignant melanoma (60%) and hepatocellular carcinoma (53%) samples and fewer of the other canine tumors had loss of immunolabeling in ≥1 MMR protein (ie, evidence of defective MMR proteins, based on the definition of dMMR in the humans). Antibodies against human MSH2, MSH6, and MLH1 were cross-reactive with the corresponding canine protein as confirmed using MMR gene knockout canine cell lines. Further studies are required to investigate the clinical outcomes in canine spontaneous tumors with dMMR to determine the potential for immune checkpoint blockade therapy for these tumor types.
Keywords
The DNA mismatch repair (MMR) system plays key roles in preserving genomic stability by identifying and repairing mismatched nucleotides in the DNA replication process. The MMR system is regulated mainly by 4 genes: mutS homologue 2 (MSH2), mutS homologue 6 (MSH6), mutL homologue 1 (MLH1), and postmeiotic segregation increased 2 (PMS2). These genes were first found as causative germline mutation in patients with Lynch syndrome in 1993. 3,8,16 Lynch syndrome is a hereditary disease and patients with this disease are prone to tumors, particularly colorectal cancer and endometrial cancer. In addition to Lynch syndrome, defects of the MMR system are also caused by somatic mutation or hypermethylation of promoter sequences and has been found in various sporadic tumors. 19,20 It is now known that MMR deficiency (dMMR) induces microsatellite instability and increases mutation rates, leading to tumor development. 5,6,25
In humans, immunohistochemical analysis for MSH2, MSH6, MLH1, and PMS2 proteins are used for detection of dMMR. 24 Several reports have shown that tumors with dMMR and/or microsatellite instability-high are sensitive to immune checkpoint blockade therapy, particularly PD-1 inhibitor therapy. 13,26,27 For instance, Le et al showed that the objective response rate in MMR-deficient colorectal cancer or MMR-proficient colorectal cancer were 40% and 0%, respectively. 15 Therefore, dMMR is implicated as a predictive biomarker for efficacy of immune checkpoint blockade therapy regardless of tumor type.
In dogs, accumulation of mutations may be associated with development of tumors. However, there are few reports about microsatellite instability and dMMR in canine cancers. A previous study confirmed using immunohistochemistry (IHC) that MLH1, MSH2, and MSH6 proteins were expressed in 44 mast cell tumor samples. 21 In a case report on 2 dogs with colon adenocarcinoma, nuclei of tumor cells in both dogs were positively labeled with IHC for MLH1 protein. 18 However, there are no reports evaluating the defects of the above-mentioned 4 MMR proteins in other types of canine tumors. In addition, 2 recent clinical studies of immune checkpoint blockade therapies using PD-1 or PD-L1 inhibitors were reported for spontaneous tumors in dogs. 10,17 In these studies, tumor response rates were approximately 20%, although the number of cases were limited. Thus, searching for predictive markers for treatment response is essential for the effective use of immune checkpoint blockade therapies. Establishment of methods to analyze MMR proteins expression and identification of tumors with dMMR in canine cancer are important for promoting immune checkpoint blockade therapies in the future.
This study aimed to evaluate the immunolabeling for MMR proteins in canine tumors and to identify the types of tumors having dMMR. Also, cross-reactivity of antibodies against human MMR proteins to canine MMR proteins was confirmed with MMR gene knockout cell lines.
Materials and Methods
Cell Culture
Human embryonic kidney (HEK) 293T cell line was cultured in Dulbecco’s modified Eagle medium (DMEM)-based complete medium (DMEM mixed with 10% fetal bovine serum [FBS], 100 units/ml penicillin-streptomycin, and 55 µM 2-mercaptoethanol). CIP-p (canine mammary gland tumor) cell line, 22 kindly provided by Dr Takayuki Nakagawa (The University of Tokyo), was cultured in RPMI1640-based complete medium (RPMI1640 mixed with 10% FBS, 100 units/ml penicillin-streptomycin, and 55 µM 2-mercaptoethanol). All cell lines were maintained at 37 °C in a humidified CO2 gas incubator.
Lentiviral Transduction of Knockout Vectors
According to our previous reports, 11 CRISPR-Cas9 small guide RNAs (sgRNA), which targets MMR genes (mlh1, msh2, and msh6), were constructed with online CRISPR design tool (http://zlab.bio/guide-design-resources). The sequences of sgRNAs of canine MLH1, canine MSH2, and canine MSH6 were GTGGTGAACCGCATCGCGGC, GCGCTTCTTCCAGGCCATGC, and GAAGTAAGGCCTAAGGTCCA, respectively. These oligonucleotides were synthesized and inserted separately into lentiCRISPR v2 plasmid, which was donated by Dr Feng Zhang (Addgene, Cambridge, MA, #52961; http://n2t.net/addgene:52961; RRID: Addgene 52961). To produce lentivirus-expressing sgRNAs that target each gene, HEK293T cells were seeded at 3.0 × 105 cells/well onto a 6-well plate a day before the transfection. On the day of transfection, 1.25 µg of each plasmid was mixed with PEI Max (Polysciences Inc, Warrington, PA) in OPTI-MEM buffer (Thermo Fisher Scientific KK), incubated for 15 minutes at room temperature, and added onto HEK293T cells. Twenty-four hours after transfection, supernatant was replaced with RPMI1640-based complete medium without antibiotics. After another 24 hours, the supernatant containing lentiviruses was collected and transferred onto CIP-p cells. Twelve hours after infection, the supernatant was replaced with RPMI1640-based complete medium, followed by addition of 7.5 µg/ml of puromycin to select stably transduced cells. Individual single clones were isolated by limited dilution method and further analyzed by western blotting to confirm the knockout of each protein.
Western Blotting Analysis
Each cell line was collected and lysed with NP-40 lysis buffer. Equal amounts of protein samples (30 µg) were electrophoresed in 7% acrylamide gel before transferring onto membranes. The membranes were incubated with each primary antibody (the same antibodies as used in IHC) diluted at 1:1000 in 0.5% skimmed milk in TBS-T overnight at 4 °C. Horseradish peroxidase (HRP)-conjugated anti-mouse IgG antibody (Merck Millipore) or HRP-conjugated anti-rabbit IgG antibody (Jackson ImmunoResearch) was used as a secondary antibody. The membranes were soaked in Western Lightning Plus-ECL (PerkinElmer) to visualize the immunoreaction. Immunoreactive bands were quantified and analyzed with AMERSHAM Image Quant 800 (Global Life Sciences Technologies Japan). The membranes were reprobed with mouse monoclonal anti-beta actin antibody (Sigma-Aldrich Japan KK) to verify equal loading of proteins in each lane.
Immunohistochemistry (IHC)
Formalin-fixed and paraffin-embedded (FFPE) canine tumor samples and testis tissue sample from a healthy dog were collected from diagnostic pathology laboratories (Yamaguchi University, Veterinary Pathology Diagnostic Center, Fukuoka, Japan; and IDEXX Laboratories, Tokyo, Japan). All samples were diagnosed by the veterinary pathologists in each laboratory. Fifteen samples were used for each tumor type (oral malignant melanoma, lymphoma, mast cell tumor, malignant mammary gland tumor, urothelial carcinoma, hepatocellular carcinoma, osteosarcoma, and hemangiosarcoma).
Sections were cut at 5 µm thickness and mounted on charged MAS-coated glass slides (Matsunami Glass). After deparaffinization and rehydration, antigen retrieval was performed in Dako Target Retrieval Solution pH9 (Agilent Technologies Japan, Ltd). The sections were incubated in 3% hydrogen peroxide in PBS to block endogenous peroxidase activity. To block nonspecific protein activity, the sections were incubated in 5% skimmed milk and 5% bovine serum albumin in PBS. Next, the sections were incubated with mouse monoclonal anti-human MLH1 antibody (G168-15, GeneTex, Inc; 1:50 dilution), rabbit monoclonal anti-human MSH2 antibody (SP46, Abcam; 1:100 dilution), mouse monoclonal anti-human PMS2 antibody (163C1251, Novus Biologicals, LLC; 1:250 dilution), mouse monoclonal anti-human PMS2 antibody (1G4E6, Proteintech Group, Inc; 1:200 dilution), or mouse monoclonal anti-human MSH6 antibody (44/MSH6, BD Transduction Laboratories; 1:250 dilution). Isotype-matched antibodies, anti-rabbit IgG antibody (DA1E, Cell Signaling Technology, Inc), and anti-mouse IgG1 antibody (P3.6.2.8.1, eBioscience, Inc) were used as negative immunolabeling control. The sections were incubated with Histofine Simple Stain mouse MAX PO (Nichirei Bioscience Inc) or Histofine Simple Stain rabbit MAX PO (Nichirei Bioscience Inc). Peroxidase Stain DAB Kit (Nacalai Tesque, Inc) was used to visualize the immunoreaction. The sections were counterstained with Mayer’s hematoxylin, dehydrated in alcohol, cleared in xylene, and mounted with xylene-based mounting solution. To bleach melanin granules, OMM samples were incubated in 10% hydrogen peroxide in PBS for 10 minutes at 65 °C before the antigen retrieval. After incubation with the secondary antibody, Simple Stain AEC Solution (Nichirei Bioscience Inc) was used to visualize the immunoreaction instead of DAB. Finally, after counterstaining with Mayer’s hematoxylin, these were mounted with water-soluble mounting solution. Immunoreactivity was considered positive if cell nuclei in the tumor cells were immunolabeled with each antibody as compared with the isotype control. We simultaneously evaluated the immunolabeling of the normal tissue area of each sample as a positive control. Immunolabeling was considered negative when the nuclei of tumor cells showed negative immunolabeling. Immunoreactivity in each sample was independently evaluated by 4 observers and dMMR was defined as a loss of immunolabeling in ≥1 MMR protein, according to the criteria used for humans. 24
Results
Immunohistochemistry of MMR Proteins in Various Canine Tumor Samples
First, we used a normal canine testis tissue to confirm the immunolabeling of each antibody. Both the anti-human PMS2 antibodies showed negative immunolabeling. Due to this result, the expression of 3 MMR proteins—MLH1, MSH2, and MSH6—were analyzed in canine tumors in this study.
Immunohistochemistry was performed on a total of 120 FFPE samples of 8 different types of canine tumors (15 samples of each tumor; Table 1).
Evidence of Mismatch Repair Deficiency (dMMR) in 8 Types of Canine Tumors. Fifteen Samples of Each Tumor Type Were Analyzed.
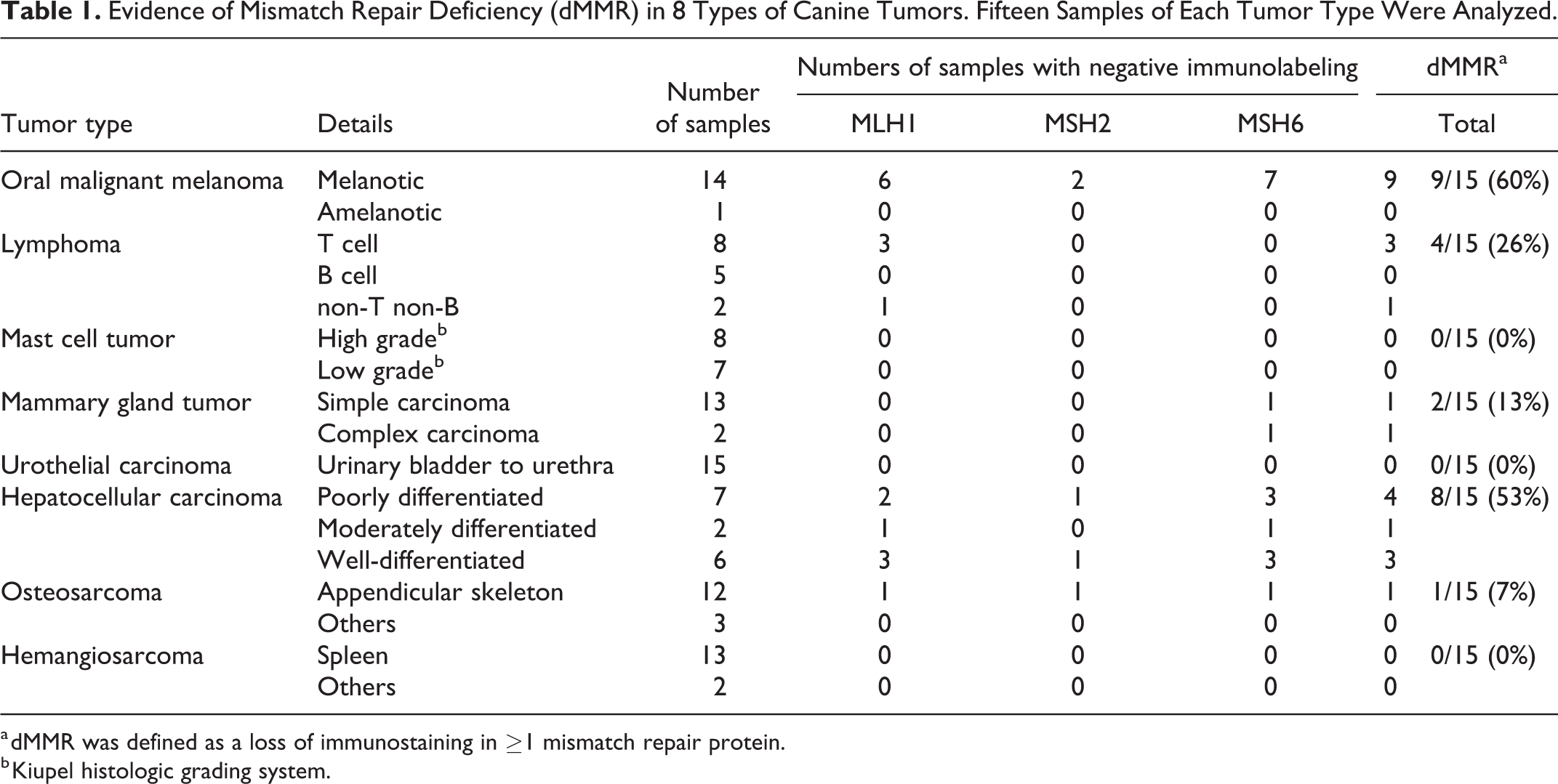
a dMMR was defined as a loss of immunostaining in ≥1 mismatch repair protein.
b Kiupel histologic grading system.
Representative results of immunostaining are shown in Figures 1 and 2. The number of samples showing negative immunolabeling for each MMR protein and number of dMMR samples in the total sample are summarized in Figure 3 and Table 1. The proportion of tumor samples with evidence of dMMR (ie, negative immunolabeling for ≥1 MMR protein) was 9/15 (60%) for oral malignant melanoma, 8/15 (53%) for hepatocellular carcinoma, 4/15 (27%) for lymphoma, 2/15 (13%) for mammary gland tumor, and 1/15 (7%) for osteosarcoma. None of the mast cell tumor, urothelial carcinoma, and hemangiosarcoma samples had evidence of dMMR (ie, all samples had positive nuclear immunolabeling for each of the 3 MMR proteins). Negative immunolabeling was more frequent for MLH1 and MSH6, and less frequent for MSH2 (Fig. 3).
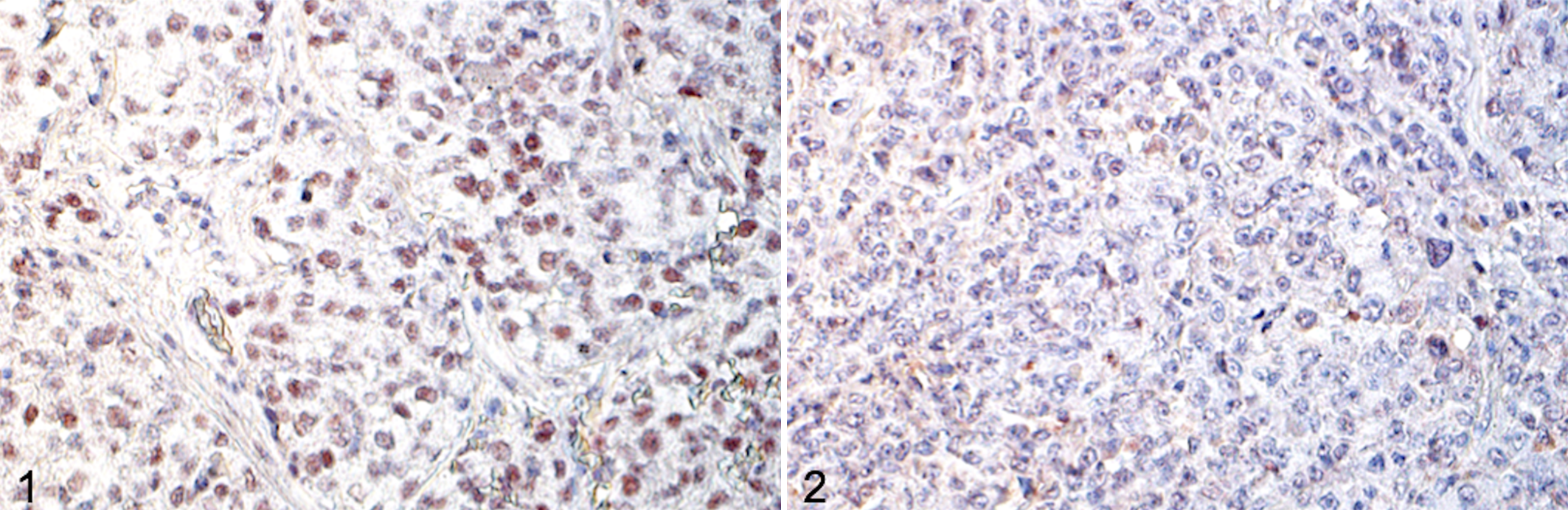
Oral malignant melanoma, dog. Immunohistochemistry (IHC). This case has positive nuclear immunolabeling for mutS homologue 2 (MSH2) protein (Fig. 1), but negative nuclear immunolabeling for mutL homologue 1 (MLH1) protein (not shown) and mutS homologue 6 (MSH6) protein (Fig. 2). Thus, the MMR system in this sample is considered deficient.
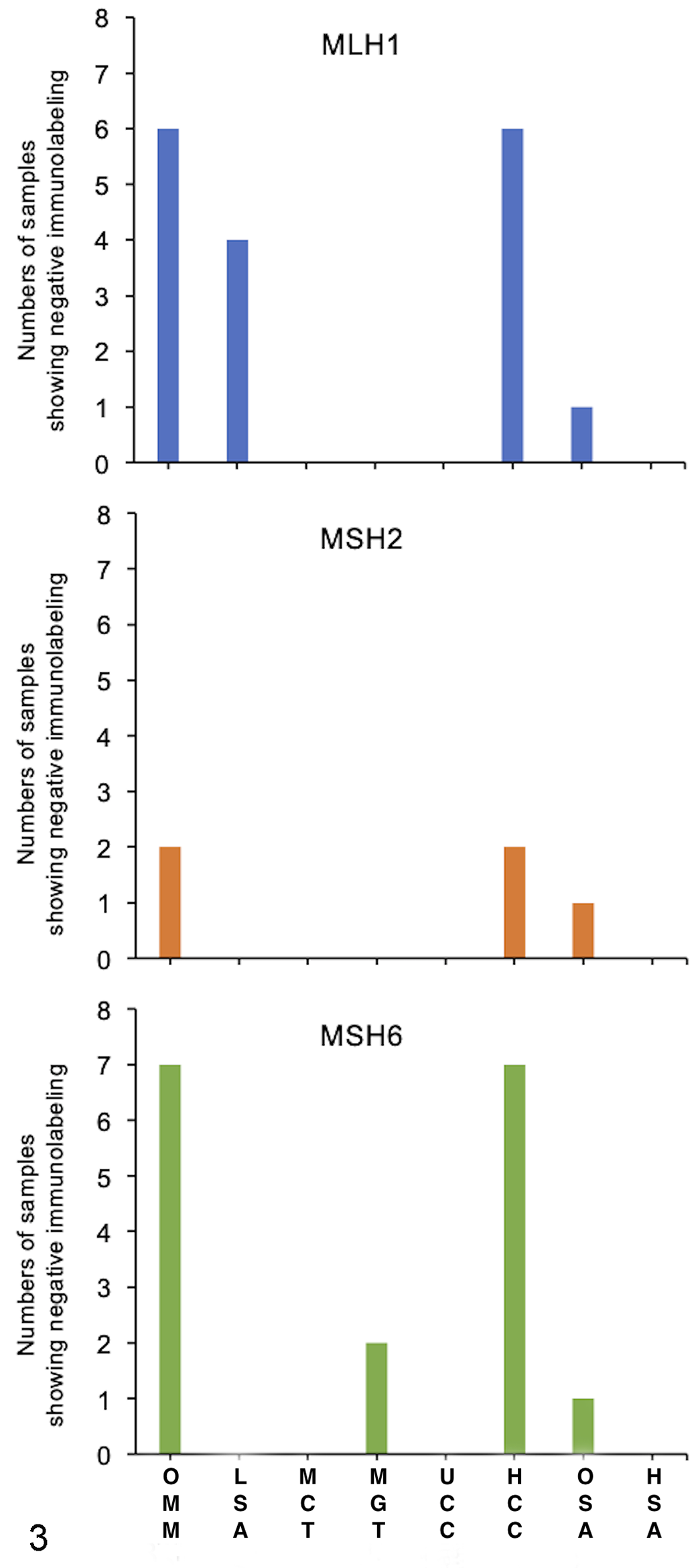
Number of samples (from 15 total for each tumor type) showing negative immunolabeling for each mismatch repair (MMR) protein (MLH1, MSH2, and MSH6). Negative immunolabeling was more frequent in canine OMM and HCC than in other tumors. Negative immunolabeling was more frequent for MLH1 and MSH6 than for MSH2. OMM, oral malignant melanoma; LSA, lymphoma; MCT, mast cell tumor; MGT, mammary gland tumor; UCC, urothelial cell carcinoma; HCC, hepatocellular carcinoma; OSA, osteosarcoma; HAS, hemangiosarcoma.
Negative immunolabeling for ≥2 MMR proteins was observed for 4/15 samples of oral malignant melanoma, 5/15 samples of hepatocellular carcinoma, and 1/15 samples of osteosarcoma (Table 2). In the 4 oral malignant melanoma samples, no MMR protein was detected in 2 samples and only MSH2 protein was detected in 2 other samples. In the 5 hepatocellular carcinoma samples, no MMR protein was detected in 3 samples and only MSH2 protein was detected in 2 other samples. In the 1 osteosarcoma sample, no MMR proteins were observed (Table 2).
The samples showing negative immunostaining for ≥2 MMR proteins.
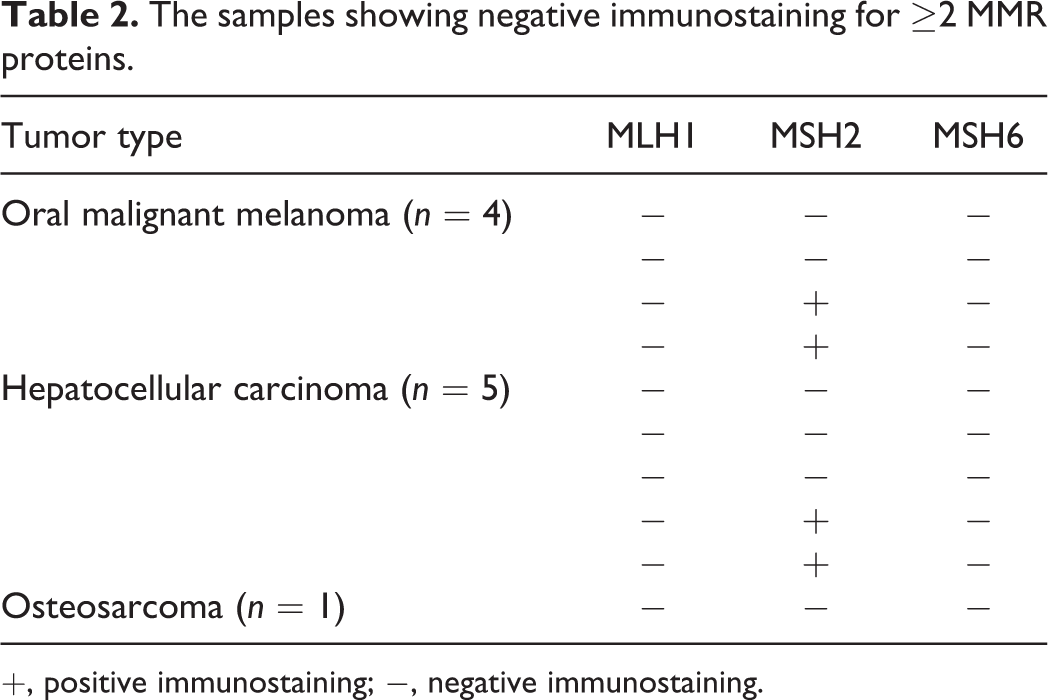
+, positive immunostaining; −, negative immunostaining.
Confirmation of Cross-Reactivity of Antibodies Against Each MMR Protein
To confirm the binding of antibodies against canine MMR proteins (MLH1, MSH2, and MSH6), we established MMR gene knockout cell lines using a CRISPR/Cas9 system. First, we confirmed that all 3 MMR proteins were detected in CIP-p cell line by western blotting (data not shown). Each antibody was found to cross-react with each canine MMR protein, as the expression of each MMR protein was lost in the cell line with knockout of the corresponding MMR gene (Fig. 4). In addition, immunolabeling for MSH6 protein was lost when the msh2 gene was knocked out in CIP-p cells, and immunolabeling for MSH2 protein was lost when the msh6 gene was knocked out in CIP-p cells.
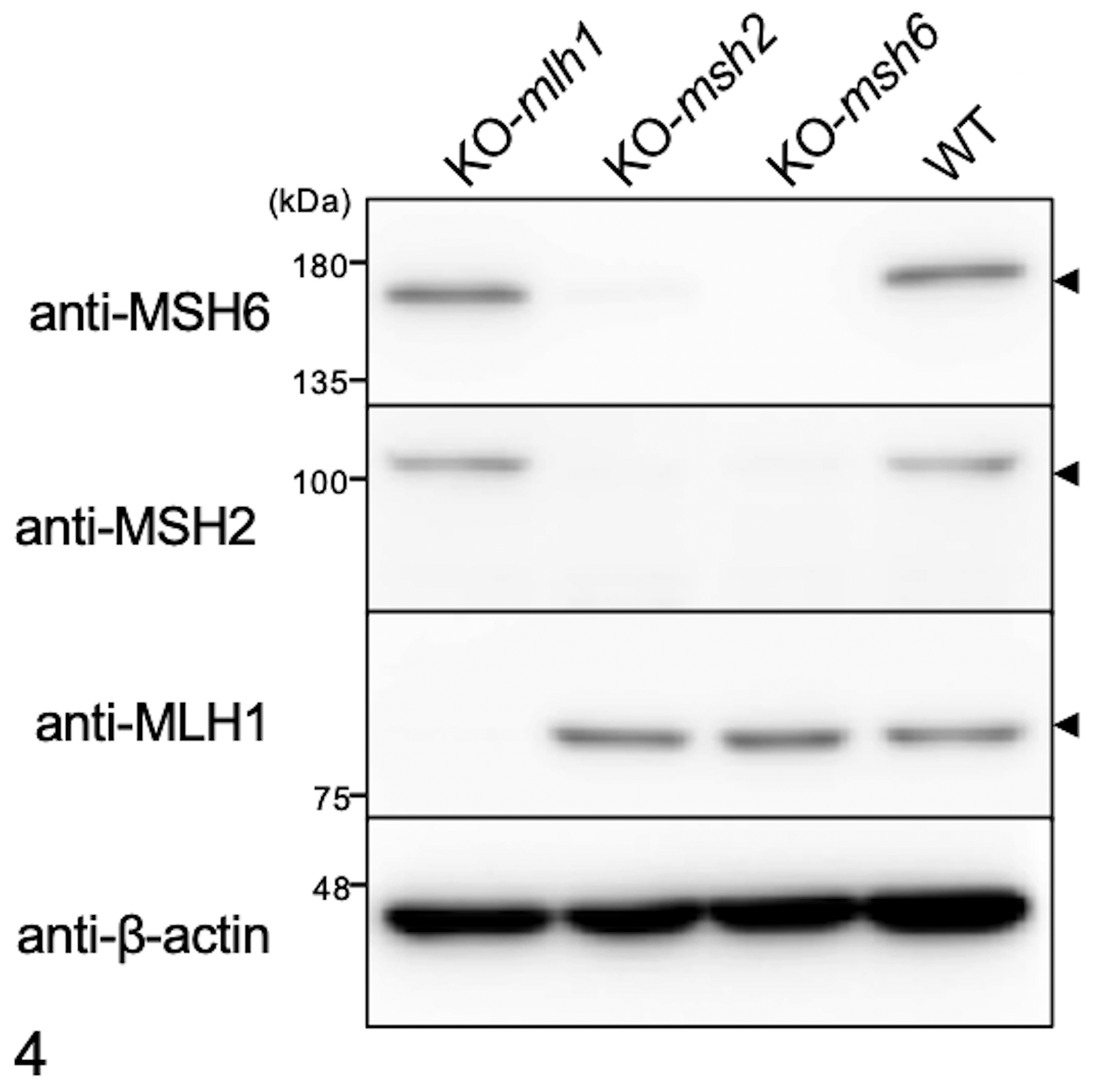
Validation of antibodies used for immunohistochemistry. Whole cell lysates were prepared from CIP-p cell lines of wild type or with knockout of canine mismatch repair genes mlh1, msh2, and msh6. Proteins were separated with SDS-PAGE, followed by western blotting with anti-human MLH1 antibody, anti-human MSH2 antibody, or anti-human MSH6 antibody. β-Actin was used as the protein loading control. These results support that these antibodies specifically reacted with canine MMR proteins.
Discussion
This is the first study to analyze deficiency of the MMR system in multiple types of canine tumors. Although we used human monoclonal antibodies to analyze dMMR, we found that these antibodies specifically reacted with canine MMR proteins, except PMS2. In the immunohistochemical analysis, dMMR tended to vary among canine tumor types. None of the mast cell tumor, urothelial carcinoma, and hemangiosarcoma samples showed a dMMR system, while oral malignant melanoma and hepatocellular carcinoma tended to have dMMR. Our results on mast cell tumor were consistent with those of a previous study 21 ; however, there are no reports on dMMR in other types of tumors in veterinary medicine.
In human malignant melanoma, a previous report 12 showed a strong tendency of dMMR (approximately 55%), which is consistent with our result on oral malignant melanoma. Tumors in humans with dMMR are associated with better response of immune checkpoint blockade. 14,15 In canine clinical studies, 10,17 PD-1 or PD-L1 inhibitor therapy seemed to be effective in some cases with oral malignant melanoma, suggesting that tumors with dMMR are possibly involved in the response to immune checkpoint blockade therapy in dogs.
In human hepatocellular carcinoma, the proportion of dMMR was reported approximately 3%. 14 Therefore, it was unexpected that canine hepatocellular carcinoma was strongly prone to a defective MMR system. Human hepatocellular carcinoma is mainly caused by hepatitis B and C virus infection, 4 whereas canine hepatocellular carcinoma is generally not caused by viral infection. Thus, the nature of HCC may be different between dogs and humans. Our results indicated that hepatocellular carcinoma might be a good candidate for the treatment of PD-1 or PD-L1 inhibitors in a future study. In addition, tumors with dMMR are known to acquire resistance to some anticancer drugs in human medicine, 1,2,7 suggesting that dMMR may be related to resistance to chemotherapy that typifies canine hepatocellular carcinoma and oral malignant melanoma.
In humans, it is known that MSH6 protein functions as a heterodimeric complex with MSH2 protein and is degraded in the absence of MSH2 protein. 27 Our IHC results supported this phenomenon because tumor samples with loss of MSH2 protein commonly showed negative immunolabeling for MSH6. However, the reverse was not necessarily true. This may be because MSH2 protein was able to form a complex with MSH3 protein in addition to MSH6 protein. 27 Results of the knockout cell lines experiment also showed that the loss of MSH2 protein expression was accompanied with a defect of MSH6 protein (Fig. 4). However, our data also showed that loss of MSH2 protein was observed in the msh6-knockout cell line. Furthermore, the tumor samples with loss of MSH2 protein immunolabeling was also accompanied with a defect of MLH1 protein in this study. Therefore, MSH2 and MSH6 proteins in dogs may have a function/mechanism that is different from the human proteins; thus, further analysis is needed.
In the present study, there are several limitations. First, 15 samples of tumor types were used in this survey may be insufficient to definitively identify the frequency of dMMR in each canine tumor types. Second, MMR proteins were used for dMMR analysis in humans, but we could not examine the expression of PMS2 protein due to the lack of a specific antibody against canine PMS2. Moreover, several other MMR proteins are also known in humans, 9,23,28 and it is possible that other proteins function as major MMR proteins in dogs. Third, we analyzed the immunolabeling of 3 MMR proteins in this study, but detecting positive immunostaining may not always relate to the functionality of the protein. Fourth, the clinical outcome in canine tumors with dMMR was not evaluated in this study. All these points are essential to be considered in a future study.
In conclusion, we found that canine oral malignant melanoma and hepatocellular carcinoma samples were prone to defective MMR proteins. Further studies to analyze microsatellite instability and to elucidate the relationship between response to immune checkpoint blockade therapy and canine tumors with dMMR are expected.
Footnotes
Acknowledgement
We thank Dr Takayuki Nakagawa for providing the canine mammary gland tumor cell lines.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
