Abstract
A 2-year-old Sprague-Dawley rat with hindlimb paralysis was diagnosed with a cerebral malignant astrocytoma. The distinctive feature of this astrocytoma was the presence of scattered binucleated cells that contained hypereosinophilic, 1–2 μm in diameter, cytoplasmic granules. The neoplastic astrocytes stained positively for vimentin (VIM), lysozyme, and phosphotungstic acid hematoxylin (PTAH). Within the binucleated cells, granules stained with PTAH and periodic acid-Schiff (PAS) before and after diastase digestion. Ultrastructurally, neoplastic astrocytes were characterized by cytoplasmic aggregates of electron-dense intermediate filaments consistent with VIM and desmin. The cytoplasm of binucleated cells contained numerous phagolysosomes enlarged by myelin figures and glycoprotein or glycolipid. Intermediate filaments were not present. This is the first description, in the rat, of a neoplasm with features resembling the human granular cell astrocytoma. Our findings suggest that an astrocytic origin should be considered for the binucleated cells in this neoplasm.
Human granular cell astrocytomas were first described by N. Sakurama et al. 11 Although their astrocytic lineage is verified, the granular cells share cytologic features and lysosomal content with granular cell tumors that occur elsewhere in the body. 6,8 In veterinary medicine, granular cell tumors are the most frequent spontaneous primary tumors of the central nervous system in rats. 2 They are considered to be of meningeal origin because of their location in the meninges and their immunohistochemical (glial fibrillary acidic protein [GFAP], protein S-100 [S-100], vimentin [VIM]), and ultrastructural properties (absence of basement membrane around neoplastic cells and presence of desmosomes). 5,10 The presence of granular cells in an astrocytoma of a rat was cited once in 1979, but details were not provided on the cell type and origin. 4
A 2-year-old Sprague-Dawley rat was presented with a history of hindlimb paralysis. The rat was euthanatized and intravenously perfused with formalin. At necropsy, the left cerebral hemisphere was partially effaced by a neoplasm. Histologic samples were fixed in 10% neutral-buffered formalin, embedded in paraffin, processed routinely, and stained with hematoxylin and eosin. Special stains included periodic acid–Schiff (PAS) with and without α-amylase digestion, VIM (Dako Corporation, Carpeinteria, CA; dilution 1/100), GFAP (Dako Corporation; dilution 1/15,000), phosphotungstic acid hematoxylin (PTAH), lysozyme (Dako Corporation; dilution 1/200), S-100 (Dako Corporation; dilution 1/2,000), pan-neurofilaments (PNF)(Zymed, South San Francisco, CA; dilution 1/1,000), and wide-spectrum cytokeratin (WSC) (Dako Corporation; dilution 1/2,000). Positive control slides consisted in rat liver (PAS), lung (VIM), brain (GFAP, PNF), heart (PTAH), spleen (lysozyme), gut (S-100), and skin (WSC). Two slides (brain astrocytoma and tissues used for positive control) were stained with diluent buffer (GFAP, Lysozyme, PNF, S-100, WSC) or mouse immunoglobulin G1 (VIM) and served as negative control slides. Multiple sections of the mass, including sections through the cerebellum, hypocampus, and cerebrum were examined microscopically. The neoplasm was nonencapsulated; effaced the region of the lateral ventricle; and extended to the corpus callosum, dentate gyri, hypocampus, third ventricle, thalamus, hypothalalmus, and overlying meninges. The neoplasm was mainly composed of sheets of neoplastic astrocytes that were round to oval, 13–20 µm in diameter, and contained a pale, eosinophilic, finely granular cytoplasm with mitotic figures ranging from 1 to 3 per high-power field. Multifocally, there were vascular proliferation, vascular necrosis, and necrohemorrhages. Occasionally, neoplastic astrocytes formed “pseudopalissades” around necrotic centers and blood vessels. A distinctive feature of this neoplasm was the presence of low number (2–4 per 40×) of large binucleated cells that were round to oval, 20–30 µm in diameter, and contained numerous bright eosinophilic, 1–2 µm, intracytoplasmic granules and occasional clear, 2–3 µm, cytoplasmic vacuoles (Fig. 1). Mitoses were not observed in these binucleated cells. In the brain tissue adjacent to the tumor, there were moderate numbers of reactive astrocytes, microglial cells, satellitosis, perivascular infiltrates of inflammatory cells, and spongiosis of the neuropil.
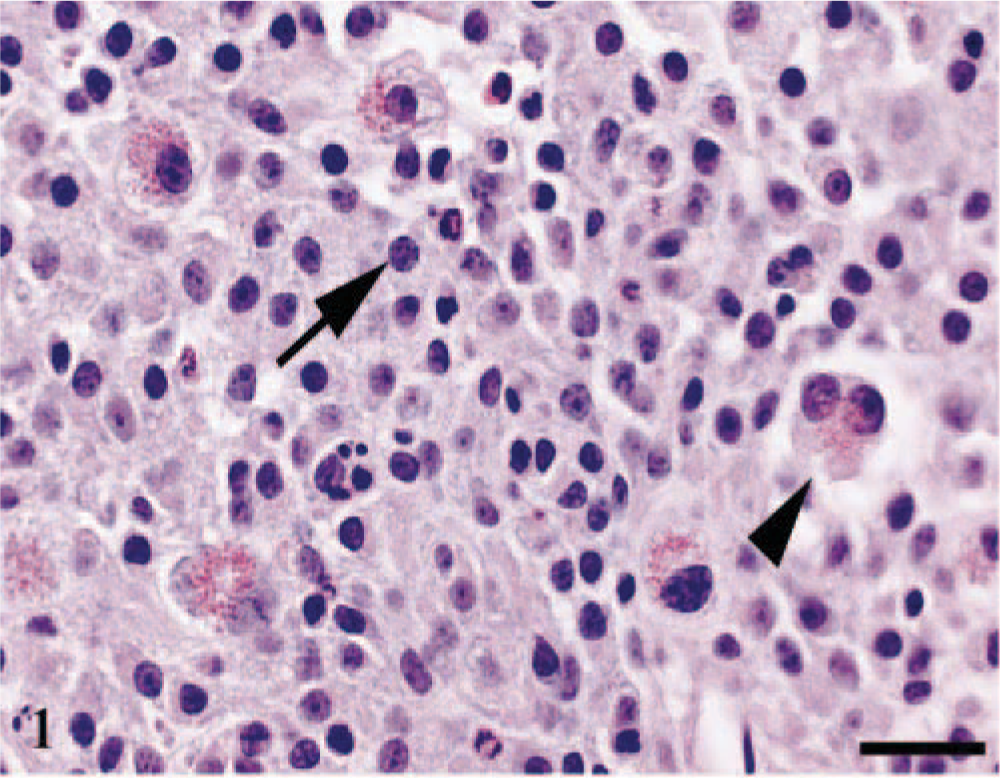
Cerebrum. Neoplastic astrocytes (arrow) are admixed with occasional binucleated cells (arrow head). HE. Bar = 25 µm.
Strong immunoreactivity for GFAP and S-100 was observed in reactive astrocytes at the tumor periphery and in occasional entrapped astrocytes, but neoplastic astrocytes and granular cells were consistently negative (Fig. 2). Strong and diffuse immunoreactivity for VIM was observed only in the cytoplasm and cytoplasmic processes of neoplastic astrocytes (Fig. 3). Strong immunoreactivity for PTAH was observed in the cytoplasmic granules of the large binucleated cells and in multifocal clusters of astrocytes. In these astrocytes, PTAH-positive staining highlighted the perinuclear region and the meshwork of cytoplasmic processes. Lysozyme staining was only observed in the cytoplasm of neoplastic astrocytes and admixed gitter cells. Strong PAS staining, before and after diastase digestion, was observed mainly in the cytoplasmic granules of binucleated cells, and to a lesser extent, in the gitter cells and few astrocytes in the necrotic areas. The neoplasm was invariably negative for WSC and PNF stainings.
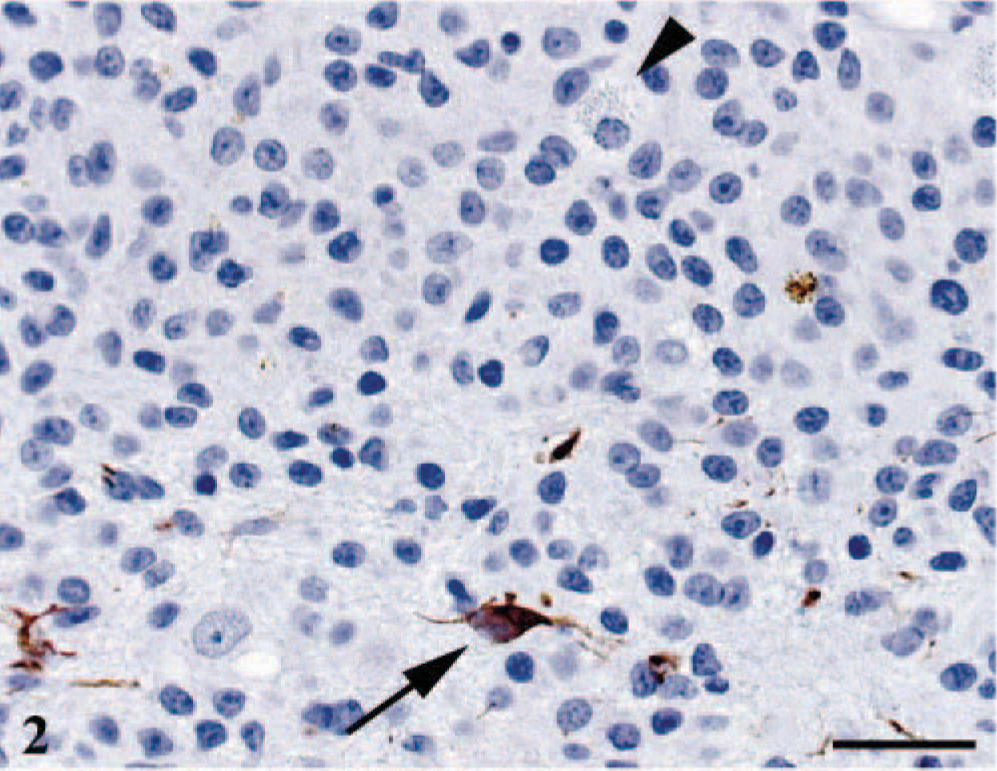
Cerebrum. Strong immunoreactivity for GFAP was observed in reactive astrocytes (arrow) but not neoplastic cells. Granular cells were consistently negative (arrow head). GFAP and hematoxylin. Bar = 25 µm.
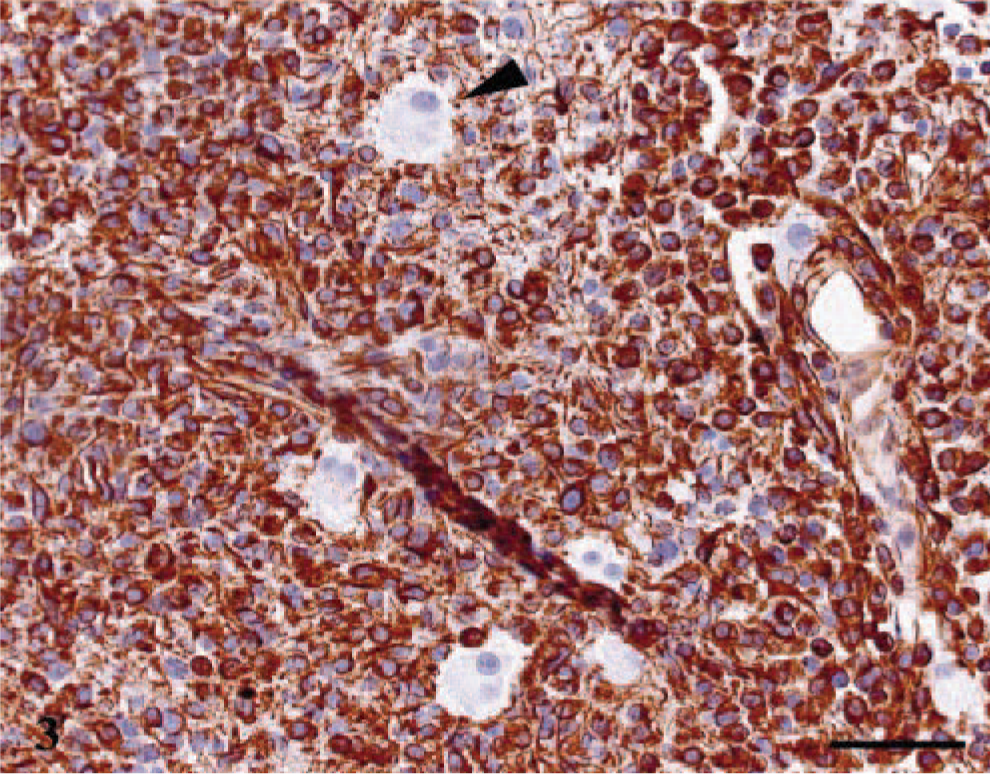
Cerebrum. Strong and diffuse immunoreactivity was observed in the cytoplasm and cytoplasmic processes of neoplastic astrocytes, whereas binucleated cells remained unstained (arrow head). VIM and hematoxylin. Bar = 25 µm.
Ultrastructurally, the neoplasm was mainly composed of neoplastic astrocytes in a background of necrotic debris, collagen fibrils, and edema fluid. Neoplastic astrocytes contained an oval to bizarre-shaped nucleus, one nucleolus, and finely granular chromatin (Fig. 4). The cytoplasm contained a well-developed rough endoplasmic reticulum (RER), moderate numbers of mitochondria, occasional pinocytic vesicles, and aggregates of 10–15 nm intermediate filaments. Cytoplasmic processes were occasionally observed and contained numerous intermediate filaments. Occasionally, the cytoplasm was expanded by confronting cisternae (multilayered RER) that consisted in a pair of closely apposed cisternae of the RER (Fig. 5). The outermost membrane bore low numbers of ribosomes on its surface, and the disintegrated ribosomes trapped between the confronting cisternae produced the dense lamina. These structures are usually seen in mitotic and interphase cells. Granular cells contained two oval and often indented nuclei and one to two nucleoli (Fig. 6). The chromatin pattern varied from finely granular to more coarsely clumped and marginated to the periphery of the nucleus. The cytoplasm was divided into organelle-free and organelle-rich regions. In the latter, there were moderate quantities of granulated and degranulated RER, mitochondria, and lipid droplets. The numerous bright eosinophilic granules observed histologically consisted of lysosomes (Fig. 6, inset). These lysosomes contain concentric membranous lamellar material and aggregates of deeply electron-dense 18–23 nm granules. The granules were also PAS positive and diastase resistant, suggesting that they could consist of glycoproteins or glycolipids. 3
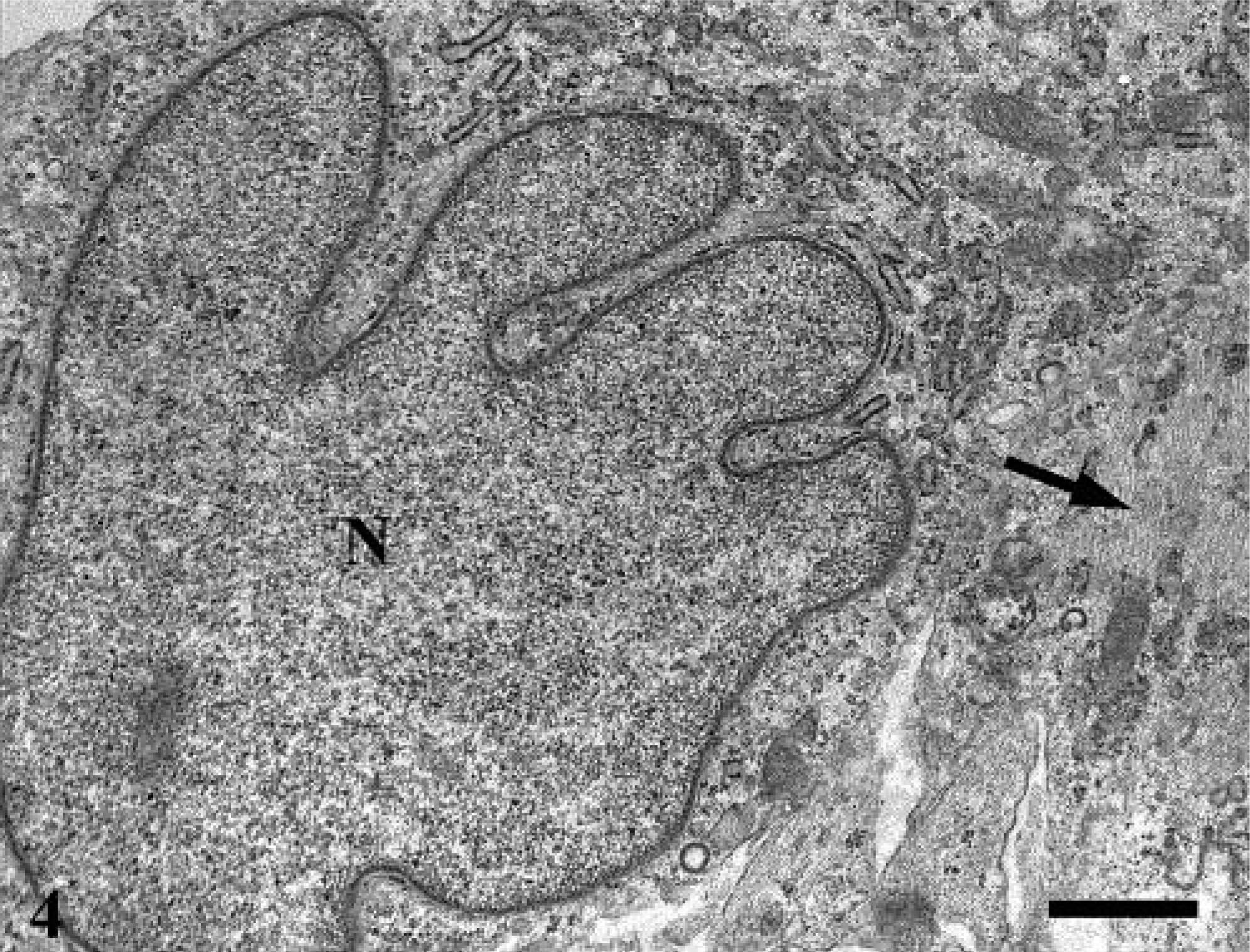
Cerebrum; electron micrograph. Neoplastic astrocytes. Nuclei (N) is irregular, and cytoplasmic processes contained intermediate filaments (arrow). Bar = 1 µm.
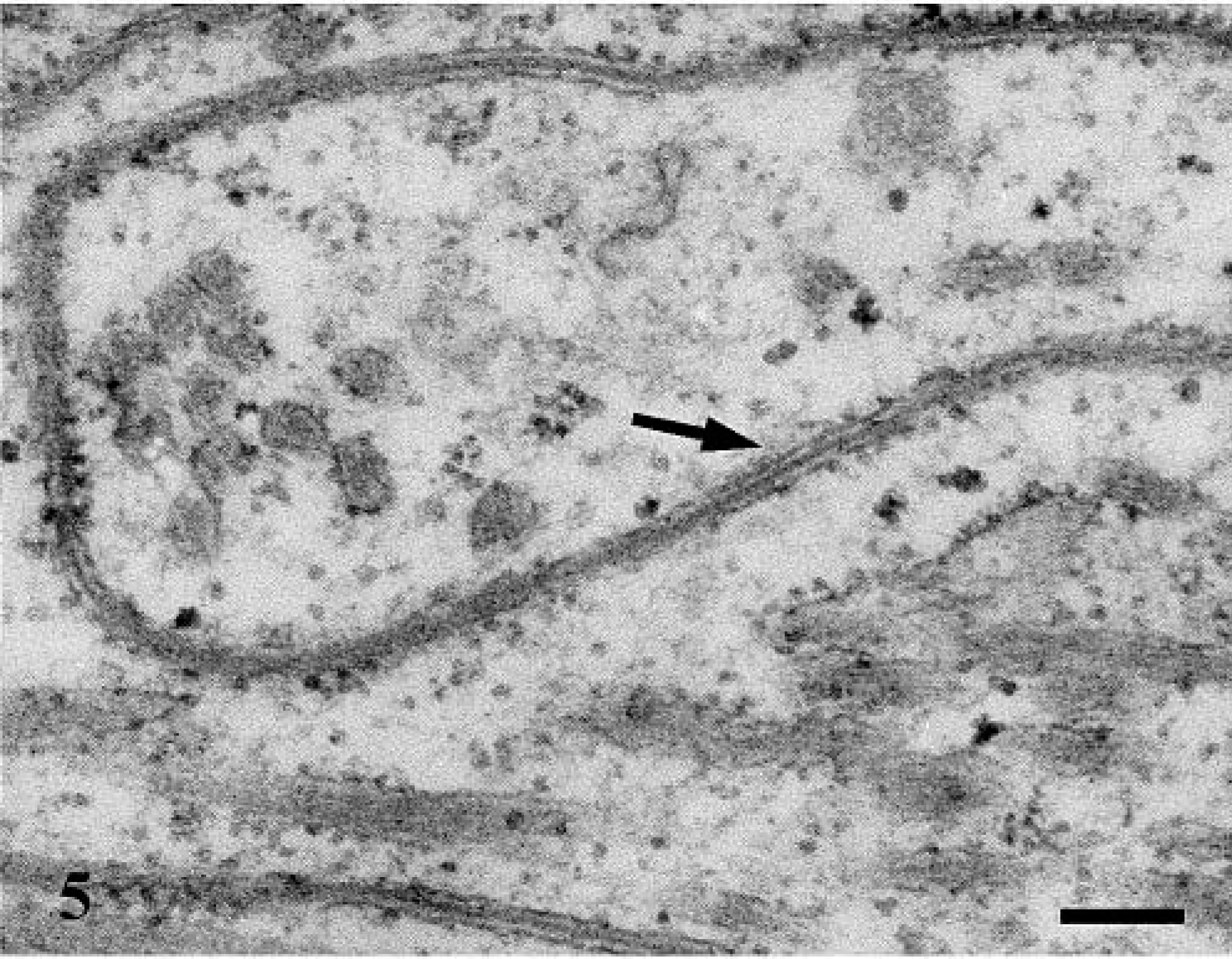
Cerebrum; electron micrograph. Confronting cisternae consist of closely apposed cisternae of the RER (arrow). Bar = 0.167 µm.
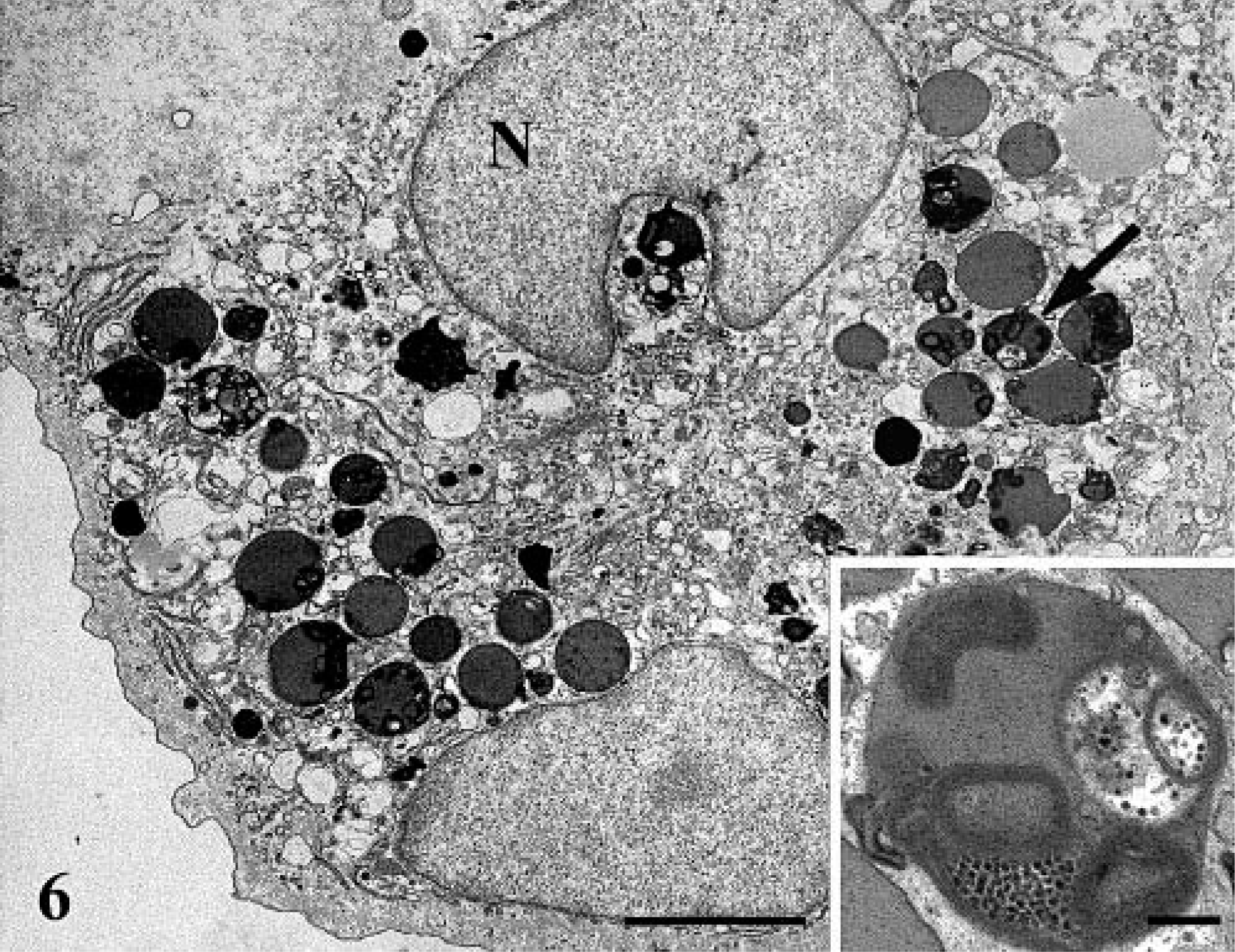
Cerebrum; electron micrograph. Binucleated (N) granular cells with numerous lysosome (arrow). Bar = 2.8 µm. Lysosomes contain glycoproteins or glycolipids (or both) and membranous whorls (Inset). Bar = 0.25 µm.
In this astrocytoma, microscopic features of high cellularity, cellular pleomorphism, nuclear atypia, mitotic activity, and more importantly, vascular proliferation, necrosis, and rare pseudopalissading of neoplastic astrocytes around necrotic areas and blood vessels are strongly indicative of anaplasia and malignancy. The negative immunohistochemistry for GFAP is consistent with a more primitive differentiation of the neoplastic astrocytes and is in agreement with previous studies, which indicate that spontaneous neoplasms of the central nervous system are often less differentiated in rodents than in humans. 9
Significant features distinguish our case from the traditional “granular cell meningioma” described in the veterinary literature. 1 First, granular cell meningioma is usually a well-circumscribed neoplasm that extends from the leptomeninges. In contrast, our astrocytoma was very infiltrative, extending from the region of the lateral ventricle in the left cerebral hemisphere, with binucleated cells found only within and around the necrotic centers. However, we cannot exclude that serial sectioning of the astrocytoma would have revealed a clear meningeal connection. Another distinction is that granular cell meningioma is traditionally composed of sheet or nests of closely packed oval cells with an abundant eosinophilic granular cytoplasm and rare mitoses, whereas our neoplasm was mainly composed of neoplastic astrocytes admixed with very low number of large binucleated cells without mitotic activity. Finally, granular cell meningioma is often GFAP positive, contrasting to the negative staining in our case.
In human medicine, the ultrastructural findings of astrocytoma with “granular cell changes” indicate that the granularity results from the accumulation of autophagocytic vacuoles due to a lysosomal enzyme defect or due to excess production of a substance normally processed in lysosome. Therefore, the acquisition of cytoplasmic granules may reflect a metabolic change in the neoplastic population. 6 In this report, neoplastic astrocytes, but not binucleated cells, were stained with lysozyme. Our hypothesis is that during tumorogenesis, astrocytes acquired a lysosomal enzymatic defect and differentiated into large granular cells with numerous autophagocytic vacuoles. The fact that the granular cells are binucleated and never showed mitotic activity could also suggest an additional mitotic defect and may explain why granular cells are found in low numbers throughout the neoplasm. Finally, absence of GFAP staining could indicate that binucleated cells derive from the neoplastic GFAP astrocytes. Alternatively, lack of GFAP staining in the granular cells could result from the crowding of the cell cytoplasm by the eosinophilic granules. A similar situation is known to occur in human pleomorphic xanthoastrocytomas, where heavily lipidized neoplastic astrocytes are usually negative for GFAP or just show GFAP positivity only in the area of the peripheral lipid-free cytoplasmic margin. 7
In conclusion, this is the first report that illustrates the morphologic characteristics of a malignant astrocytoma with binucleated granular cells in a rat. Whether the lack of VIM and lyzozyme staining in binucleated cells reflects different stages of cellular maturation, metabolic defect, or divergent cellular differentiation could not be determined. Because astrocytes were undoubtedly identified as the main component of the neoplasm and on the basis of the morphologic similarity of human granular cells astrocytoma, the assumption that, in the rat, binucleated cells are derived from astrocytes appears reasonable.
Footnotes
Acknowledgements
We thank Mary Payette, Karen Steever, and Kim Kowsz for their technical assistance.
