Abstract
Alveolar echinococcosis was diagnosed in 12 cynomolgus monkeys (Macaca fascicularis) at postmortem examination within a period of 6 years. Besides consistent involvement of the liver, parasitic lesions were also present in mesenteric lymph nodes, pancreas, lung, and kidney. In the liver, various patterns of host's responses to parasitic tissue could be distinguished. Infiltration of macrophages, often multinucleated, around usually intact metacestodes was the main feature of one pattern. A second pattern was characterized by the presence of abundant, normally degenerate granulocytes in addition to macrophages surrounding collapsed laminated structures. Finally and as a third pattern, some cysts were surrounded by marked collagen deposition, which was usually not a significant feature of the other foci. Parasitic cysts with protoscolices were observed in foci with the first and third pattern but not in the second one. The simultaneous occurrence of all three patterns was observed in most animals. Type AA amyloid was identified either in the space of Dissé, macro-phages or blood vessel walls in nine animals using immunohistochemistry. Identity of parasitic structures such as metacestodes of Echinococcus multilocularis was confirmed immunohistochemically. All animals that could be tested serologically (7/12) had detectable antibodies against the E. multilocularis-specific Em2 antigen. Liver lesions of six animals were additionally analyzed by polymerase chain reaction, yielding the amplification of a specific E. multilocularis DNA fragment in each case.
The fox tape worm Echinococcus multilocularis has a life cycle involving mainly foxes, with domestic carnivore-like dogs as final hosts and small rodents as intermediate hosts. 6 In the latter the eggs develop into metacestodes with formation of multilocular vesiculated cysts in the liver, giving the disease the name alveolar echinococcosis. These cysts have a germinal layer, which produces brood capsules containing protoscolices. 10 Other species can function as aberrant intermediate hosts, and infection with E. multilocularis larvae is reported from a variety of nonhuman primates, including gorilla (Gorilla gorilla), orangutan (Pongo pygmaeus), rhesus monkey (Macaca mulatta), and cynomolgus monkey (Macaca fascicularis). 5,15–18 Lesions include clustered cystic structures mainly in the liver with varying amounts of necrosis. These cysts correspond histologically to metacestodic larvae of E. multilocularis. Protoscolices were present in many of the primate cases, which is in contrast to findings in humans, in whom the infection is usually sterile without production of protoscolices. 6 In a recent study, the percentage of cynomolgus monkeys infected with E. multilocularis compared with other captive primates was very high (>50%). 6 Conversely, reports about the pathology and histology of alveolar echinococcosis in cynomolgus monkeys are so far restricted to one case report in a German journal about an individual monkey. 17 In our study, postmortem examination of 12 cynomolgus monkeys revealed the presence of E. multilocularis, which was confirmed histologically and immunohistochemically as well as by serology and polymerase chain reaction (PCR) technique in most cases. The objective of this study was to characterize the pathologic features of alveolar echinococcosis in the liver of these monkeys.
Materials and Methods
Liver samples from 12 cynomolgus monkeys (M. fascicularis) originating from two zoologic collections in Switzerland were included in this study. Monkeys were euthanatized or died between the years 1994 and 2001 either because of clinical signs like anorexia, cachexia, and apathy or because of dissolution of one of the colonies the age ranged from subadult to 20 years. The sex distribution was seven female and five male monkeys. For details see Table 1. All monkeys but one were necropsied at the Institut für Tierpathologie, Universität Bern, Switzerland. In case of monkey No. 8, only selected organs including the liver were submitted for further investigation. Sections of the liver and other affected organs were fixed in neutral-buffered formalin, paraffin embedded, cut, and stained with hematoxylin and eosin (HE) for routine histopathology, with Congo red to analyze the presence of amyloid and with period acid–Schiff to identify parasitic structures such as the laminated layer. The amount of fibrosis was evaluated using the Van Gieson stain. Sections were also analyzed for the presence and type of amyloid using a monoclonal antibody recognizing type AA amyloid (M0759, DAKO, Zug, Switzerland) in a dilution of 1:50 after citrate pretreatment (pH 6.0, 10 mM, 5 minutes microwave). Additional sections were stained with polyclonal rabbit antibodies against E. multilocularis in a dilution of 1:200; this antibody recognizes parasite structures such as germinal and laminated layers. 14 For the negative control the primary antibody was replaced by a preimmune rabbit serum. Biotinylated secondary antibodies, streptavidin–horseradish peroxidase, and the color indicator 3-amino-9-ethylcarbazole were applied according to the supplier's instructions (LSAB2, AEC, DAKO).
Sex, age, microscopic features in the liver and results of serology, and PCR assays in 12 cynomolgus monkeys with alveolar echinococcosis.∗
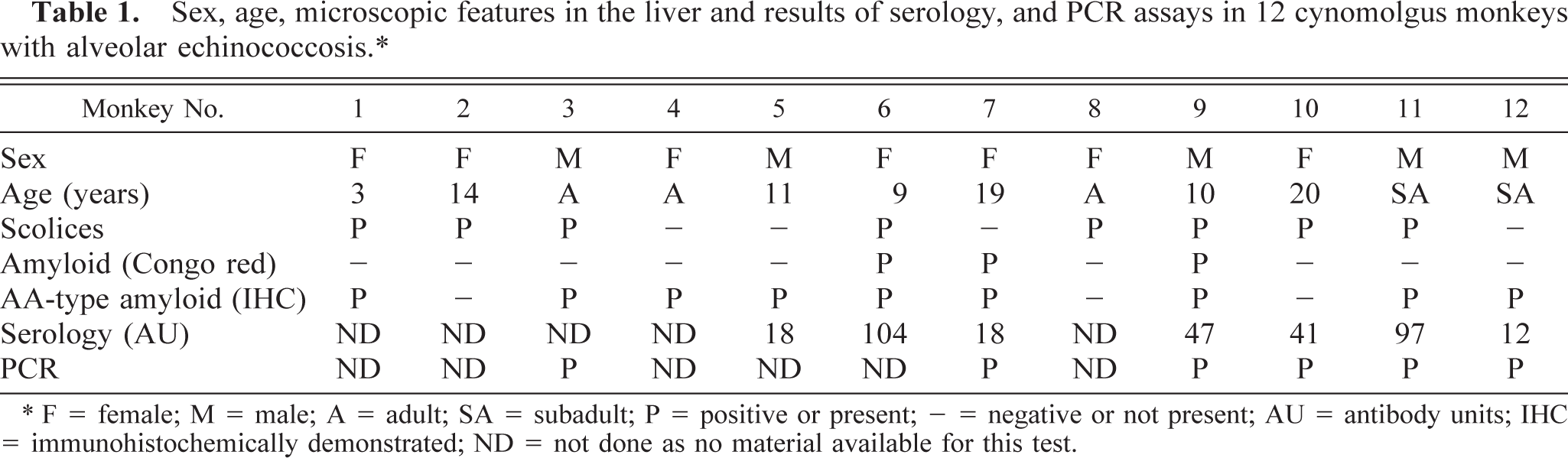
∗ F = female; M = male; A = adult; SA = subadult; P = positive or present; - = negative or not present; AU = antibody units; IHC = immunohistochemically demonstrated; ND = not done as no material available for this test.
Serum was collected at necropsy from all monkeys submitted from 1999 onward (Nos. 5–7 and 9–12) and analyzed for the presence of anti–E. multilocularis antibodies using a species-specific Em2 antigen, which is localized in the laminated layer as described. 7 The assay was adapted for the analysis of cynomolgus sera by replacing the anti-human immunoglobulin (IgG) with an anti-monkey IgG that recognizes cynomolgus monkey IgG conjugated to alkaline phosphatase (A140-102AP, Bethyl Laboratories, Montgomery, TX). Test performance, qualification (negative/positive threshold) and quantification (antibody concentration) of results were done as previously described for the corresponding serology in human patients. 12 DNA was extracted from native samples of hepatic lesions from monkey Nos. 3, 7, and 9–12 and used as a template for PCR. A primer pair BG1/BG2, which is specific for E. multilocularis, was used as described. 8,13
Results
Naturally acquired alveolar echinococcosis was diagnosed in 12 cynomolgus monkeys from two zoologic gardens at postmortem examination. The etiology was confirmed by histopathology, immunohistochemistry, serology, and PCR technique.
All monkeys had multiloculated cysts and vesiculated cystic conglomerates in the liver involving and replacing various amounts of hepatic tissue (Fig. 1). These conglomerates were up to 10 cm in size and contained usually a clear to yellow fluid. Rarely the walls of the lesions or surrounding parenchyma had hard to gritty foci interpreted as mineralization. In three animals (Nos. 6, 7, and 9) the liver was pale and waxy. Additional, similar multiloculated cysts were occasionally present in mesenteric lymph nodes (3×), lungs (3×), pancreas (2×), and kidneys (1×); however, these cysts were usually smaller than the total cystic mass present in the liver. Ascites was not a prominent feature in any monkey.
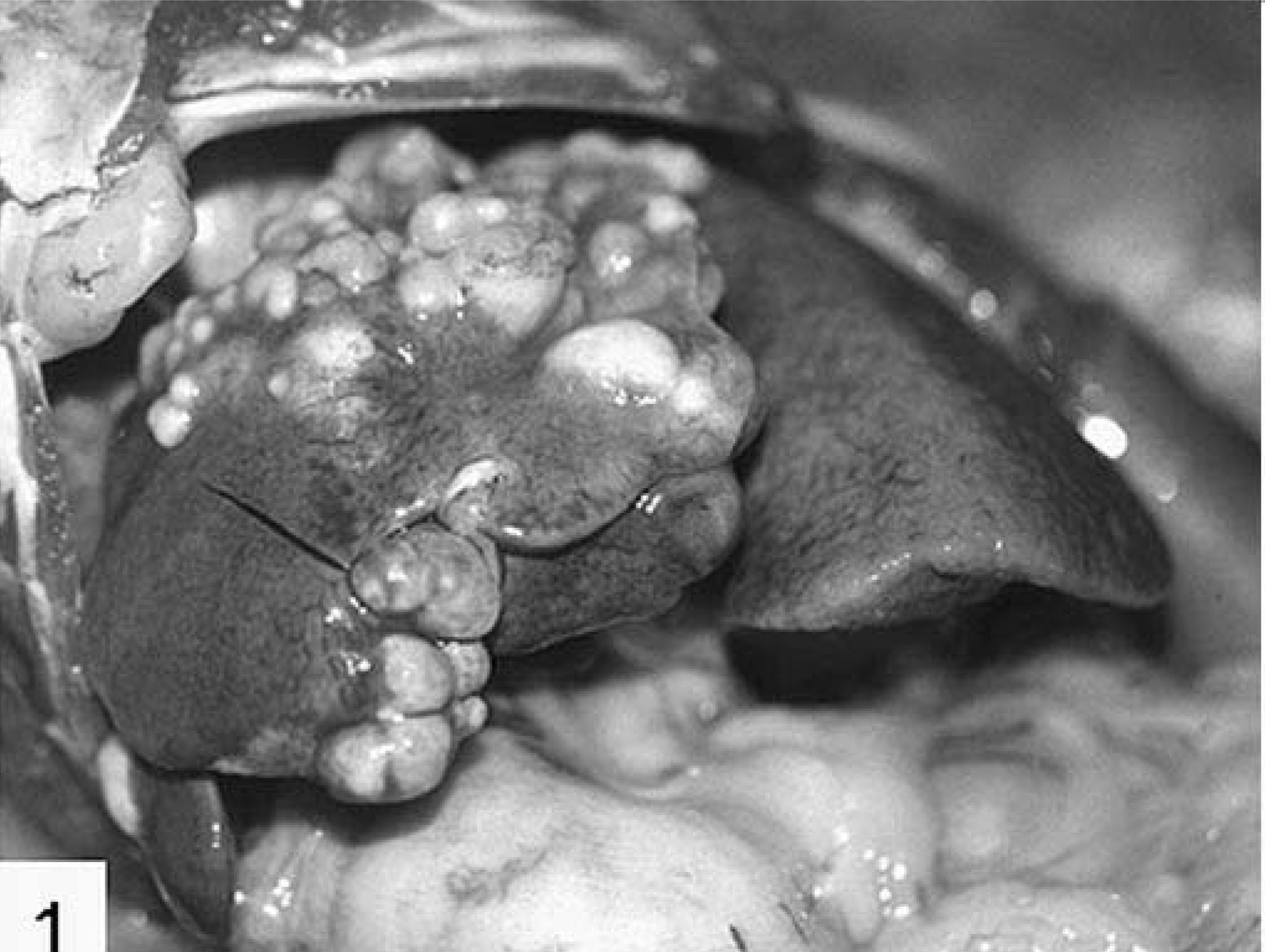
Liver; cynomolgus monkey No. 10. Hepatic alveolar echinococcosis.
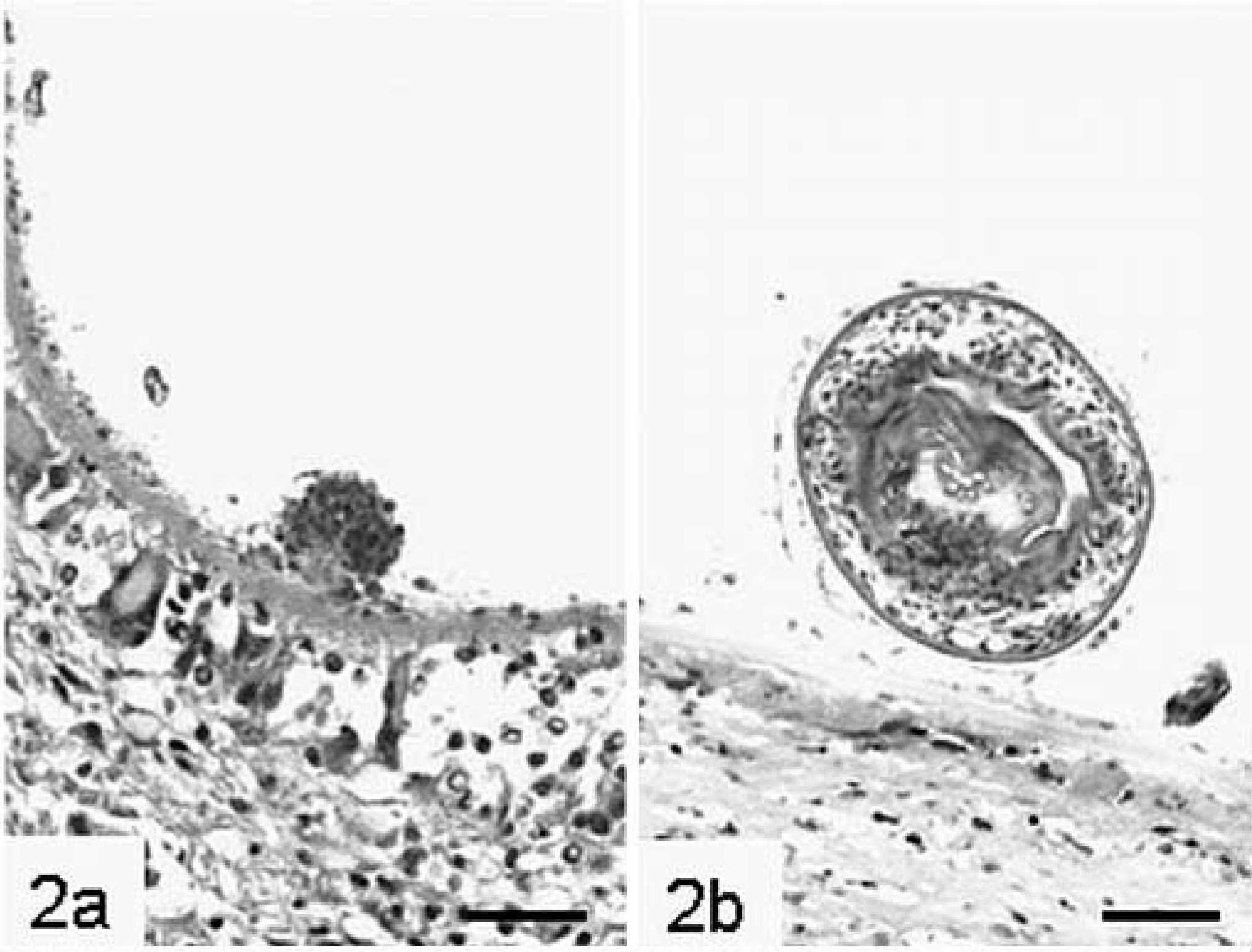
Histologic examination revealed metacestodal tissue in all liver samples. Parasitic cysts consisted of an outer laminated layer, an inner germinal layer, and protoscolices. The acelluar laminated layer was either nondisrupted and lined a cystic vesicle or was fragmented and often presented as a convoluted structure. It stained eosinophilic to rarely orange on HE stain and was mostly 2–15 µm thick. The laminated layer was strongly PAS positive. Intact laminated layers were lined on the inside by a cellular structure, the germinal layer. In some cysts the cells of the germinal layer had focally proliferated and formed internal, small, protruding buds (Fig. 2a). In nine animals (Nos. 1–3, 5, 6, 8–11) protoscolices were present in intact cysts. They were about 80–100 µm in diameter, and occasionally hooks were visible as refractile structures (Fig. 2b). Although most protoscolices were invaginated, evagination was occasionally observed. Calcareous corpuscles were present in the vicinity of the germinal layer and within protoscolices.
Histologically, various patterns of hepatic lesions could be distinguished on the basis of the presence and amount of parasitic structures, macrophages, eosinophilic and neutrophilic granulocytes, lymphocytes and plasma cells, necrosis, and fibrosis. Macrophages, as the most characteristic cell type, occurred either as mononuclear or as multinucleated cells. In the latter, the nuclei were often arranged peripherally (Langhans-type); but cells with nuclei dispersed throughout the cytoplasm (foreign body type) were also noted.
Many granulomas were composed of a central, usually nondisrupted laminated layer surrounded by macrophages with varying ratios of uninucleated and multinucleated cells (Fig. 3). These often abutted the laminated layer directly, but sometimes the macrophages were separated from the laminated layer by a zone of eosinophilic necrotic material. Protoscolices were nearly exclusively found in these foci. The surrounding hepatic tissue was often atrophic and the remainder infiltrated by varying numbers of eosinophils with fewer lymphocytes and rare plasma cells. Pericystic and intercystic fibrous connective tissue was usually not prominent. Neutrophils were not present in significant numbers.
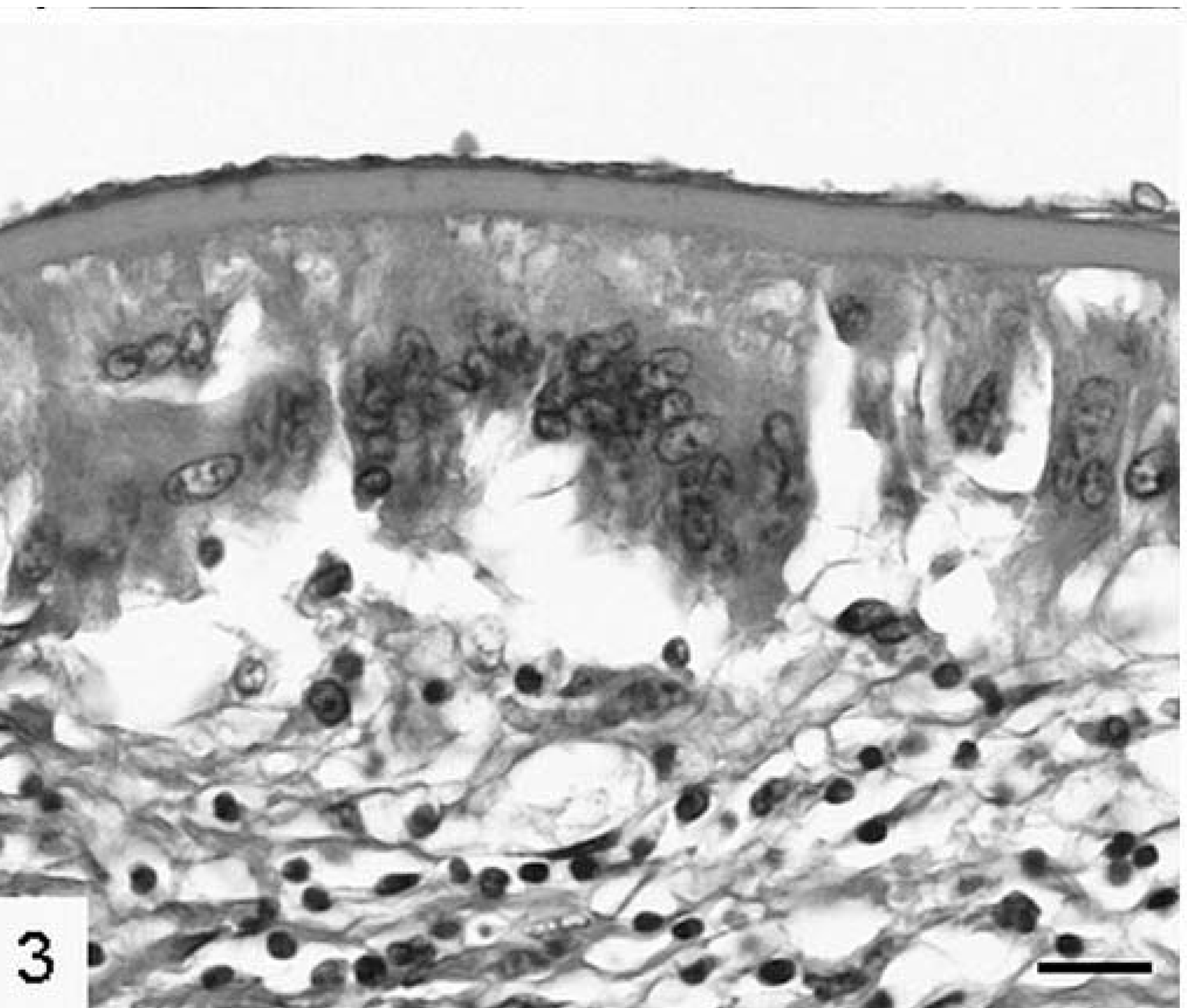
Liver; cynomolgus monkey No. 2. Laminated layer with an inner germinal layer and surrounding multinucleated macrophages. HE. Bar = 25 µm.
Another granulomatous pattern was characterized by a frequently disrupted central conglomerate of laminated layers, resembling a collapsed cyst, which was enclosed by often degenerate eosinophils and neutrophils (Fig. 4). This core was surrounded by a mantle of macrophages, sometimes multinucleated, and varying numbers of lymphocytes with fewer plasma cells. Protoscolices were not observed. The adjacent pericystic and intercystic connective and hepatic tissue was often infiltrated by eosinophils.
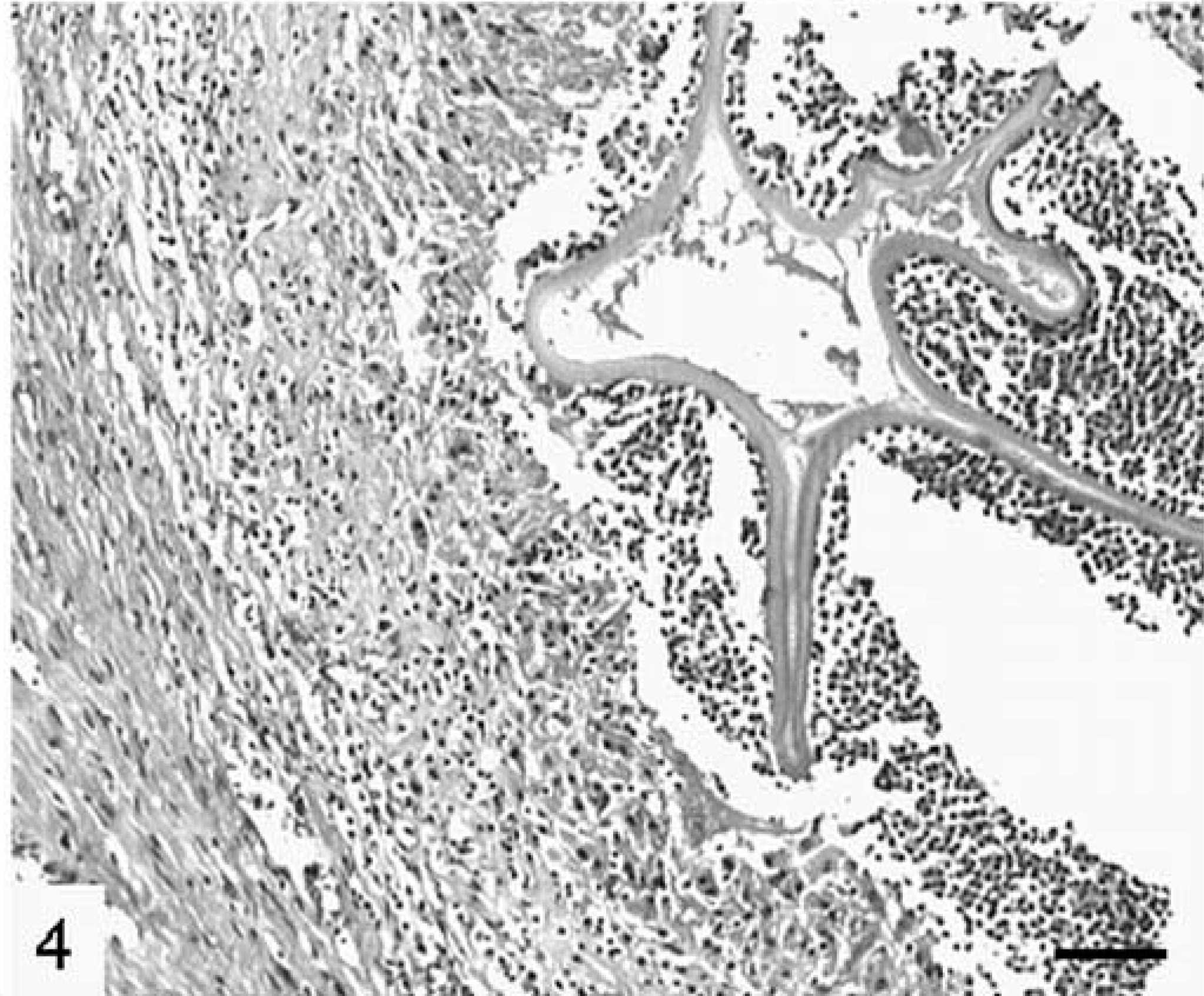
Liver; cynomolgus monkey No. 9. Collapsed cyst with a convoluted laminated layer surrounded by degenerated granulocytes, macrophages and connective tissue. Focal mineralization is present. HE. Bar = 100 µm.
Finally, in contrast to the described foci, which had none to moderate amounts of collagen, in a few foci collagen was the major feature (Fig. 5). In these foci, cell-poor and collagen-rich connective tissue surrounded parasitic structures composed of intact laminated layers and, in one case, protoscolices. Peripheral infiltration of these foci by lymphocytes and eosinophils was rare.
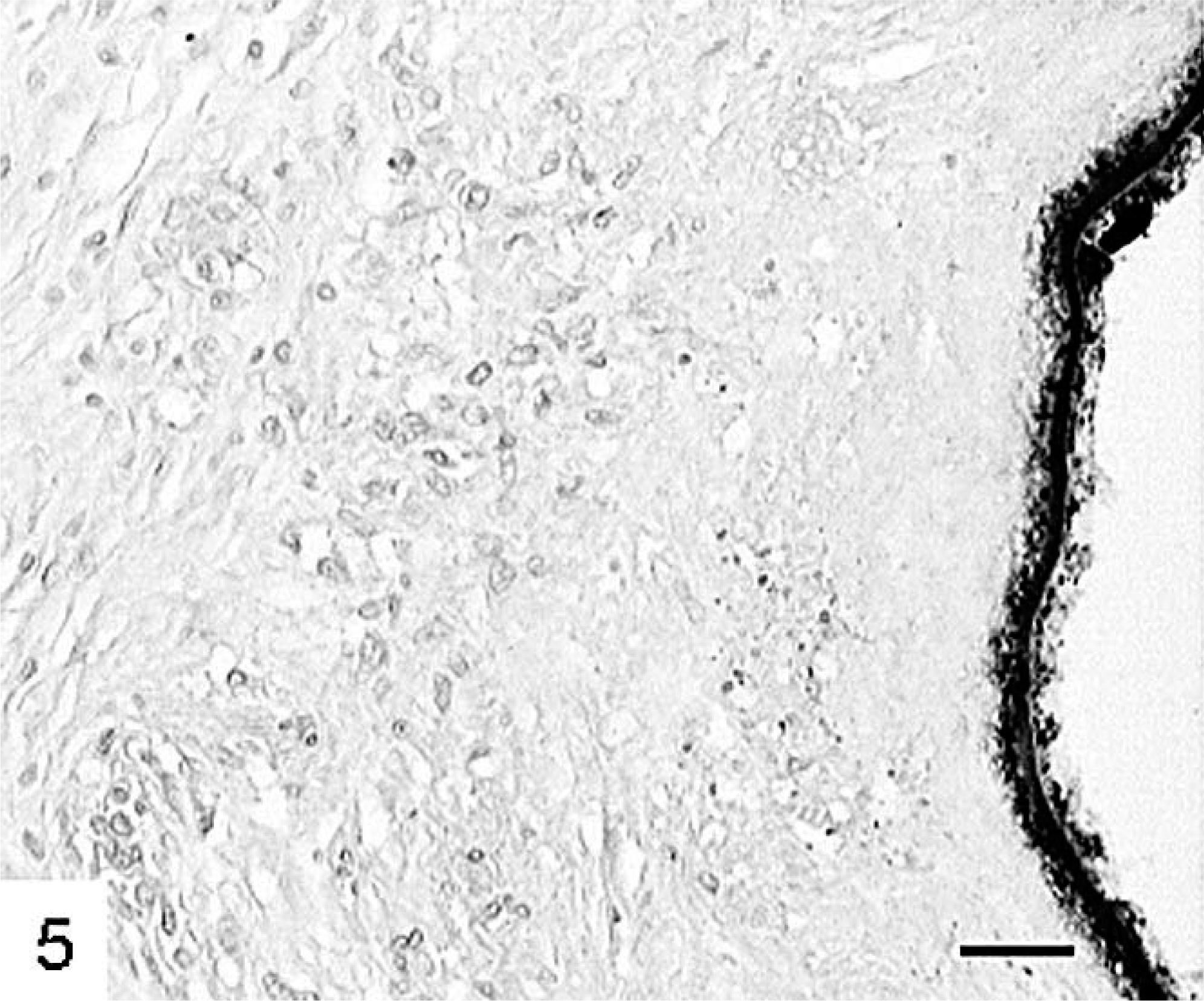
Liver; cynomolgus monkey No. 1. Collagen-rich and cell-poor connective tissue surrounding the germinal layer of a metacestode. Polyclonal anti–E. multilocularis antibody, avidin–biotin complex method. HE. Bar = 40 µm.
These various features of hepatic lesions were simultaneously present in different sites of all the individual animals. Occasionally, foci with a mixed pattern were observed within the same lesion. Mineralization was an uncommon feature in all types of lesions. Severe thickening of bile ducts due to fibrosis and epithelial proliferation, often with biliary hyperplasia and a peripheral infiltration of lymphocytes and eosinophils in the liver, was noted in some affected monkeys.
There was massive widening of the space of Dissé in three monkeys (Nos. 6, 7, and 9) attributable to an amorphous eosinophilic material, which was birefringent after staining with Congo red for amyloid. Immunohistochemically, this was identified as type AA amyloid (Fig. 6). In addition, intense granular staining was observed in the liver samples of six other monkeys (Nos. 1, 3–5, and 11–12) in individual macrophages, hepatocytes, or blood vessel walls but not in the space of Dissé.
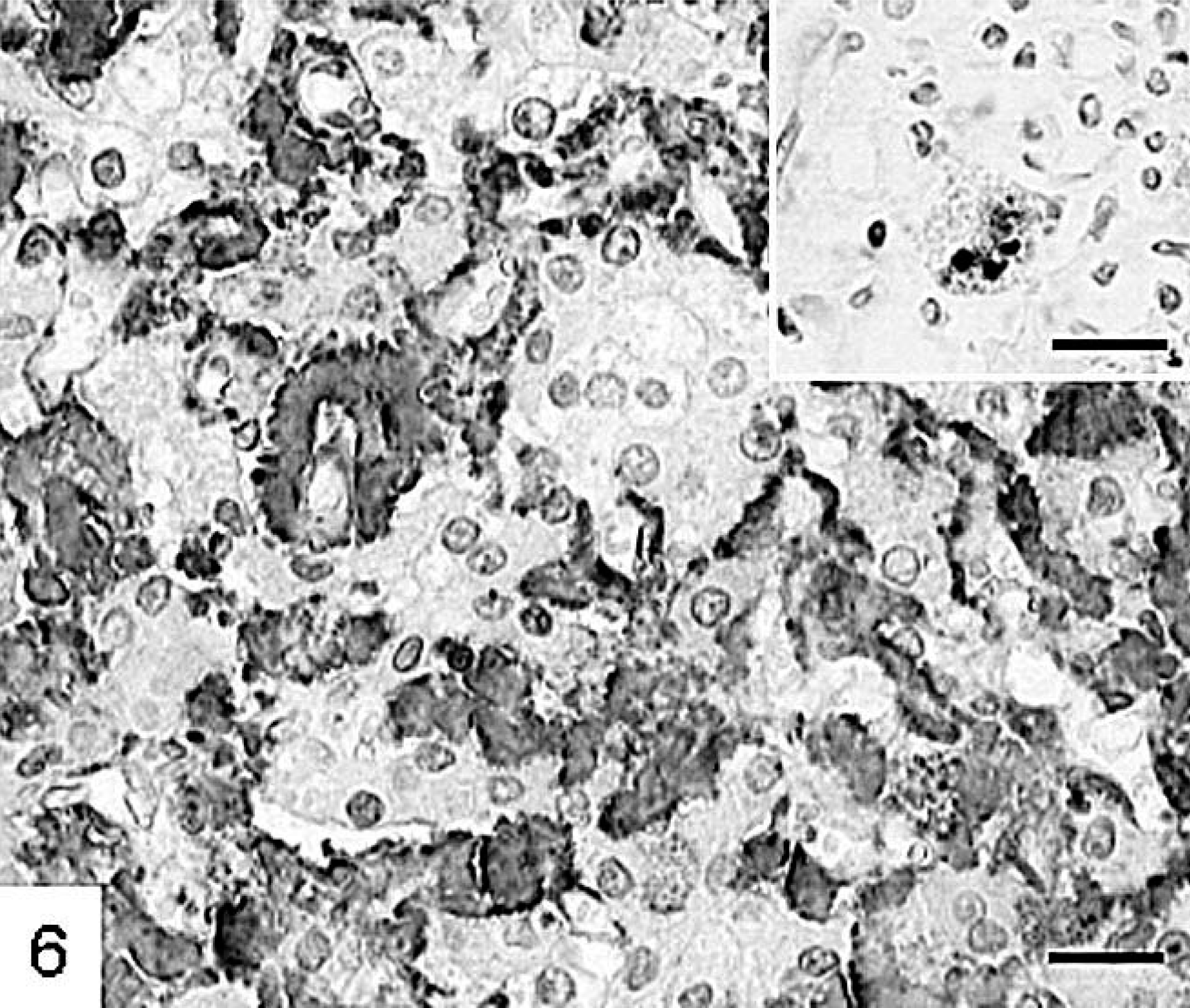
Liver; cynomolgus monkey No. 9. Type AA-amyloid deposition in the space of Dissé and intracellular in a macrophage (insert). Monoclonal antitype AA amyloid antibody, avidin–biotin complex method. HE. Bar = 25 µm, insert Bar = 30 µm.
The anti–E. multilocularis antibody stained parasitic structures with varying intensity. Staining of the outer laminated layer ranged from weak to moderate, whereas the germinal layer stained always strongly (Fig. 5). Germinal tissue usually abutted the laminated layer. Staining of protoscolices changed with advanced development. In early stages most cells were strongly positive, but in mature stages staining was usually restricted to the tegument and to the periphery of calcareous corpuscles (Fig. 7). Many macrophages contained granular material that exhibited positive staining (Fig. 8).
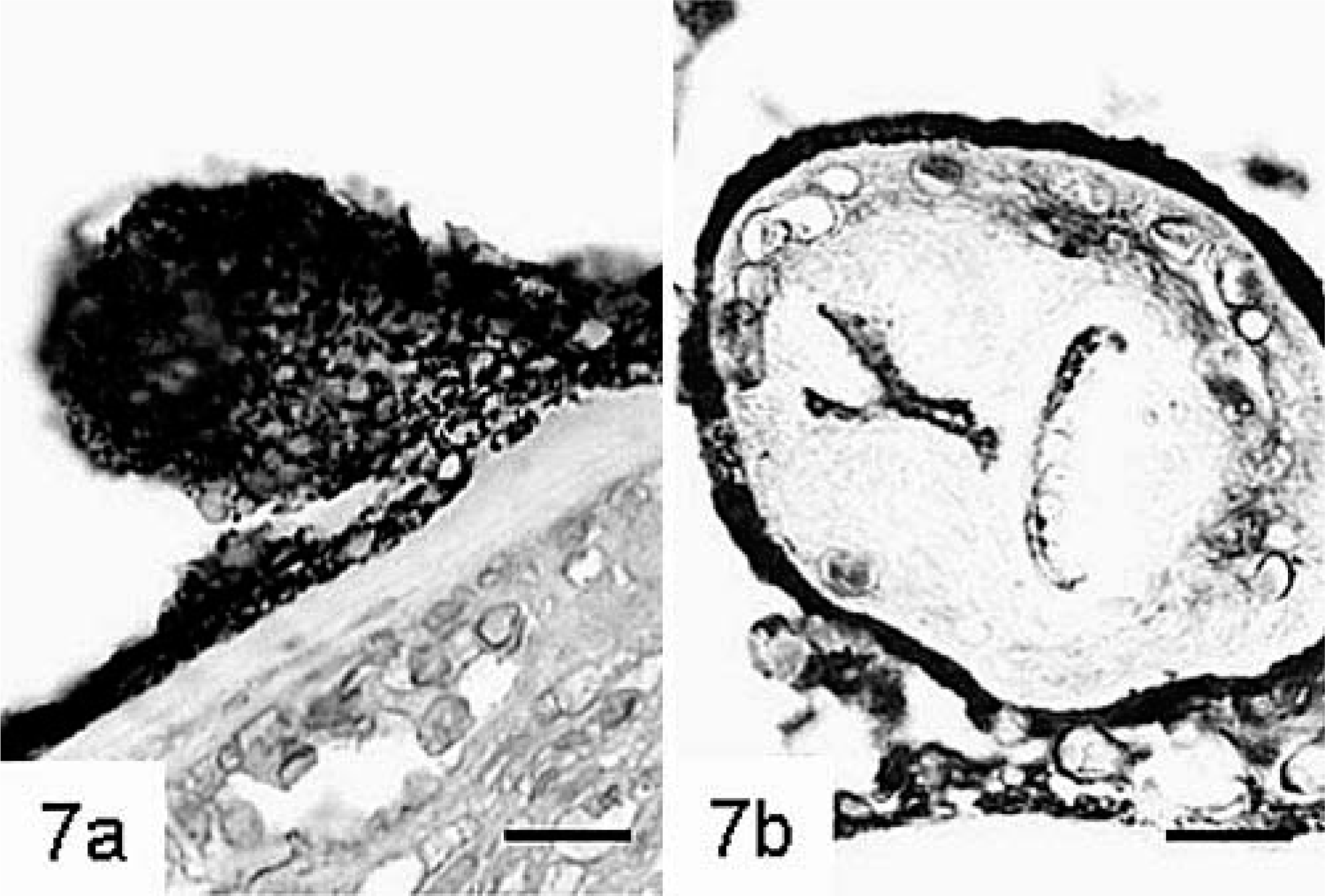
Liver; cynomolgus monkey No. 3.
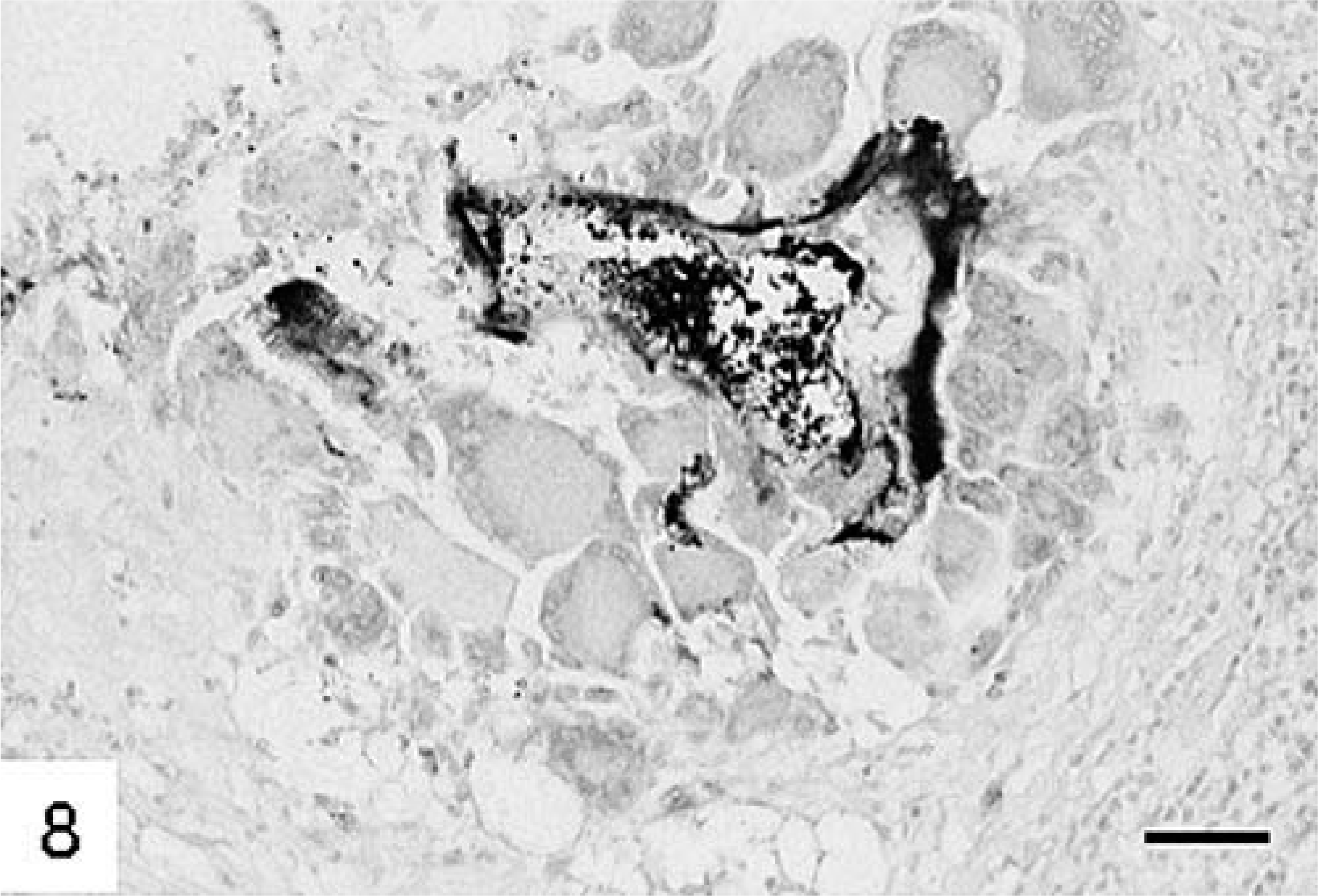
Liver; cynomolgus monkey No. 3. Macrophages contain metacestodal material. Polyclonal anti–E. multilocularis antibody, avidin–biotin complex method. HE. Bar = 70 µm.
All animals tested had serologic evidence of antibodies against the Em2-antigen. The titers ranged from 18 to 112 antibody units (AU, negative = 0 AU, positive = 1 AU, Table 1). The highest titer was observed in an animal with few cysts in the liver only, whereas the lower titers were present in animals with widespread disease and multiple organ involvement. A 2.6-kb band of E. multilocularis genomic DNA was amplified from all hepatic samples analyzed by PCR (data not shown).
Discussion
Alveolar echinococcosis was diagnosed in 12 cynomolgus monkeys at postmortem examination. Gross findings such as multiloculated cysts within the liver were usually characteristic. Histologically, the metacestode origin of these cysts became obvious, and because of their multiloculated and vesiculated nature, E. multilocularis infection was preliminarily diagnosed. In general, the hepatic lesions resembled those described for the natural intermediate hosts. 10
Three main patterns of tissue reaction to the metacestodes could be recognized. Whereas intact cysts were usually surrounded by mainly macrophages, collapsed and degenerated cysts were normally associated with a marked granulocytic infiltration. This infiltrate could represent the host's response to metabolized parasitic material. Alternatively, enzymes released from the granulocytes may attempt to dissolve the parasitic laminated layer and cause collapse of the cysts. Such an event was postulated in experimental infection of mice with E. multilocularis. 3 This hypothesis was also supported by other findings, where murine neutrophils were found firmly attached to the laminated layer of intact cysts. 2 However, in contrast to that study, in cynomolgus monkeys neutrophils were not a prominent feature in foci composed of intact cysts, which may possibly indicate that cyst wall lysis does not depend on neutrophilic enzymes. This hypothesis would match the findings obtained in an experimental murine system where neutrophil and macrophage chemotaxis appeared to have been inhibited by the parasite. 4 Because the samples were obtained from naturally infected cynomolgus monkeys, the duration of infection was impossible to know, and consequently any statement regarding the development of the lesions remains speculative.
Protoscolices were frequently present in lesions of alveolar echinococcosis of cynomolgus monkeys. This finding is in contrast to findings in humans and gorillas, in which infection is commonly sterile, and it was considered to reflect different host responses. Confirmation of this hypothesis would require further characterization of the immune response toward E. multilocularis infection in cynomolgus monkeys. The importance of cytokine production for progression or regression of disease was reported in humans. 11 Cytokines may also play a role in regulation of protoscolex formation from the germinal layer.
Severe hepatic amyloidosis was present in three animals, and deposition of type AA amyloid as a consequence of chronic infectious disease with increased production of serum protein AA was suspected. This was subsequently confirmed using immunohistochemistry. This technique additionally demonstrated intracellular type AA amyloid deposition in six other monkeys. Because other potential causes for increased serum protein AA synthesis were not evident at postmortem examination, these findings indicated that hepatic alveolar echinococcosis in cynomolgus monkeys may be frequently associated with amyloidosis, such as previously described in Alaskan patients. 1
Multiple methods were used to further confirm the identity of the parasite. Immunohistochemical demonstration of E. multilocularis using polyclonal antiserum is a straightforward method and especially suitable for the evaluation of postmortem specimens. Cynomolgus monkeys tested positive for anti-Em2 antigen when examined serologically, indicating that the presence of the parasite results commonly in antibody production in the monkeys. This suggests that this assay should be useful for antemortem detection of alveolar echinococcosis in cynomolgus monkeys. Serology for anti–E. multilocularis antibodies is described by the World Health Organization as a method of choice for the screening of larger human populations for exposure to the parasite. 9 Potentially this method could be used to investigate primate colonies as well. At the moment it is unclear whether the apparent inverse association between severity of alveolar echinococcosis and titer indicates a causal relationship. If this were the case, these results would appear to suggest that either higher titers may inhibit spread of the disease or that lower titers may be attributed to immunosuppressive activities of higher parasite masses. However, more cases need to be investigated before such a conclusion can be reached. PCR analysis of hepatic lesions demonstrated the usefulness of this technique particularly for small samples.
In summary, alveolar echinococcosis in cynomolgus monkeys was characterized by chronic granulomatous or pyogranulomatous hepatitis with varying involvement of other organs. Macroscopic and histologic findings are usually characteristic; however, if needed, various ancillary tests for confirmation of the etiology are available.
Footnotes
Acknowledgements
Parts of this work were supported by the Swiss National Science Foundation (grant no. 31-63615.00) and the EU EchinoRisk-project QLK2-CT-2001-01995 (BBW no. 00.0586-1).
