Abstract
This study investigated structural alterations and the immunohistochemical expression of androgen receptor (AR), estrogen receptor (ER), and progesterone receptor (PgR) in the mammary glands from surgically postmenopausal cynomolgus monkeys (
Introduction
Cynomolgus monkeys (
Materials and Methods
The cynomolgus monkeys ranged from 9 to 12 years old, approximating the life stage of women younger than 40 years old, 9 and had been part of a long-term study of osteoporosis (OVX monkey model). 5 The use of animals in this research complied with all relevant guidelines set by Kagoshima University. The animals were maintained in a filtered-air laminar-flow room at Shin Nippon Biomedical Laboratories, Ltd., and were given a commercial diet (Taklad Global Certified 25% Protein Primate Diet, Harlan Sprague Dawley Inc., IN, USA), and tap water ad libitum. The room temperature was maintained at 25°C ± 2°C and the relative humidity was at 55% ± 20%, with a 12-hour light-dark cycle.
Fourteen animals were divided into 2 groups of 7: one group underwent OVX, and the other group underwent a sham operation (sham). The in-life phase of the study was 18 months (78 weeks), and clinical signs were observed once weekly during this period. At 78 weeks, all animals were humanely euthanized and examined grossly. All major organs were saved and fixed in 10% neutral buffered formalin; however, specifically for this study, the mammary glands, uterus, and vagina were saved for histologic and/or immunohistologic evaluation. The pituitary and thyroid glands (included parathyroid glands), adrenal glands, kidneys, and uterus were weighed at necropsy.
All tissues were routinely processed and embedded in paraffin. Five µm sections were routinely stained with HE for histologic evaluation or were used for IHC studies. For IHC, slides were deparaffinized in xylene and rehydrated through graded alcohols. Endogenous peroxidase activity was blocked with 3.0% hydrogen peroxide in phosphate buffered saline (PBS) for 5 minutes, followed by antigen retrieval in 10 mM citrate buffer (pH 6.0) heated in the microwave. Samples were washed, and sections were blocked for nonspecific antibody binding with Block-Ace (Dainippon Sumitomo Pharma Co., Ltd., Japan) for 30 minutes. The sections were incubated overnight at 4°C with a diluted primary antibody: rabbit polyclonal AR (Novocastra Laboratories Ltd., Newcastle upon Tyne, UK, at 1 : 150), mouse monoclonal ER (Immunotech, France, clone 1D5 at 1 : 3), or mouse monoclonal PgR (Novocastra Laboratories Ltd., clone 1A6 at 1 : 200). After rinsing with PBS, the sections were incubated with the appropriate biotinylated secondary antibody for 30 minutes, washed with PBS, and incubated with a VECTASTAIN Elite ABC kit (Vector Laboratories, Burlingame, CA, USA) for 30 minutes. Immunoreactivity was visualized with 3,3'-diaminobenzidine tetrachloride that contained 0.02% hydrogen peroxide. The sections were then washed, counterstained, dehydrated, cleared in xylene, and mounted. The positive control for AR was the prostate from an intact adult male cynomolgus monkey, the positive control for ER was a mammary gland from the sham group, and the positive control for PgR was a uterus from the sham group.
IHC was evaluated by counting immunostained cell nuclei; nuclei were counted in both the acinus and the duct in 5 microscopic fields (1 field = approximately 0.0625 mm2) randomly selected. Results were expressed as a percentage of the total epithelial cell nuclei examined. The acinus and ducts in the microscopic fields contained approximately 100–450 and 50–200 epithelial cells, respectively. The mean differences were evaluated by the Student's
Results
At necropsy, the uterus was grossly smaller in the OVX animals than in those of the sham animals. This was consistent with a significant (
Effects of ovariectomy on the body and organ weights in monkeys. ∗
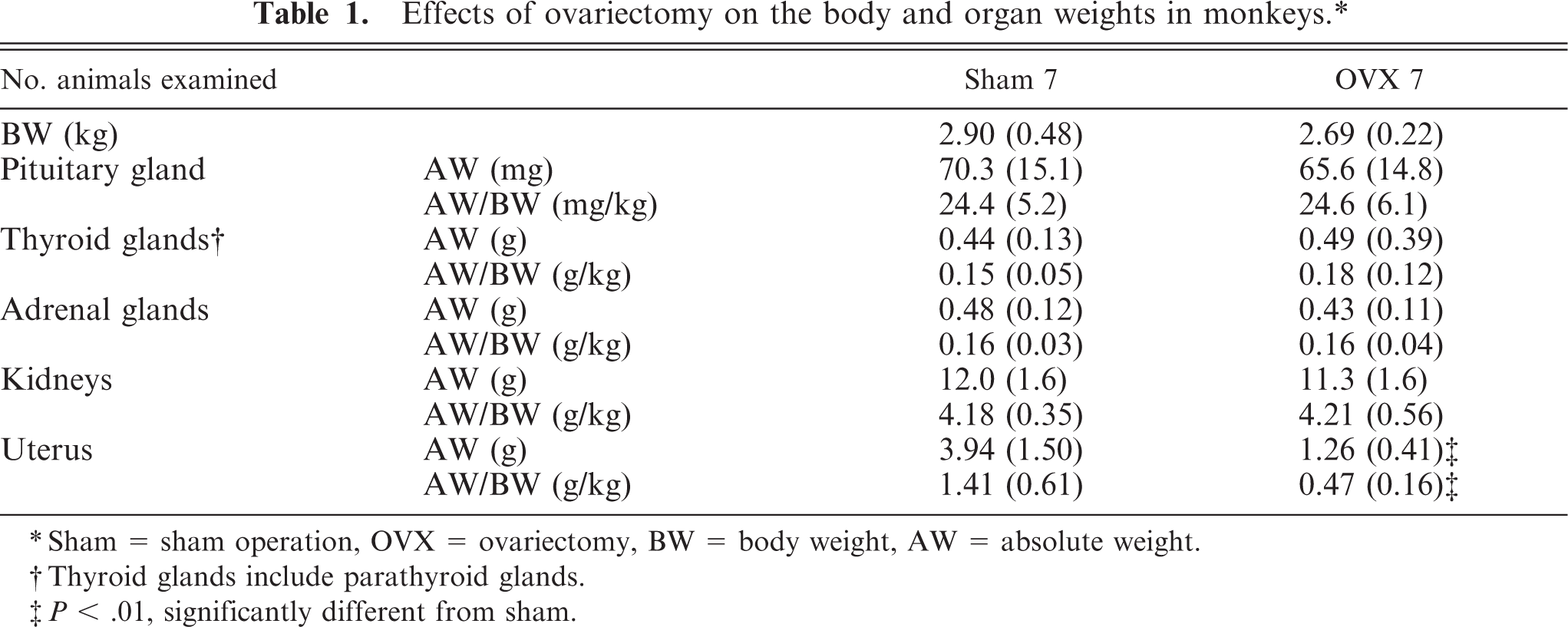
∗ Sham = sham operation, OVX = ovariectomy, BW = body weight, AW = absolute weight.
† Thyroid glands include parathyroid glands.
‡
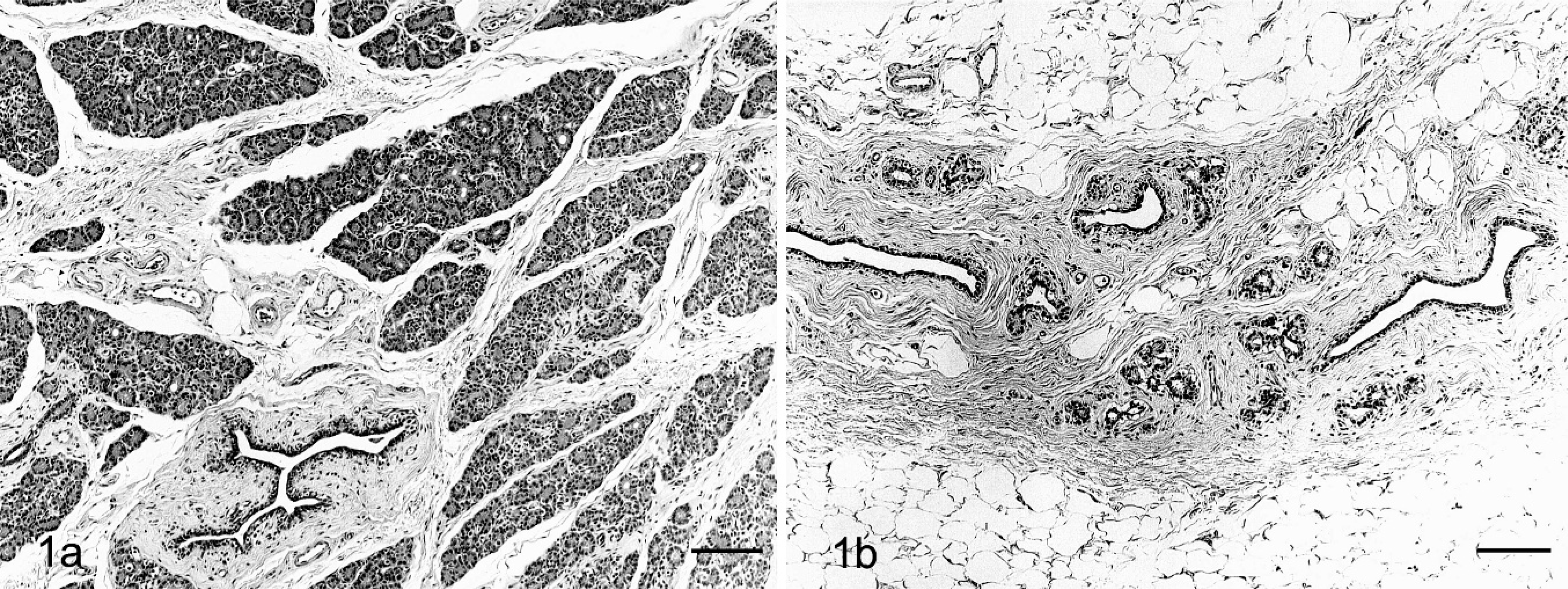
Mammary glands. HE. Bars = 200 µm. Fig. 1a. Sham monkey. No abnormal changes are seen. Fig. 1b. OVX monkey. Atrophy of mammary glands is seen; the cellularity and number of lobules are decreased, but the main duct is preserved; supporting connective tissue is decreased, and the stroma is replaced by adipose tissue.
IHC evaluation of AR, ER, and PgR in the mammary gland revealed that the percentage of AR-positive cells in acini and ducts was higher in OVX animals compared with sham animals, and the percentages of ER- and PgR-positive cells were lower in OVX animals compared with sham animals. The average percentages of AR-positive cells in the acini of OVX versus sham were of 25.6 (4.3)% versus 4.3 (1.5)%, respectively, and in the ducts were 12.5 (3.6)% versus 3.6 (2.1)%, respectively (Figs. 2a, b, and 3). The percentages of ER-positive cells in the acini of OVX versus sham were 43.6 (8.0)% versus 54.9 (9.0)%, respectively, and, in the ducts, they were 7.6 (2.6)% versus 16.0 (5.9)%, respectively (Figs. 2c, d, and 3). The percentages of PgR-positive cells in the acini of OVX versus sham were 8.6 (3.7)% versus 16.8 (8.6)% and in the ducts were 9.0 (3.2)% versus 15.4 (5.9)%, respectively (Figs. 2e, f, and 3).
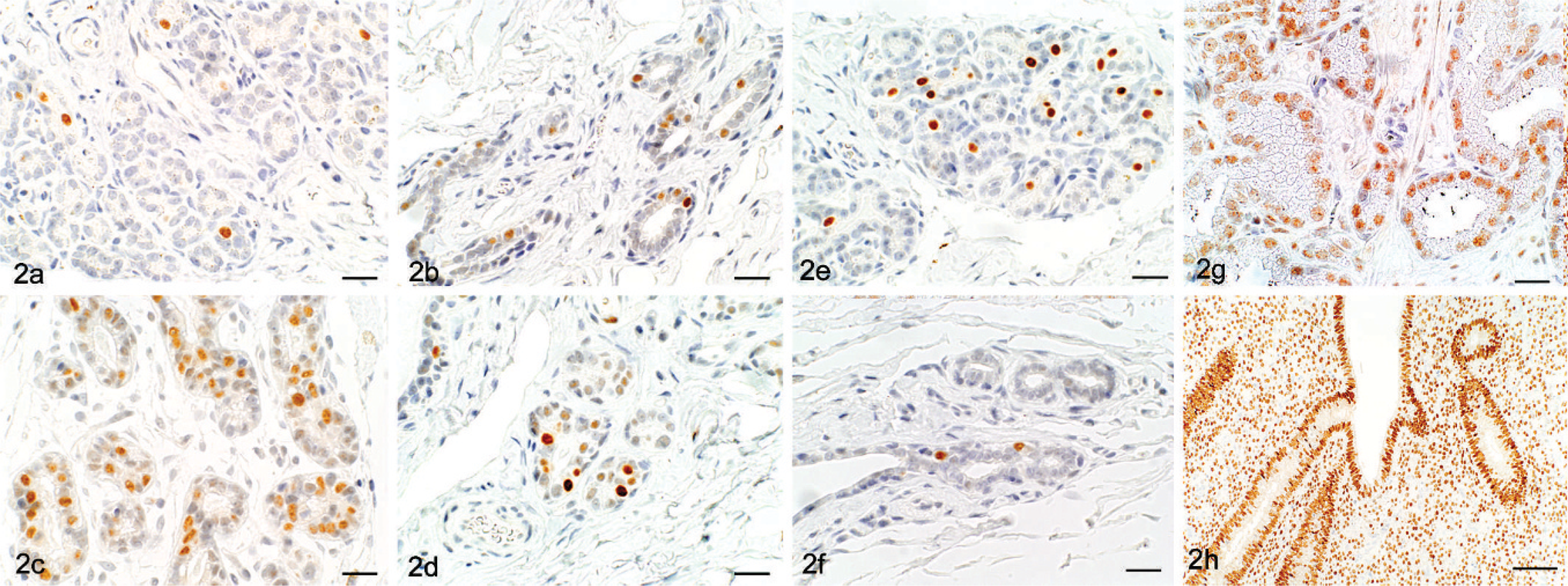
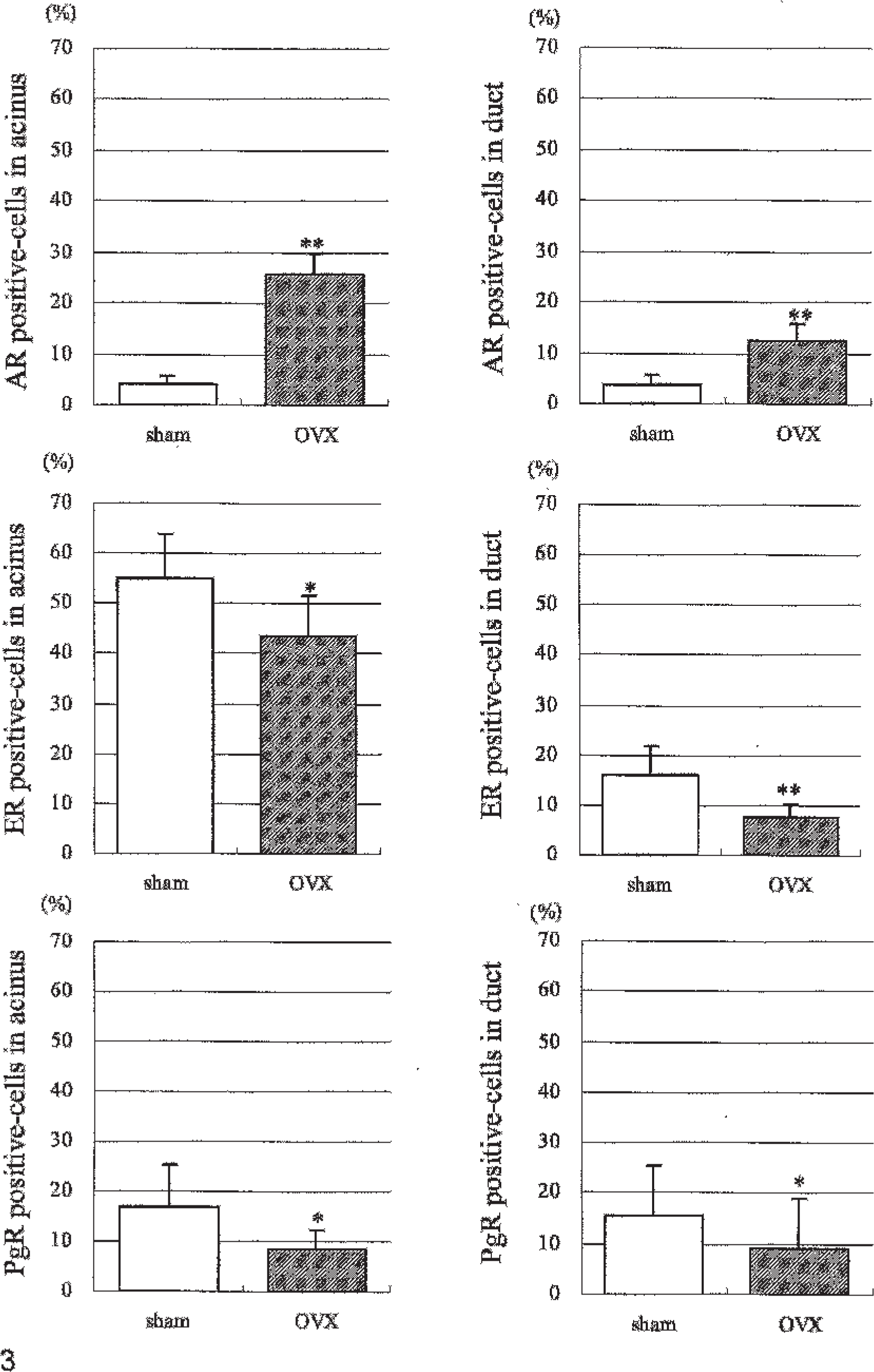
IHC examination. The proportion (%) of AR, ER, and PgR-positive cells in the acini and ducts of the mammary glands of sham and OVX monkeys. ∗
Discussion
In normal human-breast tissue, parenchymal changes during and after menopause reflect hormonal alterations in which serum estrogen and progesterone levels decline, although androgen levels, largely testosterone, are not diminished. As early postmenopausal atrophy of the human breast, general loss of acini with focal hyalinization of intralobular stroma is seen. 7 As postmenopausal involution progresses, the epithelial component completely atrophies and lobules become entirely hyalinized, forming hyaline nodules (ghost lobule). 7 The breast stroma also undergoes involutional changes in which alterations occur in elastic and collagen fibers, resulting in loss of supporting connective tissue, whereas adipose tissue increases. 7 The present study confirms that structural alterations in the mammary glands of OVX monkeys were very similar to early postmenopausal atrophy of the human breast.
IHC findings in the mammary gland of OVX monkeys yielded similarities in PgR and differences in ER between human postmenopausal mammary glands and OVX monkey mammary glands. PgR expression is less frequent in postmenopausal women compared with premenopausal women. 3, 8 Similar to this, PgR expression was lower in the mammary gland of OVX monkeys compared with sham monkeys. The mechanism of the postmenopausal PgR decrease in mammary gland is through reductions in serum estrogen. Normally, circulating estrogen binds and activates the nuclear transcription factor ER, and activated ER regulates the synthesis of PgR. 3
In contrast to the OVX monkeys, ER expression in the mammary gland of postmenopausal women has been reported to be higher than in premenopausal women. 2 In humans, the increase in ER-positive cells with age has been associated with declining serum estrogen levels. 2 We previously showed that there is a notable decrease in serum estradiol levels in OVX monkeys from 4–78 weeks after ovariectomy. 5 Therefore, the monkey mammary gland may not respond similarly to the human gland to reductions in estrogen, or there may be other factors in ER expression in the human mammary gland. Because this model of menopausal induction in monkeys is artificial, there may be hormonal functions in the young animal that are unrelated to the ovary and that are different from the natural aging process; naturally postmenopausal monkeys may have a different hormonal milieu than an OVX monkey. Evaluations of these differences would be beneficial for understanding the normal alterations in hormone receptor expression in pre- and postmenopausal women and monkeys.
Changes in the percentage of AR-positive cells in the mammary glands with aging are unclear in humans. 4 Although the expression of AR has been demonstrated in normal and cancerous breast tissue from postmenopausal women, 4 there is little information on AR expression in the postmenopausal human mammary gland. This study demonstrated that AR-positive cells increased in the mammary glands after OVX in monkeys. However, to date, the expression of AR in neoplastic monkey mammary gland lesions has yet to be reported. In humans, serum testosterone concentrations decrease after ovariectomy in women, 6 but decreases in monkeys have not been reported. Further investigation of circulating androgens in OVX monkeys and AR expression in neoplastic mammary gland lesions in cynomolgus monkeys would be valuable.
These results suggest that using an OVX primate model for hormone receptor expression has potential applications in basic human endocrinology, in particular, for the understanding of factors that contribute to hormone receptor expression in mammary glands. Further evaluations of receptor expression and circulating hormone levels in naturally postmenopausal monkeys, as well as receptor expression in mammary cancers in macaques, are an important next step for understanding the biologic relationship of this animal model to the human condition.
Footnotes
Acknowledgements
We thank Dr. K. Sato, Dr. M. Funato, Mr. S. Satake, Mr. T. Kodama, Mr. M. Souda, Mr. A. Muneoka, Mr. G. Martin, and M. Kaminishi for their valuable technical assistance. This work was supported, in part, by the Kodama Memorial Fund Medical Research.
