Abstract
Heritable, type-2 von Willebrand's disease (vWD) was studied in a line of German Shorthaired Pointers (GSPs) in which some members had a nucleotide variant in exon 28 of the von Willebrand factor (VWF) gene. A polymerase chain reaction (PCR) diagnostic test for the nucleotide variant was developed to establish the disorder's mode of inheritance and to eliminate it from the line. Thirty-six of the 49 GSPs in the line, 14 unrelated GSP controls, and 71 unrelated dogs of various breeds were tested for the presence of the variant nucleotide. All the dogs with a vWF antigen deficiency (<70% of normal) were either homozygous or heterozygous for the nucleotide variant. The variant was not located in any tested dog in the line or outside of the line with a vWF antigen value greater than 68%. Of the GSPs in the line tested, two were homozygous for the variant, 15 were heterozygous, and 19 were variant free. The collective evidence of this and other studies is consistent with the variant nucleotide being the cause of the type-2 vWD in this line of GSPs and German Wirehaired Pointers. The PCR diagnostic test for the variant nucleotide was successfully used to select and produce progeny that were variant free and vWD free. This test should be effective in the subsequent elimination of this same variant from other lines of dogs.
Von Willebrand's disease (vWD) is an acquired or inherited heterogeneous bleeding disorder common in humans and dogs, wherein there are quantitative deficiencies or qualitative abnormalities of the von Willebrand's factor (vWF) (or both). 1–5,9,10,12,16–19,22–29 Human and canine von Willebrand's factor genes (VWF) are about 85% identical in nucleotide sequence, and anti-human vWF antibodies cross-react with canine vWF, permitting the cross-species modeling of their vWDs. 3,7,20,28,29 This blood clotting factor is produced by the vascular endothelium, subendothelium, and megakaryocytes, and it stabilizes clotting factor VIII and aids platelet adhesion to the vascular subendothelium. 9,11,24 In plasma, vWF circulates as multimers of 1–20 subunits of the ∼300 kDa monomer, forming the largest protein molecules in blood. The larger the molecular weight multimer, the more effective its function appears to be. The subclassifications of vWD, types 1, 2, and 3, are on the basis of quantitative or qualitative differences (or both) in vWF. 9,11
The human VWF gene is located near the telomere of the short arm of chromosome 12 and is split into 52 exons, spanning 180 kb of deoxyribonucleic acid (DNA). 9,24 Most mutations that cause human type-2 vWD occur in VWF exon 28, a 1.4-kb-long sequence primarily encoding the mature vWF's A1 and A2 peptide domains. These two domains contain vWF-binding sites for platelet glycoprotein 1b/IX complex, sulfatides, collagen, and heparin. 9,11 Mutations in the VWF may result in the inhibition of vWF synthesis and release or the acceleration of its proteolysis (or both). Although important in blood clotting, partial or complete absence of vWF from blood is not always lethal to otherwise normal dogs, and vWD may go undiagnosed for years. 3
vWD is the most common inherited hemostatic disorder of dogs. 12 Canine type-1 vWD occurs in many breeds and has the greatest known prevalence. It is characterized by a uniform deficiency of vWF multimers and of total vWF. In the Doberman Pinscher breed, type-1 vWD is associated with VWF polymorphisms and appears to show a complex inheritance pattern, although a single mutation in the VWF gene appears to be necessary for the presentation of the disease phenotype (Venta et al., unpublished). 3,10
Type-3 vWDs are lethal, simple autosomal recessive disorders in the Shetland Sheepdog, Chesapeake Bay Retriever, Scottish Terrier, and Dutch Kooiker breeds, characterized by the complete absence of detectable vWF. 22,25,28,29 In the Scottish Terrier, the vWD is the result of single nucleotide deletion in the exon region encoding vWF prepeptide. 28 In Dutch Kooiker dogs, the variant VWF contains a splice site mutation in intron 16, which results in a splicing error in the exon region encoding for the vWF's propeptide. 22
Type-2 vWD is associated with a deficiency of plasma vWF and its large multimers. The mode of inheritance in German Wirehaired Pointers (GWHP) has been characterized as simple autosomal recessive, with vWF antigen assay percentage (vWF:Ag) of <1, as determined by enzyme linked immunosorbent assay (ELISA). 1,4,5,27 Individual cases of type-2 vWD, without evidence of inheritance or the identification of a mutation, have been reported in two German Shepherd dogs, a Shetland Sheepdog, a Pembroke Welsh Corgi, a Miniature Pinscher, a Rottweiler, and a mixed breed dog. 14 The vWF:Ag values of these individual vWD-affected dogs ranged from 9.6 to 76.9 (with the normal range defined as 100, SD 23.7). In another report, three female German Shorthaired Pointers (GSP) were reported to have type-2 vWD. Rocket immunoelectrophoresis vWF assay values of 20, 12, and 19 were reported for these dogs, with the author's normal range stated as 50–160. 12 Abnormal bleeding occurred in two of the dogs (those with 20 and 12) but not in the third dog that was a sister of the clinically affected dog with the vWF:Ag of 12. There was no other evidence that the disease was inherited.
One of us (J. W. Kramer) was aware of a GSP that had been diagnosed with vWD on the basis of an abnormal bleeding incident and a vWF:Ag value of 1. Other members of this line were discovered to be deficient in vWF:Ag. We hypothesized that the vWD in this line of GSPs was the result of a VWF mutation in exon 28, where most mutations causing type-2 vWD in humans are found. 9,24
We proposed to establish the subtype of vWD observed in this line of GSPs by vWF multimer analysis and its pattern of inheritance in the line and to determine whether vWD-affected dogs had one or more nucleotide variants in exon 28 of the VWF gene that might account for the disease. If a VWF nucleotide variation was identified, we further proposed to develop a polymerase chain reaction (PCR) diagnostic test to determine whether the variant cosegregated with the vWD phenotype and decreased vWF:Ag values and to further determine the test's utility for selecting dogs free of the variant VWF and vWD.
Materials and Methods
Subjects
The proband, a 1-year-old female GSP with an episode of persistent bleeding from skin scratches, was presented to the Veterinary Teaching Hospital of Washington State University. The vWD diagnosis was on the basis of a normal coagulation time, normal platelet concentration, a prolonged bleeding time, and a vWF:Ag value of 1. The proband's owner and breeder requested assistance in clearing his line of GSPs of the disorder.
Breeders and owners provided the records of members of the proband's pedigree. Owners were interviewed to establish the health of their dogs and to obtain their consent for inclusion in the study. When an owner reported observing abnormal bleeding and had consulted a veterinarian, the attending veterinarian also was interviewed. No vWD-affected dogs or potential carriers were prospectively bred for the purpose of the study. Type-2 vWD was defined as having abnormal, persistent bleeding episodes and deficiencies of large–molecular weight vWF multimers and plasma vWF:Ag.
ELISA vWF:Ag assay
Venous blood samples were drawn directly into citrate or ethylenediamine-tetraacetic acid anticoagulant. The plasma was isolated immediately by centrifugation and frozen. The frozen sample was sent to a commercial veterinary laboratory for vWF:Ag determination (Comparative Coagulation Section, Diagnostic Laboratory, Cornell University, Ithaca, NY). The interassay coefficient of variation for the ELISA has been reported to be 4, with a lower limit of detection of 0.1. 1,3 The vWF:Ag value of 100 was established by determining the vWF:Ag of a pooled plasma sample from clinically normal dogs. The commercial laboratory's normal canine range is defined as 70–180% of the pooled sample. The lower the vWF:Ag value, the greater is the risk of hemorrhage. The vWF:Ag intra- and interassay reproducibility are good, but the value of a single sample assay at the carrier to normal threshold is limited by the individual dog's daily and weekly vWF:Ag variation. 3,17,27
Mutimeric analysis
Plasma vWF multimeric analysis was performed to establish the line's vWD subtype. Citrated blood samples were collected from the proband's vWD-affected sibling, sire, and a VWF variant–free GSP dog; the plasma was collected by centrifugation, immediately frozen at <−20 C, and analyzed 2 weeks later. Control plasmas from a variant-free dog, a normal individual, and an individual with type-1 vWD were assayed in parallel with the two canine subjects of the line.
vWF multimeric analysis was performed by a commercial medical laboratory (Quest Diagnostics Inc., San Juan Capistrano, CA) by a modification of the Brosstad method. 4,6,30 Briefly, the plasma vWF multimers were separated by sodium dodecyl sulfate agarose electrophoresis and transferred to a nitrocellulose membrane. A chemiluminescent immunostaining procedure was used to detect the vWF multimers in which rabbit, anti-human vWF immunoglobulin G (IgG), and a secondary goat anti-rabbit IgG conjugated to luciferase were used. Anti-human vWF antibodies cross-react with canine vWF multimers. 4,13 Results were recorded with a digital camera and the multimer bands counted. No attempt was made to quantify the individual vWF bands.
Sequence analysis
Cells for DNA analyses were collected from brushings of the proband's buccal epithelium and eluted from the brush. 21 A 1,334-bp section of the canine VWF gene, corresponding to most of human exon 28, was PCR amplified using two primers: P1, 5′GTCATTGTGATGGTGTCAACTT and P2, 5′AGCACCTCTGTAGCACCA. The targeted DNA was amplified in a 25-µl reaction using 50 mM KCl, 10 mM Tris buffer (pH 8.3 at 20 C), 1.5 mM MgCl2, 100 µM diethylnitrophyenyl thiophosphate, 10 µM of each primer, and 0.25 U of Taq DNA polymerase. Cycling conditions were: 1 minute, 94 C; 2 minutes, 63 C; 3 minutes 72 C, repeated for 50 cycles (PTC-100, MJR Research Inc., Watertown, MA). The amplified product was separated on a 1% agarose gel and purified using a commercially available kit according to the instructions of the manufacturer (Qiagen, Chatsworth, CA). The purified PCR product was directly sequenced using a Thermo Sequenase cycle sequencing kit according to the instructions of the manufacturer (USB Corp., Cleveland, OH). The proband's sequence was compared with the known normal sequence of this region (GenBank accession numbers AF099154, L76227, U66246, and L16903).
Diagnostic test for the variant
A variant nucleotide was identified in the proband's VWF, resulting in a Bsp1286 I restriction site that is absent in the normal allele. Genotype determination from the product amplified with primers P1 and P2 was difficult. Therefore, a third primer (P3, 5′GCGAGGTCCTACAGCAGGT) that pairs with P2 was designed to produce a smaller 333-bp PCR product for a diagnostic test. Digestion of the amplified product produced a 203-bp band common to all genotypes and a 130-bp band for the normal allele, which was further reduced to 69- and 61-bp bands for the PCR product containing the nucleotide variant found in the proband. Cycling conditions were 94 C, 1 minute; 57 C, 1 minute; and 72 C, 1 minute, for 50 cycles. AmpliTaq Gold (PE Biosystems, Foster, CA) was used to suppress some extraneous bands that were observed when standard Taq DNA polymerase was used for PCR amplification. All other conditions were the same as those described above for the identification of the proband's VWF nucleotide variant. Band sizes were determined on 5% polyacrylamide gels by comparison with a commercially available DNA size standard.
Cheek swabs were used to collect DNA samples from members of the proband's pedigree and 14 unrelated GSP. Samples were also collected for vWF:Ag from the related dogs and four unrelated GSP dogs. Seventy-one DNA samples banked in the laboratory from a previous study of 10 other breeds (six Cocker Spaniels, seven Greyhounds, four Doberman Pinschers, eight Siberian Huskies, 10 Labrador Retrievers, eight Collies, 10 Scottish Terriers, eight German Shepherd Dogs, 10 Beagles, and seven Pointers) were surveyed for the nucleotide variant to determine whether it might be a common polymorphism.
Statistical analysis
The independence of the amount of vWF:Ag and genotype was tested using the Wilcoxon rank-sum test.
Results
Dogs
Of the nine vWD-affected GSPs identified, only two, the proband and a female sibling, were alive when the VWF nucleotide variant was identified and the DNA-based diagnostic test became available. No sex predisposition for vWD was noted. Some members of families 1 and 2 (Fig. 1) were whelped before the beginning of the study, and not all owners could be contacted. During the study, owners and breeders were informed of the status of their dog's test results as soon as the information became available. Owners of dogs with vWF:Ag values lower than the normal dog range, <70, were counseled not to breed their dogs. Despite the counseling, families 3, 4, and 6 were produced by one or more parents with the VWF nucleotide variant identified in the proband.
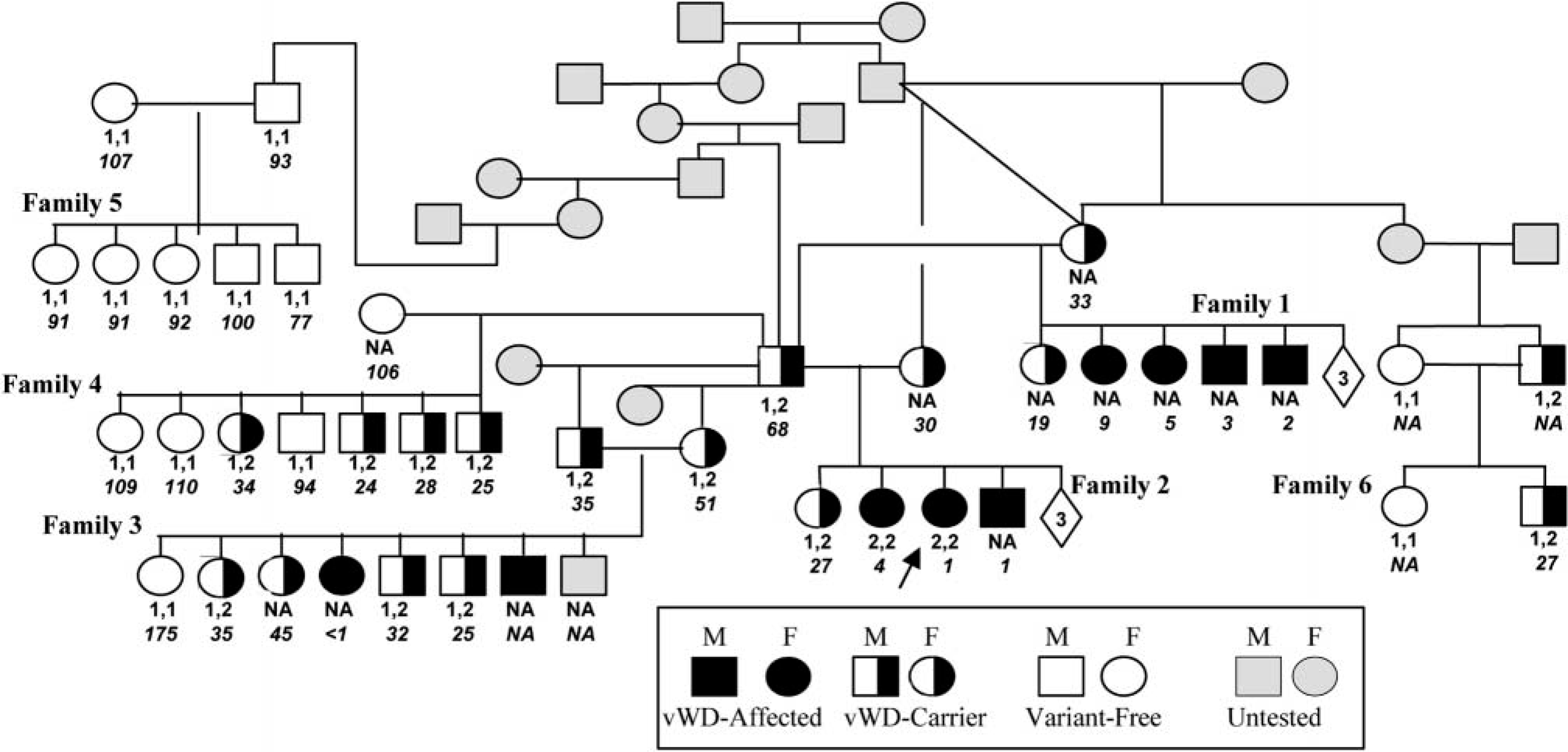
The pedigree of a vWD-affected line of GSP with genotypes, phenotypes, and vWF:Ag concentrations. The arrow points to the proband. Only dogs for which a history was available are included. Numbers enclosed in diamonds indicate the number of unavailable siblings in the litter. Status is based on clinical history, vWF:Ag, or genotype (or all). The normal nucleotide is indicated by 1 and the VWF variant nucleotide by 2. Allele genotypes (e.g., 1,1) are shown directly below the symbols for each dog. The vWD:Ag values are italicized below the genotypes. NA indicates that a sample was not available for factor or genotype analysis (or both).
vWF multimer analysis
Multimer analysis of plasma of normal variant-free GSP (No. 5-P; family 5, parent) (Table 1) revealed a normal distribution of vWF multimer for dogs. 3,13 In contrast, large vWF multimer deficiencies were observed in both the vWD-affected siblings (No. 2-2; family 2, second offspring from the left in Fig. 2) and its carrier sire (No. 2-P) (Table 1) serving to define the GSP's vWD as type 2.
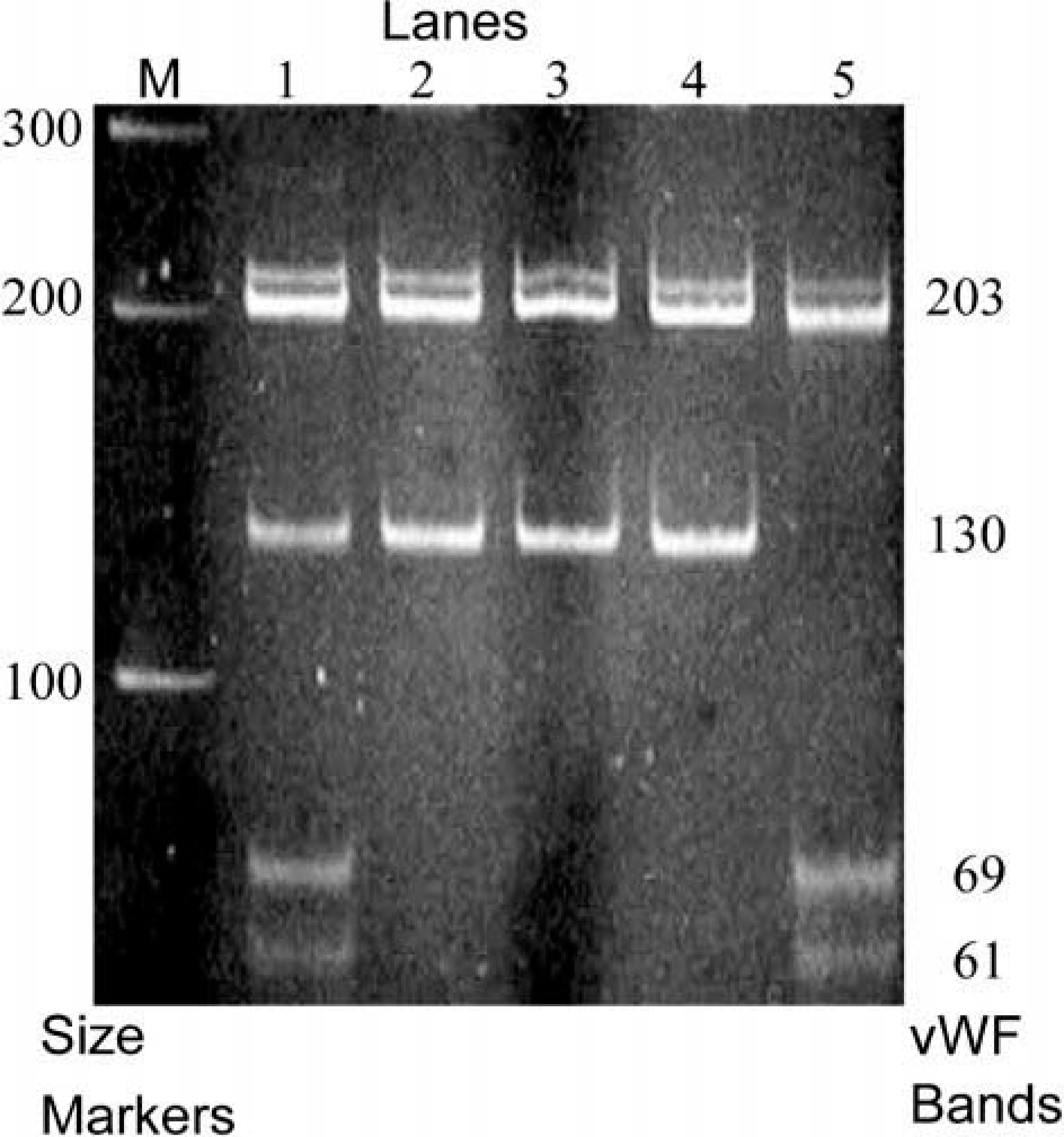
Acrylamide gel electrophoresis banding patterns. Lane M is a 100-bp ladder marker lane. The DNA sizes shown on the left of the figure represent the size marker bands in lane M and those on the right represent VWF band size markers. As with Fig. 1, the VWF variant allele contained a Bsp1286 I restriction site and was designated 2 and the normal allele 1. Lane 1 represents the heterozygous, (1,2), vWD-carrier dog No. 2-P and lanes 2–4 the three normal variant-free (1,1), dogs. Lane 5 represents the homozygous (2,2) vWD-affected dog No. 2–3. A common 203-bp band occurs in all the numbered lanes. The normal allele is represented by the 130-bp band in lanes 1, 2, 3, and 4 but is absent from lane 5, which represents the vWD-affected dog. In lanes 1 and 5, the digested variant allele is represented by the 69- and 61-bp bands, indicative of the VWF N883S substitution.
∗ Absent from Fig. 1. Not all of the dogs in this list appear in Fig. 1. Some were too distant from the proband to include in the figure.
† Variant status is based on clinical history, vWF:Ag, and/or genotype.
‡ Genotype abbreviations NA = not available for testing; 1,1 = variant free; 1,2 = heterozygous for the VWF variant; and 2,2 = homozygous for the VWF variant.
§ The individual dog's number consists of the family number followed by a hyphen and then the number corresponding to the dog's position from the left in its family in Fig. 1. i.e., Dog 2–3, the litter proband, the third sibling in family 2, and is marked by an arrow. The letter P denotes a parent of the litter.
VWF nucleotide analysis
The sequence analysis of VWF exon 28 from the clinically affected type-2 vWD proband and its sibling in family 2 (Fig. 2) revealed a homozygous nucleotide variant compared with the previously determined canine sequences (Fig. 3). PCR amplification and restriction enzyme digestion (Bsp1286 I) of the product produced a 203-bp band common to all genotypes, and a 130-bp band for the normal allele. The 130-bp band normal allele was reduced to 69- and 61-bp bands for the proband's VWF variant allele. Of the 34 dogs genotyped, only the two GSPs homozygous for the variant allele (vWD affected) had clinical bleeding episodes (Fig. 1, family 2). The variant allele does not appear to be a common polymorphism because it was absent from the 14 unrelated GSPs and the 71 dogs of the 10 other breeds examined.
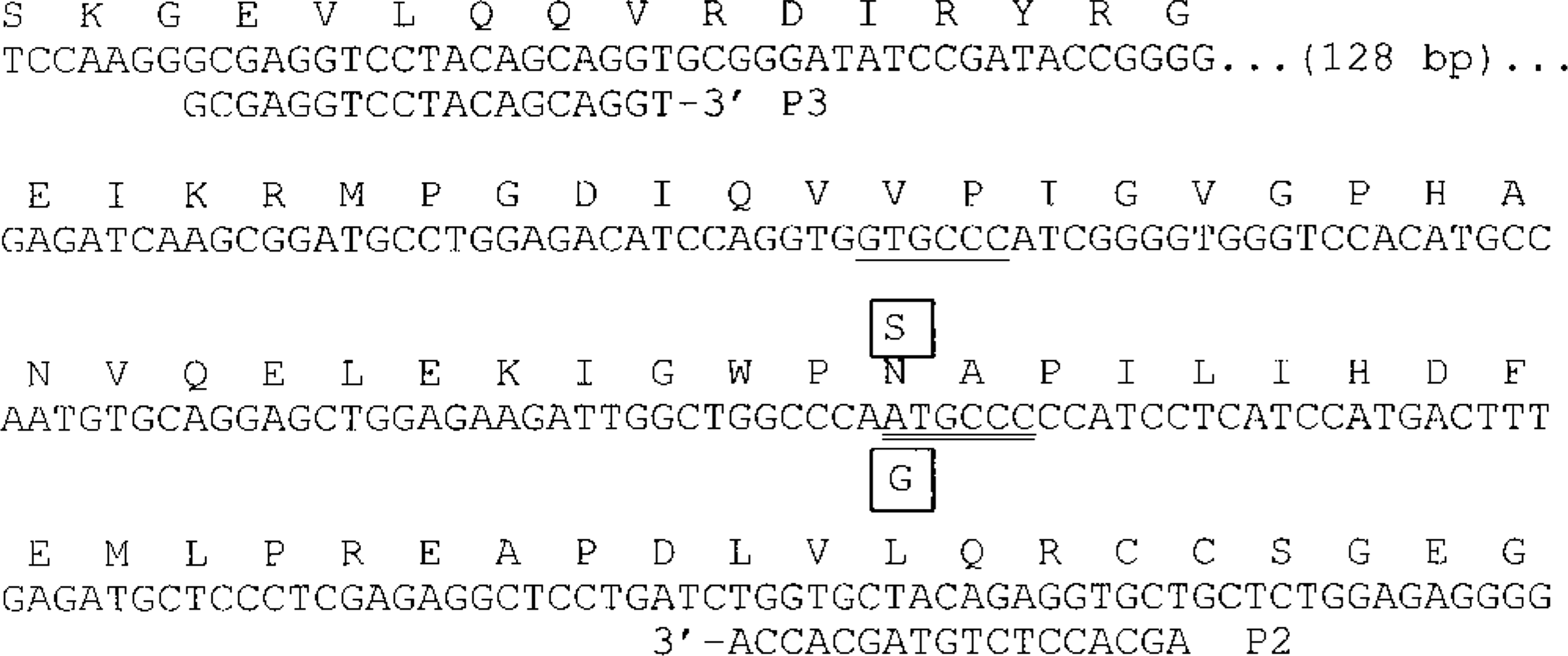
Location of GSP nucleotide variant found in vWD-affected GSPs and PCR primers used in the diagnostic test. The canine nucleotide sequence for approximately the last half of the encoded vWF A2 domain is shown, with the inferred, single letter abbreviation amino acid sequence given above it. The boxed S is the inferred amino acid substitution caused by the G substitution (also boxed) that was found in vWD-affected GSPs. This same mutation was independently discovered in vWD-affected GWHP by van Oost and colleagues and reported at a scientific meeting. The nonvarying Bsp1286 I restriction enzyme site (GDGCHC [where D = G, A, or T, and H = C, A, or T]) that is common to all dogs studied is underlined, and the diagnostic Bsp1286 I site, which occurs only in the variant allele, is double underlined. The PCR primer sequences (P2 and P3) used in the diagnostic test are shown below their respective primer target sequences.
vWF: Ag analysis
The four unrelated variant-free GSP control's vWF:Ag values were 93, 105, 117, and 131. All the variant-free GSPs of this line had vWF:Ag values of 69–175 (mean 106, SD 24). All but one variant-free GSP's vWF:Ags were within the commercial laboratory's range of a normal canine population (70–180) and normal GWHPs (mean 104, SD 20) (Table 1). 4 The vWF:Ag threshold between heterozygous and homozygous (normal) for the VWF variant allele in the GSPs in this study occurred between 68 and 69. All 19 GSPs heterozygous for the VWF variant had vWF:Ag ranging from 19 to 68 and were inferred to be vWD carriers (Table 1). The vWF:Ag of eight of the nine vWD-affected GSPs identified was <10 (Table 1). The ninth vWD-affected GSP (No. 3-7) was not tested.
Variant VWF diagnostic test
The results of the Wilcoxon rank sum test were consistent with a significant association (P < 0.001) between the vWF:Ag values and the presence of the variant allele, as determined by the PCR diagnostic test developed to distinguish the variant-free GSPs from vWD-affected and vWD-carrier GSPs.
Clinical history
Thirty-four of the 49 members of this GSP line were available for testing for both the vWF:Ag and the VWF variant (Table 1, Fig. 1). The absence of an apparent decrease in litter size at birth of litters containing the variant VWF suggests that no prenatal losses occurred because of the variant allele. Conventional partial amputation of the tails and dewclaws of 1-week-old puppies resulted in no reports of abnormally long bleeding episodes. Reports of persistent spontaneous bleeding episodes were confined to GSPs homozygous for the variant VWF or with a vWF:Ag <10, or both.
Of the nine vWD-affected GSPs identified, seven died before the variant VWF was discovered and the PCR diagnostic test developed (Table 1, Fig. 1). Two vWD-affected dogs were available for the study—the proband, No. 2-3, and its vWD-affected female sibling, No. 2-2. These two vWD-affected dogs were 9 years old at the time of this report and, with the exception of the vWD, are in good health. The proband was diagnosed with vWD at 18 months of age on the bases of persistent bleeding from minor skin abrasions and a vWF:Ag of 1. The proband's female sibling underwent an unremarkable ovariohysterectomy at 18 months of age. At 8 years of age, vWD was diagnosed after surgical removal of an anal gland adenocarcinoma was complicated by excessive bleeding. The owner of dog No. 2-2 reported that minor skin scratches resulted in prolonged bleeding that stopped within 24 hours.
Only three of the nine owners of the vWD-affected GSPs recognized a bleeding disorder before a veterinarian diagnosed vWD after attending a major bleeding episode in which routine hemostatic procedures failed. These bleeding episodes were associated with severe traumatic incidents, such as automobile accidents or ovariohysterectomies. Death generally occurred before the vWF:Ag results became available (Table 1). Of the nine vWD-affected GSPs, one, No. 3-7, vWF:Ag was not measured (Table 1). This dog's vWD was diagnosed on the basis of a history in which routine clinical hemostatic care failed to control hemorrhage arising from a traumatic incident and from its vWD-carrier parents producing a litter (Fig. 1, family No. 3) containing one other vWD-affected puppy and four vWD-carriers.
In general, hemorrhage associated with small wounds was slow to clot but self-limiting. When trauma, such as surgery, resulted in large wounds, routine hemostatic practices by a veterinarian were frequently unsuccessful.
Genetics
The VWF variant allele was observed in these families in ratios consistent with the Mendelian segregation of a simple, autosomal recessive trait (Fig. 1). Family 3's vWD-carrier parents produced seven offsprings with an observed variant-free:vWD-carrier:vWD-affected ratio of 1:4:2, a ratio consistent with the expected segregation ratio, 1:4:2 or 2:4:1, of a simple autosomal recessive trait. Family 4, a backcross of vWD-carrier:variant-free parents, contained seven siblings, three variant-free and four vWD-carriers, with ratio of 3:4. This is again a ratio consistent with the expected ratios of 3:4 or 4:3 of a simple autosomal recessive trait in a litter of seven. Family 5's variant-free parents produced only variant-free offspring, as would be expected with a simple autosomal recessive trait.
Discussion
The bleeding disorder in this line of GSPs with deficiencies of large vWF multimer and vWF:Ag associated with a VWF variant nucleotide in exon 28 with a high level of statistical confidence established the disorder to be an inheritable type-2 vWD. When this study of GSP was nearing completion, the identical VWF variant nucleotide was reported to be associated with type-2 vWD in GWHP. 8,15,29 Thus, the clinical history and deficiencies of multimers and vWF:Ag and the discovery of the variant VWF nucleotide in vWD-affected dogs in one study independently corroborates the other study.
The founder effect is known to be the cause of a relatively high breed-specific incidence of some inherited canine diseases. 19 In Germany, in the 19th century, the GWHP breed was established by crossbreeding GSPs with members of other established breeds. This crossbreeding may have resulted in passage of the variant vWF from parent GSP stocks to the GWHP breed.
The collective results from this study and others are consistent with a single nucleotide defect in which the identified VWF variant nucleotide is the cause of type-2 vWD observed in GSPs and GWHPs. 4,8,12 The evidences supporting this conclusion are: the location of the variant amino acid within the VWF A2 domain is in the same general location in which most human type-2A vWD mutations occur, lack of the commonality of the VWF variant allele in breeds other than the GSP and GWHP breeds, the complete concordance of the vWF:Ag concentration with the variant nucleotide's presence, and the observed results of the population genetics of the disorder in GSP and GWHPs. 1,11,29
The vWF:Ag assay continues to be a valuable screening test for type-2 vWD when there is hemorrhage and a clear threshold between the vWF:Ag of vWD affected and vWD carriers. 1,5,17 In contrast, there is less confidence in separating vWD carriers from variant-free dogs because the vWF:Ag threshold for GSPs and GWHPs, 68–69 and 68 or 71, respectively, is small, and temporal variations confound the ability to determine an individual dog's genotypic status with a single vWF:Ag test with a high degree of accuracy. 1,5,17 Temporal variations should not limit a PCR-based variant VWF diagnostic test.
Further research is needed to establish the importance of this vWF variant allele's function in these two breeds. It is unlikely that concerns over intellectual property rights would prohibit further research on this canine VWF variant by a wide range of researchers. The original sequence of the canine VWF exon 28 was publicly available many years ago (GenBank accession number L16903), and the variant nucleotide was publicly announced several years ago. 8
The variant VWF diagnostic test developed herein successfully tracked this variant allele in the line of vWD-affected GSP and provided the basis for the selection of variant-free parents to produce vWD-free progeny. The prevalence of the type-2 vWD variant VWF described herein in the general GSP and GWHP and the canine population at large remains to be established. This PCR-based test for the variant VWF nucleotide will be useful in the detection of this same variant in the general population of the GSPs and GWHPs and the other breeds of dogs.
Footnotes
Acknowledgements
We thank the GSP owner and veterinarian participants, and Dr. Jane Wardrop for her comments.

