Abstract
Three horses died as a result of eating grass hay containing summer pheasant's eye (Adonis aestivalis L.), a plant containing cardenolides similar to oleander and foxglove. A 9-year-old thoroughbred gelding, a 20-year-old appaloosa gelding, and a 5-year-old quarter horse gelding initially presented with signs of colic 24–48 hours after first exposure to the hay. Gastrointestinal gaseous distension was the primary finding on clinical examination of all three horses. Two horses became moribund and were euthanatized 1 day after first showing clinical signs, and the third horse was euthanatized after 4 days of medical therapy. Endocardial hemorrhage and gaseous distension of the gastrointestinal tract were the only necropsy findings in the first two horses. On microscopic examination, both horses had scattered foci of mild, acute myocardial necrosis and neutrophilic inflammation associated with endocardial and epicardial hemorrhage. The third horse that survived for 4 days had multifocal to coalescing, irregular foci of acute, subacute, and chronic myocardial degeneration and necrosis. A. aestivalis (pheasant's eye, summer adonis) was identified in the hay. Strophanthidin, the aglycone of several cardenolides present in Adonis spp., was detected by liquid chromatography-mass spectrometry-mass spectrometry in gastrointestinal contents from all three horses. Although Adonis spp. contain cardiac glycosides, cardiac lesions have not previously been described in livestock associated with consumption of adonis, and this is the first report of adonis toxicosis in North America.
Keywords
Adonis spp. have long been used in European folk medicine for their cardiac-enhancing effects. 1,6 Native to Europe and Asia, Adonis aestivalis (summer pheasant's eye, summer adonis) was introduced into North America as a garden plant but escaped cultivation and is now abundant in disturbed sites and open forests in the western United States. 2 Adonis spp. are members of the Ranunculaceae family, one of 11 plant families with 34 genera known to contain cardiac glycosides. 10,12 Animals consuming plants containing cardiac glycosides typically develop fatal digestive and cardiac disturbances. 7 Some of the cardiotoxic plants found in North America include oleander, milkweed, foxglove, dogbane, lily of the valley, yew, avocado, rhododendron, Star of Bethlehem, periwinkle, hyacinth, and death camas. Plants containing cardiac glycosides most commonly associated with poisoning in livestock are oleander (Nerium spp.), milkweed (Asclepias spp.), and foxglove (Digitalis spp.). 2,10,12 There are no reports of adonis toxicosis in North America. In fact, reports of toxicosis in livestock associated with consumption of Adonis spp. are rare throughout the world. 4,13,14 In addition, although Adonis spp. contain cardiac glycosides, myocardial degeneration has not been described in livestock associated with consumption of adonis. This report describes digestive and cardiac disturbance in three horses that consumed hay contaminated with summer adonis.
Materials and Methods
Clinical history
Three horses were submitted to the California Animal Health and Food Safety Laboratory for necropsy. Twenty horses were housed at a ranch that had changed hay 2 days before the appearance of clinical signs. Many of the horses refused to eat the hay despite the apparent normal appearance of the hay. Three of 17 horses exhibited severe clinical signs 24–48 hours after first exposure to the hay and were ultimately euthanatized. The only clinical sign noted in the 17 remaining horses was feed refusal. The first horse, a 9-year-old thoroughbred gelding (horse No. 1), exhibited signs of acute colic and shock. Exploratory coeliotomy revealed gastric impaction with tympany, moderate gaseous distension of the small intestine and cecum, and generalized ileus. After surgery, the horse continued to show signs of shock despite intensive medical therapy and was euthanatized 12 hours later. The second horse, a 20-year-old appaloosa gelding (horse No. 2), was presented with lethargy and anorexia. Physical examination revealed that the horse was dehydrated and had dark mucus membranes and moderate gaseous distension of the large colon. Rapid deterioration was accompanied by muscle tremors, tachycardia, weak peripheral pulse, ileus, and decreased anal tone. The horse collapsed in the stocks and was euthanatized. Both horses were euthanatized within 24 hours of first exhibiting clinical signs. A third horse, a 5-year-old quarter horse gelding (horse No. 3), was examined the next day. Ileus, muscle tremors, and weakness were present, and a foul odor was evident upon passage of a nasogastric tube. Treatment for the next 3 days consisted of intravenous fluids, broad-spectrum antibiotics, botulinum serum, and activated charcoal. Heart rate became irregular with premature ventricular contractions. The horse became progressively weaker, was recumbent and painful, and was euthanatized 4 days after first exhibiting clinical signs. There was no response to botulinum antitoxin or administration of activated charcoal.
Diagnostic workup
Complete necropsies were performed on all three horses (horse Nos. 1–3) with subsequent histologic evaluation of tissues. Portions of brain, heart (right and left atria, ventricular septum with papillary muscle and right and left ventricular free wall), liver, kidneys, lungs, spleen, stomach, small intestines, large and small colons, adrenal glands, skeletal muscle, and diaphragm were collected from all horses and immersed in 10% buffered neutral formalin for 24 hours. Four-micrometer sections of fixed tissues were stained with hematoxylin and eosin and examined by light microscopy. Fifteen additional sections of the heart from right and left atria and ventricles and septum were examined from horse No. 3. For each horse (horse Nos. 1–3), colon contents were collected in selenite broth and cultured for Salmonella, and lungs and spleen were cultured on split MacConkey/blood agar plates and incubated under aerobic conditions. Selenium and heavy metal analyses (arsenic, molybdenum, zinc, lead, cadmium, copper, iron, and mercury) were performed on liver and kidney using inductively coupled plasma emission spectrometry. Brains from horse Nos. 1 and 2 were tested for cholinesterase activity.
Stomach and cecal contents from all three horses (horse Nos. 1–3) were tested for oleandrin, gitoxin, digitoxin, gitoxigenin, and grayanotoxins I and III using two-dimensional thin-layer chromatography (TLC). 8 In brief, cardiotoxins were extracted with dichloromethane and cleaned up using charcoal and reverse-phase solid-phase extraction (SPE) columns. Analysis was performed by two-dimensional TLC on silica gel plates and visualized using aluminum chloride followed by chloramine T spray (detection limits for oleandrin, gitoxin, and remaining toxicants were 0.05, 0.1, and 0.2 µg/g, respectively, in gastrointestinal contents).
Hay submitted with the first two horses was examined for animal parts. When none were found, portions of the fines from the hay, as well as liver and stomach contents from the first two horses (horse Nos. 1 and 2), were tested for botulinum toxin using mouse bioassay. The hay was screened for mycotoxins, which included aflatoxin B1, zearalenone, ochratoxin A, T-2, DAS, and vomitoxin, using TLC as described previously. 15 In brief, hay was extracted in acetonitrile–4% aqueous KCl, and extracts were processed by SPE before detection by TLC. In addition, the grass hay was examined for toxic plants and condition.
Stomach and cecal contents of all three horses (horse Nos. 1–3) were analyzed for strophanthidin using liquid chromatography–mass spectrometry (Filigenzi, personal communication). In brief, the strophanthidin glycosides were hydrolyzed and extracted from plant material and ingesta with methanol, water, and hydrochloric acid. The resulting extracts were cleaned up using C18 reverse-phase SPE cartridges. High-pressure liquid chromatography (HPLC) separation was performed by reverse-phase HPLC on a C18-column, and strophanthidin was determined by positive-ion electrospray on an ion trap spectrometer. The method detection limit for strophanthidin in ingesta was 0.075 µg/g, whereas the limit of quantitation was 0.24 µg/g.
Results
Pathology
On necropsy of the thoroughbred gelding (horse No. 1), there were several liters of red-brown cloudy peritoneal fluid and the diaphragm had fibrin tags on the peritoneal surface. There was thick white froth in the tracheal lumen, and the lungs were noncollapsable and wet. The soft tissues of the head and neck (particularly the retropharyngeal soft tissue and ligamentum nuchae) had pockets of yellow gelantinous edema. The pericardial sac contained approximately 30 cc of red-brown clear fluid. The stomach was distended with green fluid, and the colon contained scant ingesta. Ecchymotic hemorrhages were present on the serosa of the large colon and on the parietal pleura adjacent to the spinal column. There were hemorrhages along the coronary groove of the heart, and dry, ecchymotic endocardial hemorrhages were seen in the atria and ventricles.
The appaloosa (horse No. 2) had similar gastric distension with green fluid but no mucosal lesions. The small and large colons contained firm, dry balls of ingesta. The pleural cavity contained red-brown clear fluid, and there were dry ecchymotic hemorrhages on the parietal pleural of the diaphragm and along the spinal column. The pericardial sac contained red-brown fluid. There was ecchymotic hemorrhage along the coronary groove of the heart and extensive dry suffusion hemorrhage in the endocardium of the ventricles and atria. In the quarter horse (horse No. 3), the stomach was distended with fluid, fine chop, and charcoal. The cecum was also distended with similar contents, and the cecal mucosa was edematous. The tracheal lumen contained thick, white froth, and the lungs were uniformly wet and heavy. The kidneys were swollen and pale. Ecchymotic hemorrhages were present in the parietal pleura along the spinal column. There was red-brown clear pericardial fluid and hemorrhage along the coronary groove of the heart. The endocardium had foci of dry hemorrhage that extended into the myocardium as well as delineated, irregularly shaped, hemorrhagic foci that were interspersed with distinct pale foci and were most prominent in the papillary muscles in the ventricular septum and left ventricular free wall (Fig. 1).
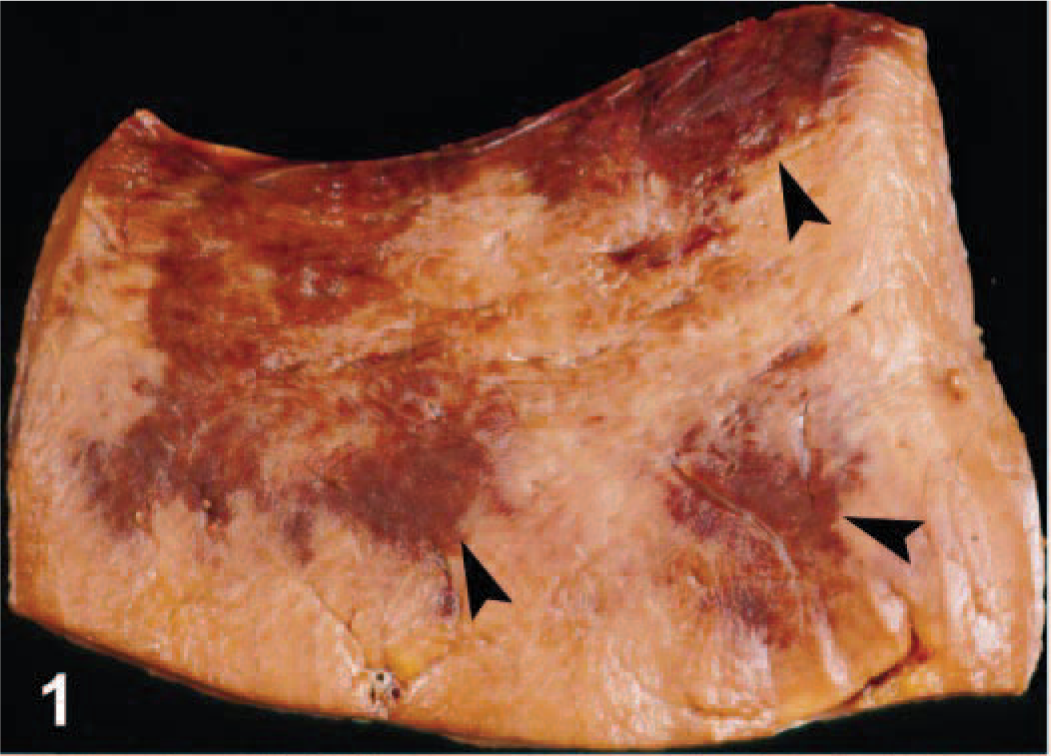
Left ventricular free wall; horse No. 3. Dark foci (arrows) are foci of myocardial hemorrhage, necrosis, and collapse. Foci were most prominent in the left ventricular free wall and septum on gross examination.
Histopathology
Microscopically, the first two horses (horse Nos. 1 and 2) had diffuse endocardial and epicardial hemorrhage with pockets of fibrin that extended into the underlying myocardium. The adjacent myocardium had mild interstitial edema with light neutrophilic infiltrates and rare foci of acute myocardial necrosis (Fig. 2). The remainder of the myocardium was unremarkable in both horses (horse Nos. 1 and 2). The third horse (horse No. 3) had severe, subacute, multifocally extensive, necrohemorrhagic, and pleocellular (neutrophils, lymphocytes, and macrophages) myocarditis and myocardial collapse. Microscopic changes in the liver were compatible with heart failure and characterized by marked congestion, hepatocellular degeneration, and bile stasis in centrilobular to midzonal regions. Pulmonary edema with pulmonary arterial thrombosis and multifocal acute renal tubular necrosis were also evident microscopically in horse No. 3.
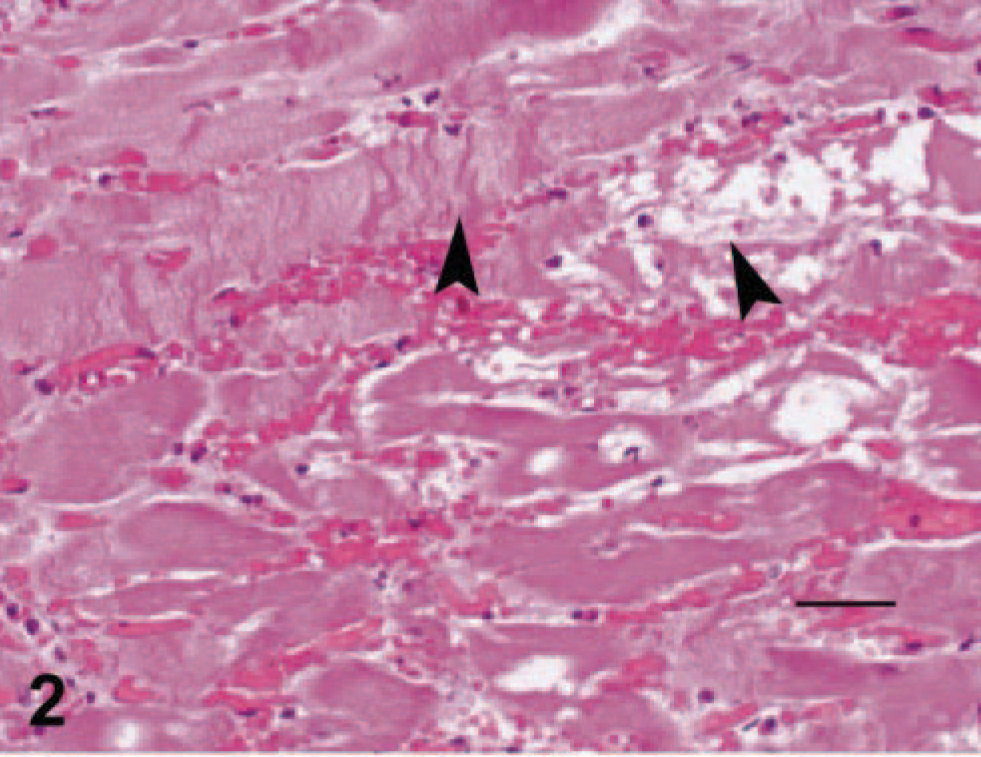
Left ventricle; horse No. 1. Photomicrograph of the left ventricle with mild interstitial edema, neutrophilic infiltrates and myocardial necrosis. The arrows demonstrate contraction band necrosis and fragmentation of the sarcoplasm. HE. Bar = 100 µm.
In the 15 additional sections of myocardium examined from horse No. 3, foci of acute myocardial necrosis were multifocally interspersed with subacute and chronic changes and primarily located in the ventricular septum, papillary muscles, left ventricular free wall, and atria. Foci of endocardial hemorrhage extended deep into the interstitium of the underlying myocardium. Foci of acute myocardial necrosis were characterized by myofiber contraction band necrosis, swollen myofibers with fragmented and hyalinized sarcoplasm and pyknotic nuclei, interstitial hemorrhage, edema, and pleocellular inflammation (multiple foci with variable combinations and quantities of lymphocytes, plasma cells, macrophages, and neutrophils). Subacute and chronic changes were characterized by scattered variably sized foci of myofiber collapse and “drop-out” and satellite cell hypertrophy, and fibroplasia, respectively (Fig. 3). Mononuclear inflammatory cell infiltrates and fibrosis were pronounced surrounding Purkinje fibers in the septum.
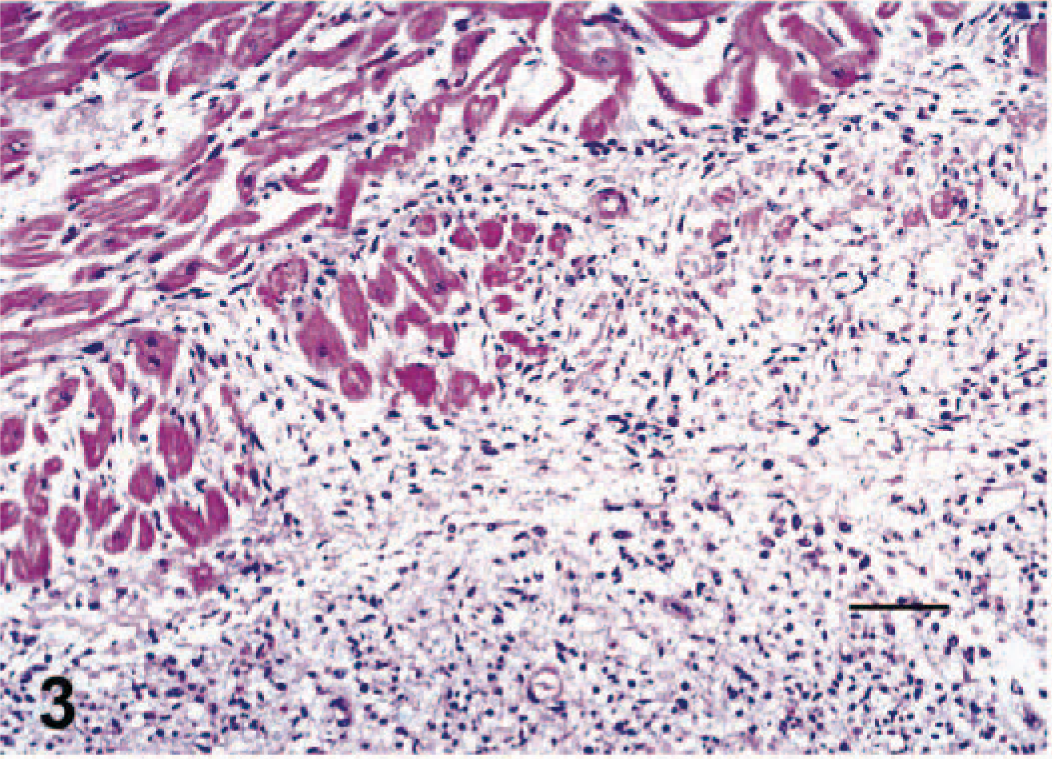
Left ventricle; horse No. 3. Left ventricle with large regions of myofiber collapse and satellite cell hypertrophy. HE. Bar = 200 µm.
Microbiology and toxicology results
Liver and stomach contents (horse Nos. 1 and 2) and hay fines were negative for botulinum toxin by the mouse bioassay. Oleandrin, gitoxin, digitoxin, gitoxigenin, and grayanotoxins I and III were not detected in stomach or cecal contents tested from all horses (horse Nos. 1–3) at a concentration greater than the minimum detection limit. Aerobic cultures of the lungs and spleen from the horses (horse Nos. 1–3) were negative for bacterial growth. Cholinesterase activities of the brains of all three horses were within expected limits (horse Nos. 1, 2, and 3: 2.7, 3.0, and 3.7 µM/g/min, respectively; normal > 2.1 µM/g/min). Heavy metal analyses of the livers and kidneys (horse Nos. 1–3) yielded tissue levels within normal limits. Liver selenium concentrations were slightly low in all three horses (horse Nos. 1, 2, and 3: 0.180, 0.185, and 0.182 ppm, respectively; normal = 0.3–1.0 ppm).
Listed mycotoxins were not detected in the submitted hay sample. A. aestivalis (summer adonis, summer pheasant's eye) (Figs. 4, 5–6) was found in the hay in addition to dallisgrass, orchardgrass, field pennycress, and alfalfa. A. aestivalis has erect stems with terminal solitary flowers. The flowers have oblanceolate orange petals with central purple-black anthers. The leaves are simple, alternate with blades two or three times pinnately dissected into linear segments. Percent contamination could not be estimated because of fragmentation of the dried plant as well as patchy distribution throughout the hay sample. Strophanthidin was detected in the stomach and cecal contents of horse Nos. 1 and 2 and in the cecal contents but not in the stomach contents of horse No. 3.

Summer pheasant's eye (Adonis aestivalis). Flowers have waxy, orange petals and purple-black anthers. The arrow shows the characteristic pod (seed head).
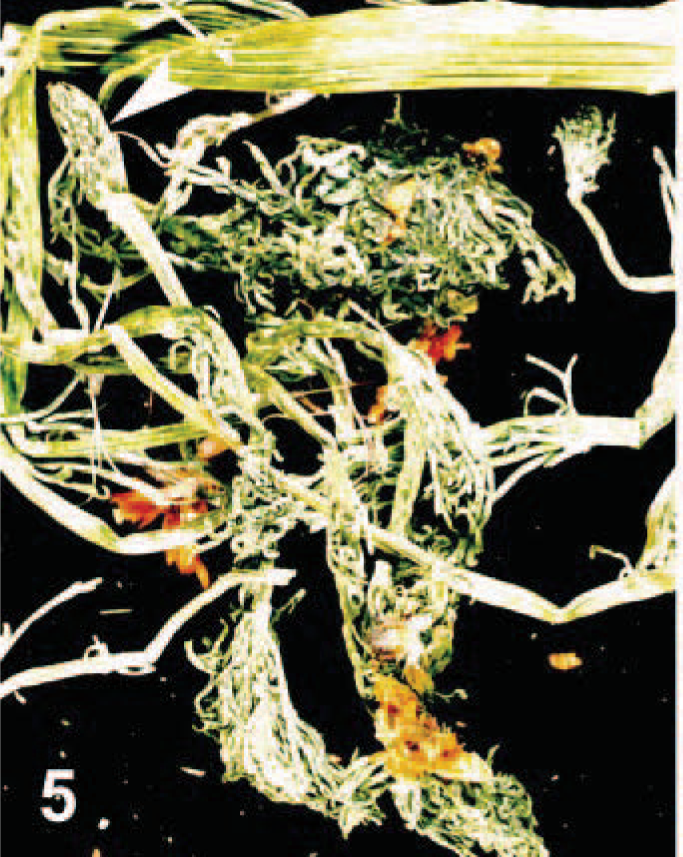
Adonis aestivalis. Dried A. aestivalis found in grass hay consumed by horse Nos. 1, 2, and 3. Note the linear ridges in the stems, the feathery foliage, and the orange flowers. The arrow demonstrates the pod characteristic of this plant.

Stand of Adonis aestivalis; Canby, Modoc County, California.
Discussion
The three horses described in this report developed gastrointestinal disturbances and myocardial necrosis after consumption of hay contaminated with summer pheasant's eye (A. aestivalis). Adonis species contain a mixture of cardiac glycosides, including adonitoxin, strophanthin, vernadigin, and cymarin. 2,5 Strophanthidin, the aglycone of several cardenolides present in adonis species, was detected in the stomach and cecal contents from the first two horses that died 24–48 hours after initial exposure to the hay and only in the cecal contents of the third horse that died 4 days later. Strophanthidin was not detected in the stomach contents of the third horse that died 4 days after initial exposure likely because of movement of the ingesta down the gastrointestinal tract. In addition, although selenium levels were low in all three horses and selenium deficiency is associated with myocardial degeneration, levels were only slightly low. Selenium deficiency may have contributed to increased susceptibility of the heart to the cardiac glycosides.
A. aestivalis was introduced into North America as a horticultural plant, escaped cultivation, and is now naturalized in the western United States (California, Oregon, Washington, Idaho, Montana, and Utah), Missouri, and New York. 2,16 Adonis autumnalis and Adonis vernalis, which also contain cardiac glycosides, are typically found in the south-central and southeastern regions of the continent. A. aestivalis has been well established in some northern California counties for more than 80 years 17 and was documented in Modoc County, California, in 1924. Despite the length of time this plant has been naturalized, there are no previous cases of adonis toxicosis reported in the United States. All Adonis species are considered unpalatable, which is probably why reports of poisoning are rare. 2,9 This is also likely the reason for feed refusal displayed by many of the horses on the ranch and why only three horses were ultimately affected.
In other parts of the world, there are a few reports of mortality in livestock associated with ingestion of Adonis spp. In a 1952 report from Germany, feed refusal and oral mucosal irritation were the only signs reported in four separate cases in which horses were fed alfalfa hay contaminated with A. aestivalis. 13 Hay samples contained less than 7% summer adonis in two, 7% in one, and 17% in one of the cases. Oral mucosal irritation did not occur in the three horses in this study. The moisture content of the alfalfa hay in the 1952 study was not reported, but the grass hay used in this study was dried. The irritant compound in Adonis spp. responsible for oral mucosal irritation may be unrelated to the toxins that target the heart and intestinal motility and may be volatile and therefore no longer present in the dried state. This volatile compound may also be responsible for mucosal irritation to the gastrointestinal tract, causing gastroenteritis noted in the literature 4,11 but not seen in these horses.
Four of six horses fed hay containing 8.5% A. aestivalis died in 1931 in Budapest. 4 Two of the six horses developed severe diarrhea but survived. Hemorrhagic gastroenterocolitis and endocardial hemorrhage were noted on postmortem examination of the only horse that was necropsied in this group. Although this is nonspecific and not definitive evidence of myocardial damage, it is the only report in the literature of endocardial hemorrhage associated with ingestion of adonis. Endocardial hemorrhage was the most significant lesion seen on gross examination of all three horses in this study. The endocardial hemorrhagic foci had a slightly dry, rough surface, suggesting that the lesion was more significant than endocardial hemorrhage commonly seen as a terminal event. Hemorrhagic gastroenteritis was not seen in any of the horses in the current case. Several horses died in 1912 in New South Wales, Australia, after eating alfalfa hay that was contaminated with A. autumnalis. 14
In addition to horses, Adonis spp. appear to be toxic to other livestock. Feed refusal, vomiting, dyspnea, and death occurred in a group of growing and finishing pigs fed a diet containing seeds of Adonis microcarpa. 3 Although experimental feeding of sheep with Adonis annua proved lethal at 1% body weight (450 g of plant material), feeding of cattle with doses up to 2.7 kg for several days failed to elicit clinical signs and death. 9
Plants of the Adonis genus contain a series of cardenolides similar to oleander (Nerium spp.) and foxglove (Digitalis spp.). 2 Although Adonis spp. contain potent cardiac glycosides, most reports of adonis toxicosis in livestock describe digestive disturbances, and cardiac lesions with subsequent heart failure have not been well documented. Cardenolides are highest in the leaves and flowers, 5 both of which were present in the hay in apparent low quantities. Some Adonis spp. are used in herbal medicine to treat “flabby heart” and are sometimes used where digitalis fails because they appear to be 10 times more “powerful” than digitoxin. 1,6 At therapeutic doses, the mode of action is similar to digitoxin, causing positive inotropic effects by way of binding and inhibition of membrane-bound Na-K-ATPase and ultimately increased intracellular calcium and the force of myocardial contraction. 10 At toxic doses, inhibition of the sodium-potassium pump ultimately depletes intracellular potassium and increases serum potassium, resulting in a progressive decrease in electrical conductivity, severe dysrhythmias, and eventual blockage of cardiac activity. Increased intracellular calcium leads to cardiocyte death. In addition, these glycosides act directly on the gastrointestinal tract, causing hemorrhagic enteritis and diarrhea, and in fact, most reports of poisoning in livestock are of gastrointestinal disturbances. The initial clinical sign in all three horses was decreased intestinal motility with gaseous distension. However, no mucosal microscopic lesions were evident in the gastrointestinal tracts of any of the horses.
There are no reports that document histopathology associated with adonis toxicosis. Microscopic changes seen with adonis poisoning were similar to those documented for oleander toxicosis, 7 and in fact, necessitated analysis of the stomach and cecal contents from all three horses for oleandrin, the toxic compound of oleander, of which none was detected. In addition to endocardial and epicardial hemorrhage, the initial microscopic change evident in the hearts of the horses that consumed the adonis was inflammatory in nature with mild interstitial edema, light neutrophilic inflammation, and occasional pockets of fibrin in the myocardium subtending the foci of hemorrhage. Foci of myocardial necrosis were rare in the two horses that died 24 hours after exposure. The initial mild interstitial inflammatory response is similar to that described in horses that die of oleander toxicosis. Over time, lesions of myocardial damage and collapse were extensive, with foci of acute myocardial necrosis interspersed with chronic changes of myofiber collapse and fibrosis seen in the horse that survived for 4 days. Mononuclear inflammatory cell infiltrates and fibrosis surrounding Purkinje cells of the bundle of HIS are similar to that described with oleander toxicosis. 7
In summary, consumption of A. aestivalis was associated with digestive disturbance and myocardial necrosis in three horses from the same premises. The principle toxins that are present in all Adonis spp. are cardiac glycosides, which cause myocardial necrosis through inhibition of Na-K-ATPase. Although typically unpalatable and not likely consumed in pasture, poisoning of livestock is becoming more feasible through contamination of fields harvested for animal feed. Poisoning may become more common as this plant becomes better established (naturalized) in the western region of North America.
Footnotes
Acknowledgements
We extend special thanks to Dr. Dale Woods for photographs of A. aestivalis taken in Modoc County, California (Figs. 4, ![]() ).
).
