Abstract
An intraocular mass from a 13-year old Husky-mix dog was diagnosed as retinal astrocytoma. The mass arose from the ganglion layer of the retina and occupied 50% of the vitreous space. The mass was immunoreactive for neuron-specific enolase, S-100, vimentin, and glial fibrillary acidic protein. The neoplasm had characteristics similar to solitary retinal astrocytomas of humans but lacked the marked vascularity.
Keywords
In domestic animals, primary tumors of the globe can include melanocytic tumors, iridociliary epithelial tumors, medulloepithelioma, feline posttraumatic sarcoma, retinoblastoma, and astrocytoma. 4,11 Astrocytoma originating from the retina is a very rare neoplasm that is not well described in the veterinary literature. 6 In this study, we characterize the microscopic and immunohistochemical features of a retinal astrocytoma in a dog.
A 13-year-old Husky-mix dog presented to the referring veterinarian for progressively decreased vigor and mobility related to advanced age. Physical examination revealed a pale fleshy intraocular mass in the vitreous space of the right eye. The eye was enucleated and sent to Iowa State University's Surgical Pathology Service for histopathologic diagnosis. The mass occupied approximately 50% of the vitreous space and was attached to the posterior retina (Fig. 1). The eye was processed routinely and stained with hematoxylin and eosin for microscopic evaluation.
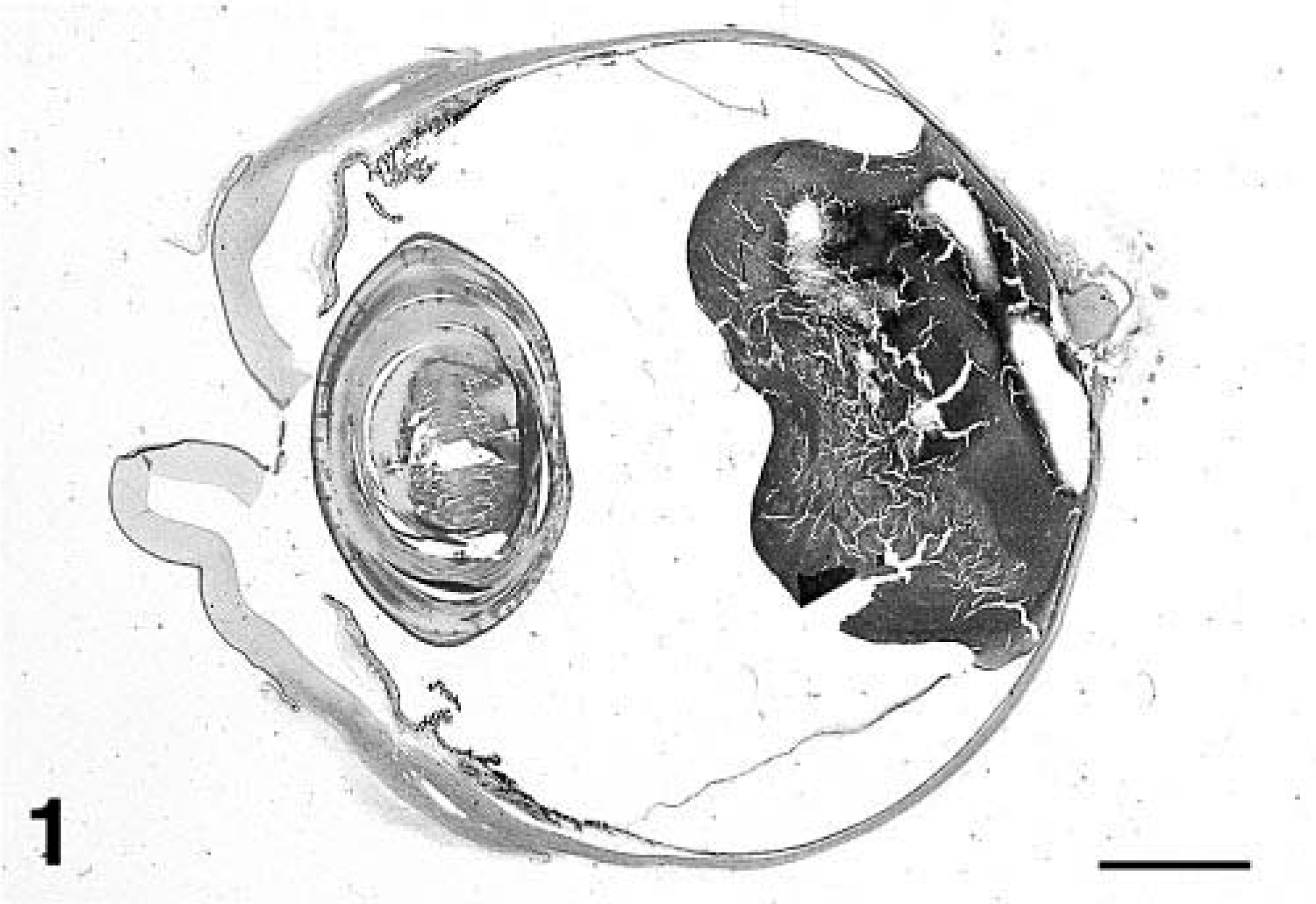
Eye; dog. Sagittal section of eye and posterior retinal mass. HE. Bar = 4 mm.
A circumscribed, nonencapsulated mass was attached to the posterior retina centered over the optic disk. This mass appeared to arise from the superficial aspect of the ganglion layer (Fig. 2). It was composed of palisading sheets of bipolar spindle cells aligned perpendicular to the fibrovascular stroma. The neoplastic cells had a single oval nucleus located at one end of the cell, one to three small nucleoli, a moderate amount of finely fibrillar eosinophilic cytoplasm, and indistinct cell borders. In addition, the neoplastic cells had cytoplasmic processes that extended toward adjacent vessels. Anisokaryosis, nuclear pleomorphism, and large nucleoli were seen infrequently. However, there were a few small foci of necrosis, which were surrounded by vague palisades of neoplastic cells. The mitotic rate was moderate (1/40× field). The area of the retina adjacent to the mass was atrophied. The retina was detached, and the pigmented epithelium was hypertrophied. Lesions were not evident in other segments of the eye.
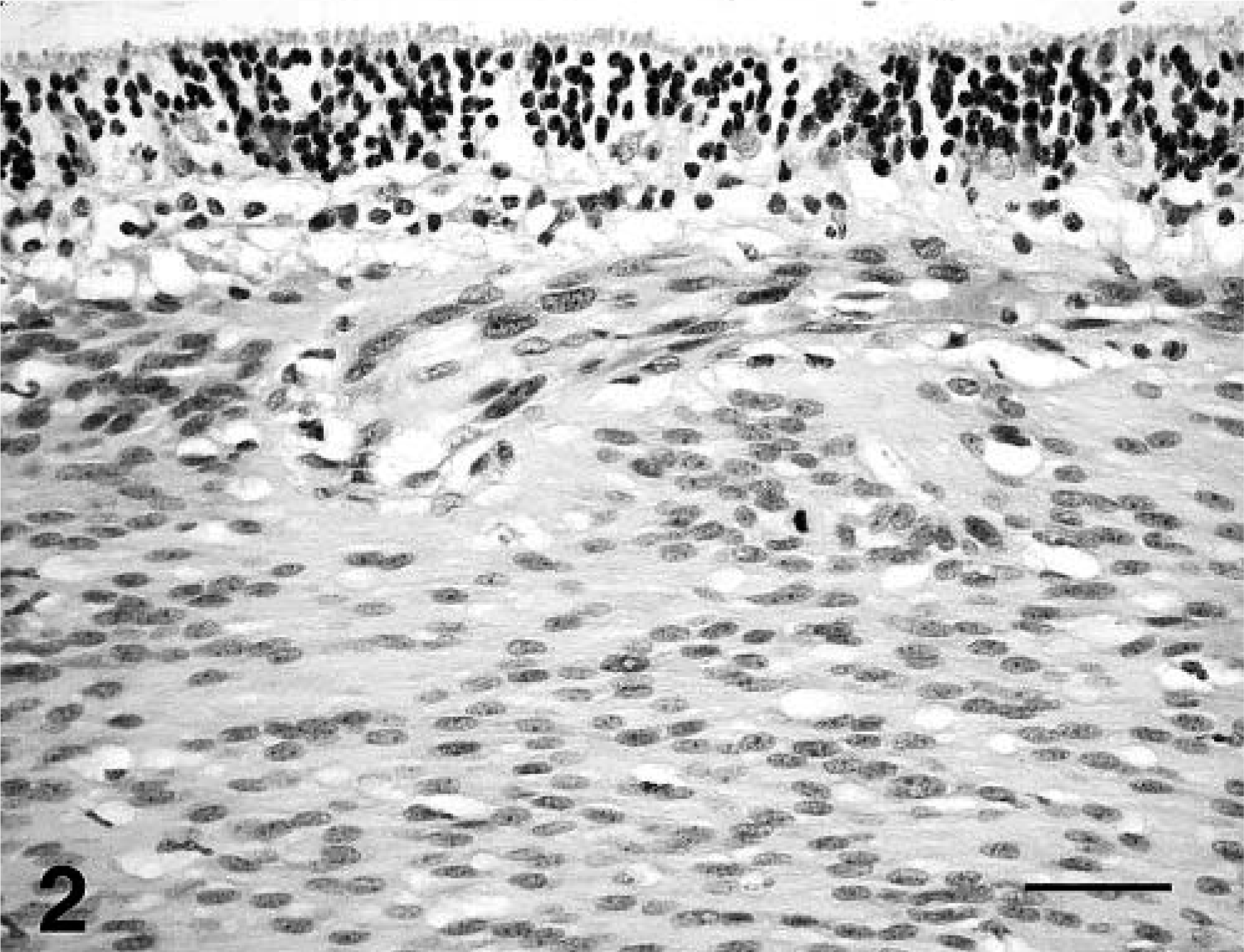
Retina; dog. Neoplastic astrocytes arise from the ganglion layer of the retina. HE. Bar = 25 µm.
The mass was further characterized by immunohistochemistry. The neoplastic cells had strong diffuse cytoplasmic immunoreactivity for neuron-specific enolase (NSE), S-100, and vimentin. In addition, throughout the section, about 30% of neoplastic cells had moderate immunoreactivity for glial fibrillary acidic protein (GFAP). GFAP immunoreactivity was concentrated in the cytoplasmic processes that extended toward vessels (Fig. 3). Neoplastic cells were not immunoreactive for neurofilaments or melan A.
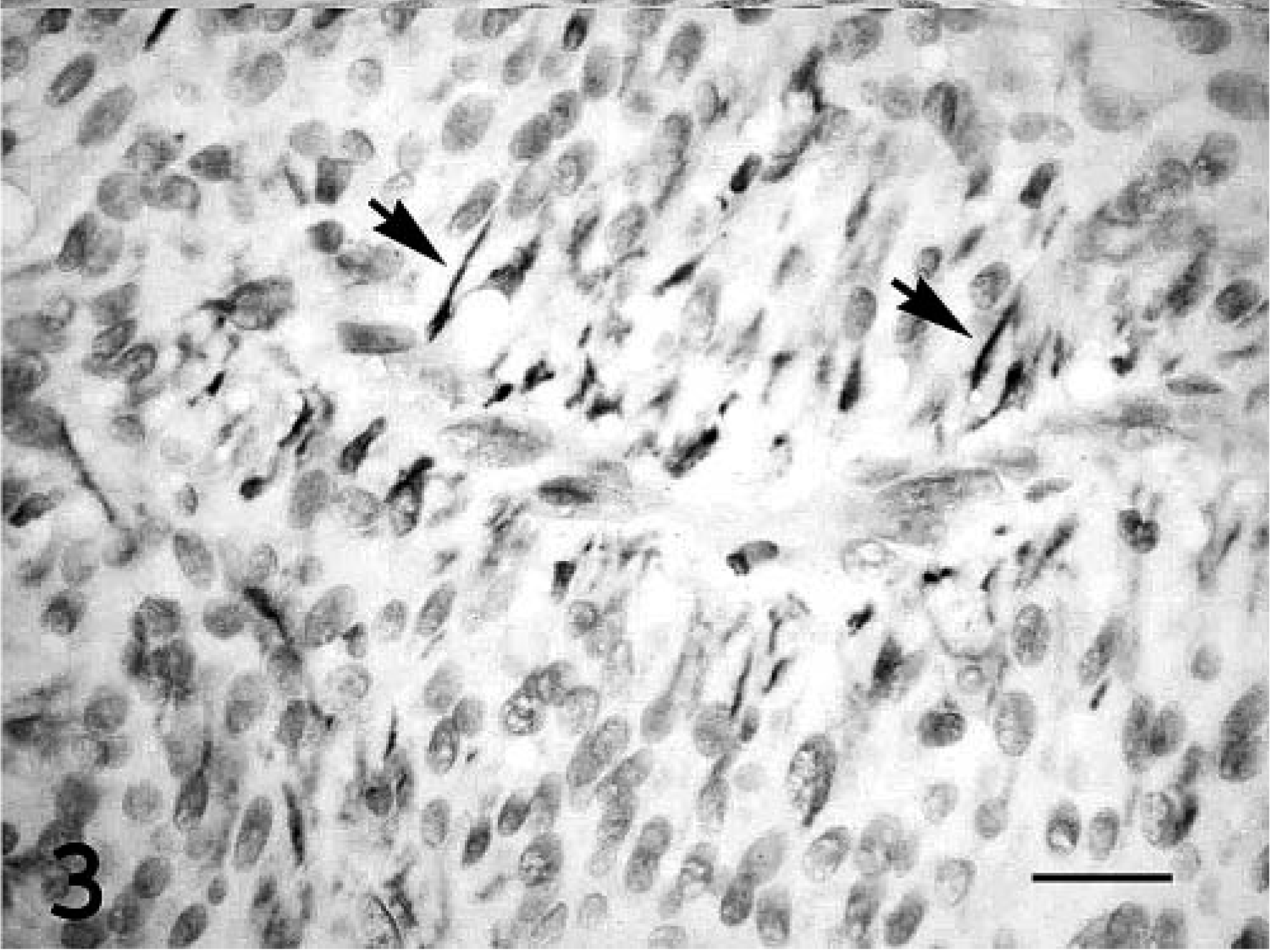
Retinal astrocytoma; dog. Prominent GFAP immunoreactivity is present in the cytoplastic processes (arrows) of neoplastic cells adjacent to vessels. Streptavidin–biotin–peroxidase method, Harris' hematoxylin counterstain. HE. Bar = 10 µm.
In veterinary medicine, ocular astrocytomas are rare and present usually as a retrobulbar mass originating from the optic nerve. 2,9,10 In one dog, an astrocytoma of the optic disk formed a protuberance into the vitreous space with optic nerve invasion. 3 Canine and feline astrocytomas originating from the retina have been reported; however, these cases are not well characterized in the literature. 4,6
In human medicine, there are two general types of retinal astrocytic proliferations, astrocytic hamartoma and solitary retinal astrocytoma. Astrocytic hamartomas are the most common type and are frequently associated with tuberous sclerosis or neurofibromatosis. Astrocytic hamartomas are considered benign lesions and some may regress over time. 5 Solitary retinal astrocytomas are rare and occur spontaneously with the histologic appearance and behavior of neoplasia. This is clinically associated with intraocular damage such as retinal detachment, central retinal vein occlusion, neovascular glaucoma, and necrosis. Histologically, this form can have large multiple nucleoli, mitotic figures, cellular pleomorphism, and marked tumor vascularity. Extra ocular invasion is rare. GFAP immunoreactivity especially in the cytoplasmic processes is characteristic for these retinal astrocytic proliferations. 1
In this case of retinal astrocytoma in a dog, some histologic features (mitotic rate, multiple nucleoli, foci of necrosis, lack of invasion) and intraocular changes (adjacent retinal atrophy, retinal detachment, and retinal epithelial pigment hypertrophy) were similar to solitary retinal astrocytoma of humans. The astrocytoma appeared to originate from the ganglion layer of the retina. The retinal neurons are supported by a network of astrocytes and Müller cells, and so it is reasonable that astrocytomas can originate in the retina. 5 Previously reported retinal astrocytomas in the dogs and cats have shown local invasion in the optic nerve and choroid, but there was no recognized recurrence after surgical removal. 6 In this study, the astrocytoma extended into the vitreous space with no apparent choroid invasion or optic nerve involvement. This feature along with the behavior in previous case reports suggests that retinal astrocytomas have the potential for local destruction and that surgical resection may be beneficial therapy.
The leading differential diagnoses for the posterior retinal mass were tumors of neuroepithelial origin including retinoblastoma. In the original case report, canine retinoblastoma was composed of abundant amounts of undifferentiated small cells that arose from the retina, and these had scant cytoplasm and hyperchromatic nuclei with admixed foci of differentiation into Flexner–Wintersteiner rosettes. GFAP immunoreactivity was present in only a few cells and reflected either localized reactive gliosis or glial differentiation. 11 Recently, a primitive neuroepithelial tumor was described in a dog retina with retinoblastoma- and medulloepithelioma-like components and no GFAP immunoreactivity. 7 In this case of retinal astrocytoma, immunohistochemistry was useful for confirming the diagnosis. Immunoreactivity in canine astrocytomas can include GFAP (especially in the cytoplasmic processes), S-100, and vimentin. 3,8,12 NSE, although typically accentuated in neurons and their processes, has been localized to reactive astrocytes and astrocytomas in humans. 13
Footnotes
Acknowledgements
We thank Dr. Nasreen A. Syed and Dr. Greg Hageman (University of Iowa) for assistance with immunohistochemistry and microscopic interpretation.
