Abstract
Astrocytoma is one of the most common tumors of the central nervous system in animals. Of the domesticated animal species, most examples are seen in dogs, and the spectrum that has been described is quiet broad. Previous studies have revealed morphologic similarities between human and animal astrocytomas. Human astrocytomas are often associated with genetic alterations that determine the clinical behavior and therapy outcome. The purpose of this study was to further characterize astrocytomas in dogs and to determine whether there are genetic changes similar to those in the human counterpart.
Thirty-one canine astrocytomas were analyzed retrospectively from archival tissues available from three veterinary laboratories. These astrocytomas were classified based on their phenotype (morphology and immunohistochemistry), anatomic localization, and breed incidence. Two additional astrocytoma subtypes, which are not included in the present World Health Organization (WHO) classification, are described in this study. Thirty-five percent of astrocytomas exhibited positive immunolabeling for genetic alterations of p53, and 23% were positive for alterations of epithelial growth factor receptor genes (EGFR). A single point mutation of p53 was detected in one dog from 12 deoxyribonucleic acid (DNA) samples analyzed. Morphologic patterns of astrocytomas in animals were similar to those of human astrocytomas, and genetic alterations were mostly associated with malignant forms. Results generated from this study of microscopic and genetic alterations of central nervous system astrocytoma hopefully will contribute to a classification system of this neoplasm in veterinary medicine that will better reflect their biologic behavior and treatment responses to novel therapies.
Astrocytoma is one of the most common and most frequently malignant neoplasms of the central nervous system in animals. Of the domesticated animal species, most examples are seen in dogs, with much lower occurrence in other species. 10–13,18,30,34,40,41,44 Intracranial neoplasia occurs more frequently in dogs than in humans (14.5 per year per 100,000 canine populations at risk compared with 4–5 per 100,000 humans). 11,18
Canine intracranial astrocytoma occurs most commonly in brachycephalic breeds, in particular the Boston Terrier and Boxer. No sex predilection has been reported, and they are recognized with greater incidence in animals over 6 years of age. The neoplasm is most commonly located in the pyriform lobes, cerebral hemispheres, thalamus, hypothalamus, and brain stem. Clinical signs of astrocytomas may vary with tumor location, and neurologic deficits reflect direct nervous tissue involvement as well as secondary effects of peritumor edema, necrosis, hemorrhage, compression, herniation, or obstructive hydrocephalus. 10,13,23,34
Cerebrospinal fluid findings in animals with astrocytoma are usually nonspecific and include increased protein concentration and mixed-cell pleocytosis that reflect disturbance of the blood-brain barrier. 10,13,30,34 Computed tomography (CT) and magnetic resonance imaging are routinely available to veterinary practitioners for diagnosis to localize intracranial lesions. CT characteristics of some canine brain tumors have been reported to be similar to those in humans. 40,41
In veterinary medicine, histopathology still remains the principal tool for intracranial neoplasia characterization and classification. 8,9,12,14,17,24,44 In humans, however, both microscopic characteristics and genetic alterations are of considerable importance for prognosis and appropriate patient management. 5,16,28,31,45,46 The relationship of genetic alterative changes associated with intracranial neoplasias is still novel in veterinary medicine.
Loss or mutation of the p53 tumor suppressor gene occurs in approximately one-third of human astrocytomas, suggesting an early genetic event in tumor development. An important consequence of the loss of wild-type p53 activity is increased genomic instability, which appears to accelerate neoplastic progression. 16,46,47 EGFR gene is the most frequently amplified oncogene in human astrocytic tumors and is amplified in one-third of glioblastomas (GMs) and in fewer anaplastic astrocytomas. 2,3,6,16,19,33 Unfortunately, there are no published reports regarding genetic changes associated with astrocytomas in the veterinary literature. The frequent occurrence of p53 and EGFR changes in human astrocytoma therefore stimulated our interest to investigate the possible occurrence of similar genetic changes in animal tumors.
The aim of this study was to further characterize the microscopic features of canine astrocytomas and to correlate these results with the immunophenotype, genotype, and changes in expression of p53 and EGFR genes.
Materials and Methods
Tissue source
All the 31 dogs studied were retrieved from the files of three veterinary pathology laboratories: Texas A&M University Department of Veterinary Pathobiology (11 dogs); Texas Veterinary Medical Diagnostic Laboratory (eight dogs); and University of Georgia Athens Diagnostic Laboratory (12 dogs). Twenty-eight samples were derived from necropsy cases, and three samples were derived from surgical biopsies. Specimens were formalin fixed, paraffin embedded, and collected over a 10-year period.
Histology and morphologic diagnosis
In this study, astrocytomas were classified according to the recentWHO classification of the central nervous system tumors of domestic animals 17 except for the suggested addition of three types that occur mostly in humans. Typing was based on the assessment of the predominant cell type by conventional light microscopy of hematoxylin eosin (HE)–stained sections and by immunohistochemistry using specific cell type biomarkers as detailed below.
Immunohistochemistry
The indirect alkaline phosphatase technique using VECTASTAIN ABC-AP avidin-biotin system (Vector Labs, Burlingame, CA) was used as previously described. 37,44 Tissues were deparaffinized and incubated in a moist chamber at room temperature with pepsin (Sigma 7012), 1 mg/ml in 0.01 N chlorhydric acid for 10 minutes at 37 C. Nonspecific binding (2% bovine serum albumin, 30 minutes) was applied before primary antibody incubation. Deparaffinized tissue sections (5 µu;m thick) from each tumor were incubated with primary antibodies against glial fibrillary acidic protein (GFAP) (GFAP-rabbit polyclonal, 1 : 400, 1 hour; Sigma Chemical Company, St. Louis, MO), S-100 protein (S-100, rabbit polyclonal, 1 : 50, 1 hour; DAKO Corporation, Carpinteria, CA), p53 protein (p53-rabbit polyclonal, 1 : 50, 2 hours; Santa Cruz Biotechnology, Santa Cruz, CA), and EGFR-rabbit polyclonal, 1 : 100, 2 hours; InnoGenex). Goat anti-rabbit IgG-biotin and anti-mouse IgG-biotin (Vector Labs), 1 : 400 dilution for 30 minutes, served as secondary antibodies. In the amplification step, a VECTASTAIN ABC-alkaline phosphatase kit was used, followed by substrate solution (red chromogen kit, Vector Labs, or 3,39-diaminobenzidine tetrachloride-hydrogen peroxide). Before mounting, the sections were counterstained with methyl green, 1% for 5 minutes. For negative control slides, the first antibody was substituted with phosphate-buffered saline and normal rabbit serum. A positive internal control for GFAP was staining of the glia limitans.
The percentage of p53 positively immunostained tumor cell nuclei was determined by dividing the number of immunolabeled nuclei by the total number of nuclei, both immunostained and unstained. A minimum of 100 tumor cells was scored. Those astrocytomas with p53 immunostaining of at least 10% of the cell nuclei were considered to have p53 overexpression, whereas those with less than 10% p53 immunostained nuclei were considered to be within normal range of p53 expression. 26,47
p53 gene sequencing
Analysis of p53 gene was performed by direct sequence of complementary DNA amplified by polymerase chain reaction (PCR) products, as described previously. 15,20,27,29,45 All DNA samples were obtained from formalin-fixed, paraffin-embedded tissues. Control DNA was obtained from cardiac muscle of a freshly necropsied Labrador Retriever. After microscopic examination, areas of tumor tissue of interest were selected for dissection from paraffin blocks. Paraffin was removed from the 10-µu;m sections by soaking in xylene and then in ethanol. The tumor was microdissected from each slide into 50 µu;l of lysis buffer (10 mM Tris-HCl pH 8.0, 100 mM KCl, 2.5 mM MgCl2, 2.5 mg/ml proteinase K) and then digested overnight, followed by standard phenolchloroform extraction. The DNA was resuspended in 10 mM Tris-HCl (pH 8.0), and DNA concentration was measured by reading absorbance at 260 nm.
PCR was used to amplify p53 gene exons 5–8. Four different sets of 20- to 22-mer oligonucleotide primers were designed using the genomic sequence of p53 (GenBank accession number: U62133). Primers were designed to span each exon of the p53 open reading frame and sufficient bases of the intronic sequence to ensure that the splice donor and splice acceptor sites were included for analysis (Table 1). Oligonucleotide primers were synthesized by a contract laboratory (Advanced DNA Technologies, Texas A&M University). PCR was performed in a reaction buffer (100 µu;M Tris-HCl, pH 8.3, 500 µM KCl) containing each diethylnitrophyenyl thiophosphate at 200 µM final concentration, 1.5 µM MgCl2, 2.5 units pfu polymerase (Stratagene, La Jolla, CA), 0.2 µM primer, and 50–100 ng template DNA in a total reaction volume of 100 µl. PCR reactions were carried out using 30 cycles of denaturation (94 C, 30 seconds), annealing (30 seconds at the appropriate temperature), and extension (72 C, 30 seconds), followed by a final 10-minute extension at 72 C on a PTC-100 thermal cycler (MJ Research, Inc., Watertown, MA). Amplified PCR products were separated on 1.5% agarose gels and viewed by ethidium bromide staining. p53 genomic DNAs could be amplified and used for further analysis from only 12 of 31 dogs. Gel-purified DNA fragments were cloned with the TOPO TA cloning kit, according to the instructions of the manufacturer (Invitrogen, San Diego, CA) after adding 39 adenines. Positive plasmid clones were identified by restriction analysis. Six clones of each sample were combined for DNA sequencing. The Dye Terminator Sequencing Kit (Perkin-Elmer, Foster City, CA) was used to determine the sequence of the clones.
Primers used for the amplification and analysis of the canine p53 gene.

Results
A total of 31 canine astrocytomas were investigated in this study. All samples were derived from necropsy cases except for three samples, which originated from surgical biopsies. The classification of all tumors is included in Table 2. Signalment, morphologic appearance, location, and immunohistochemical results are listed in Table 3. The sex distribution of astrocytoma in this study was 19 females (60%) and 12 males (40%), and the age ranged from 1 to 18 years, with a mean of 8 years and a mode of 7 years. In this study, the highest incidence (seven, 21%) of astrocytoma occurred in the Boxer. The anatomic location of the neoplasm was predominantly in the brain, with cerebral hemispheres being the most frequent location. In one case (dog No. 16, Table 3), astrocytoma was located in the spinal cord of a 1-year-old dog.
Classification of 31 canine astrocytomas.

∗ Tentatively included in this classification. WHO classification includes this tumor in the category of neuroepithelial tumors of uncertain origin. 16
Morphology and immunohistochemistry results in dog astrocytomas.
Astrocytomas, in our study, were diagnosed based on their microscopic and immunohistochemical characteristics. Unequivocal positive GFAP staining of tumor cells was seen in 28 (84%) tumors. The staining pattern for GFAP varied with the area examined; less differentiated areas composed of rounded cells with little cytoplasm often showed poor staining, whereas more differentiated cells had more distinctive staining.
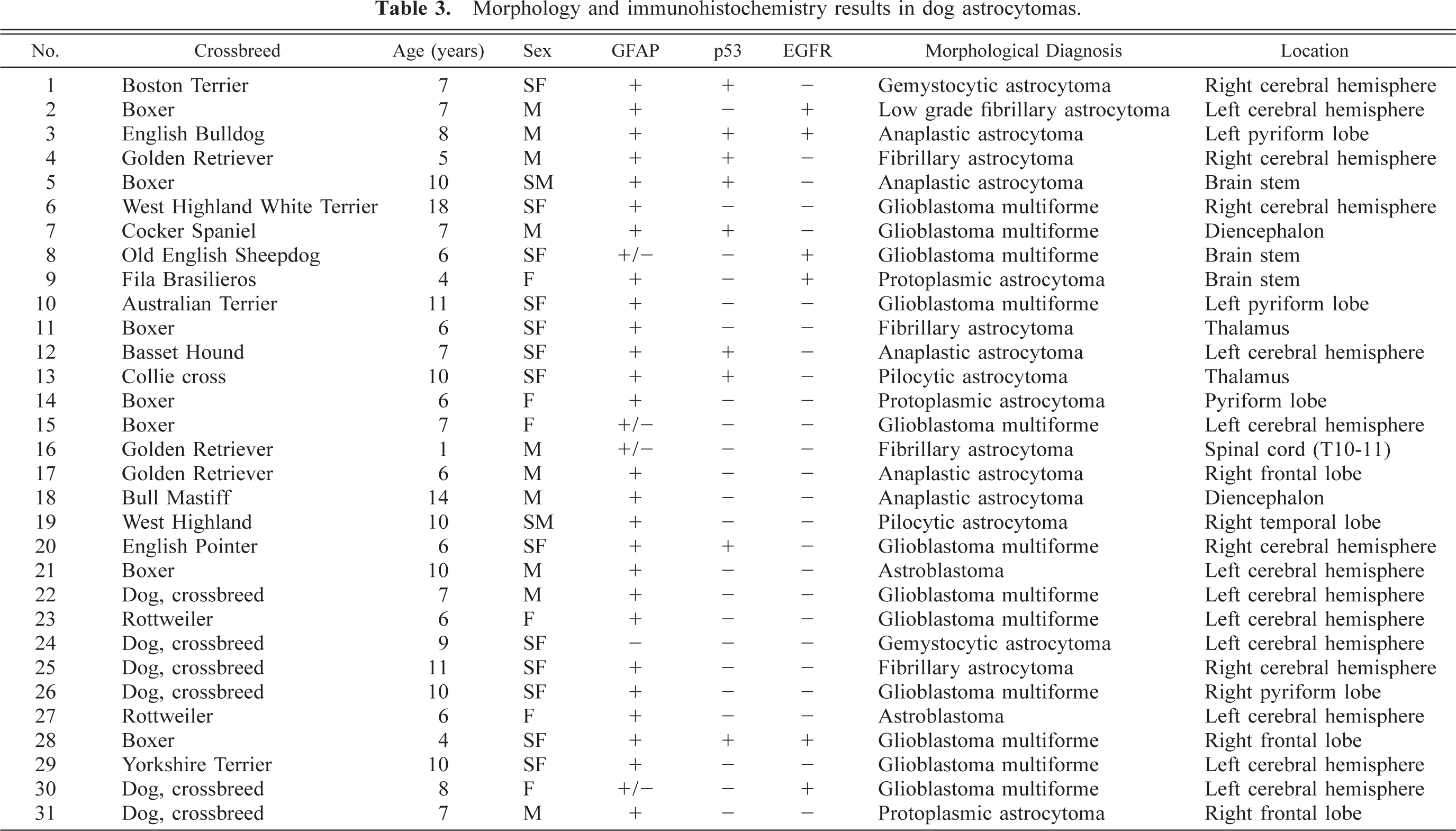
The most common astrocytic neoplasm in this cohort was classified as astrocytoma (12 dogs). Astrocytomas could be subdivided histologically into fibrillary (Fig. 1), protoplasmic (Fig. 2), gemistocytic (Fig. 3), and pilocytic subtypes (Table 2). Microscopic features of GM multiforme (12 dogs) included necrosis with pseudopalisading of cells, abundant vascular proliferation, hypercellularity with increased mitosis, and anaplasia (Fig. 4).
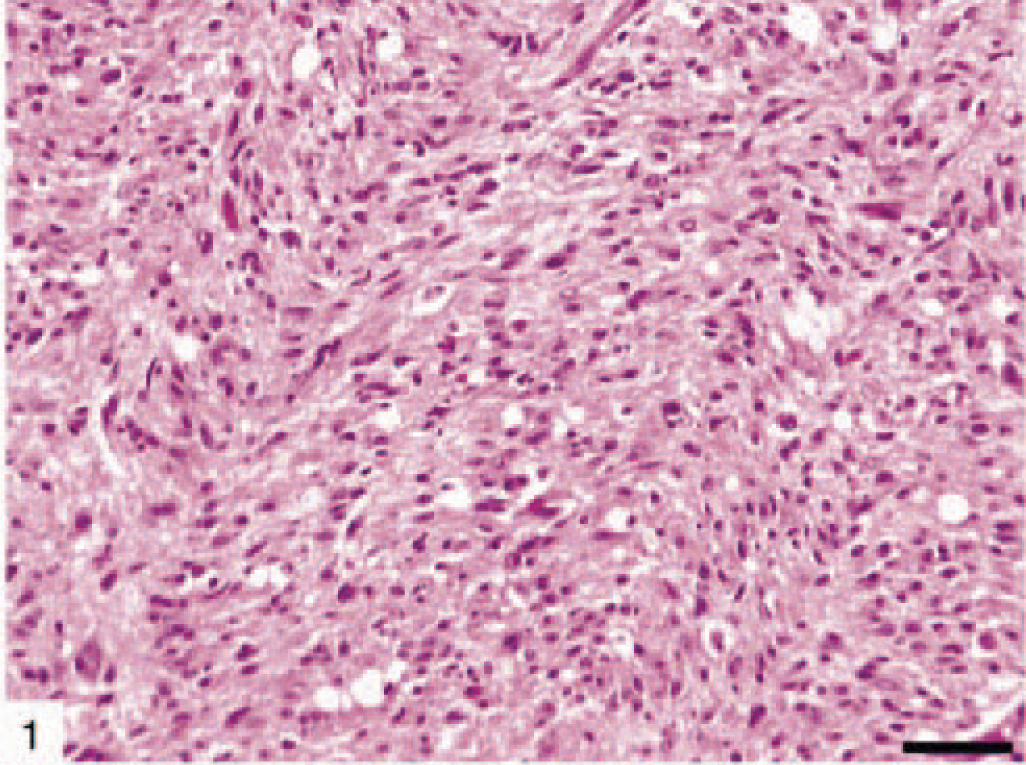
Fibrillary canine astrocytoma (dog No. 4) showing moderate cellularity and minimal nuclear atypia. HE. Bar = 150 µm.
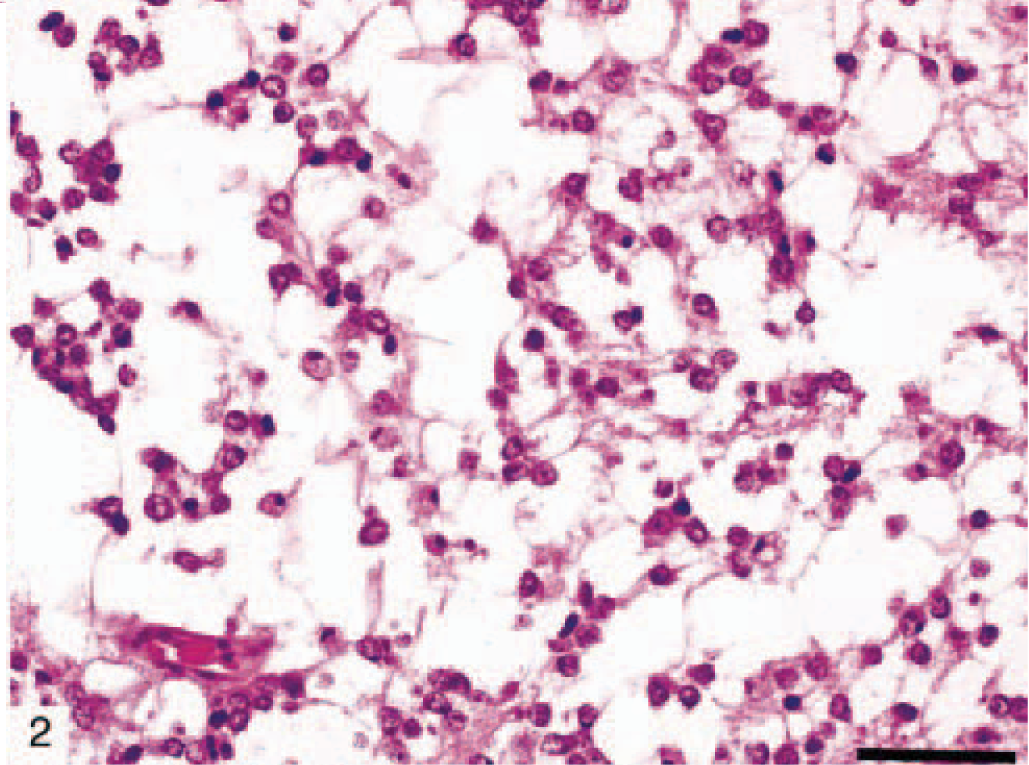
Protoplasmic canine astrocytoma (dog No. 9) consisting of relatively homogenous small astrocytic cell population with short, delicate processes. HE. Bar = 150 µm.
Gemistocytic canine astrocytoma (dog No. 24) showing large cells with eosinophilic cytoplasm and nuclei displaced to the periphery. HE. Bar = 150 µm.
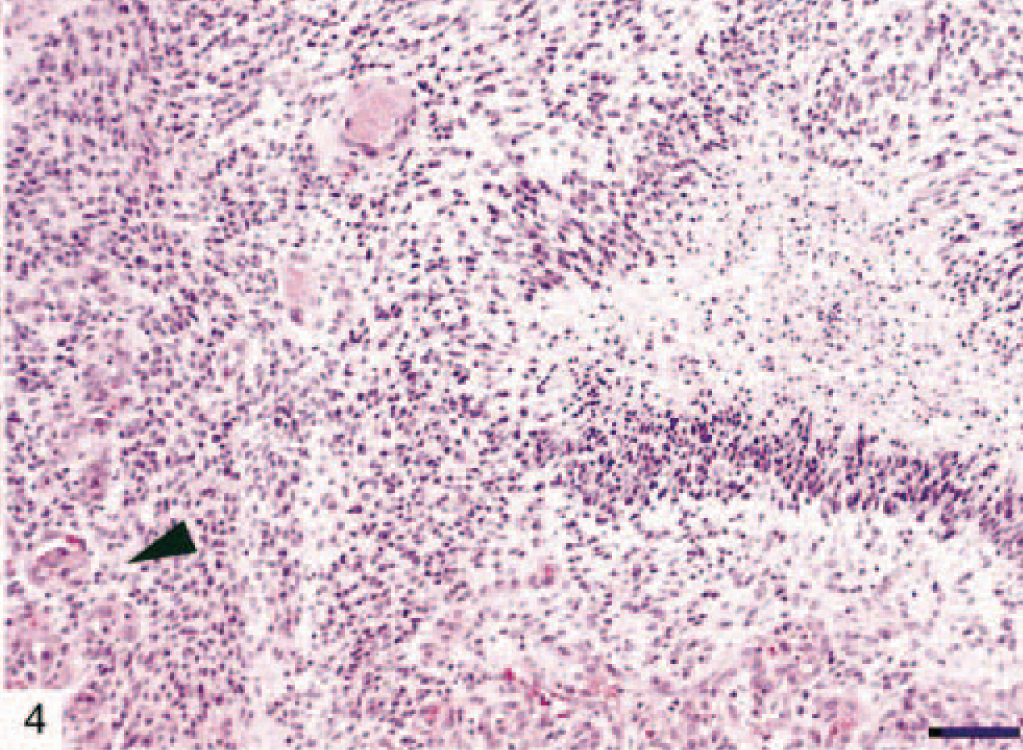
Canine glioblastoma multiforme (dog No. 28) showing characteristic features such as pseudopalisading around a zone of necrosis and glomeruloid-like vascular proliferation (arrow head). HE. Bar = 150 µm.
We encountered in our study some astrocytic tumors that are rarely described in veterinary literature. These were astroblastoma and pilocytic astrocytoma. Astroblastomas (Dog Nos. 21 and 27) showed a papillary growth pattern, radial arrangement of elongated tumor cells around a central vessel, and extensive vascular sclerosis (Figs. 5, 6). These tumors stained positively for GFAP and S-100 protein. No vascular endothelial proliferation, nuclear pleomorphism, or mitotic activity was observed in these two astroblastomas.
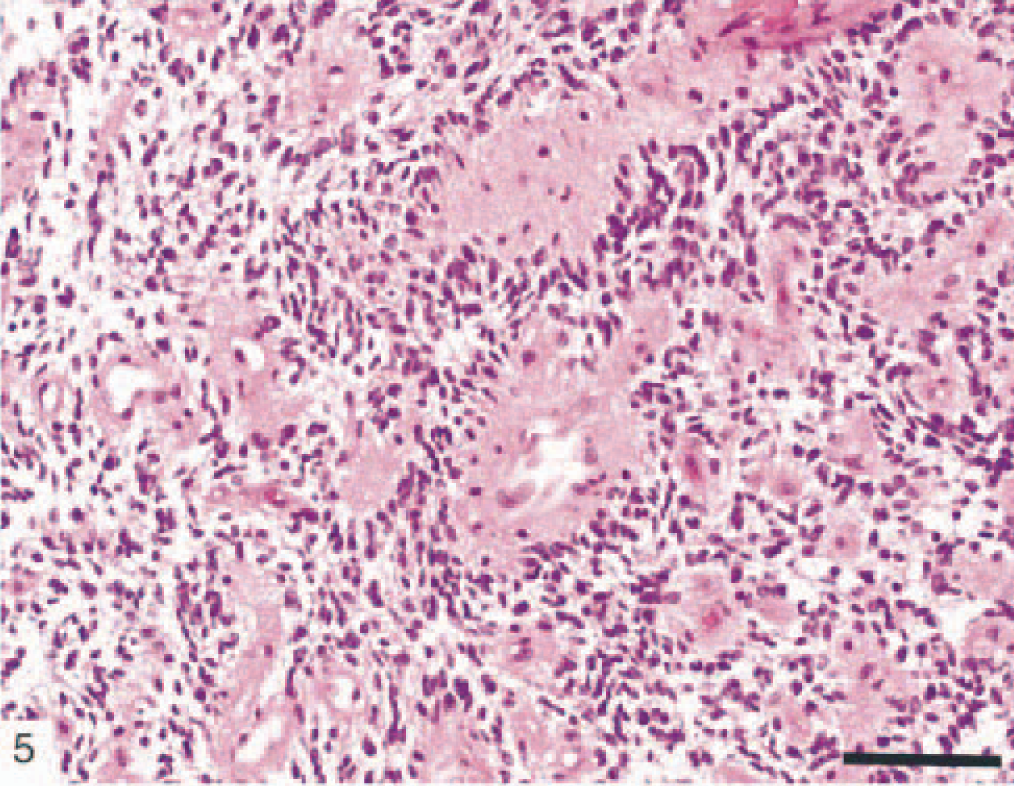
Canine astroblastoma (dog No. 27) showing a perivascular arrangement of radiating astrocytes and extensive vascular sclerosis. HE. Bar = 150 µm.
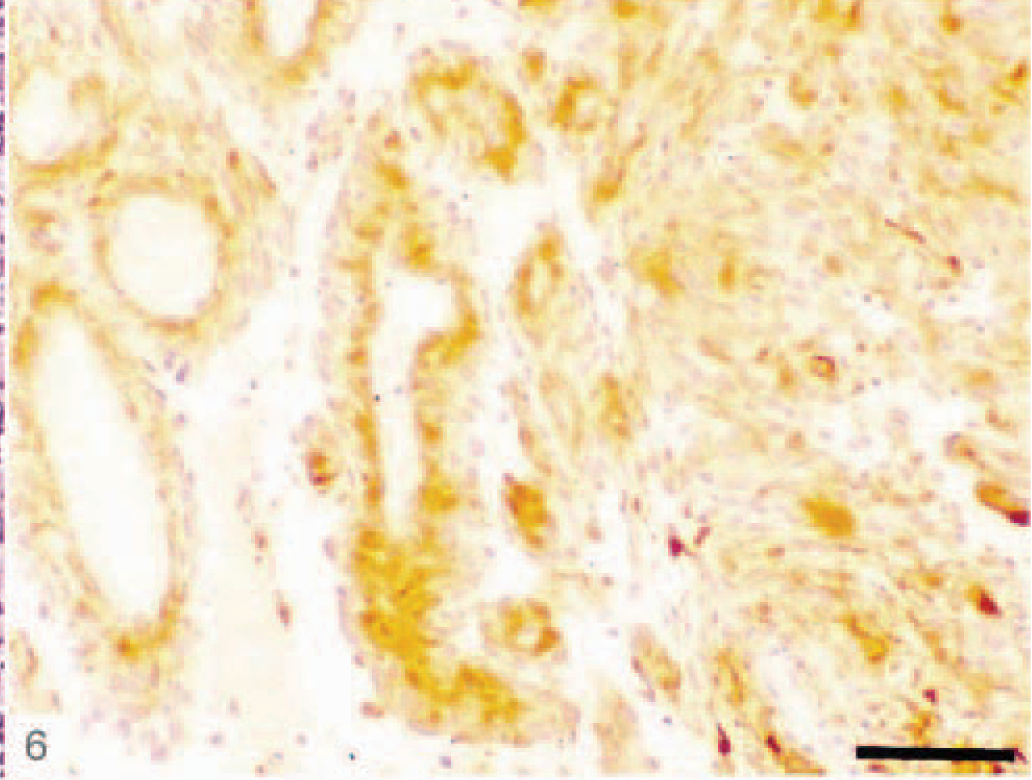
Canine astroblastoma (dog No. 27) showing the perivascular cells strongly immunoreactive for GFAP. ABC immunoperoxidase with methylene blue counterstain. Bar = 150 µm.
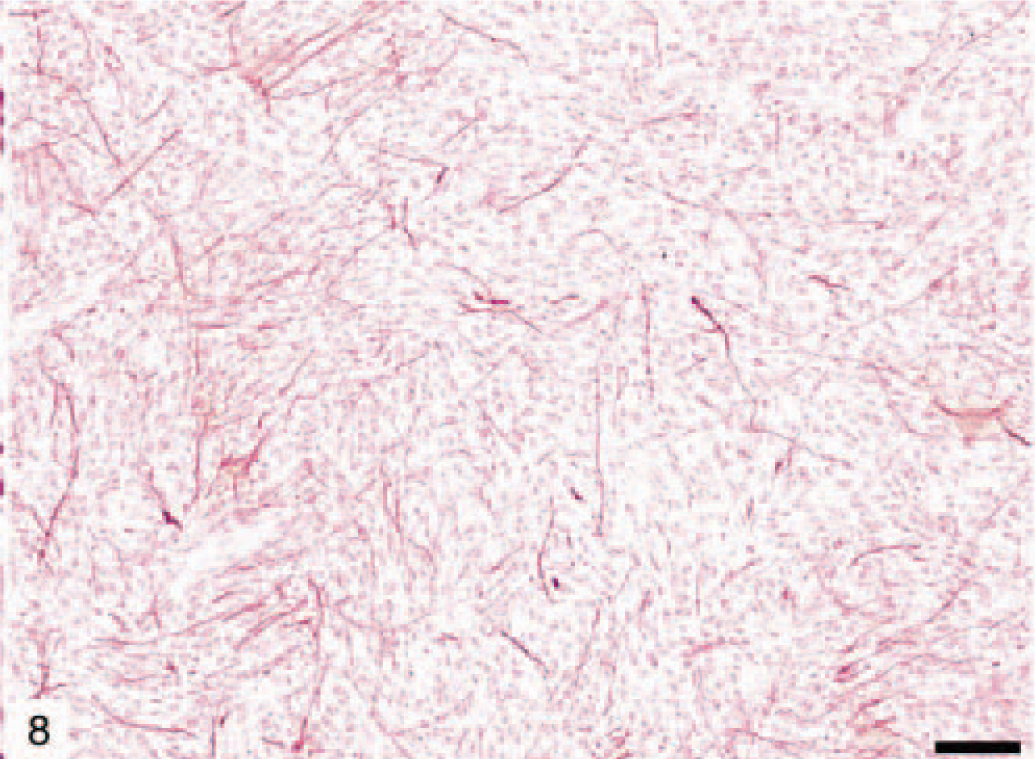
Pilocytic astrocytoma was found in two dogs (Nos. 13 and 19). On microscopic examination, these tumors were relatively well circumscribed, but not encapsulated, and showed little tendency to invade adjacent structures. This rare astrocytic tumor was characterized by low cellularity and predominantly bipolar, fibrillated, and elongated (piloid, hairlike cells) cellular morphology (Figs. 7, 8). The piloid astrocytes were often packed in bundles, usually with a perivascular distribution. Rosenthal fibers, granular hyalin droplets, eosinophilic intracytoplasmic bodies, microcyst formation, and glomeruloid capillaries were common, although mitotic figures were not observed. The pilocytic cells were positive for GFAP protein, although Rosenthal fibers and eosinophilic bodies demonstrated variable GFAP immunoreactivity.
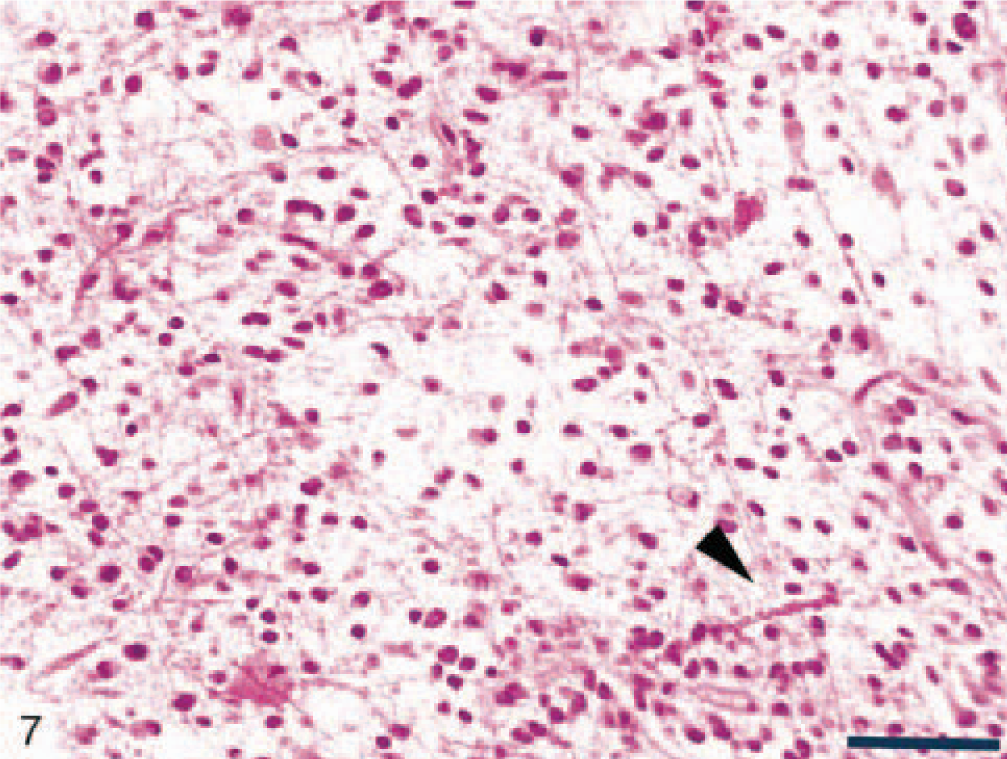
Pilocytic canine astrocytoma (dog No. 13) showing bipolar, elongated (piloid) astrocytes and Rosenthal fibers (arrow head). HE. Bar = 150 µm.
Pilocytic canine astrocytoma (dog No. 13) demonstrating GFAP immunoreactivity for piloid astrocytes. Immunohistochemical staining: streptavidin–alkaline phosphatase method. Bar = 150 µm.
Anaplastic astrocytomas (five dogs) were classified according to WHO criteria 17 and were characterized by the presence of focally or diffusely increased cellularity, nuclear atypia, and mitotic activity without endothelial proliferation or necrosis (Figs. 9, 10). The percentage of cells with nuclear staining for p53 varied from 0 to 50%, with anything above 10% considered indicative of overexpression. 47 Immunohistochemical staining revealed that 11 of 31 (35%) astrocytomas in this study had p53 protein overexpression, which was exclusively nuclear and abundant in the less differentiated areas (Fig. 11). It is noteworthy that p53 protein overexpression was predominately seen in anaplastic astrocytomas and GM multiforme, suggesting a relationship with degree of differentiation or malignancy, or both (Table 3). In 7 of 31 (23%) tumors there was an overexpression of the EGFR in the plasma membrane of astrocytic cells (Fig. 12). In three dogs, there was overexpression of both p53 and EGFR proteins.
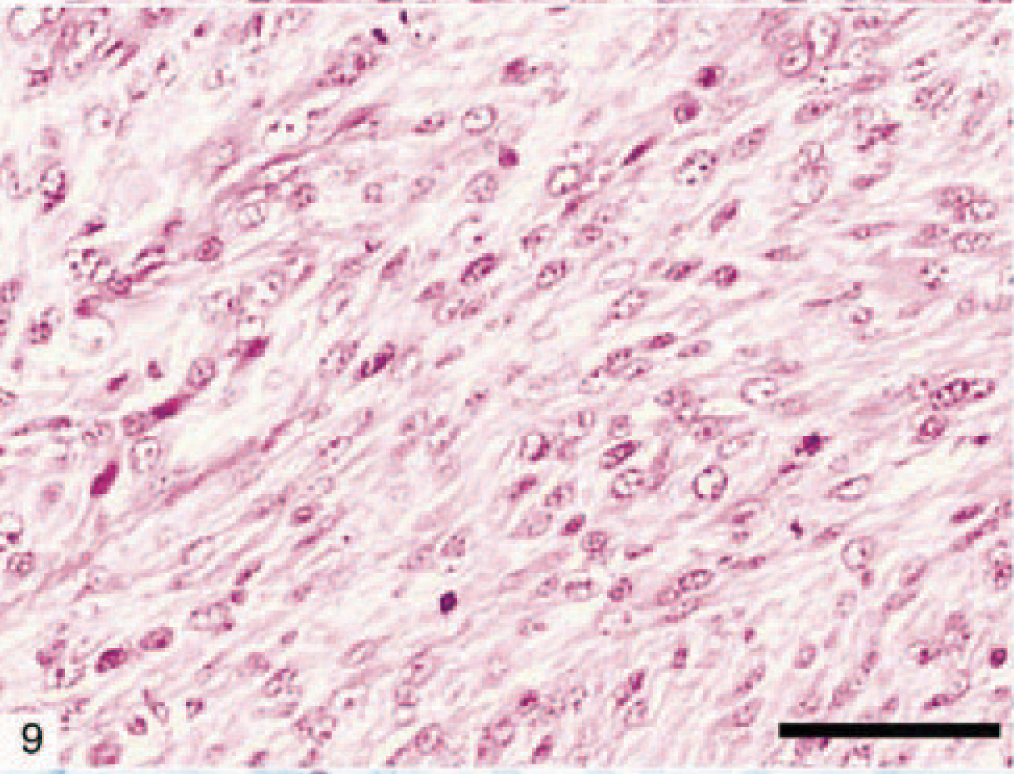
Canine anaplastic astrocytoma (dog No. 5) characterized by increased cellularity, nuclear atypia, and mitotic activity. Bar = 150 µm.
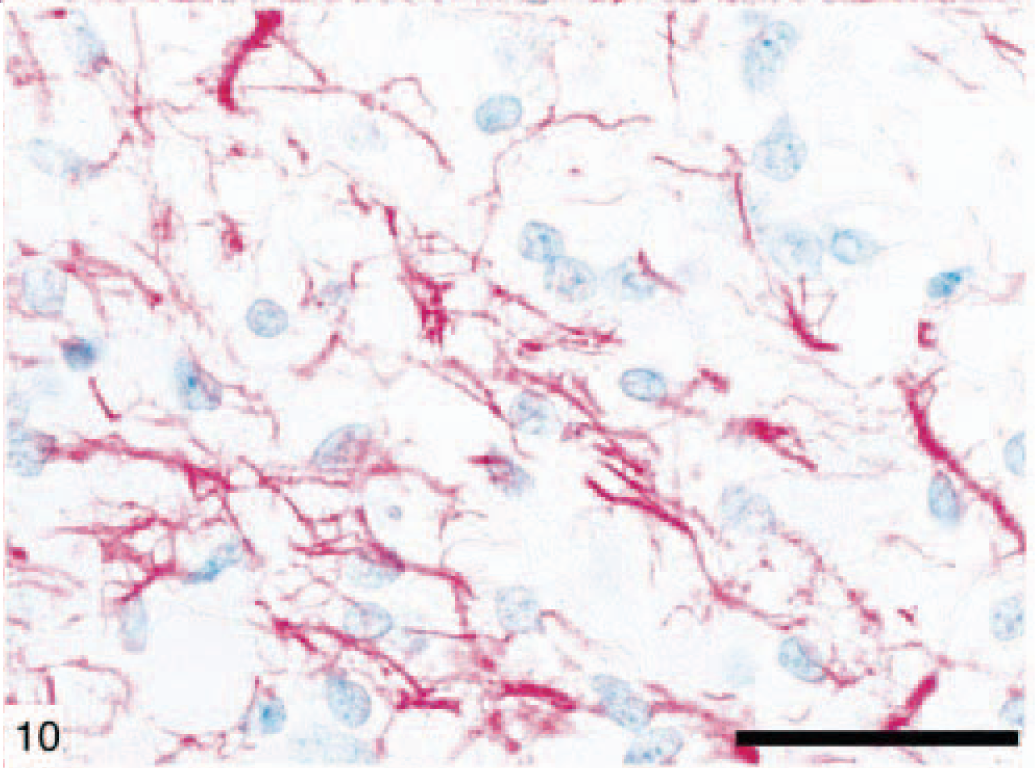
Canine anaplastic astrocytoma (dog No. 5) demonstrating GFAP immunoreactivity. Immunohistochemical staining: streptavidin–alkaline phosphatase method. Bar = 150 µm. regardless of the role of p53 gene mutation for oncogenesis and the progression of these tumors. 19,29 In contrast, there is a general consensus that EGFR over-expression is a significant predictor of poor radiographically assessed radiation response, particularly in older patients. 2,6,19 In the present study of 31 canine astrocytomas, p53 (35%) and EGFR (23%) gene products, assessed by immunohistochemistry, were over-expressed. EGFR is over expressed mainly in the malignant forms of astrocytomas and in relatively older dogs.
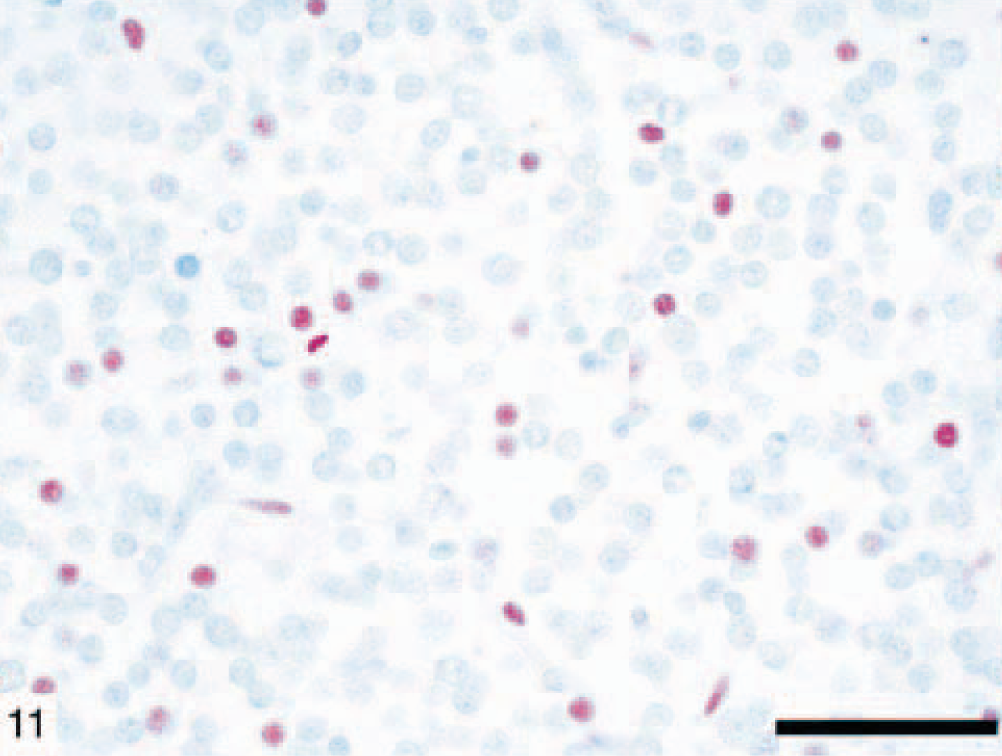
Canine glioblastoma multiforme (dog No. 7) showing abundant nuclear immunolabeling for p53 protein. Immunohistochemical staining: streptavidin–alkaline phosphatase method. Bar = 150 µm.
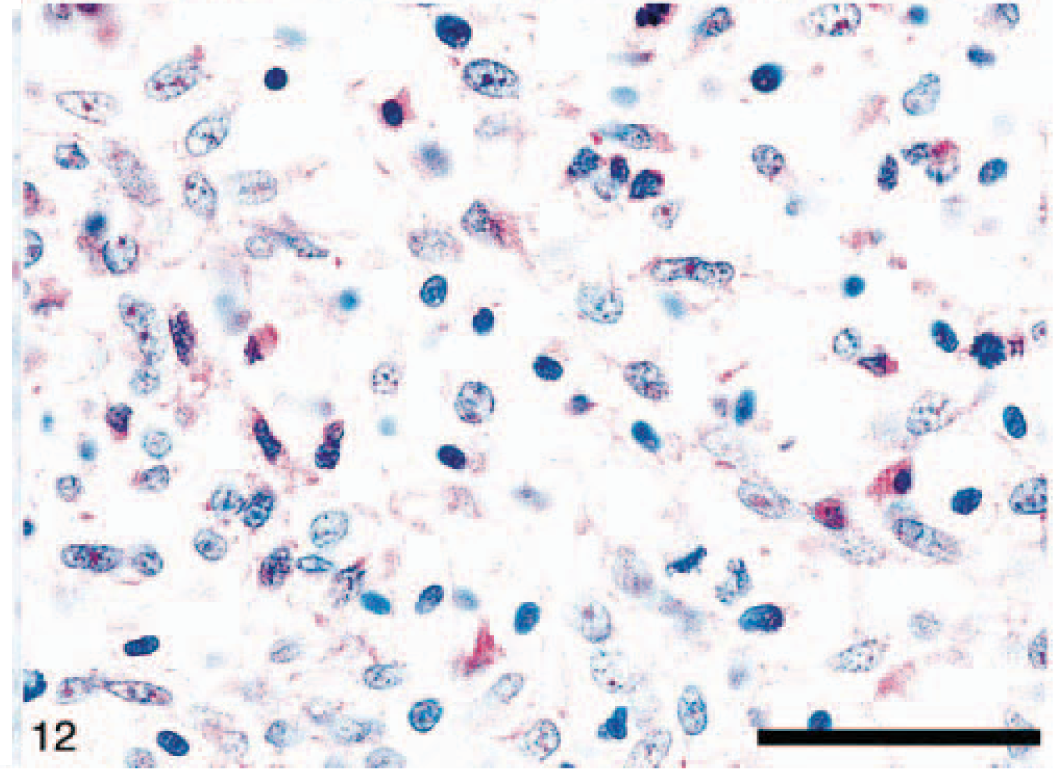
Anaplastic astrocytoma immunostaining (dog No. 12) showing overexpression of EGF receptor protein in the plasma membrane of neoplastic astrocytes. Immunohistochemical staining: streptavidin–alkaline phosphatase method. Bar= 150 µm.
DNA was extracted from all 31 samples, but the DNA was suitable for genetic analysis from only 12. A single-nucleotide substitution mutation in the canine p53 gene was detected in only one (No. 7) of these 12 dogs. A somatic mutation was located at codon 253 (ACT → GCT) in exon 7. The tumor was a GM multiforme that showed immunohistochemical overexpression for p53 but was negative for EGFR.
Discussion
In the veterinary literature there is ample morphologic characterization of typical astrocytomas in animals, 10–14,17,18,23,30,34,38,40,41,43,44 although little in comparison with other species. This study supports the conclusion that there is great morphologic and immuno-histochemical similarity between canine and human astrocytomas. This study also identified two astrocytic tumor types that are not contained in the recent “Histological Classification of Tumors of the Nervous System of Domestic Animals”. 17 These tumors are astroblastoma (two dogs) and pilocytic astrocytoma (two dogs), and their classification was based primarily on their similarity to the morphologic and immunohistochemical characteristics described to occur in humans. 16,25,46
Astroblastomas are considered to be very rare human glial tumors. They have been described and characterized by Bailey and Cushing 1 according to specific morphologic criteria and they constitute a distinct type of glial tumor. They are usually circumscribed and supratentorial in young human adults, with a characteristic microscopic pattern of perivascular arrangements of tumor cells. They are thought to derive from the astrocytic lineage, but their definitive origin and subsequent differentiation have not been elucidated using current methods of immunohistochemistry and electron microscopy. 21 The recent WHO classification includes this tumor in the category of neuroepithelial tumors of uncertain origin. 16 This conclusion is based on the fact that their precise histogenesis remains enigmatic, despite their undisputed neuroepithelial origin. Until the histogenesis of astroblastoma is clarified, this tumor can be included in the category of neuroepithelial tumors of uncertain origin as it is in the human classification.
Pilocytic astrocytoma has not been recognized to occur in animals according to the new classification of CNS tumors in domestic animals. 17 The histologic pattern encountered in two dogs during this study fits the description of pilocytic astrocytoma in humans. 16,25 Characteristic histologic features of human pilocytic astrocytoma are the predominant hairlike (piloid) pattern plus the presence of Rosenthal fibers, granular hyalin droplets, and eosinophilic intracytoplasmic bodies. 25 Pilocytic astrocytoma, based on its phenotype and biologic behavior, should be included in the low-grade astrocytoma category (WHO classification).
One of the main aims of applying immunohistochemical methods in human astrocytic tumors is to clarify the clinicopathologic significance of oncoprotein expression for prognostic value. Attempts have been made to identify biologic markers that could be useful in predicting prognosis in these tumors. For example, the relationship between p53 status and radio-resistance in tumors and tumor cell lines is a complex one. The normal pathway leading to apoptosis after DNA damage involves p53 function, including p53-dependent upregulation of p21. In GM cell lines that express wild-type p53, induction of p21 does not take place after irradiation, 2,3,7,19 suggesting that other defects in the p53 pathway may exist in these tumors. There is a relative general consensus in human literature that p53 overexpression has a questionable prognostic value for the whole spectrum of astrocytomas, regardless of the role of p53 gene mutation for onco-genesis and the progression of these tumors. 19,29 In contrast, there is a general consensus that EGFR over-expression is a significant predictor of poor radio-graphically assessed radiation response, particularly in older patients. 2,6,19 In the present study of 31 canine astrocytomas, p53 (35%) and EGFR (23%) gene products, assessed by immunohistochemistry, were over-expressed. EGFR is over expressed mainly in the malignant forms of astrocytomas and in relatively olderdogs.
The results of this study indicate that p53 alterations may be involved in development of astrocytomas in the dog. A tumor-specific somatic mutation (ACT-GCT) in codon 253 was found in one dog with a GM multiforme. Codon 253 is located in a highly conserved region and one of the seven recognized hot-spots for mutations of the p53 gene and it is therefore likely that this mutation is associated with cancer progression in this tumor. One such alteration in the conserved region of p53 could interfere with p53-DNA binding or with internal stabilization of p53. This mutation diminishes the ability of the mutant protein to carry out its activities and can bestow new dominant negative or gain-of-function properties such as neoplastic progression. 16 Comparison of immunostaining and DNA sequence alterations of p53, however, showed no significant association between overexpression and mutation. Indeed, upregulation of p53 expression does not necessarily indicate the presence of a mutation but may reflect genotoxic stress or additional genetic alterations. It also might be that there are mutations in other exons than those that were explored.
As reported in human astrocytomas, this study also demonstrated little immunohistochemical correlation between p53 and EGFR expression. Recent studies have shown that expression of EGFR is upregulated in more than 60% of human cases and constitutes a hallmark of primary GM. 16 It is also recognized from the human literature that loss or mutation of the p53 tumor suppressor gene has been detected in many types of glioma and represents an early genetic event in a subset of astrocytomas. It may be that EGFR alterations and p53 inactivation may represent distinct molecular pathways leading to astrocytic tumor formation. 16,17,31
The most commonly altered oncogene-encoded proteins in human gliomas are vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), phosphatase and tensin homologue deleted chromosome ten (PTEN), p53, and EGFR. 3,22,32,35,39 There are distinct genetic pathways leading to the GM, the most malignant astrocytic brain tumor. Primary GM usually develops in older patients and is characterized by EGFR amplification-overexpression, p16 deletion, and PTEN mutation. Secondary GM, which progresses from low-grade or anaplastic astrocytomas, develops in younger patients and frequently contains p53 mutation. 33 Genetic alterations commonly seen in GMs can have a combined effect to increase tumor progression and malignancy. For example, PTEN mutation can cooperate with EGFR activation to increase VEGF messenger ribonucleic acid level by transcriptionally upregulating the proximal VEGF promoter through the PI3K-Akt pathway. 32 Previous studies have shown that glioma cell migration, invasion, and growth are stimulated by EGFR. 42 PDGF plays an important role in the development of a subgroup of malignant astrocytic tumors that do not contain amplification of EGFR. PDGF exerts a differentiating role compared with EGFR that mainly has a mitogenic function. 35 From this study and others it seems that a certain proportion of canine astrocytomas is less differentiated tumors compared with their human counterpart. 4,23,36 Interestingly, one of these tumors, a GM (dog No. 15) that was equivocal (±) for GFAP expression in vivo, became positive for GFAP and vimentin after culture in vitro (G. Stoica, unpublished). It may be that these cells derive from a less differentiated glial precursor cell (stem cells), which on histology shows a less differentiated astrocytic pattern.
In summary, this and other studies reveal that a broad spectrum of astrocytomas occurs in dogs with phenotypic and genetic alterations similar to those in humans. Our findings correlate the morphology with the immunophenotype, genotype, and overexpression of p53 and EGFR genes. Because owners increasingly request biopsy, surgical removal, and therapy of brain tumors in their pets, there is a need in veterinary neurooncology for an improved classification including a grading system that is based on new knowledge related to the genetic events that are pertinent to the biologic behavior and clinical responses of these tumors to novel therapies. Investigation of spontaneous brain tumors in animal species may also be used as diagnostic and therapeutic models for comparable human neoplasms.
Footnotes
Acknowledgements
This work was supported in part by an interdisciplinary enhancement grant from Texas A&M University. We thank Mrs. Wollmar Rosemary and Dr. Sandy Ambrus for their technical assistance with histology and immunohistochem-istry.
