Abstract
Tumors of astrocytic lineage are among the most common primary brain neoplasms in people and dogs. Current understanding of the pathogenesis of astrocytic tumors is limited in dogs compared with humans. In dogs, critical biological data concerning the natural history of disease progression, tumor imaging features, and response to therapeutic intervention are lacking. This review outlines the clinical, genetic, immunologic, and histopathologic characteristics of astrocytic tumors in dogs with special focus on comparative neuro-oncology. Common problems associated with the diagnosis of these neoplasms are summarized. Traditional veterinary histologic typing and grading of astrocytic tumors must be updated and supplemented with molecular data so that future studies directed toward therapeutic intervention and outcome can be optimized.
Astrocytic tumors in dogs are made up of neoplastic cells that phenotypically resemble astrocytes. The cell of origin of these tumors is still a matter of controversy. This uncertainty about the cell of origin is due to the fact that no premalignant states are recognized, as is the case in some solid epithelial tumors. The presence of cancer stem cells in canine glioblastoma multiforme (GBM) cell lines suggests that some astrocytic tumors might originate from preexisting neural stem cells; the subventricular zone of the lateral ventricles is a known location for resident neural stem cells. 73,85 The hypothesis that the subventricular zone is a potential target for gliomagenesis is supported by older literature demonstrating that a single dose of the alkylating agent ethylnitrosourea to pregnant rats during the late stage of gestation selectively induced periventricular brain tumors of a variety of phenotypes in the offspring. 37,39
Histologic classification of canine astrocytic tumors depends on the recognition of areas of tumor cells that express phenotypic characteristics of astrocytes and the immunohistochemical detection of glial fibrillary acidic protein (GFAP) in astrocytes. No antigen is exclusively expressed by astrocytic tumors. The presence or absence of GFAP might help in the classification of astrocytic tumors, but it cannot be considered an absolute criterion, especially in poorly differentiated tumors.
The veterinary World Health Organization (WHO) classification of 1999 designated five astrocytic tumors (some with subgroups) based on histopathologic characteristics. Three malignancy grades were recognized by the veterinary WHO system: low-grade, medium-grade, and high-grade astrocytomas. 38 However, little information exists in the veterinary literature regarding their malignant progression and biological behavior.
In contemporary veterinary medicine, efforts are being made to provide high-quality care for companion animals by using advanced imaging technologies, such as computerized tomography (CT) and magnetic resonance imaging (MRI), to detect brain tumors. The request of pet owners for biopsy, surgical removal, and therapy for brain tumors has increased in recent years. Additionally, members of the biomedical community perceive dogs with naturally occurring primary brain tumors to be a potential preclinical model for therapeutics that may be developed for human disease. Therefore, there is a need to improve our understanding of the biology of canine astrocytic tumors and their classification.
Incidence, Clinical Features, and Anatomical Location of Astrocytic Tumors
A large body of literature exists concerning the incidence of cancer in people. The Central Brain Tumor Registry of the United States pools data from 44 cancer registries of newly diagnosed human primary brain and central nervous system (CNS) tumors. Between 2004 and 2005, the overall incidence of primary brain and CNS neoplasms in people in the United States was reported as 18.2 per 100,000. 7 Tumors of astrocytic lineage had an incidence of 4.56 per 100,000, with GBM representing 70% of newly diagnosed astrocytic neoplasias. 7 In Rochester, Minnesota, the incidence of astrocytic brain tumors in people between 1950 and 1990 was 4.9 per 100,000. 61 Data from psychiatric patients in the Northeast United States that were autopsied between 1884 and 1954 indicated 928 of 44,780 (2.1%) had primary brain tumors. 35
The true incidence of primary brain tumors is challenging to determine, especially in veterinary medicine. A study of dogs living in California between 1963 and 1967 indicated that the incidence of peripheral and CNS tumor was 14.5 per 100,000. 69 More recent data obtained from dogs living in the United Kingdom between 1997 and 1998 reported the incidence of brain tumor to be 20 per 100,000. 19 A retrospective single-center study indicated that in a population of 6,175 dogs undergoing necropsy, 118 (1.9%) had primary brain tumors, 70% of which were gliomas. 51 Astrocytic tumors have been estimated to constitute 17 to 28% of all canine primary CNS tumors. 25,71
In people, astrocytic tumors appear to be much more common in Caucasians than in other races. In 38,453 people studied between 1973 and 2001, the relative risk of malignant brain tumor was 1.86 in Caucasians compared with African Americans. 15 A study of people living in Memphis, Tennessee, showed that the Caucasian:African American incidence ratio for low-grade astrocytoma was 3.5:6.2 for GBM. The underlying reasons for racial predilections in people are unknown. 64
In dogs, breed predispositions for several brain tumor types have been identified. 25,76 Boxers and Boston Terriers have a high frequency of astrocytomas, with a relative risk of 5.2 and 23.3, respectively, of developing any glial tumor, compared with reference breeds. 25
In people, the incidence of astrocytic neoplasia is related to sex, and it increases with age. GBM is found more commonly in males than females. 7,58 The incidence of GBM in individuals aged 20 to 34 years is 0.4 per 100,000, whereas the incidence in individuals aged 75 to 84 years is 14.61 per 100,000. 7 In addition, the mean age of individuals developing low-grade astrocytoma is substantially younger than those with anaplastic astrocytoma or GBM. 7,44,58 The risk for developing glioma in dogs also increases with age. 25 In one study of dogs, the mean age (± standard deviation) at the time of astrocytoma diagnosis was 8.6 ± 3.3 years. 71 No relationship between sex and astrocytic tumor development has been recorded for the canine species. 25,71,72
The clinical signs associated with dog astrocytic tumors are diverse and so reflect the neuroanatomical distribution of the lesion. In general, astrocytic tumors result in progressive, lateralized neurologic deficits. Even highly malignant tumors such as GBM do not commonly appear to result in multifocal neurologic signs. 42,47,70 –72,79,80 Data from two large retrospective studies suggest that in dogs, intracranial astrocytomas are far more common than those arising within the spinal cord (79 versus 10 reported, respectively). 25,72 The most common clinical signs associated with intracranial astrocytic tumors in dogs are mentation changes (14 of 30), seizures (12 of 30), vestibular disturbances (9 of 30), and vision loss (7 of 30). 71
The telencephalon and diencephalon are the most common sites for intracranial astrocytic tumors in dogs. In one retrospective study, 23 of 31 dogs had lesions arising in the telencephalon and 4 of 31 from the diencephalon; no dogs were described as having more than one anatomic area invaded by tumor. 72 Similarly, a study on 5 dogs with GBM noted that all masses were restricted to either the prosencephalon or the caudal brainstem. 47
Magnetic Resonance Imaging
MRI has dramatically enhanced the detection and characterization of astrocytic tumors in people, and it will likely provide the same benefits for dogs. In people, diffuse astrocytoma (WHO grade II) appears as a homogeneous intra-axial mass that has high T2 signal characteristics and variable contrast enhancement; typically, there is limited peritumoral T2 hyperintensity. 31 GBM appears as a large mass of heterogeneous T2 signal on MRI. 31 Signal heterogeneity is believed to be due to combinations of edema, necrosis, and hemorrhage, which are common in this tumor type. GBM may have areas of cystic change (high T2 signal and low T1 signal). Mass effect is significant; tumor margins are often indistinct; and enhancement is typically present but heterogeneous. 31 Whereas the ability of MRI to group brain tumors into broad diagnostic categories (eg, meningeal, glial, metastatic) is excellent, traditional MRI techniques are limited in their ability to accurately assign histologic tumor type and grade. 14,33,74 For example, in one large retrospective study, the ability of sensitivity and specificity to correctly categorize a tumor as low-grade astrocytoma was 14% and 97%, respectively. 33 Relative cerebral blood volume maps, MRI volumetric texture, and magnetic resonance spectroscopy may hold promise for more objectively differentiating astrocytoma type and grade. 22,45
Information concerning the MRI appearance of astrocytic tumors in dogs is limited. Low-grade astrocytoma has been described as being intra-axial with high T2 signal and isointense or hypointense T1 signal; contrast enhancement has been described as being mild, and peritumoral high T2 signal is limited. 41 A case series of 5 dogs with GBM indicated that all tumors appeared intra-axial and had heterogeneous T2 signal, low to isointense T1 signal, and significant peritumoral edema. 47 Contrast enhancement was variable, and most tumors had sharp margins.
Stereotactic Brain Biopsy
Until the middle of the 20th century, obtaining brain tissue from people in vivo to allow for histopathologic diagnosis was challenging. Open approaches to the brain could be used, but visualizing deeply located lesions was complicated by a lack of multiplanar imaging and the ability to locate abnormal structure. By the 1980s, the development of stereotactic brain biopsy and CT/MRI made it possible to reach various intracranial lesions in people, with an accuracy of 1 mm or less. 34 The reported diagnostic accuracy of stereotactic biopsy samples, when compared with samples acquired through open surgery, is 91 to 97%. 46,62 Reported morbidity (1 to 13%) and mortality (0 to 3%) from biopsy are low across multiple human studies. 1,46,62,84
Several articles have reported the use of frame-based stereotactic systems in dogs, with varying accuracy. The Kopf system was recently utilized in small dogs and found to correspond to selected supratentorial targets within 0.9 ± 0.9 mm. 78 An adaptation of the Pelorus III device 36 was accurate to 3.5 ± 1.6 mm, and a manufactured device described by Moissonnier et al 53 had a reported accuracy of 2.9 ± 1.0 mm. Despite numerous reports on the validation of frame-based systems in canine cadavers, there is only one large study 36 describing clinical findings from neurologically affected dogs. In that report, there was agreement between stereotactic biopsy and open biopsy or necropsy in 20 of 22 cases (91%). 36 Adverse events associated with biopsy were recorded in 5 of 41 animals (12%), with 3 animals (7%) requiring euthanasia. 36 In a smaller report, adverse events associated with stereotactic biopsy were described in 6 of 23 dogs (27%), with mortality occurring in 2 of 23 dogs (8%); diagnostic accuracy could not be determined. 53
It seems likely that in veterinary medicine, frameless stereotactic brain biopsy will replace frame-based approaches, as has occurred in people. These systems are more flexible in species with varying head shape; they are easier to operate; and they are likely to offer accuracy that is equivalent to frame-based techniques. 13,84
Genomics and Tumor Genetics
In human neuro-oncology, there are a number of familial syndromes with an increased incidence of astrocytic tumors, such as neurofibromatosis type 1 and 2 and Li Fraumeni syndromes. 50 In veterinary medicine, no such genetic associations have been described, although some brachycephalic breeds are overrepresented among dogs with astrocytoma. 76
A recent microarray-based comparative genomic hybridization analysis of 25 canine gliomas (9 of astrocytic origin) showed chromosome copy number aberrations (CNAs) that share evolutionarily conserved synteny with those previously reported in their human counterpart. 77 For example, CNAs were found in the canine orthologue to the human 7p chromosome, which contains the EGFR gene; CNAs in this region were present in a large proportion of high-grade canine gliomas, as is the case in people. There were also CNAs in canine chromosomal regions containing the MYC oncogene in 5 of 9 astrocytic tumors. As is the case in people, cytogenetic alterations were more strongly associated with glioma histologic grade than type.
Histopathology of Canine Astrocytic Tumors
Differentiated, low-grade astrocytoma
The veterinary WHO classification characterizes differentiated, low-grade astrocytoma into fibrillary, protoplasmic, and gemistocytic subtypes. In fibrillary astrocytomas, the tumor cells exhibit a phenotype that is characteristic of astrocytes with no nuclear atypia and limited perinuclear cytoplasm. These tumors generally express S-100 protein and GFAP. Microcysts may be present, and the neoplastic cells diffusely infiltrate the brain parenchyma to varying degrees. Protoplasmic astrocytoma is less common than the fibrillary subtype of astrocytic tumors and is composed of small rounded astrocytes with scant GFAP expression and with mucoid degeneration and microcyst formation. Cellularity is low and mitotic activity absent.
Large cells with a peripherally located nucleus and abundant eosinophilic cytoplasm characterize gemistocytic astrocytoma. The gemistocytic neoplastic astrocytes consistently express GFAP in their cytoplasm and cell processes. In people with WHO grade II astrocytoma (diffuse astrocytoma), malignant progression is more rapid in the gemistocytic subtype compared with fibrillary subtype, despite the fact that gemistocytes are in a nonproliferative state, suggestive of terminal differentiation. 50
Pilocytic astrocytoma is reported to occur in dogs, 72,76 but it is not included in the veterinary WHO classification; pilocytic astrocytomas are classified as WHO grade I in people. Some reports include this type of astrocytic tumor as a histologic differentiating pattern within fibrillary astrocytoma. 76 Despite its rarity in the dog, we consider pilocytic astrocytoma to be a specific entity and consequently believe that it should be included in the veterinary classification of the astrocytic tumors as it is in the current human WHO classification. 50
Hypercellularity characterizes the anaplastic astrocytomas, and they demonstrate cellular pleomorphism, distinct nuclear atypia, mitotic activity, and occasional multinucleated tumor cells. Anaplastic astrocytoma retains histologic and immunohistochemical characteristics of astrocytes without evidence of necrosis or abnormal neovascularization. Anaplastic astrocytoma is classified as medium-grade astrocytoma in dogs and grade III astrocytoma in the human WHO classification.
Glioblastoma (GBM)
Canine GBM is categorized as high-grade astrocytoma in veterinary medicine (WHO, 1999) and grade IV malignancy in the human classification (WHO, 2007). 38,50,76 GBM is the most malignant variant of astrocytic tumors and is characterized by its cellular heterogeneous phenotype, necrosis with pseudopalisading of tumor cells around necrotic areas, marked microvascular proliferation, and occasional multinucleated cells. As the term suggests, the microscopic pathology of GBM can be extremely variable. The pathognomonic feature of GBM is intratumoral necrosis of varying extent and distribution. No histologic feature is more powerful in predicting poor prognosis. 48 Some GBMs exhibit acute areas of ischemic necrosis due to occlusive fibrin thrombi. However, the most classic pattern consists of branching serpentine channels of necrosis delineated by palisading glial cells oriented perpendicular to this necrotic area. The microvascular proliferation occurs at the periphery of necrotic areas or in long ribbon-like aggregates around cystic areas or near the tumor–brain parenchyma interphace. 4,72,76 However, such features are sometimes seen in high-grade oligodendrogliomas and ependymomas. 76 This heterogeneous cellular phenotype suggests a common histogenesis, most likely from stem cells located in the subventricular region or from the preexisting neoplastic glial cells reprogrammed to stemness attributes. 12
Differentiated tumor cells in canine GBM, such as gemistocytes or anaplastic fibrillary astrocytes, are immunoreactive for GFAP and vimentin. There is a large population of undifferentiated rounded tumor cells in canine GBM that exhibit inconsistent GFAP staining but consistent immunoreactivity for vimentin. Although vimentin expression is not specific for astrocytes, its expression with GFAP in these tumors is suggestive of their glial origin. 50 Among the tumor models studied, the spontaneous canine GBM is the only one that exhibits endothelial cell proliferation, as demonstrated by Von Willebrand factor and CD34 staining of endothelial cells. 4,73 It is also significant that canine GBM is highly invasive, exhibiting the classic pattern of human GBM invasion, which is characterized by the diffuse spread of neoplastic cells within the brain parenchyma. 11,32,47,72,73 The GBM infiltrative path into the normal brain parenchyma is not random; it often follows white matter tracts and extends along perivascular spaces, the glial limitans externa, and the subependyma. Human and canine GBM exhibit similar histopathologic features—namely, nuclear atypia, cellular pleomorphism, mitotic activity, vascular thrombosis, microvascular proliferation, multinucleated giant cells, and necrosis. Glioblastomas in people are recognized as primary de novo tumors without recognizable precursor lesions and as secondary tumors that develop from preexisting diffuse astrocytoma (WHO grade II) or anaplastic astrocytoma (WHO grade III). Multifocal glioblastomas are also described in people, which may imply multiple independently arising gliomas. 48,50,58 No such subclassification is recognized in veterinary medicine.
Astroblastomas
Astroblastomas are rare brain tumors sporadically reported to occur in dogs. 10,72 Astroblastoma is believed to be an astrocytic tumor of uncertain histogenesis. 50 In this tumor, the astrocytes are palisading around blood vessels in radial (cartwheel-like) patterns. Astroblastoma with extensive vascular sclerosis can be a common occurrence. Cytoplasmic immunoreactivity for vimentin, S-100 protein, and GFAP is characteristic of astroblastoma. The histogenesis of astroblastoma is controversial, with some authors suggesting the tanycyte as the cell of origin. 50 In people, this rare glial neoplasm mainly affects children and young adults and has been associated with certain chromosomal abnormalities not found in other glial tumors, such as astrocytomas or ependymomas. 3 The biological behavior is variable, and in the absence of sufficient clinical–pathologic data, the WHO grade is not established. Differentiating astroblastoma and ependymoma is challenging because both demonstrate similar biomarkers and histopathologic patterns. Electron microscopic examination of tumor samples could be helpful. Electron microscopy of astroblastoma shows abnormal blood vessels with abundant basal lamina and collagen formation. The neoplastic cells, often radially oriented toward the blood vessels, contain intermediate filaments of 10 nm, in addition to the usual cell organelles. 44 Ependymomas maintain characteristic ultrastructural properties of ependymal cells, such as cilia, blepharoblasts, and microvilli located at the luminal surface; junctional complexes at the luminal surface; and lack of a basement membrane at the internal surface. 50
Variants of glioblastomas
Variants of glioblastomas, such as giant cell glioblastoma, are sporadically reported to occur in dogs, 79 and they also are not included in the veterinary classification of CNS tumors. This variant is characterized by bizarre multinucleated cells and expression of a reticulin network. In people, they are interpreted as WHO grade IV malignancy with frequent p53 mutations. 50 Gliosarcoma is another rare variant of glioblastoma, which is occasionally reported in the veterinary literature 80,81 and not included in the present classification of astrocytic tumors.
Gliomatosis cerebri
Gliomatosis cerebri is a well-recognized entity in people, characterized by diffuse, widespread infiltration of the brain with neoplastic glia; gross structural architecture is preserved. 50 According to the WHO, gliomatosis cerebri in people and dogs is currently classified as a neuroepithelial tumor of unknown origin. 38,50 An astrocytic origin is suspected, and although differentiation from diffuse astrocytoma can be challenging, gliomatosis cerebri is distinguished from astrocytoma by more widespread neuraxial involvement and preservation of brain structure. 60
Spongioblastoma
In people and dogs, spongioblastoma is a CNS neoplasm of presumed neuroglial lineage. The histology of this tumor is poorly defined. It arises near the third ventricle and has a characteristic pattern of palisaded, slender fusiform cells. A few spongioblastomas have been described in dogs but were diverse with respect to age at presentation, architecture, and cellular morphology. 76
Mixed glioma
Mixed glioma represents a tumor with neoplastic oligodendrocytes and astrocytes separated or intermingled within the tumor mass. Oligoastrocytoma corresponds histologically to WHO grade II. 50 The histogenesis of this mixed tumor of the CNS is unresolved, but derivation from multiple potent progenitor cells able to undergo astrocytic and oligodendroglial differentiation is a tenable hypothesis. 50
Astrocytic Tumor Biomarkers
The expression of biological molecules (biomarkers) in tumor tissue is of increasing interest. Biomarkers may provide diagnostic, prognostic, or predictive information. 86 Diagnostic biomarkers can be used as adjuncts to traditional histopathology in tumor typing and grading. Prognostic biomarkers are associated with survival or remission duration at the time of initial diagnosis. Finally, predictive biomarkers can assist in the selection of a therapeutic modality. 9 In naturally occurring and induced astrocytic tumors, various biomarkers have been associated with neoplastic transformation, tumor invasiveness, tumor progression, and angiogenesis.
Epidermal growth factor receptor
Epidermal growth factor receptor (EGFR) has received substantial attention because mutations are common in human malignant astrocytomas and seem to be involved in the progression between tumor grades. Activating mutations and gene amplification enhance EGFR signaling in glioblastoma. Increased EGFR activity facilitates the Shc-Grb2-Ras and PI3K pathways, enhancing mitogenesis and angiogenesis while reducing apoptosis. 48,86 Additionally, overexpression of EGFR seems to facilitate glioblastoma leading edge invasion via upregulation of metalloproteinases and collagens. 24,43,48,52 In a subset of dogs with malignant astrocytomas, EGFR alteration has been associated with tumor invasiveness, but the predictive or prognostic value of EGFR status has not been examined. 72 Increased EGFR expression was recently demonstrated by tissue microarray immunophenotyping in 57% (4 of 7) of GBMs, 40% (2 of 5) of grade III astrocytomas, and 28% (2 of 7) of grade II astrocytomas. 26 In people with glioblastoma, the presence of EGFR mutation may be predictive of response to tyrosine kinase blockade, as seen in individuals with other genotypic alterations. 28,58,86
Vascular endothelial growth factor and its receptor
Data from the human literature have demonstrated that vascular endothelial growth factor (VEGF) and its receptor (VEGFR) support tumor neovascularization, vascular permeability, and mitogenesis. These events may enhance peritumoral edema and alter intracranial blood flow. Additionally, the expression of VEGF can be upregulated through a multitude of mechanisms, including EGFR or p53 mutation and activation of the PI3K pathway. 30,83 VEGF is released by tumor cells, and its expression may be facilitated by hypoxia secondary to necrosis. 20 The effects of VEGF are mediated through VEGFR-1 (also referred to as FLT-1) and VEGFR-2 (also referred to as KDR), which are expressed on blood vessels within astrocytic tumors and at tumor margins. 8,27,48 Normal human brain endothelial cells are largely devoid of both VEGFR subtypes. 8,27,48
In people with astrocytic neoplasia, VEGF expression is greatest in high-grade tumors, especially GBM, where immunostaining is intense in glial cells palisading around necrosis. 40,59 As is the case in people, the concentration of intratumoral VEGF in dogs with astrocytic neoplasia increases with tumor grade. 16,65 Three canine VEGF mRNA isoforms (VEGF120, VEGF164, VEGF188) are recognized in astrocytic tumors, with VEGF164 having the highest relative expression. 16 The expression of VEGF mRNA is significantly greater in tumor tissue of dogs with GBM (grade IV) compared with dogs with grade II astrocytoma. 17 High-grade human astrocytic tumors, especially GBM, have greater concentrations of VEGFR-1 and VEGFR-2 mRNA, compared with low-grade astrocytomas. 27 Both VEGFR-1 and VEGFR-2 mRNA have also been identified in astrocytic tumors from dogs, although among 23 kinds of canine astrocytic neoplasia, expression levels did not significantly differ on the basis of tumor grade. 16 In people, astrocytic tumor VEGF expression has not been shown to be an independent prognostic biomarker associated with survival. 21,30,40,55 The value of VEGF and VEGFR as predictive or prognostic biomarkers in dogs with astrocytic neoplasia has not been examined.
p53 and platelet-derived growth factor receptor
In addition to EGFR, VEGF, and VEGFR, both p53 and platelet-derived growth factor receptor (PDGFR) have been investigated in canine astrocytoma. The p53 tumor suppressor normally functions to halt the cell cycle at G1, induce apoptosis, or arrest proliferation by binding promoter regions of various effector genes. 48 In people, loss of p53 function appears to be an early event in malignant transformation of astrocytes and is therefore present in low- and high-grade neoplasia. 48,49 Cases of canine astrocytoma with p53 mutations have been identified, although attempts to determine diagnostic, prognostic, or predictive value have not been made. 72 Mutation of p53 or alteration of the p53 pathway in human astrocytoma appears to be of limited diagnostic, prognostic, or predictive value because it is relatively ubiquitous even in diffuse astrocytoma. 86
PDGFRα is normally expressed on neural stem cells within the subventricular zone and glial precursors; stimulation of these populations with PDGF leads to glioma-like growths in rodents. 23 PDGFRβ is found on endothelial cells associated with glioblastoma, and agonists of this receptor may facilitate angiogenesis. Overexpression of PDGFRα in human diffuse and anaplastic astrocytoma has been reported. 29,63,82 In contrast, in a population of 11 dogs with varying grades of astrocytoma, minimal increases in PDGFRα mRNA were detected in tumor tissue compared with controls. 16 Recently, overexpression of PDGFRα was demonstrated by tissue microarray immunophenotyping in 43% (3 of 7) of GBMs, 20% (1 of 5) of grade III canine astrocytomas, and 14% (1 of 7) of grade II canine astrocytomas. 26
In people with astrocytoma, the prognostic and predictive value of PDGFRα expression within tumor tissue is controversial and clouded by inconsistencies in patient populations, analytic methods, and study design. For example, in diffuse astrocytoma, one report showed that high PDGFRα was associated with prolonged survival, whereas another showed that overexpression was correlated with shorter survival. 67,68,83
Insulin-like growth factor-binding protein 2
In addition, overexpression of insulin-like growth factor–binding protein 2 (IGFBP2) was detected in 71% (5 of 7), 60% (3 of 5), and 28% (2 of 7) of GBMs, grade III canine astrocytomas, and grade II canine astrocytomas, respectively. 26 The incidence of overexpression of EGFR, PDGFRα, and IGFBP2 in these canine gliomas closely parallels that in human tumors of similar type and grade. 26 Overexpression of IGFBP2 in human higher grade astrocytomas is predictive of poorer prognosis. In human GBMs, there is a significant correlation between overexpression of IGFBP2 and elevated MMP-2 expression, which relates to increased invasiveness and malignancy. Table 1 outlines the immunohistochemical markers observed in canine astrocytic tumors.
Histopathologic and Immunohistochemical Features in Canine Astrocytic Tumors a
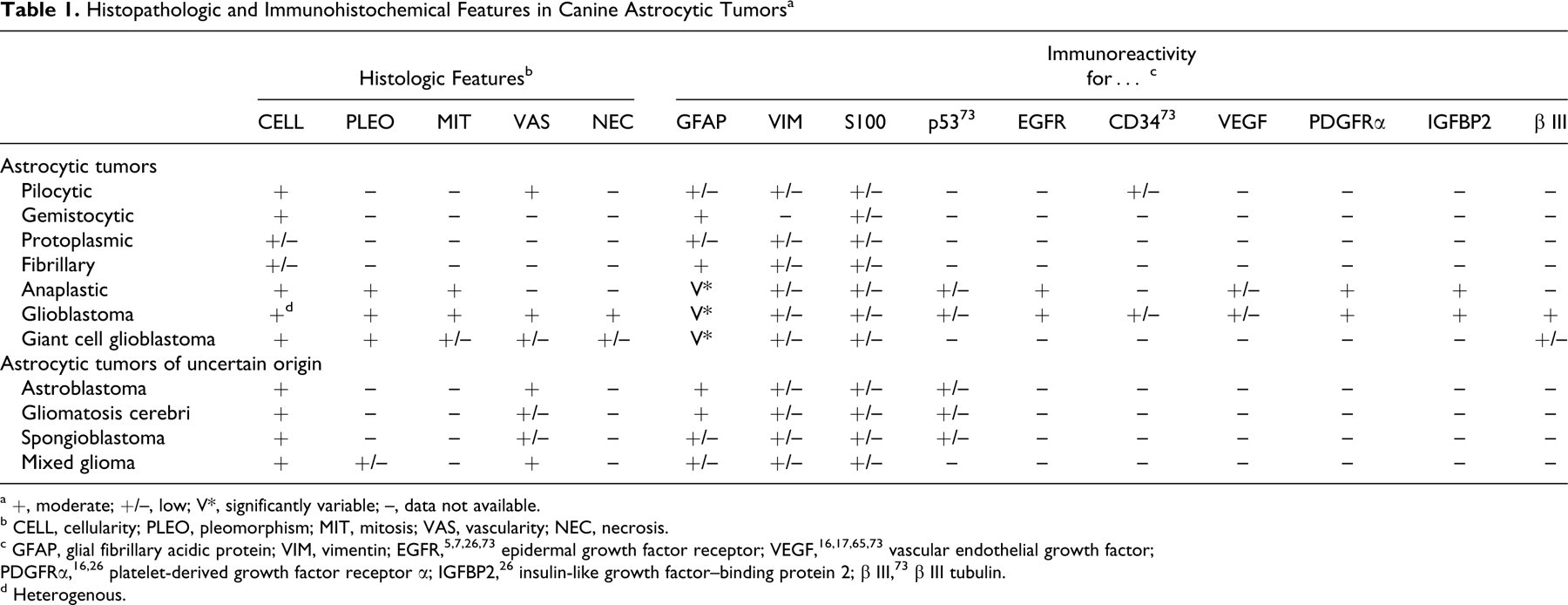
a +, moderate; +/–, low; V*, significantly variable; –, data not available.
b CELL, cellularity; PLEO, pleomorphism; MIT, mitosis; VAS, vascularity; NEC, necrosis.
c GFAP, glial fibrillary acidic protein; VIM, vimentin; EGFR, 5,7,26,73 epidermal growth factor receptor; VEGF, 16,17,65,73 vascular endothelial growth factor; PDGFRα, 16,26 platelet-derived growth factor receptor α; IGFBP2, 26 insulin-like growth factor–binding protein 2; β III, 73 β III tubulin.
d Heterogenous.
Prognostic Factors
No large retrospective or prospective studies have been performed concerning survival in dogs with astrocytoma. The evaluation of chemotherapy and surgery to treat canine astrocytoma is largely limited to case reports, mostly dating from the early 1990s. Survival time has ranged from 3 to 8 months when chemotherapy (usually, alkylating agents such as carmustine) has been given alone or in combination with radiotherapy or surgical debulking. 18,32,54,66 Whether therapeutic intervention substantially enhances survival in canine astrocytoma beyond supportive measures (eg, anticonvulsive drugs, corticosteroids) is unknown because controlled studies are not available and many reports on treatment have not relied on histopathologic diagnosis. 2 In contrast, treatment protocols and outcome data are well established for people with astrocytic tumors.
The poor outcomes associated with high-grade astrocytic tumors in people have stimulated interest in biomarker-targeted therapies. Inhibitors of receptor tyrosine kinases such as EGFR, PDGFRα, and VEGFR have received significant attention. Whereas trials with single-receptor inhibitors have been somewhat disappointing, drugs capable of multireceptor blockade may offer more promise. 56,67,68,75 Viral and nonviral gene transfer has been successfully performed in the canine brain in vivo and in canine glioma cells in vitro, suggesting that the dog may be an excellent animal model for targeted therapy strategies. 5,57
Perspective
Despite the dismal prognosis associated with high-grade glioma in people and dogs, the molecular biology and genetics underlying astrocytic oncogenesis have advanced substantially. New molecularly targeted therapies—such as monoclonal antibodies reactive with growth factors and their receptors, small molecule signal transduction inhibitors, improvement of cellular immunotherapy, the use of neural stem cells as therapy, and the targeting of tumor cancer stem cells—have been tried with variable degrees of success in people with glioma. 9,21,24,52,58,68 With all these advances, the clinical significance of molecular parameters for diagnostic and prognostic purposes is still limited in veterinary medicine. The ultimate goal is to find ways of uniquely eradicating tumor cells, leaving the normal brain undamaged.
In a recent article, Cardiff et al emphasized that “the veterinary and medical pathology professions are steeped in a rich history of ‘One Medicine,’ but they have paradoxically parted ways, leaving the discipline of pathology poorly positioned to contribute to contemporary science.” 6 This truth is no better reflected than in the discrepancy between the WHO human and veterinary classification of nervous system tumors. The similarity of primary CNS tumors in people and dogs is consistently reported. Despite all the similarities, the WHO document detailing tumors of the CNS in domestic animals, published more than 10 years ago (1999), is not keeping up with the WHO human classification, which has been updated twice since that time. Unfortunately, there is no scientifically validated data for canine CNS tumors that relate a classification or grading system to biological behavior. In conclusion, a scientifically based classification of astrocytic tumors in dogs with diagnostic and clinical prognostic significance needs to be developed to recognize the value of comparative pathology.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
