Abstract
Twelve coho salmon, approximately 8 weeks old, were each observed to have a single neoplasm involving the dorsolateral axial skeletal musculature. The neoplasm was closely associated with the vertebrae in all cases. The neoplasm was composed of islands containing small cells with round and occasional spindeloid morphology. Neoplastic cells had basophilic cytoplasm and vesicular nuclei. These cells exhibited immunopositivity only for vimentin and S-100 protein. Ultrastructurally, neoplastic cells had nuclei with a predominance of euchromatin, cytoplasm containing marked amounts of rough endoplasmic reticulum, scant amounts of smooth endoplasmic reticulum, and scattered mitochondria. Rudimentary cell junctions were occasionally observed between adjacent neoplastic cells. Based on the close association of these neoplasms with the vertebrae as well as the histologic, ultrastructural, and immunohistochemical findings, these neoplasms were considered to all be primitive neuroectodermal neoplasms.
Keywords
Although neoplasms are rather uncommon in fish, some neoplasms have been well documented. Dermal sarcomas in walleye are associated with a retrovirus. 2 Dermal fibromas have been reported in a redband parrotfish. 3 Epithelial neoplasms and dermal sarcomas have been demonstrated in rainbow smelt. 6,7 Carcinoma of the pancreas has been reported in medaka and coho salmon. 4
This is the first report of a primitive neuroectodermal neoplasm in coho salmon, Oncorhynchus kisutch. Twelve coho salmon, approximately 8 weeks old, were submitted live to the Animal Disease Diagnostic Laboratory, Purdue University, in West Lafayette, Indiana. Fish were from a cohort of approximately 150,000 same-aged fish and were being raised at an Indiana state fish hatchery for stocking into Lake Michigan. All fish were spawned from adult broodstock captured from Lake Michigan watersheds. All affected fish had a very similar gross lesion of a single, round to oval, slightly pink, raised, nonlobulated, fluctuant mass that was just beneath the integument. Masses were approximately 1.5 cm in diameter and extended along the right or left dorsolateral aspect at the level of the dorsal fin and caudally to the midceliac region of the fish. The masses extended into the dorsolateral axial skeletal musculature in all fish (Fig. 1).

Whole body; coho salmon. A single raised, oval, fluctuant mass is present on right lateral body wall.
Histologically, neoplasms observed in all the affected fish were similar. The neoplasms were nonencapsulated expansile masses that extended from the superficial dermis into the axial skeletal musculature and were very closely associated with the dorsal or lateral or both aspects of the vertebrae (Fig. 2). Neoplastic cells formed islands that were often confluent into a solid sheet within a scant fibrovascular stroma. The neoplasm was composed of a monomorphic population of cells having either small, round or, less commonly, spindeloid morphology (Fig. 3). Neoplastic cells contained vesicular nuclei with inapparent nucleoli. Round cells had markedly basophilic cytoplasm, and spindeloid cells contained bipolar basophilic cytoplasm. “Pseudorosette” formation of these cells was observed but was a very infrequent finding. Mitotic index was approximately 4 mitoses per high power field (40×).
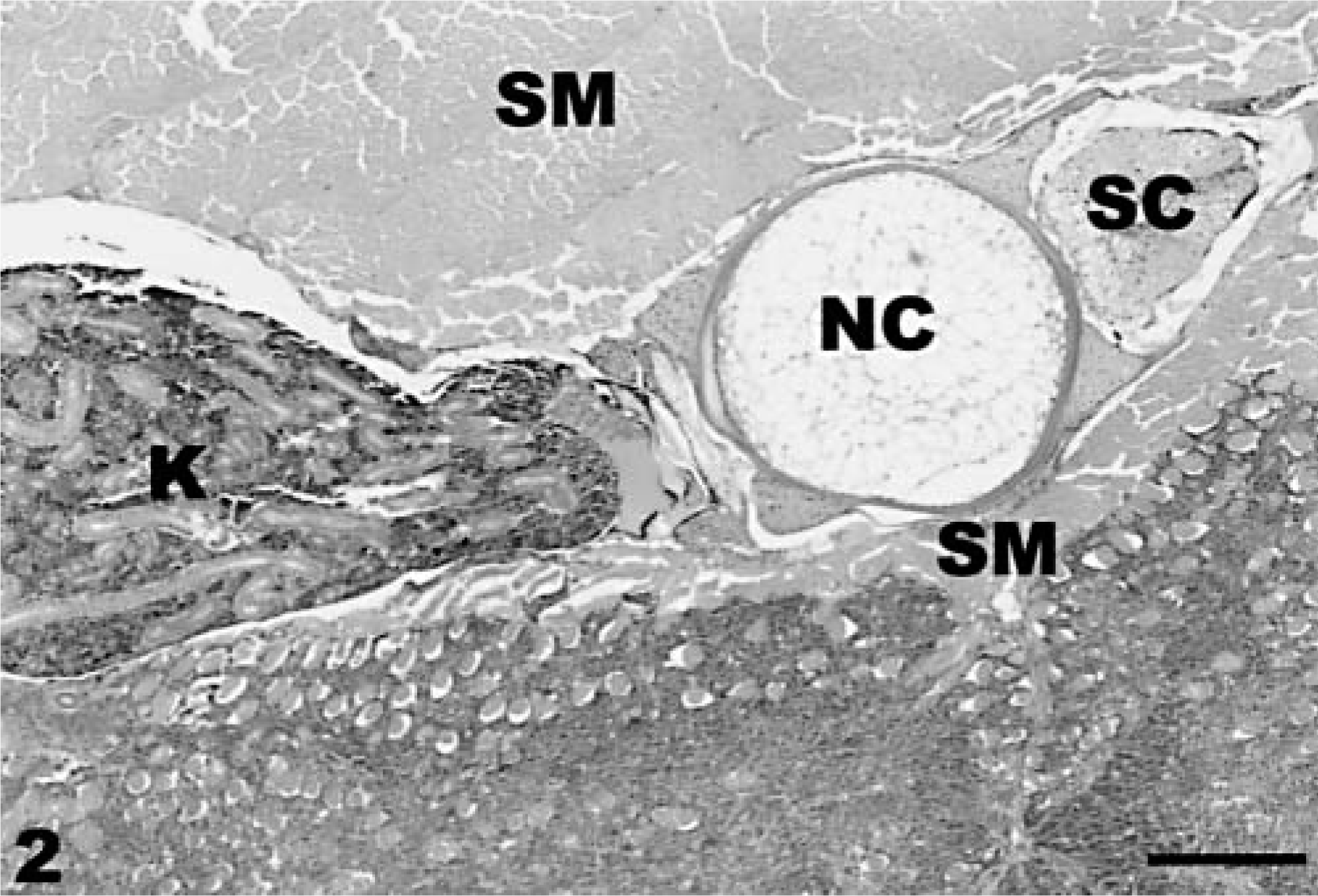
Neuroectodermal neoplasm; coho salmon. Neoplasm is observed in the lower portion of photo adjacent to kidney (K), abdominal skeletal muscle (SM), notochord (NC), and spinal cord (SC). HE stain. Bar = 400 µm.
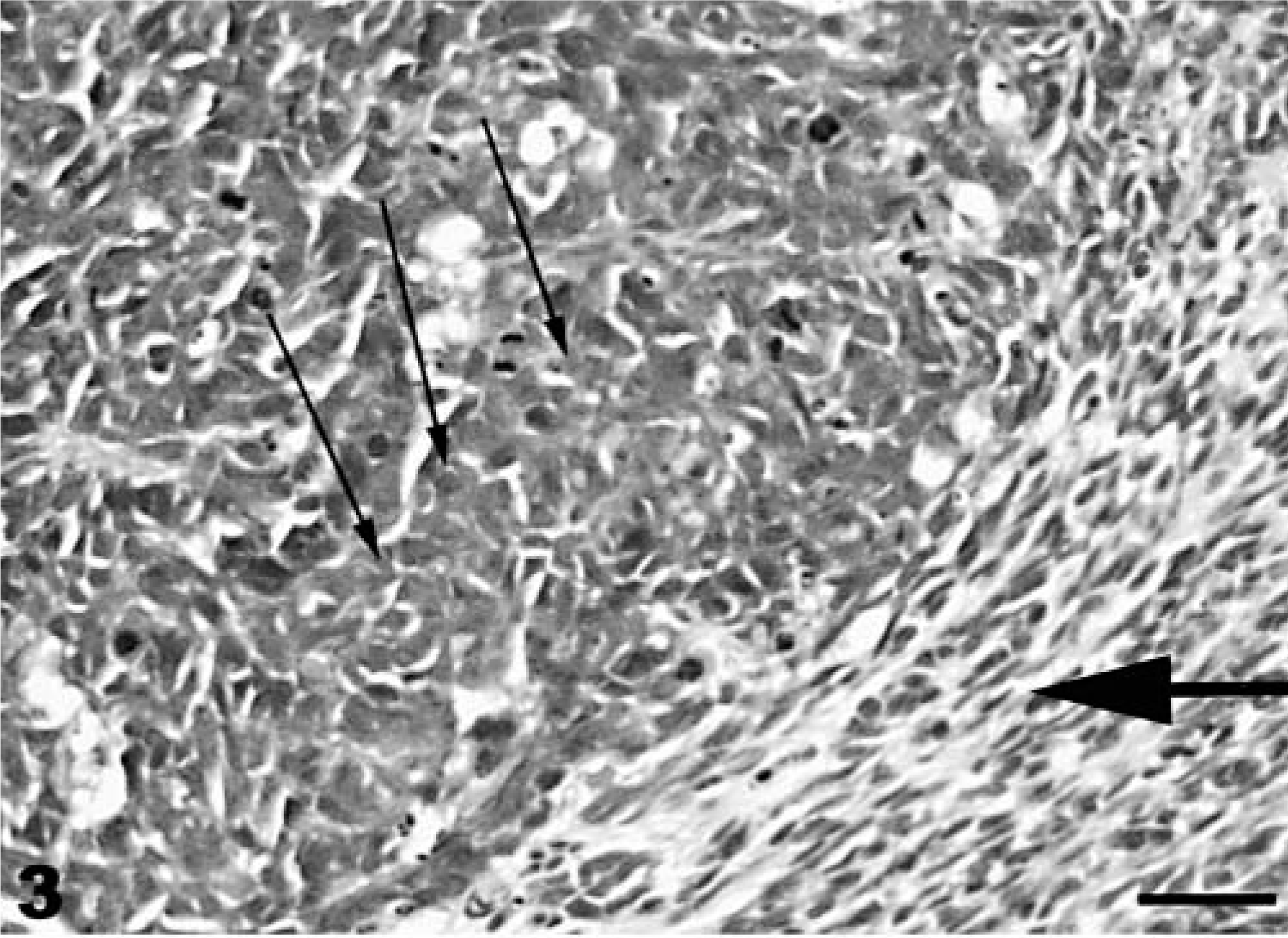
Neuroectodermal neoplasm; coho salmon. The majority of the cells are small, round cells (small arrows), with occasional spindeloid (large arrow) cells observed. HE stain. Bar = 100 µm.
Immunohistochemical staining was performed by placing paraffin-embedded sections of the neoplasm on positively charged slides and staining sections with antibodies to S-100 protein, cytokeratins (AE1/AE3), vimentin, neuron-specific enolase, chromogranin A, and synaptophysin. Staining was performed using a labeled streptavidin–biotin method with diaminobenzidine as the chromogen. 8 For negative controls, the primary antibody was replaced with normal rabbit serum for the polyclonal antibodies and normal mouse serum for the monoclonal antibodies. Age- and species-matched fish tissues were used as positive controls. Immunohistochemical staining revealed moderate positive cytoplasmic staining of the neoplastic cells with vimentin and S-100 protein (Figs. 4, 5). Positive staining with vimentin was punctate to linear within the round cells and linear only in the spindeloid cells. Staining of cells with S-100 protein was more apparent in the spindleloid cells as compared with the round cells. The neoplasm was negative for neuron-specific enolase, cytokeratins (AE1/AE3), chromogranin A, and synaptophysin. Age-and species-matched positive control fish had predictable normal staining of these immunohistochemical reagents.
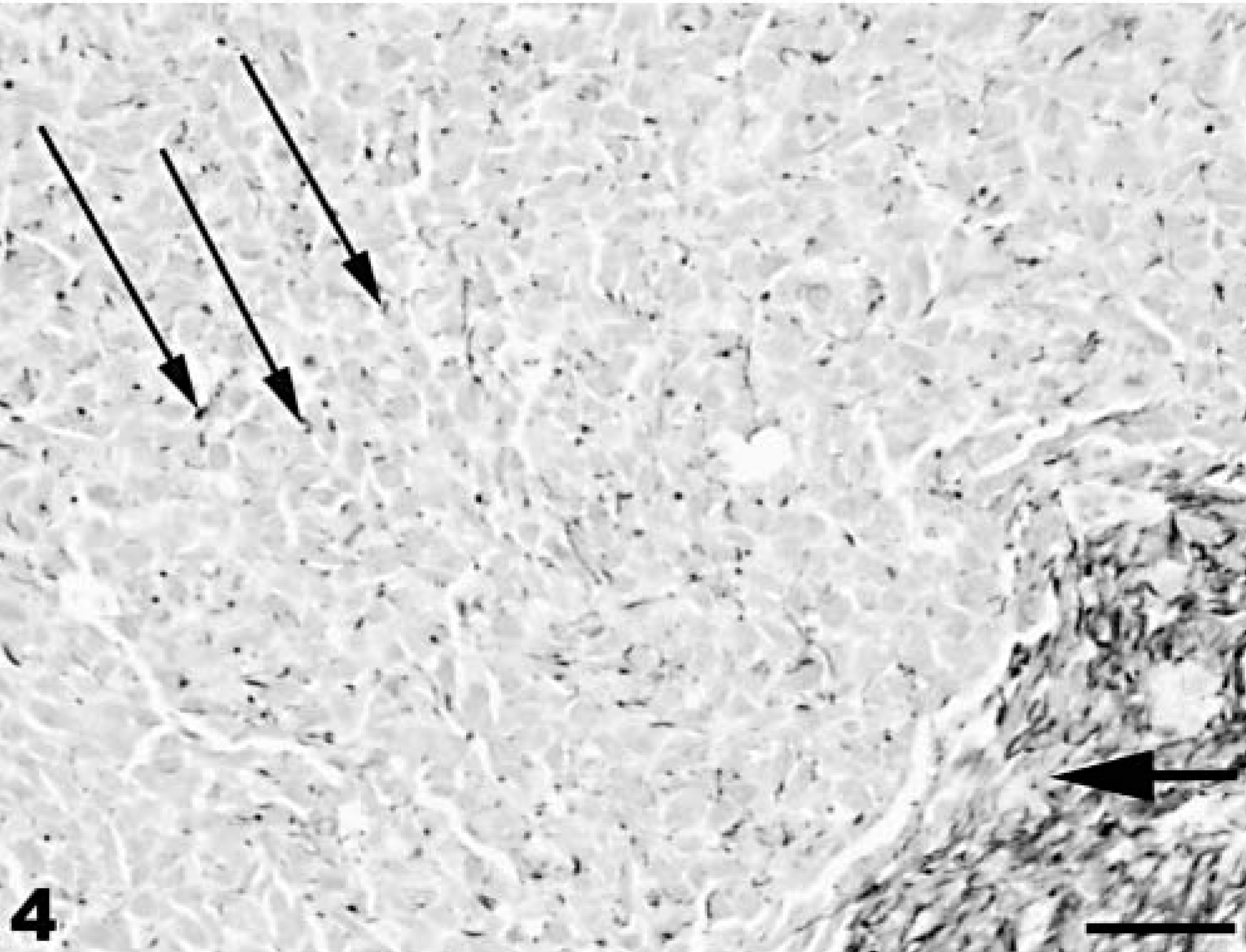
Neuroectodermal neoplasm; coho salmon. Small, round neoplastic cells (small arrows) and spindeloid cells (large arrow) are positive for vimentin. Mayer's hematoxylin counterstain. Bar = 100 µm.
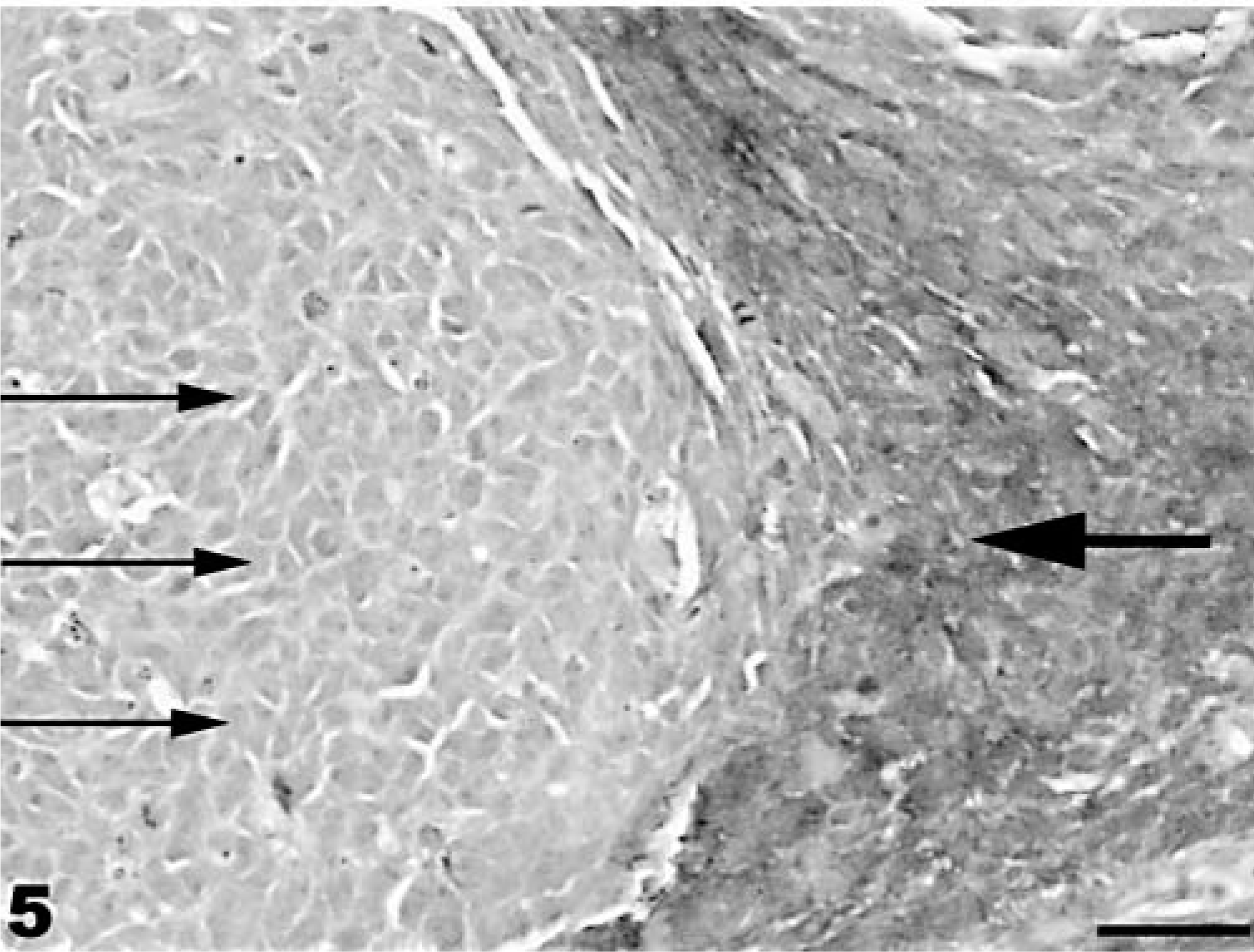
Neuroectodermal neoplasm; coho salmon. Spindeloid cells (large arrow) are positive for vimentin, whereas small, round neoplastic cells (small arrows) show a faint positive reaction for S-100 protein. Mayer's hematoxylin counterstain. Bar = 100 µm.
Electron microscopy was performed by placing formalin-fixed tissues of the neoplasm in 3% glutaraldehyde solution followed by postfixation in 2% aqueous osmium tetroxide, with dehydration in graded ethanols and propylene oxide. Sections were embedded in epoxy resin, sectioned at 80 nm, and stained with uranyl acetate and lead citrate.
Ultrastructurally, neoplastic cells had nuclei with a predominance of euchromatin, cytoplasm containing marked amounts of rough endoplasmic reticulum, scant amounts of smooth endoplasmic reticulum, and scattered mitochondria. Cytoplasmic processes of adjacent cells were interdigitated, and occasional rudimentary cell junctions were observed between adjacent neoplastic cells (Fig. 6).
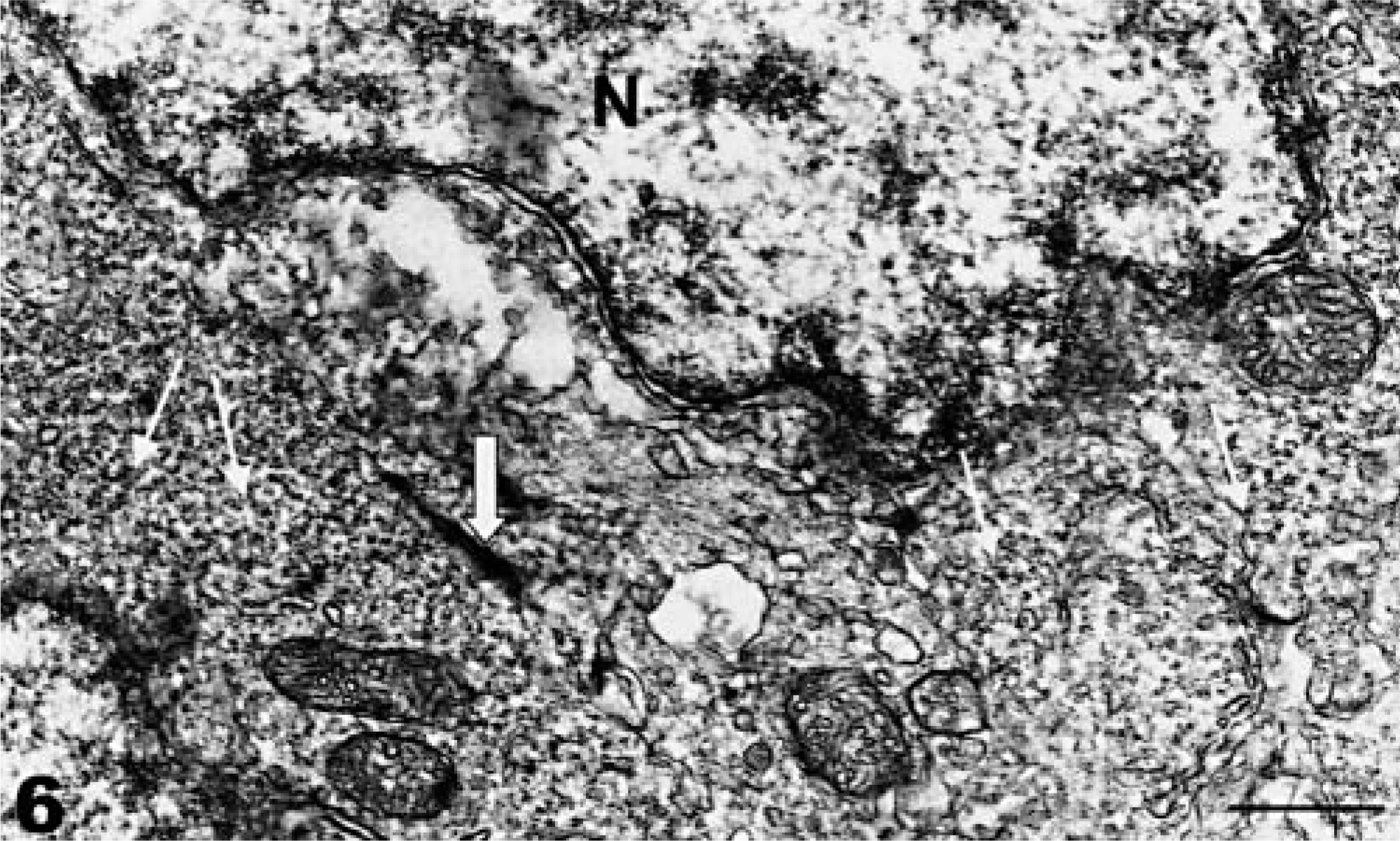
Neuroectodermal neoplasm; coho salmon. Transmission electron micrograph. Features of neoplasm include nucleus (N) with abundant euchromatin, rudimentary cell junction (thick arrow), and smooth endoplasmic reticulum (thin arrows). Uranyl acetate and lead citrate. Bar = 0.25 µm.
On the basis of the close proximity of this neoplasm to the dorsal midline of the vertebral column and the histologic, ultrastructural, and immunohistochemical findings, these neoplasms were considered to be primitive neuroectodermal tumors.
Primitive neuroectodermal neoplasms have been best documented in the bone and soft tissue of children and young adults but can occur at other sites. 9 In humans, these neoplasms have a histologic appearance similar to the neoplasm described here, with the exception of the rosette or pseudorosette formation being observed in some cases. These neoplasms are commonly positive for vimentin and S-100 protein as well as neuron-specific enolase. Ultrastructurally, some of the more differentiated neoplasms in humans contain rare neurosecretory granules. 1,5,10 Although the cause of this neoplasm in these 12 fish could not be determined, it was considered to be a congenital defect of these fish forming during embryological development. The neoplasms most likely arose as a consequence of incomplete formation of the neural tube, whereby a small “embryonic rest” of tissue undergoes a neoplastic transformation later in life. Even though this is the first time these fish have been observed to have visible neoplasms, the prevalence rate of 12 fish in a population of 150,000 is only 0.08%. These fish would probably not have survived in the watersheds of Lake Michigan because of the increased visibility of this mass making them more susceptible to predation as well as possible swimming abnormalities associated with the presence of this neoplasm within the axial skeletal musculature precluding escape from predation.
Footnotes
Acknowledgements
I thank Dr. Jose A. Ramos-Vara of the Veterinary Medical Diagnostic Laboratory, University of Missouri-Columbia, College of Veterinary Medicine, for performing immunohistochemistry. This work was supported, in part, by funding from the Indiana Department of Natural Resources, Fish and Wildlife Division, Grant No. E2-2-A386.
