Abstract
Hard pad disease represents an uncommon manifestation of canine distemper virus (CDV) infection with a still uncertain pathogenesis. To study the pathogenesis of this uncommon, virally induced cutaneous lesion, the footpads of 19 dogs with naturally occurring distemper were investigated for histologic changes and distribution pattern of CDV antigen. All dogs displayed clinical signs of distemper, which had lasted from 10 to 75 days. Overt digital hyperkeratosis was observed in 12 animals (group A), whereas the footpads of the remaining seven dogs appeared normal macroscopically (group B). Orthokeratotic hyperkeratosis (12/12; 100%), irregular acanthosis (11/12; 92%), thickened rete ridges (10/12; 83%), and mild mononuclear perivascular (10/12; 83%) and periadnexal (7/12; 58%) dermatitis were the most common findings in dogs with hard pad disease. Surprisingly, orthokeratotic hyperkeratosis (5/7; 71%), irregular acanthosis (5/7; 71%), and thickened rete ridges (4/7; 57%) were also seen in the dogs without clinical evidence of digital hyperkeratosis. CDV-specific inclusion bodies and ballooning degeneration were not observed in the footpad epidermis of the 19 dogs. Immunohistochemistry revealed that CDV antigen was most frequently found in the stratum spinosum and granulosum and in the epithelial cells of the eccrine sweat glands and only rarely in the basal layer. Fibroblasts, pericytes, endothelial cells, and hair follicles were also positive in some animals. Despite the obvious difference regarding the macroscopic picture, the microscopic changes were less prominent between the animal groups. The selective infection of keratinocytes in the stratum spinosum might be the key event for the development of hard pad disease in the dog.
Canine distemper virus (CDV) is a single-stranded RNA virus that belongs to the genus morbillivirus of the paramyxovirus family. 6 CDV causes a highly contagious disease in the domestic dog worldwide. 6,7 Recently, cases of canine distemper (CD) have been reported even in fully vaccinated dogs in various countries, 5,10 including Greece. 11
CDV infection in dogs may be manifested by a combination of respiratory, ocular, gastrointestinal, neurologic, and cutaneous signs or lesions (or both) that appear simultaneously or sequentially. 6 Clear epitheliotropism associated with cutaneous lesions have been described in a variety of canine viral infections, such as mumps virus, parapoxvirus, orthopoxviruses, and papillomavirus infections 14,17 ; according to their cellular tropism, the associated lesions may vary from papillomatous to vesicular and ulcerative. CD-associated skin lesions include impetigo and nasal-digital hy-perkeratosis. 10,14 The latter represents the hallmark of hard pad disease, which has been considered a rather uncommon form of CD. 8 The grossly affected pads are thickened and fissured or are unusually hard and unyielding when pressed from the side. 8 Digital hyper-keratosis is often accompanied or soon followed by the appearance of various neurologic signs. 6 Histologic skin lesions in hard pad disease are characterized by severe orthokeratotic hyperkeratosis and the presence of acidophilic inclusion bodies in the footpad epithelium. 9,14,20 Although the pathogenesis of digital hyper-keratosis is still uncertain, numerous mechanisms have been proposed to explain how viruses may dysregulate the cell cycle and affect keratinocyte proliferation. 13 In humans, the macular and papular rash associated with measles virus (closely related to CDV) is probably mediated by the deposition of virus-antibody complexes or is a direct effect of the virus on epithelial and vascular endothelial cells. 3
In an attempt to elucidate the pathogenesis of hard pad disease in the dog, the scope of the present study has been the investigation of the tropism of CDV in the footpads and the associated histologic lesions in dogs with and without digital hyperkeratosis.
Materials and Methods
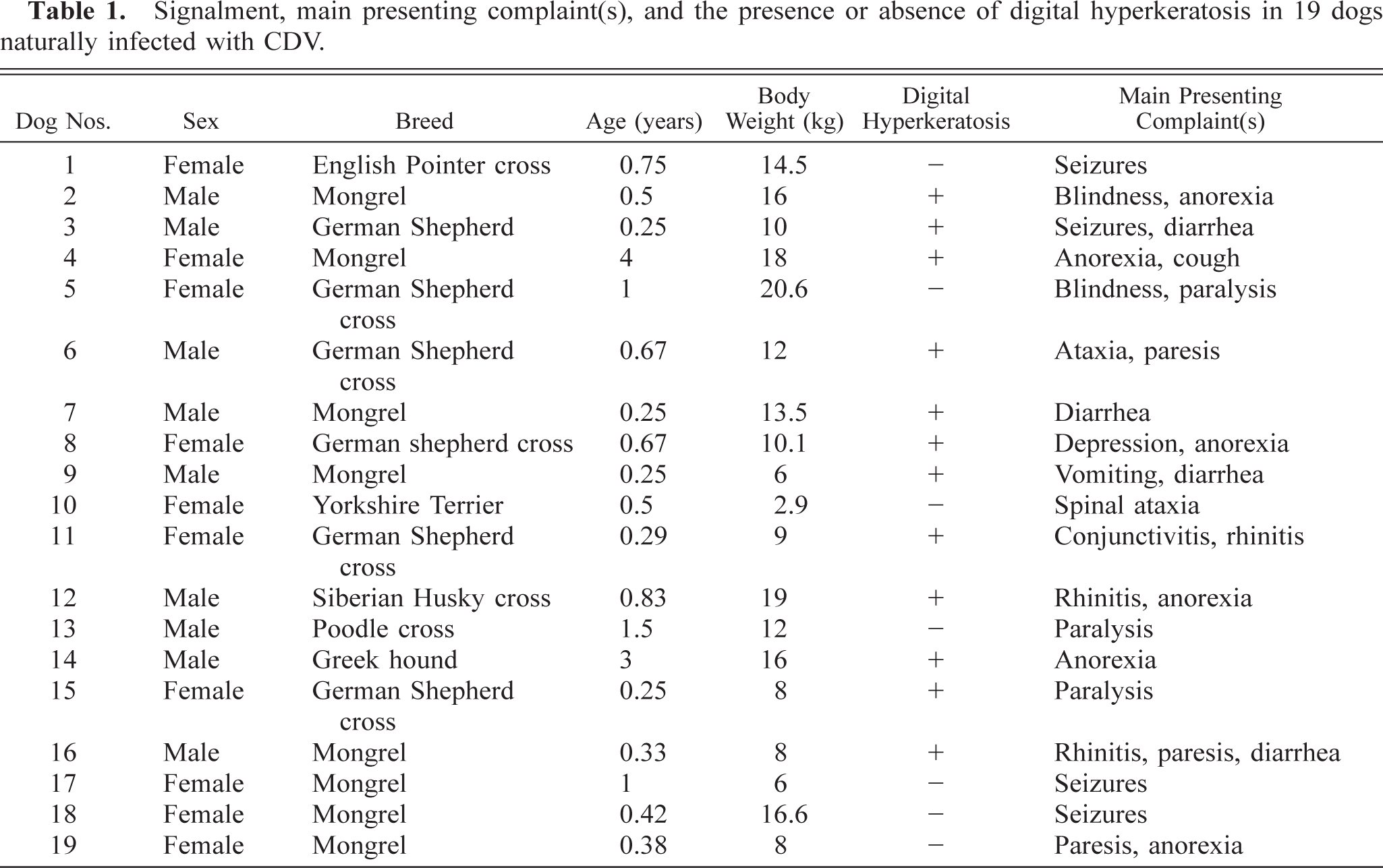
A total of 19 dogs naturally infected with CDV were used in the study (Table 1). These dogs were selected from a total of 34 dogs with CDV-compatible clinical signs because of the presence of viral antigen expression in their footpad skin. All these animals had been admitted to the Clinic of Companion Animal Medicine, School of Veterinary Medicine, Aristotles University of Thessaloniki, for a 4-year period (1996–1999). Nine dogs (47.4%—dog Nos. 2, 3, 6, 7, 9, 12–14, 16) were males and 10 (52.6%) females (dog Nos. 1, 4, 5, 8, 10, 11, 15, 17–19). Three (15.8%) were purebreds (dog Nos. 3, 10, 14), 8 (42.1%) crossbreds (dog Nos. 1, 5, 6, 8, 11–13, 15), and 8 (42.1%) mongrels (dog Nos. 2, 4, 7, 9, 16–19). The age of these animals ranged from 3 months to 4 years (mean: 10.6 months) and their body weight from 2.9 to 20.6 kg (mean: 11.9 kg). The majority of the dogs had not been vaccinated against CDV (14/19; 73.7%—dog Nos. 1, 3–6, 8–10, 13–17, 19) and enjoyed an outdoor life-style (13/19; 68.4%—dog Nos. 1–6, 11, 12, 14, 15, 17–19). The allocation of the 19 animals into two groups was based on the presence (group A—dog Nos. 2–4, 6–9, 11, 12, 14–16) or absence (group B—dog Nos. 1, 5, 10, 13, 17–19) of grossly visible digital hyperkeratosis.
Signalment, main presenting complaint(s), and the presence or absence of digital hyperkeratosis in 19 dogs naturally infected with CDV.
Under general anesthesia (3/19—dog Nos. 4, 11, 14) or immediately after euthanasia (14/19—dog Nos. 1, 2, 5–8, 10, 12, 13, 15–19) or natural death (2/19—dog Nos. 3, 9), 8-mm punch biopsy samples were obtained from the center and the periphery of metacarpal or metatarsal footpads and submitted for histopathology and immunohistochemistry.
Footpad biopsy samples were fixed in 10% nonbuffered formalin, embedded in paraffin, cut into 2- to 4-µm-thick sections and stained with hematoxylin and eosin (HE). The most important histopathologic findings (orthokeratotic hyperkeratosis, irregular acanthosis, thickened rete ridges, perivascular and periadnexal mononuclear dermatitis) were, subjectively and independently, scored by two of the authors (W. Baumgärtner and D. Tontis), who were blinded regarding the presence of hard pad disease, as 0 (absent), 1 (mild), 2 (moderate), or 3 (severe). The differentiation of epidermal acanthosis and hyperkeratosis of footpad skin from normalcy was mainly based on the histologic comparisons made between the CDV-affected animals and 10 age-matched dogs the footpads of which looked quite normal.
CDV protein was demonstrated by a standard avidin–biotin complex (ABC) method. 2 Briefly, paraffin-embedded tissue was dewaxed in xylene and hydrated through graded alcohols. Endogenous peroxidase activity was suppressed by 0.5% hydrogen peroxide in methanol. Sections were incubated with a CDV nucleoprotein-specific monoclonal antibody (NP-2; mab, clone 3991, kindly provided by Dr. C. Örvell, Central Microbiological Laboratory, Stockholm Country-Council, Sweden), 12 as primary antibody, overnight at 4 C, followed by biotinylated horse anti-mouse serum as link antibody and the ABC (Vector Laboratories; Bulingame, CA, USA) for 30 minutes at room temperature. Positive antigen-antibody reactions were observed by incubation with 3,3-diaminobenzidine-tetrahydrochloride (DAB-H2O2) in 0.1 M imidazole, pH 7.1, for 10 minutes, followed by slight counterstaining with hematoxylin. Sections were mounted with Corbit-Balsam (Hecht, Kiel-Haesse, Germany). Positive and negative controls included in vitro CDV-infected Vand, noninfected Vero cells, as well as tissue from CDV-positive (experimentally or naturally infected) and -negative dogs. The CDV infection was confirmed by in situ hybridization or reverse transcriptase–polymerase chain reaction. Additional controls included omission of primary antibody, link antibody, or ABC. The CDV antigen expression at the various layers of the epidermis, fibroblasts, and pericytes/endothelial cells of the superficial dermis and eccrine sweat glands was scored subjectively (W. Baumgärtner) as 0, 1, 2, 3, or 4, corresponding to absent, minimal, few, moderate, or numerous immunopositive cells, respectively.
The presence of the various histologic lesions in footpads and the expression of CDV antigen in epidermal keratinocytes and dermal fibroblasts, pericytes/endothelial cells, and eccrine sweat glands were compared between group A (dog Nos. 2–4, 6–9, 11, 12, 14–16) and group B (dog Nos. 1, 5, 10, 13, 17–19) dogs using Fisher's exact test. Furthermore, the t-test was used to compare the severity of histologic lesions and the intensity of CDV antigen expression between these groups.
Results
History and physical examination revealed that all 19 dogs exhibited neurologic, ocular, respiratory, or gastrointenstinal signs (or all), with a duration of 10–75 days (median: 10 days) before presentation (Table 2). The most common clinical signs included poor body condition (14/19), conjunctivitis (14/19), fever (13/19), myoclonus (12/19), depression-lethargy (12/19), rhinitis (11/19), and spinal ataxia (10/19). Moderate to severe digital hyperkeratosis was seen only in group A dogs (12/19—dog Nos. 2–4, 6–9, 11, 12, 14–16). The metacarpal or metatarsal pads of all these animals displayed a firm consistency at the center, along with excess keratin tissue, fissures, and erosions, that appeared more pronounced at the edges of the footpad (Fig. 1). Superficial pustular dermatitis or impetigo on the ventral abdomen (2/12 group A dogs; 16.7%—dog Nos. 11, 16) and nasal hyperkeratosis (9/12 group A dogs; 75%—dog Nos. 2, 4, 6–9, 12, 14, 16 and 2/7 group B dogs; 28.6%—dog Nos. 18, 19) were additional cutaneous findings.
Clinical signs in 19 dogs with natural CDV infection.
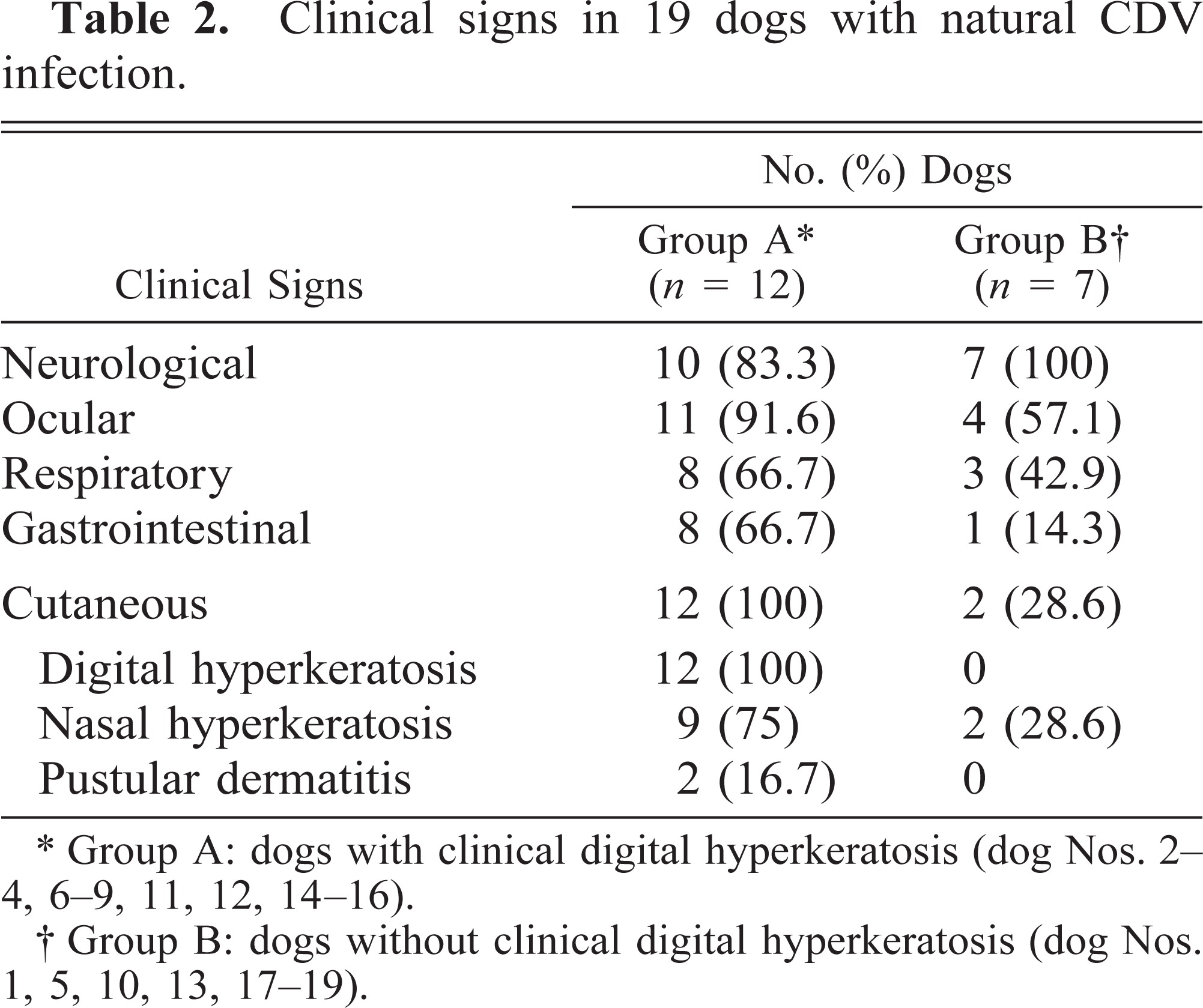
∗ Group A: dogs with clinical digital hyperkeratosis (dog Nos. 2–4, 6–9, 11, 12, 14–16).
† Group B: dogs without clinical digital hyperkeratosis (dog Nos. 1, 5, 10, 13, 17–19).

Metatarsal and digital pad hyperkeratosis in the hind limbs; group A dog with CDV infection (hard pad disease); dog No. 4. Excessive keratin, especially at the periphery of the pads, with fissuring. The central area of the pads is harder than normal.
No significant differences were found between group A and group B dogs regarding the presence and the severity of the histologic lesions (Table 3), although the lesions generally seemed to be more severe in group A dogs. Orthokeratotic epidermal hyperkeratosis was present in all group A (dog Nos. 2–4, 6–9, 11, 12, 14–16) and in five of 7 group B dogs (dog Nos. 1, 5, 13, 18, 19) (Table 3, Fig. 2). Irregular ac-anthosis with thickened and occasionally fused rete ridges (Fig. 3) was more common in group A animals. The number of dogs with mild mononuclear perivascular (Fig. 4) and periadnexal cellular infiltration was similar in both groups. The inflammatory infiltrate was mainly composed of histiocytes, with only a few lymphocytes and plasma cells scattered in between. Pigmentary incontinence was seen in five group A (dog Nos. 2, 4, 6, 7, 9) and two group B dogs (dog Nos. 5, 10) dogs. Changes in eccrine sweat glands and their ducts were either absent or consisted of single-cell necrosis. There was no evidence of CDV-specific cytoplasmic inclusion bodies or vacuolar degeneration in the keratinocytes of the stratum basale and the stratum spinosum.
Histological findings in the footpads of 19 dogs with naturally occurring CDV infection. Significant differences were not detected between groups A and B.
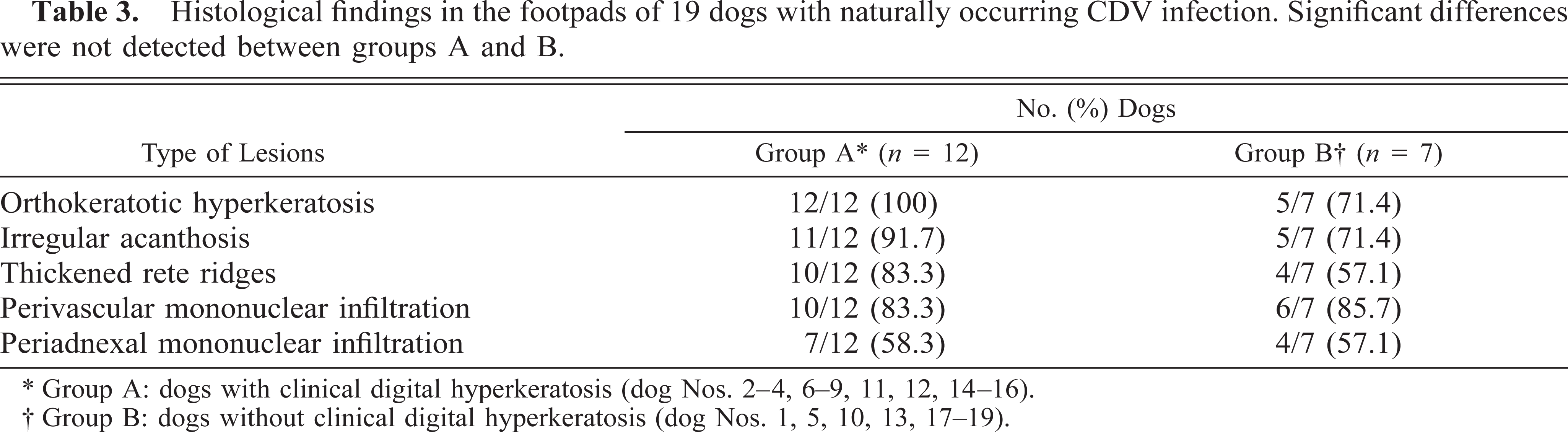
∗ Group A: dogs with clinical digital hyperkeratosis (dog Nos. 2–4, 6–9, 11, 12, 14–16).
† Group B: dogs without clinical digital hyperkeratosis (dog Nos. 1, 5, 10, 13, 17–19).
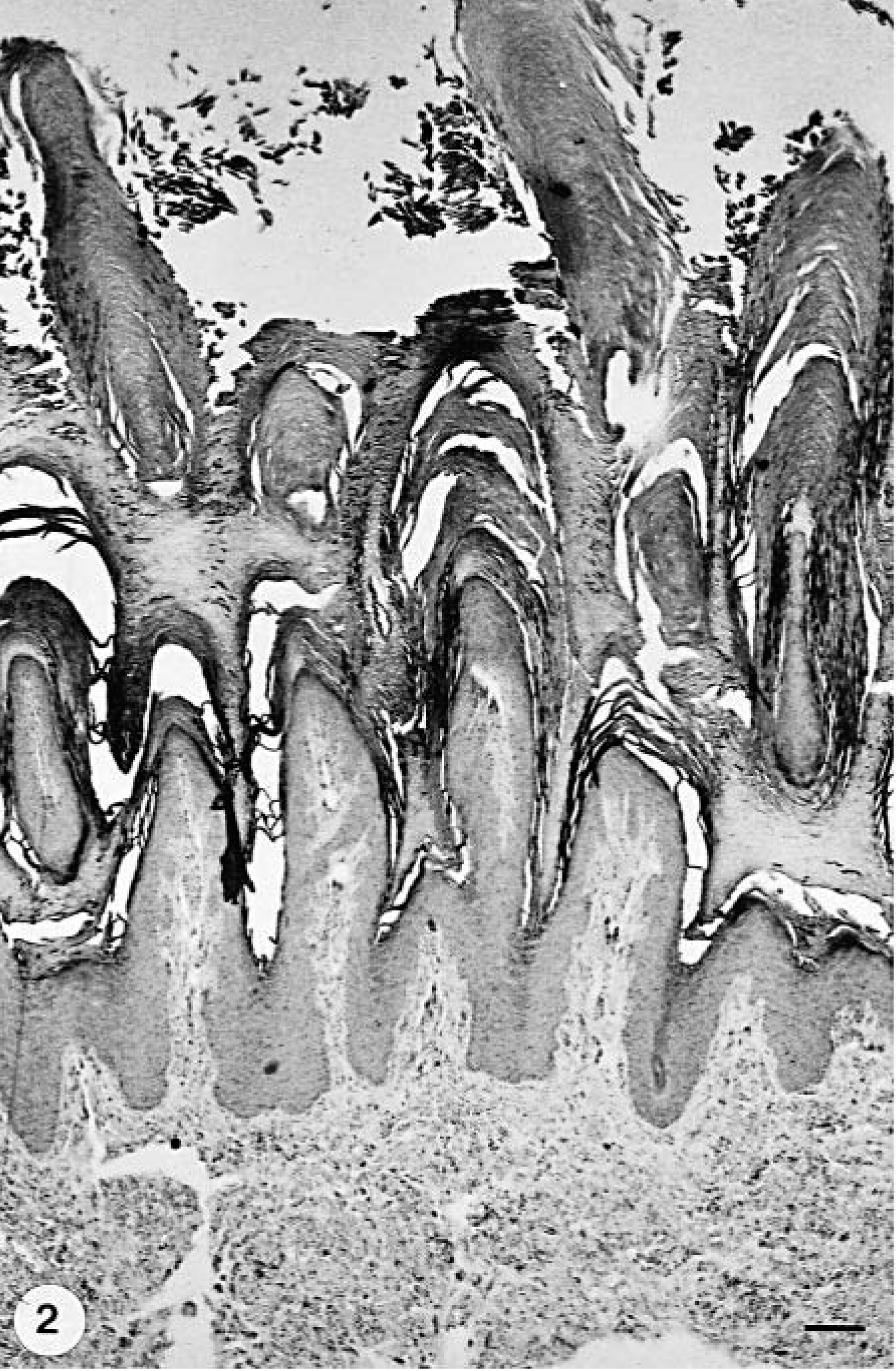
Footpad epidermis; group B dog with CDV infection but without digital hyperkeratosis (hard pad disease); dog No. 1. Prominent orthokeratotic hyperkeratosis and normal epidermis. HE stain. Bar = 138.8 µm.
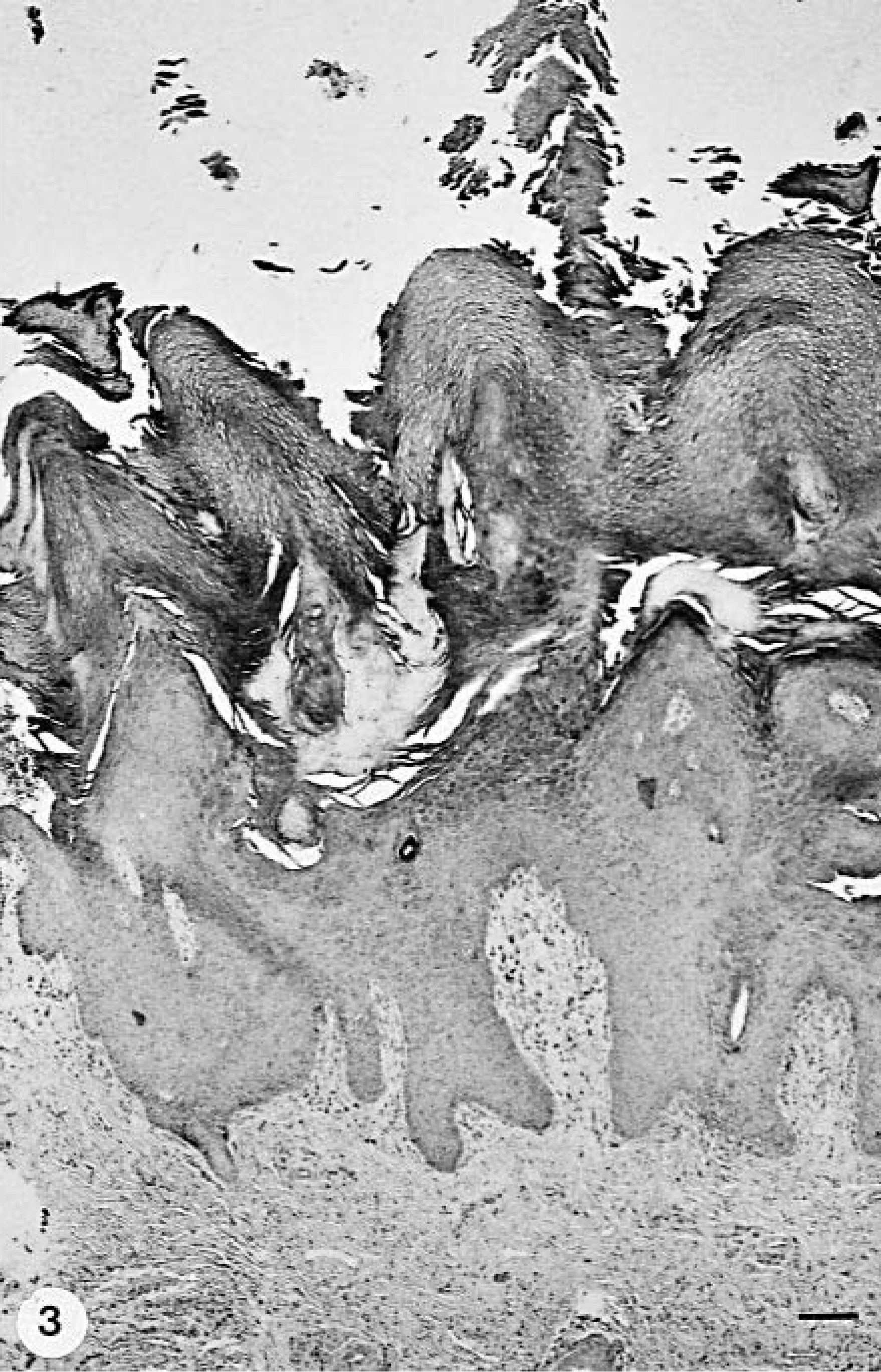
Footpad epidermis; group A dog with CDV infection and digital hyperkeratosis (hard pad disease); dog No. 7. Epidermal hyperplasia with thickened and fused rete ridges and compact orthokeratotic hyperkeratosis. HE stain. Bar = 128 µm.
Footpad skin; group A dog with CDV infection and digital hyperkeratosis (hard pad disease); dog No. 16. Mild perivascular mononuclear infiltrates in the superficial dermis. HE stain. Bar = 29.4 µm.
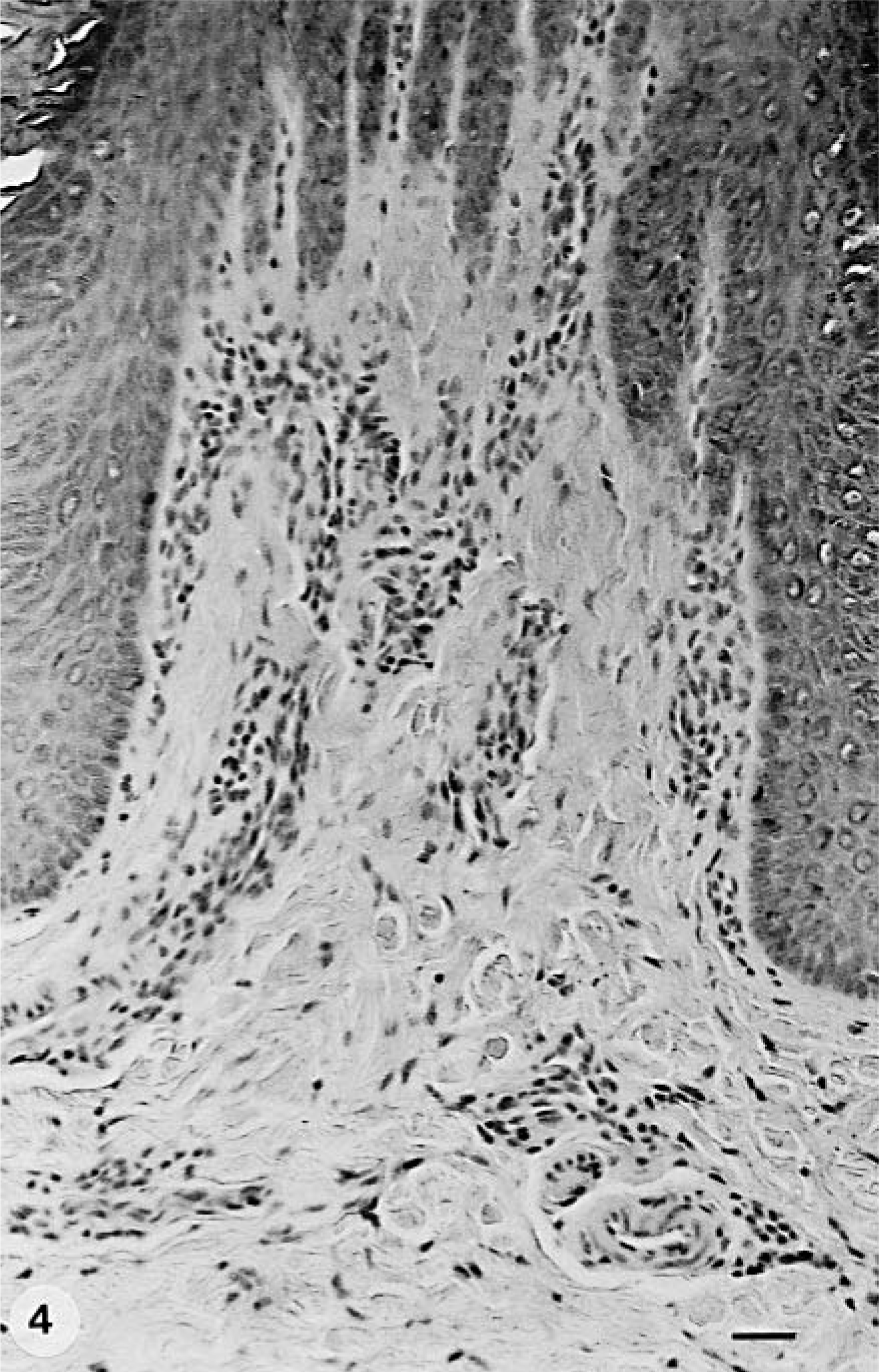
CDV antigen expression and distribution were similar in both groups. Cytoplasmic and intranuclear CDV antigen expression was mainly found in the stratum spinosum and granulosum (Fig. 5), as well as in fibroblasts, pericytes, and endothelial cells of the superficial dermis and in epithelial cells of the eccrine sweat glands and, to a lesser extent, in their ducts (Table 4, Fig. 6). In five animals (three from group A—dog Nos. 6, 8, 11 and two from group B—dog Nos. 13, 19), viral antigen expression was also seen in the hair follicles of the neighboring pedal skin. The number of CDV antigen–positive animals did not differ significantly between the groups. However, in group A dogs CDV antigen was found more often in the stratum spinosum (83%—dog Nos. 2–4, 7–9, 11, 12, 14, 15) compared with group B dogs (57%—dog Nos. 1, 5, 18, 19); quite the opposite was true regarding its presence in the dermis (Table 4). Surprisingly, in both groups CDV immunoreactivity was seen only rarely in the stratum basale and never in the stratum lucidum and stratum corneum. Two dogs in each group lacked viral antigen expression in the epidermis (dog Nos. 6, 10, 13, 16). The immunoreactivity in epidermis, dermis, and adnexae (Fig. 7) appeared to be of higher intensity in group A. However, this observation could not be substantiated statistically.
CDV antigen detection in the footpads of 19 dogs with naturally occurring CDV infection. Significant differences were not detected between groups A and B.

∗ Group A: dogs with clinical digital hyperkeratosis (dog Nos. 2–4, 6–9, 11, 12, 14–16).
† Group B: dogs without clinical digital hyperkeratosis (dog Nos. 1, 5, 10, 13, 17–19).
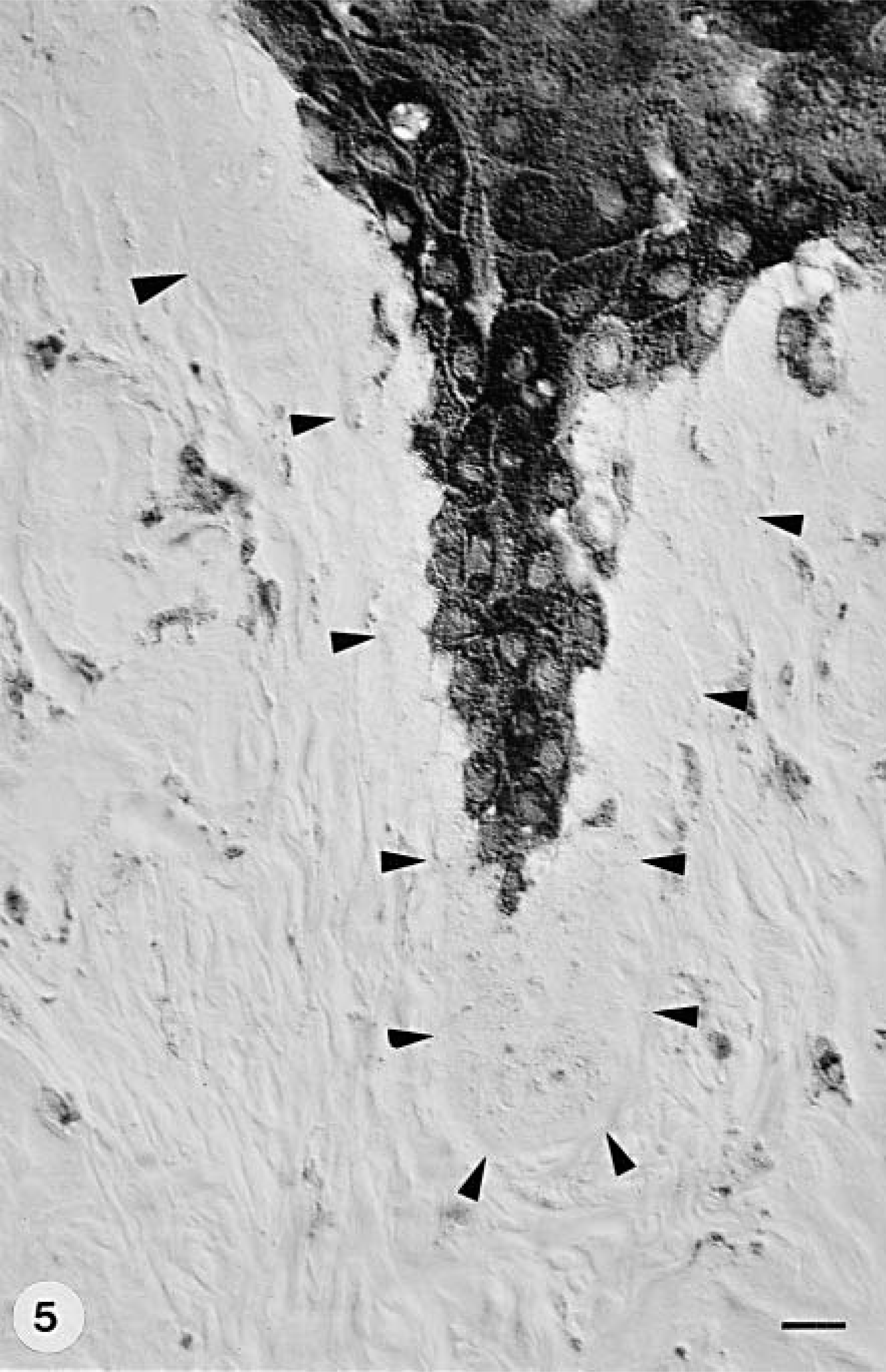
Footpad skin; group A dog with CDV infection and digital hyperkeratosis (hard pad disease); dog No. 9. Immunostaining shows CDV expression in the epidermal stratum granulosum and spinosum and in some dermal fibroblasts. The arrowheads indicate the dermoepidermal junction. Anti–NP-2 antibody, ABC method, mild hematoxylin counterstain. Nomarski illumination. Bar = 11.2 µm.
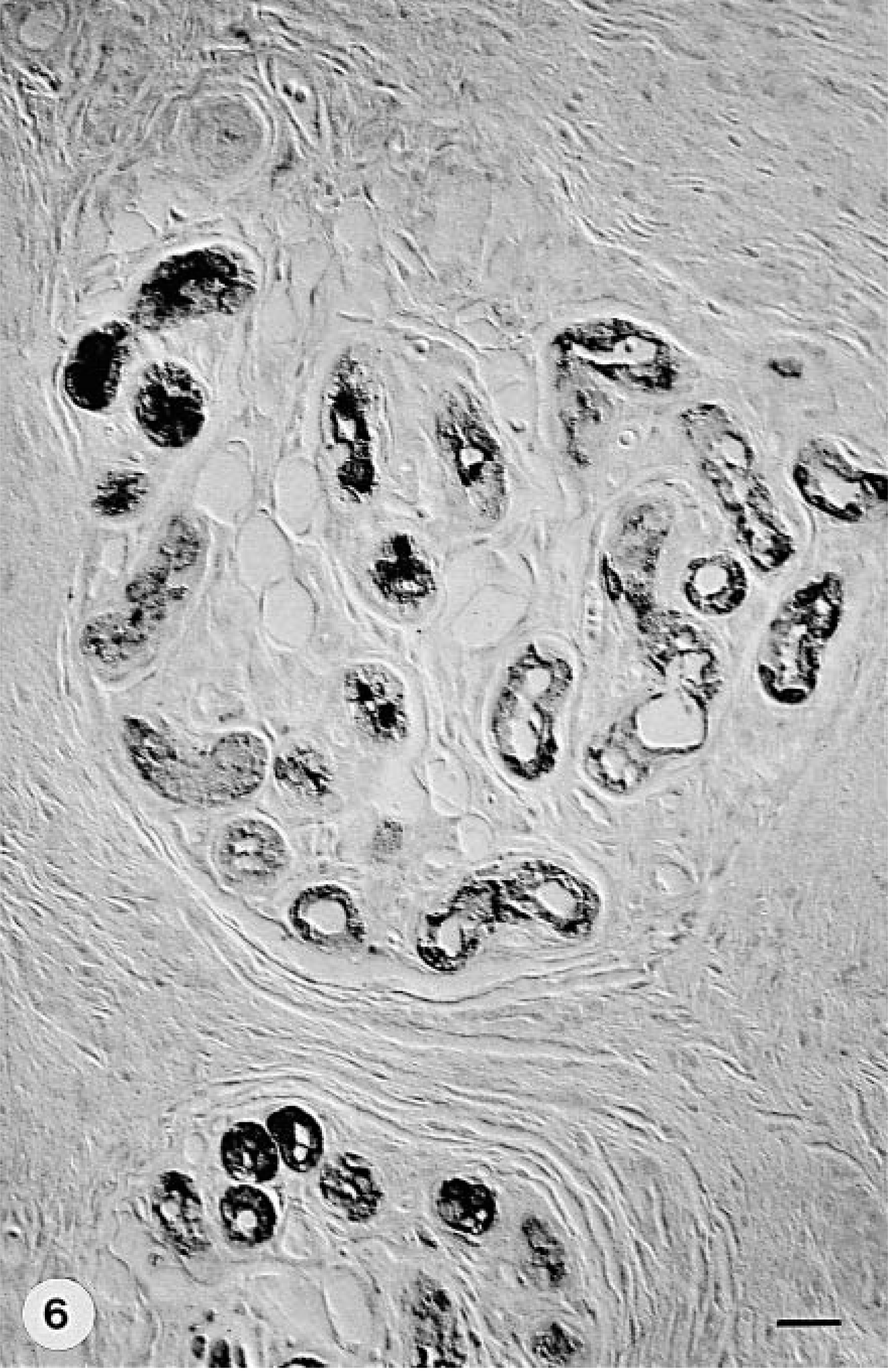
Footpad skin; group A dog with CDV infection and digital hyperkeratosis (hard pad disease); dog No. 3. Immunostaining shows moderate CDV expression in the epithelial cells of eccrine sweat glands and their ducts. Anti–NP-2 antibody, ABC method, mild hematoxylin counterstain. Nomarski illumination. Bar = 21.7 µm.
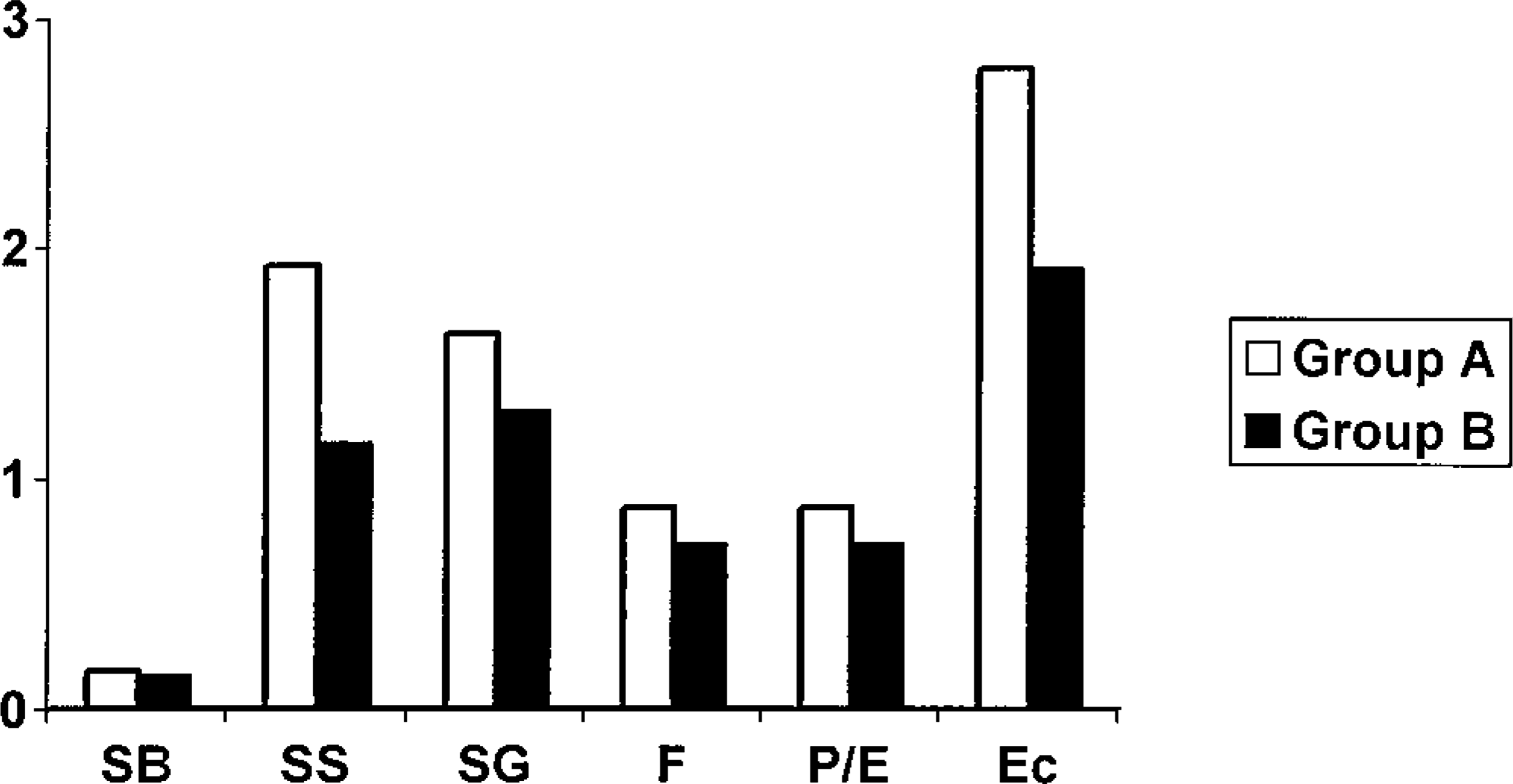
Degree of intensity of CDV antigen expression in the footpads of 19 dogs with naturally occurring CDV infection. SB = stratum basale; SS = stratum spinosum; SG = stratum granulosum; F = fibroblasts; P/E = pericytes/endothelial cells; Ec = eccrine glands. 0 = absent, 1 = minimal, 2 = few, 3 = moderate, and 4 = numerous immuno-positive cells. No significant differences were found between the groups.
Discussion
In the present study the histopathologic changes in the footpads of CDV-infected dogs, with and without grossly visible digital hyperkeratosis (hard pad disease), were correlated with the virus antigen expression. Surprisingly, despite the obvious clinical difference of digital hyperkeratosis between the two groups, their associated histopathologic and immunohistochemical findings were quite similar. As expected, orthokeratotic hyperkeratosis and acanthosis were the most prominent histopathologic findings. Immunohistochemistry, on the other hand, disclosed that the stratum spinosum and granulosum keratinocytes were the main cell targets of CDV.
Hard pad disease is usually associated with the appearance of neurologic signs, as was the case in 10/12 (dog Nos. 2–4, 6–8, 12, 14–16) group A dogs and in all group B dogs (Table 2). In group A dogs the contact surfaces (center) of digital, metacarpal, or metatarsal pads appeared less affected, although they felt harder, probably because friction during walking wore down the keratin. 14 The popular belief is that clattering sound or discomfort while walking on hard floors is common in dogs affected with hard pad disease, but it was actually noticed only in a few animals.
Clinical diagnosis of CDV infection was confirmed by immunohistochemistry in all 19 cases. This technique is considered valuable in the antemortem diagnosis of CD. 7,10 The design of the present study precludes any comment on viral persistence in footpad tissues; however, the history of these dogs indicates that foot pad infection may have occurred at various time intervals from the onset of the clinical signs. In dogs with chronic infection, it is still questionable whether CDV can persist in footpads throughout the evolution of the disease or simply represents a late event. Other investigators believe that CDV can persist for at least 60 days after infection in the footpad epithelium and the haired skin as well. 6
CDV antigen showed an intracytoplasmic and intra-nuclear localization in both epidermal and dermal cells. 9 Interestingly, the expression of viral antigen was mainly in the stratum spinosum and granulosum and only rarely in the stratum basale. The fact that papil-lomaviruses also preferentially infect the stratum spinosum and granulosum keratinocytes 15,16,18 suggests that epitheliotropic viruses may find these epidermal layers more suitable for replication. Another option could be the innate resistance of the keratinocytes of the stratum basale and stratum corneum to viral infections. This selective localization of CDV may have an impact on keratinocyte differentiation that finally leads to excessive orthokeratotic hyperkeratosis and eventually to the establishment of hard pad disease. Viruses, such as polyomaviruses, adenoviruses, and papil-lomaviruses, can affect the cell cycle through molecular mechanisms that override restriction and DNA damage checkpoints. 13 Unfortunately, no data exist regarding the possible effects of CDV or other morbil-liviruses on keratinocyte cycling.
Two group A dogs (dog Nos. 6, 16) and one group B dog with orthokeratotic hyperkeratosis (dog No. 13) did not express CDV antigen in their epidermis. It remains unclear whether the virus had already been eliminated at the time when the biopsy samples were obtained (dog Nos. 6, 16) or epidermal infection had not yet been established (dog No. 13). Immunohistochemistry revealed that viral antigen was more often found in the dermis of dogs without hard pad disease than in those with it. This observation supports the possibility of hematogenous spread of the virus to the footpads. In the dermis the presence of CDV antigen in blood vessel pericytes and endothelial cells may have been the result of viremia. The involvement of fibroblasts merits further investigation because these cells may influence the proliferation and differentiation of keratinocytes. 19 The same could also apply to eccrine sweat glands. Interestingly, CDV antigen was detected in all 14 dogs that contained eccrine sweat glands (dog Nos. 1–4, 6, 8, 9–11, 13, 15–17, 19). The ensuing quantitative and qualitative changes in their secretion would further modulate keratinocyte kinetics by altering the hydration status of the epidermis. The hair follicles, present only in a few peripheral biopsy samples (dog Nos. 6, 8, 11, 13, 19), always expressed viral antigen, as has been described previously. 7 Cytoplasmic acidophilic inclusion bodies are reportedly a common occurrence in keratinocytes, and sebaceous and sweat glands of the haired skin of CDV-infected dogs. 9,14 CDV inclusion bodies, occurring in various tissues in 15–73% of CD cases, 1 are not only usually detected late in the course of the disease but are also relatively transient. 6,7 This may explain their total absence in the keratinocytes of the dogs in the present study. Also, they are hardly specific for CDV because other intracellular structures may look similar. 7 The highly diagnostic intranuclear inclusion bodies, on the other hand, are seen only rarely, at least in the skin. 14 Ballooning degeneration of keratinocytes, a rather specific feature of pox viral infections, 14 was also absent in all 19 animals, although it has been reported in CD footpads, especially in the stratum spinosum. 20 The lack of CDV-specific changes and the abundant expression of CDV antigen underlines the value of immunocytochemistry as a sensitive and specific tool. 4,6,10 Group A and group B dogs were not significantly different with regard to the presence of CDV antigen–positive cells, the intensity of their immunoreactivity, and the type and severity of histologic lesions. This suggests a common underlying pathologic mechanism, irrespective of the presence or absence of overt digital hyperkeratosis. It remains unclear whether subtle changes, which can be easily missed by light microscopy, would be the differentiating point between the clinical and the subclinical disease.
In conclusion, the hyperkeratosis on the footpads of dogs with hard pad disease is probably the result of virus localization in the stratum spinosum and granulosum. In contrast to what was thought before, the presence of CDV inclusion bodies and the ballooning degeneration of keratinocytes is quite uncommon, at least in the footpad epidermis. In dogs with the nervous form of CD and grossly normal footpads, the histopathologic and immunohistochemical features of footpad epithelium are almost identical to those of hard pad disease.
