Abstract
We evaluated thyroid gland lesions in beluga whales (Delphinapterus leucas) from the St. Lawrence Estuary (n = 16) and Hudson Bay (n = 14). Follicular cysts and nodules of adenomatous hyperplasia of the thyroid gland were found in eight and nine adults from the St. Lawrence Estuary (n = 10), respectively, and in four and six adults from Hudson Bay (n = 14), respectively. The total volume of the lesions of thyroid adenomatous hyperplasia was positively correlated with age in both populations. Comparison between populations could not be performed because of differences in age structures of sample groups. Beluga whales from both populations have unique thyroid lesions among marine mammals.
Major concerns have been raised about endocrine-disrupting chemicals in the environment and their effects on human and wildlife health. 12 Suspected effects include decreased fertility and reproductive abnormalities and abnormal thyroid function in birds 44 and fish. 7,33 Similar abnormalities, associated with exposure to environmental contaminants, have also been reported in a limited numbers of marine mammals. 5,8,9,43
The St. Lawrence waterway is a highly contaminated ecosystem inhabited by an isolated and endangered population of beluga whales of approximately 650 individuals. In 1983, a research program based on the postmortem examination of stranded beluga whales from the St. Lawrence Estuary (SLE) population was initiated at the Faculté de Médecine Vétérinaire (FMV) of the University of Montreal. Among the major results of this research program was the finding of a high prevalence of cancers and high tissue levels of chemical contaminants, many of which are known to have endocrine-disrupting effects. 27,36
Lesions suggestive of endocrine disruption in the SLE beluga whale population include hyperplastic and degenerative changes of the adrenal gland, 17,29 two “adenomas” of the thyroid gland, a case of true hermaphroditism, 18 one case of male pseudohermaphroditism (I. Mikaelian, unpublished data), three cases of mammary carcinomas, 38 an uterine adenocarcinoma, 30 and a high prevalence of fibroleiomyomas of the female tubular genitalia. 39 We herein compare histologic lesions of the thyroid glands of beluga whales from the SLE and from the Hudson Bay in the Canadian Arctic.
Beluga whales found dead stranded on the shores of the St. Lawrence between May 1996 and December 1998 were transported to the FMV where complete postmortem examination was carried out. Beluga whales from Hudson Bay were collected through subsistence hunting in August 1995 and postmortem examination was carried out on site.
Sections of major organs and all lesions for animals from both populations were fixed in 10% neutral buffered formalin. Serial sections (5 mm thick) of the thyroid gland were made to detect any macroscopic anomaly. Formalin-fixed tissues were routinely processed, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin-phloxine-saffron (HPS). Aging of whales was carried out by counting dentine growth layers on longitudinal sections of teeth, adopting the standard of two growth layer groups per year. 22
The diameter (D) of cystic and hyperplastic lesions was determined on each histologic section. The volume (V) of these lesions was determined as follows: V = πD3/6.
The relationship between age and the total volume of cysts and adenomatous hyperplasia was tested using Spearman rank correlation. The level of significance was set at P = 0.05. Statistical analysis were performed using the SAS 6.12 software (SAS Institute Inc., Cary, NC).
Nine of 16 SLE beluga whales were affected by adenomatous hyperplasia (Table 1). These lesions appeared macroscopically as white nodular masses, up to 17 mm in diameter, that distorted the thyroid gland (Fig. 1). Six of 14 beluga whales from Hudson Bay were affected by similar but smaller (up to 7 mm in diameter) lesions. These lesions were found in animals aged 9 years or older. Among adult animals (older than 6 years), the lesions were observed in 6 of 13 Hudson Bay and 9 of 10 SLE belugas. Some of these masses were cystic to polycystic and were filled with a thick viscous amber-colored material. Histologically, these lesions consisted of multiple expansive nodules. A thin fibrous capsule often surrounded these nodules (Fig. 2). The smaller nodules consisted of variably sized follicles with a cuboidal to columnar epithelium. This epithelium often formed papillary projection into the lumen of the follicles. The larger nodules were often surrounded by a thick capsule and markedly compressed the surrounding parenchyma. These larger nodules were composed of small follicles separated by wide solid areas and were often subdivided by thick fibrovascular septa. They were generally densely cellular, although some were partly cystic. Cells in these nodules were closely packed and polygonal to fusiform. They had a moderately abundant acidophilic and delicately granular cytoplasm. Their nucleus was basal in columnar cells to central in fusiform cells. The nucleus was round with a coarsely granular chromatin. The center of the larger nodules was often necrotic and hemorrhagic.
Total volume (mm3) of thyroid lesions in beluga whales from Hudson Bay and the St. Lawrence Estuary.
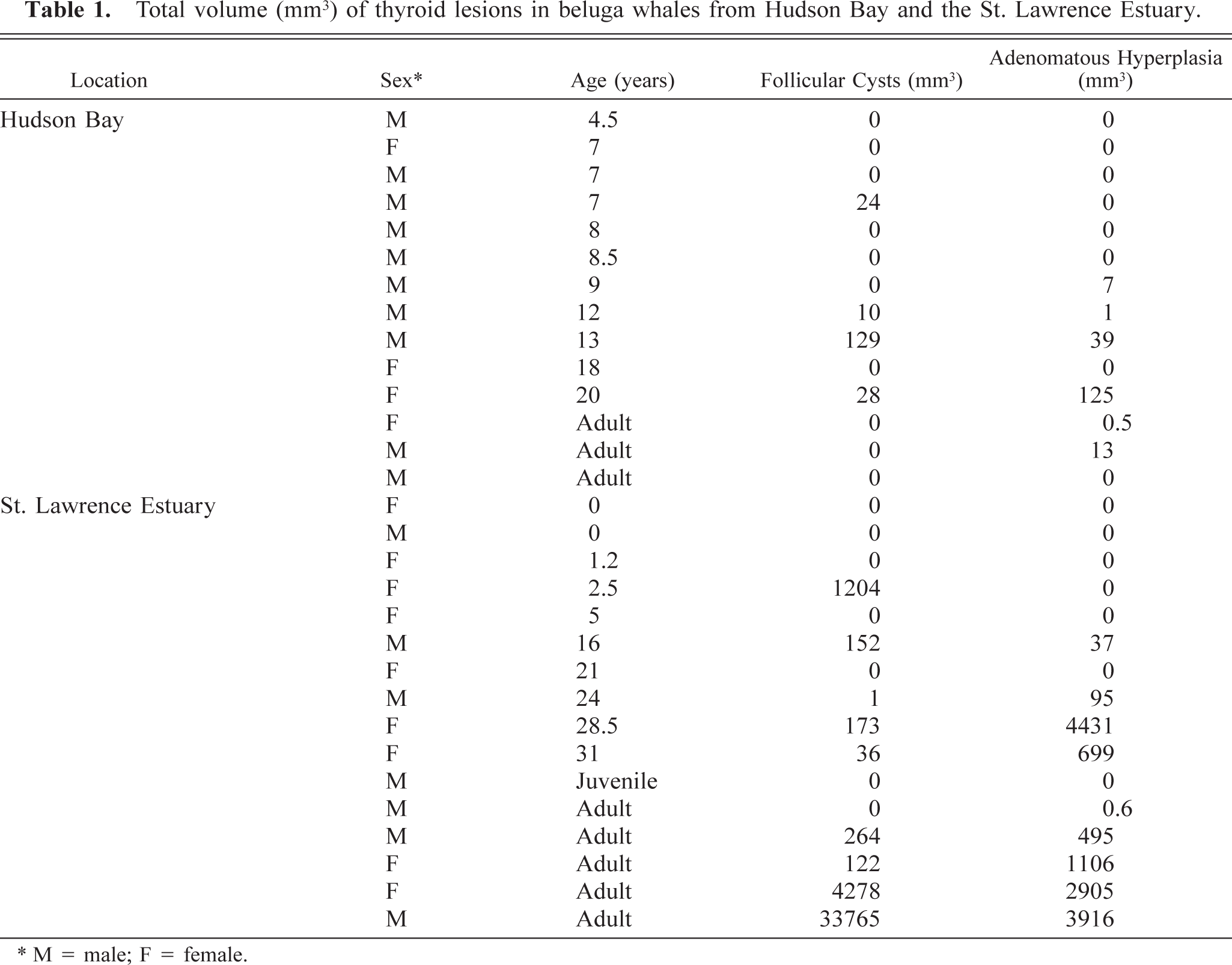
∗ M = male; F = female.

Thyroid gland, beluga whale from the St. Lawrence Estuary. Two large nodules of adenomatous hyperplasia markedly distort the architecture of the thyroid gland. One of the nodules is composed of multiple smaller coalescing nodules. Formalin-fixed tissue. Bar = 5 mm.
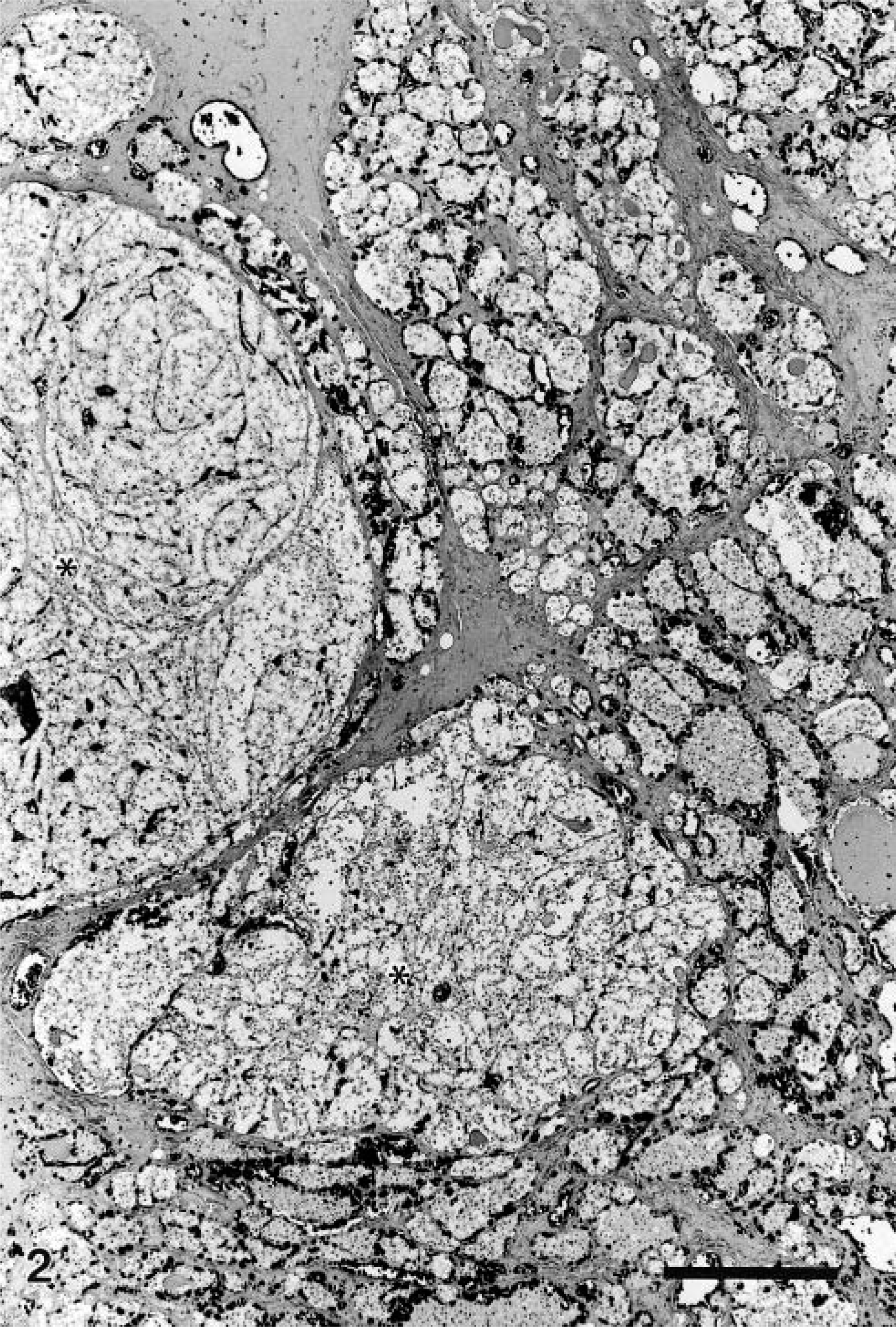
Thyroid gland, beluga whale from the St. Lawrence Estuary. The thyroid contains two small nodules of adenomatous hyperplasia (∗), one of which is surrounded by a delicate fibrous capsule. HPS. Bar = 200 µm.
Follicular cysts measuring up to 25 mm in diameter (Fig. 3) were found in nine of 16 beluga wales from the SLE. Smaller follicular cysts, up to 7 mm in diameter, were found in four of 14 beluga whales from Hudson Bay. Cysts were found in animals as young as 2.5 years old. Among adult animals (older than 6 years), cysts were observed in 4 of 13 Hudson Bay and 8 of 10 SLE belugas. These cysts contained an amber-colored gelatinous material. Histologically, follicular cysts were lined by a flattened follicular epithelium and were filled with colloid. The surrounding connective tissue was often mildly thickened by collagen fibers.
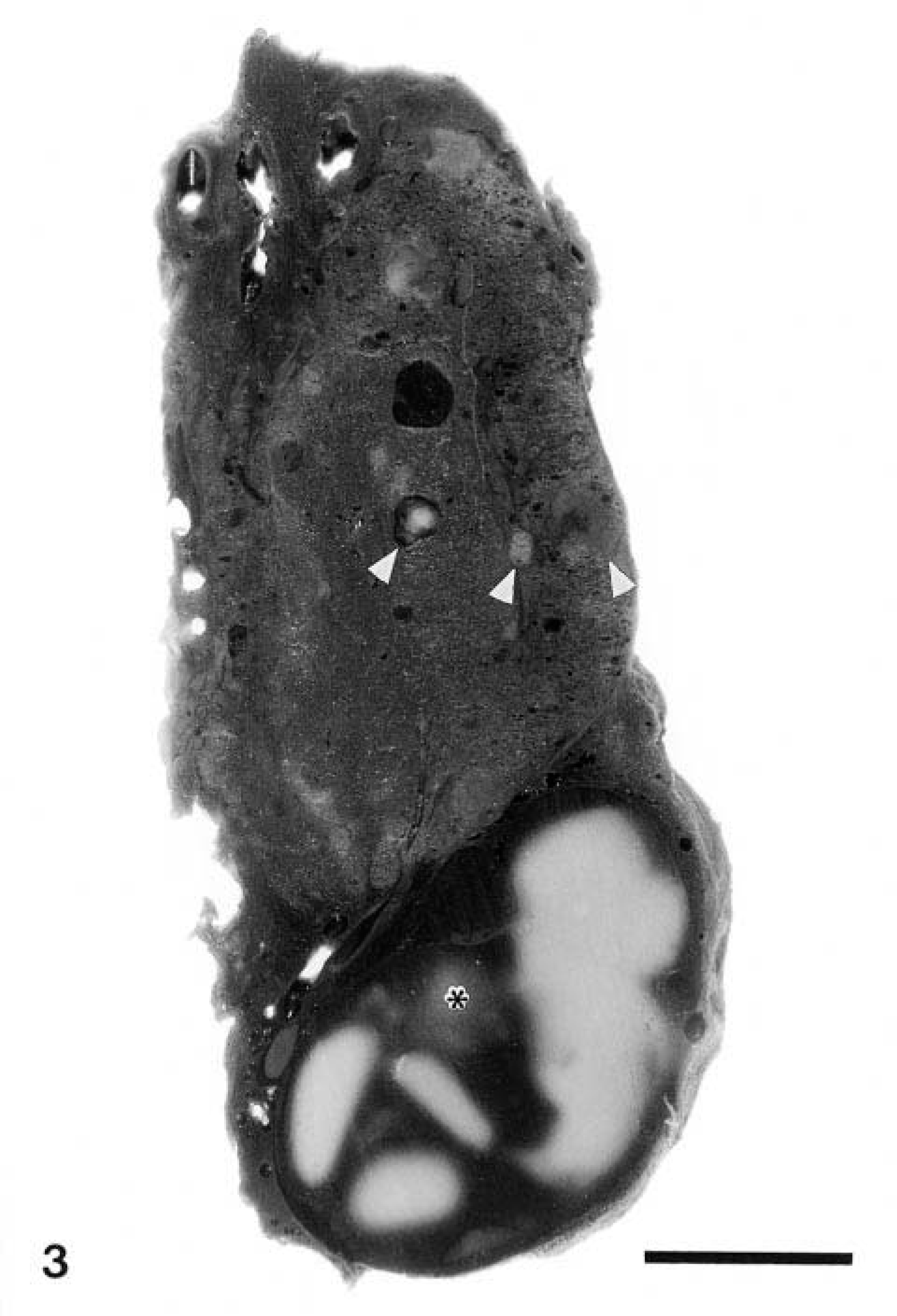
Thyroid gland, beluga whale from the St. Lawrence Estuary. The thyroid gland contains a large cyst filled with coagulated colloid (∗). Smaller cysts and minute nodules of adenomatous hyperplasia (arrowheads) are scattered throughout the thyroid gland. Formalin-fixed tissue. Bar = 8 mm.
The volume of the lesions of adenomatous hyperplasia was significantly positively correlated with age in both populations (P < 0.05). There was no relation between age and the volume of cysts.
The pituitary glands of beluga whales from the SLE was serially sectioned every 1.5–2 mm. Pituitary tumors were not detected. Pituitary glands of beluga whales from the Hudson Bay could not be examined because of field conditions.
The present study showed that hyperplastic and cystic lesions of the thyroid gland are common in adult beluga whales from two populations with different tissue levels of environmental contaminants. The only proliferative lesions of the thyroid gland reported in marine mammals prior to this study were two “follicular adenomas” in an SLE beluga whale. Hyperplastic lesions of the thyroid gland in beluga whales were categorized as adenomatous hyperplasia in accordance with the human 24,35 and veterinary literature. 34 Because some of the nodules were encapsulated, differential diagnosis of adenomatous hyperplastia of the thyroid gland should include thyroid follicular adenoma. However, hyperplastic thyroid lesions were always multiple, which is not consistent with a diagnosis of adenoma. Also, it is now accepted that hyperplastic lesions and adenomas of the human thyroid gland cannot be differentiated histologically and belong to the same process. 19 Thus, the lesions diagnosed earlier as thyroid adenomas in beluga whales would more adequately be categorized as adenomatous hyperplasia.
Thyroid lesions are rare in marine mammals other than SLE beluga whales and consist of colloid goiter, colloid depletion, and fibrosis. 14,15,42 Thyroid lesions were not detected in surveys of the thyroid gland of various species of marine mammals, 16 including beluga whales from Hudson Bay. 46
In humans and domestic animals, factors other than environmental contaminants that are implicated in the etiology of thyroid hyperplasia and neoplasia include iodine deficiency or excess, lymphocytic thyroiditis, and aging. Iodine deficiency is unlikely in a marine mammal species because sources of iodine are abundant in the marine environment, and iodine excess has not been reported in these species. Lymphocytic thyroiditis was absent in the animals from the present study. Therefore, the major causes of proliferative lesions of the thyroid gland in man and domestic animals, other than aging, are presumably not implicated in the occurrence of these lesions in beluga whales.
Polychlorinated biphenyls (PCBs) are one of the major recognized contaminants causing hyperplastic and neoplastic lesions of the thyroid gland. 1,3 Tissue concentrations of PCBs in beluga whales from the present study were not evaluated. However, earlier studies have indicated that tissue levels of PCBs are approximately 10 times higher in beluga whales from the SLE than from Hudson Bay. 6,37
The effects of PCBs on thyroid hormone homeostasis have been extensively investigated in rodents. Polychlorinated biphenyls affect thyroid hormone homeostasis by interfering with thyroid hormone synthesis, secretion, metabolism, and transport system (Table 2). The end result of endocrine disruption by PCBs is a decrease in plasma concentrations of thyroid hormones followed by a compensatory increase in thyroid-stimulating hormone (TSH) secretion by the hypophysis. 26 Permanent stimulation of the thyroid by TSH leads to thyroid follicular hyperplasia and subsequently to thyroid follicular neoplastic transformation. The greater sensitivity of the rodent thyroid to drugs, chemicals and physical perturbations, compared with the human thyroid, is related to the shorter plasma half-life of thyroxine, which results in higher basal TSH levels and therefore increased thyroid stimulation. 20,22 No data are available on the thyroid hormone plasma half-life in beluga whales. However, plasma half-life of thyroid hormones in two dolphin species is much shorter than in humans. 47 A short half-life of thyroid hormones in dolphins may increase the risk of developing proliferative thyroid lesions through mechanisms similar to that of rodents.
Mechanisms of polychlorinated biphenyl interference with thyroid homeostasis.

Alteration of the thyroid function has been documented in humans contaminated by PCBs. 4 Hyperplastic thyroid lesions, thought to be associated with environmental contaminants, are found in fish and aquatic birds 21 from the Great Lakes, which are drained by the St. Lawrence River. 23,28,31,41 The occurrence of thyroid lesions in populations of whales with radically different levels of exposure to contaminants indicates that, in beluga whales, PCBs should not be considered as the only possible cause of hyperplastic lesions of the thyroid gland.
Thorough statistical comparison between the two populations was not possible because beluga whales from Hudson Bay were significantly younger than those from the SLE. Further studies using age-matched groups are needed to compare thyroid lesions of SLE beluga whales with those of other beluga whale populations.
Follicular cysts were found in beluga whales from both populations. Cowan described severe colloid goiter in four of 55 pilot whales (Globicephala melaena) from Newfoundland 15 . Follicular cysts in beluga whales histologically resemble the lesions in pilot whales. However, follicular cysts in beluga whales did not result in an overall enlargement of the thyroid gland. The cause of these lesions remains undetermined.
Footnotes
Acknowledgements
We are thankful to S. Lair for participating in the sampling of beluga whales in the Arctic and to J. Cardin, J. Deslandes, and M. Langlois for technical assistance. This study was funded by the Canadian Cooperative Wildlife Health Centre and the Centre Québécois sur la Santé des Animaux Sauvages.
