Abstract
Benign mammary mixed tumors in dogs resemble human salivary pleomorphic adenomas with regard to their histogenesis, including the occurrence of cartilaginous or bony metaplasia as well as the expression pattern of cytoskeletal proteins in proliferative myoepithelial cells. Recently, a monoclonal antibody specific for class II β-tubulin has been developed. The epitope it recognizes was determined to be the hepta-peptide Glu-Glu-Glu-Glu-Gly-Glu-Asp, which is the common sequence found among the canine, rat, mouse, and human class II β-tubulin-specific regions. We carried out immunohistochemical studies on mammary mixed tumors obtained from three female dogs using this the monoclonal antibody. The antibody to class II β-tubulin reacted intensely with proliferative myoepithelial cells in canine mammary mixed tumors, whereas staining was barely detectable in normal myoepithelial cells surrounding alveoli and alveolar ducts within the tumor and adjacent normal tissue. Proliferative myoepithelial cells also expressed vimentin, but α-smooth muscle actin (αSMA) staining was barely detectable. Immunoblot analysis showed that class II β-tubulin and vimentin were expressed in myoepithelial cell lines prepared from the three mammary mixed tumors. On the other hand, only one cell line, which was negative for αSMA, produced cartilage-specific type II collagen. These results suggest that class II β-tubulin could be a new molecular marker of proliferating myoepithelial cells in canine mammary mixed tumors and that differential expression of cytoskeletal components is associated with cartilaginous metaplasia of proliferative myoepithelial cells in mixed mammary tumors.
In humans, pleomorphic adenoma is the most common tumor of the major salivary glands. It is characterized by epithelial and myoepithelial proliferation and an abundant myxoid or chondroid matrix. Previous studies demonstrated the expression pattern of cytoskeletal proteins in proliferative myoepithelial cells 6,16,18,31 and suggested the involvement of these cells in the formation of metaplastic cartilage and bone. 11,17,26,27 The chondroid matrix in canine mammary mixed tumor has also been considered to develop from metaplasia of proliferative myoepithelial cells, 19,21,22,24 but these cells have not yet been fully characterized. In early stages of mammary mixed tumor development, myoepithelial cells begin to proliferate in the area between the epithelium and basement membrane. Proliferative myoepithelial cells form a nodular mass, then scatter in the myxomatous substance they secrete, and finally, type II collagen–containing cartilaginous matrix accumulates around these cells. 3
Myoepithelial cells are known to originate from the ectoderm, 11 but these cells have both epithelial and mesenchymal characteristics in terms of cytoskeletal proteins. 7,13,16,31 Alpha-smooth muscle actin (αSMA) is usually detected in normal myoepithelial cells; other smooth muscle–specific proteins, such as smooth muscle myosin heavy chain and calponin, are also present. 7 Myoepithelial cells coexpress cytokeratin, vimentin, and, occasionally, glial fibrillary acidic protein (GFAP) intermediate filaments. 6,18,24 In addition, desmin is expressed in proliferative myoepithelial cells in human salivary pleomorphic adenoma. 31
Tubulin is the major component of microtubules; it is a heterodimer of two polypeptide chains, designated as α- and β-tubulin. In higher vertebrates, β-tubulins have been classified into seven isotypes, which have different cell type distributions and different stabilities. 10,15,25,30 These β-tubulin isotypes are highly conserved across vertebrate species and differ from each other predominantly at the carboxyl terminus. 5,23 Each isotype has a unique tissue distribution: classes I and IVb are constitutive in all cell types, classes III and IVa are brain specific, and the class VI is expressed only in hematopoietic cells. Class II is a major brain tubulin but is found at low levels in a wide range of tissues. 1,2,10 Recently, expression of the class II isotype in some mesenchymal cells, including chondrocytes, vascular smooth muscle cells, and endothelia, has been described. 2 However, no information on the expression of class II β-tubulin isotype in myoepithelial cells in normal or pathologic state is available.
In this study, expression of the class II isotype of β-tubulin in proliferative myoepithelial cells in canine mammary mixed tumors was examined immunohistochemically using the epitope-defined monoclonal antibody, MAb58A. 2 In addition, the production of selected cytoskeletal proteins and cartilage-specific type II collagen in cultured cells derived from mammary mixed tumors was evaluated.
Materials and Methods
Analysis of the specificity of anti–class II β-tubulin antibody
To determine whether the monoclonal antibody, MAb58A, is specific for canine class II β-tubulin, immunochemical analysis with synthetic peptides corresponding to the five isotypes (I, II, III, IVa, and IVb) of canine β-tubulin was performed using the SPOTs kit (Genosys Biotechnology, Geselshaft fur Biotechnologische Forschung, Braunschweig, Germany) using F-moc amino acid derivatives as follows. The nucleotide sequences and deduced amino acid sequences of isotype-specific regions corresponding to canine classes I, II, III, IVa, and IVb β-tubulin were reported previously, 1 and these sequences appeared in the DDBJ/EMBL/GenBank databases under the accession numbers AB022054 (class I), AB022055 (class II), AB022056 (class III), AB022057 (class IVa), and AB022058 (class IVb). Isotype-specific peptides corresponding to the five canine isotypes of β-tubulin (as shown in Fig. 1) were synthesized on the SPOTs membrane (Genosys). The synthesized peptides on the membrane were acetylated with 20% piperidine-dimethylformamide and then treated by side-chain protection with 48.8% dichloromethane-48.8% trifluoroacetic acid-2.4% triisobutylsilane. The resulting membrane was incubated with a blocking buffer (Genosys) and then incubated with MAb58A at room temperature for 3 hours. After washing with 0.15 M NaCl-50 mM Tris-HCl, pH 7.6 (Tris saline), containing 0.05% Tween 20, the membrane was incubated with alkaline phosphatase–labeled secondary antibody at 37 C for 30 minutes and finally immersed in 0.01% nitroblue tetrazolium (NBT, Promega Co., Madison, WI) and 0.005% 5-bromo-4-chloro-3-indolyl phosphate (BCIP, Promega) diluted with 100 mM Tris-HCl, pH 9.5, containing 100 mM NaCl and 5 mM MgCl2.
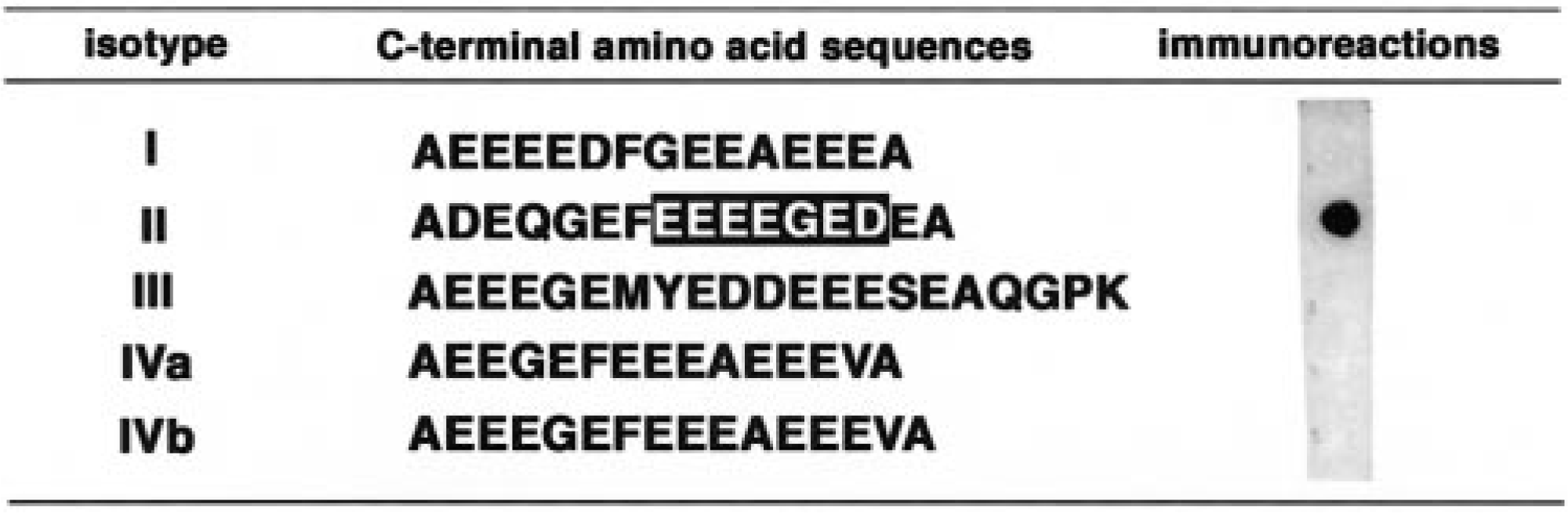
Immunoreaction of MAb58A with synthetic peptides corresponding to the isotype-specific partial sequences of five canine classes of β-tubulin (I, II, III, IVa, and IVb). Open letters in the isotype II peptide sequence indicate the epitope sequence of the antibody.
Tissue specimens
Surgical samples of benign mammary mixed tumors that developed in three aged, female mongrel dogs (dog Nos. 1–3) were obtained, and each tumor specimen was divided into two parts under sterile conditions. A part of the tumor tissue was fixed in methanol Carnoy solution for immunohistochemistry, and the remaining tissue was used for preparation of cell culture.
Cell culture
Cell lines were prepared from the three mammary mixed tumors (dog Nos. 1–3) by outgrowth from the explants as described previously. 4 The cell lines were termed MME-1, MME-2 and MME–3, respectively. Cells were maintained in Dulbecco modified Eagle medium (Life Technologies Inc., Grand Island, NY) containing 10% fetal calf serum (Life Technologies), penicillin (50 units/ml, Sigma Chemical Co., St. Louis, MO), streptomycin (50 µg/ml, Sigma), and neomycin (100 µg/ml, Sigma). These cells were grown to confluence and used for immunoblot analysis.
Immunohistochemistry
Paraffin-embedded 3-µm sections were stained with hematoxylin and eosin for histopathologic studies. Immunoperoxidase staining of selected serial sections was performed using the peroxidase-antiperoxidase method. 28 After treatment with 0.5% periodic acid to inactivate endogenous peroxidase, deparaffinized sections were incubated first with MAb58A or with monoclonal antibodies to pig vimentin (1 : 100, DAKO, Glostrup, Denmark), pig glial GFAP (1 : 100, Sigma), or human αSMA (1 : 1,000, DAKO) overnight at 4 C; then with a secondary antibody (1 : 100, DAKO) at 37 C for 30 minutes; and finally, with peroxidase-antiperoxidase complex (1 : 100, DAKO) at 37 C for 30 minutes. Each section was immersed in 0.02% 3,3′-diaminobenzidine–Tris saline supplemented with 0.03% H2O2 and counterstained with Mayer's hematoxylin.
Immunoblot analysis
A crude cytoskeletal fraction was prepared from an adult canine brain as described previously. 1 In brief, tissue was homogenized in Tris saline containing a protease inhibitor cocktail (5 mM ethylenediamine tetraacetic acid–2 mM N-ethylmaleimide–1 mM phenylmethylsulfonyl fluoride) to eliminate water-soluble proteins. The insoluble pellet was washed twice with ice-cold acetone, once with ethyl ether, and then air-dried. The resulting dried powder was dissolved in 8 M urea–40 mM dithiothreitol–Tris saline containing protease inhibitor cocktail (urea buffer).
Cultured cells were directly dissolved in the urea buffer after washing the cell layer twice with phosphate-buffered saline (PBS). Preparation of the collagen fraction was performed as described previously. 12 In brief, the medium and cell layer were collected in the same tube and sonicated on ice in the presence of protease inhibitors, and then collagenous proteins were precipitated by the addition of ammonium sulfate to a final concentration of 176 mg/ml. These precipitates were dissolved in 0.5 M acetic acid and digested with pepsin (1.1 mg/ml, Roche Diagnostics GmbH, Mannheim, Germany) at 4 C for 6 hours. After inactivation of pepsin by neutralizing with NaOH, soluble fractions were dialyzed against 5 mM acetic acid and lyophilized. The cytoskeletal and collagenous fractions were subjected to 10 or 5% sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS-PAGE), respectively, and the separated protein bands were electrophoretically transferred to a cellulose nitrate membrane. Total protein bands were observed by 0.1% Coomassie brilliant blue R-250 staining. After blocking with 1% casein, the membrane was incubated with antibodies to class II β-tubulin (MAb58A), 2 pig vimentin (1 : 500, DAKO), human αSMA (1 : 5,000, DAKO), or α1 chain of canine type II collagen (α1[II]) 4 overnight at 4 C and then with alkaline phosphatase–labeled or peroxidase-labeled secondary antibody at a dilution of 1 : 5,000 at 37 C for 1 hour. After washing with 0.05% Tween 20–PBS, the resulting membrane was immersed in NBT–BCIP in a similar manner as for epitope analysis. To detect the signal for type II collagen, a horseradish peroxidase–based chemiluminescent detection system (ECL, Amersham Pharmacia Biotech, Buckinghamshire, UK) was used according to the manufacturer's protocol.
Results
Characterization of the monoclonal antibody
The monoclonal antibody, MAb58A, recognized a single 55-kd band of the expected size in adult canine brain extracts on western blot (Fig. 2). Immunoreaction with synthetic peptides derived from isotype-specific regions of five isotypes of canine β-tubulin demonstrated that MAb58A was specific for the class II–derived synthetic peptide and did not react with other isotypes of canine β-tubulin (Fig. 1). Thus, the antibody was judged to be specific for the class II isotype of canine β-tubulin.
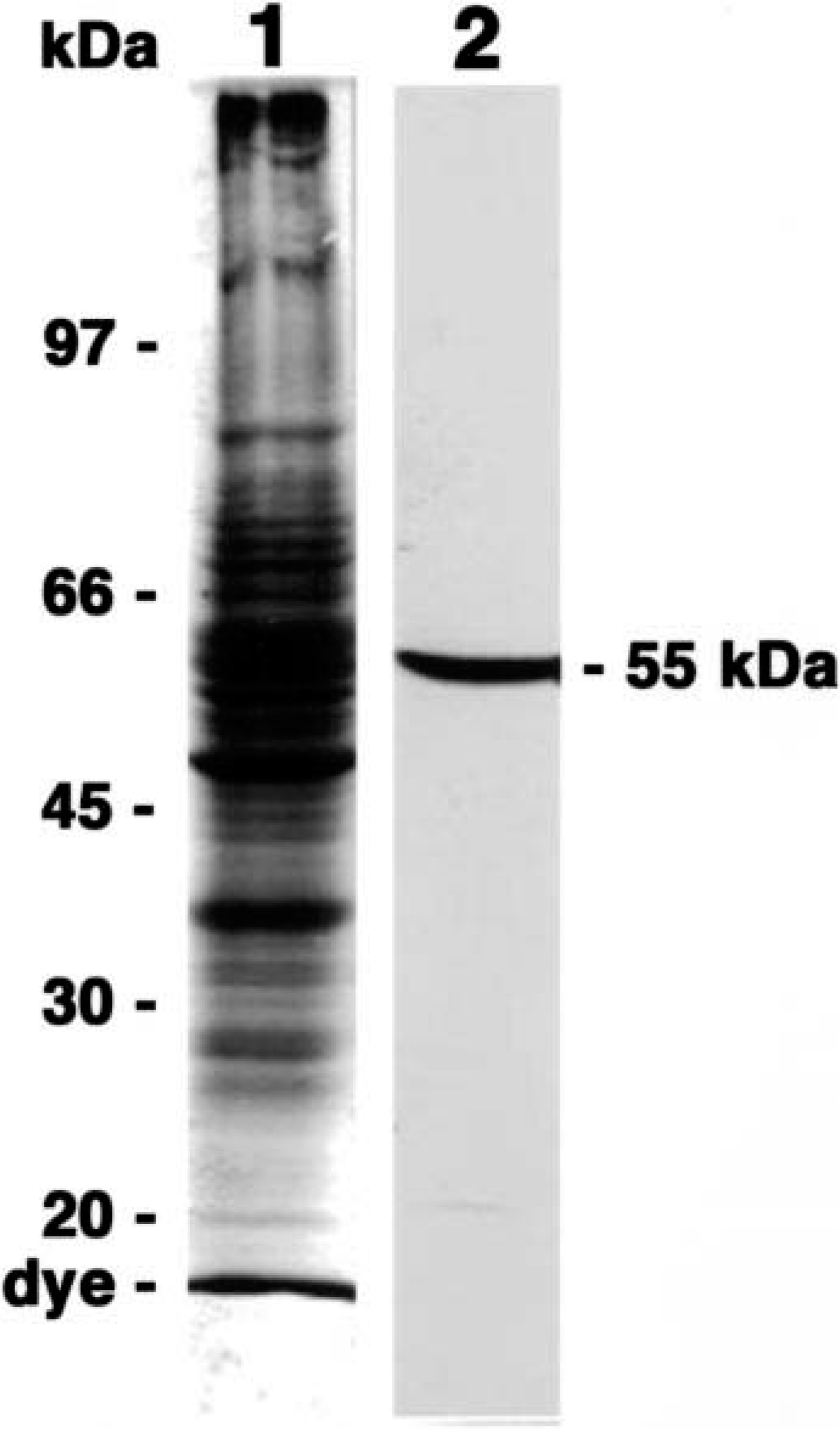
Coomassie brilliant blue R-250 staining (lane 1) and immunoblotting with MAb58A (lane 2) of the crude cytoskeletal fraction extracted from an adult canine brain. MAb58A recognized 55-kd band as the expected size for β-tubulin. The molecular weight of size markers is indicated (left).
Immunohistochemical studies
In dog No. 1, proliferative myoepithelial cells formed a small mass adjacent to an alveolar duct. These myoepithelial cells were intensely stained by MAb58A (Fig. 3A, arrowheads), but the majority of myoepithelial cells in the nodules were negative for αSMA (Fig. 3B, arrowheads). Normal-appearing myoepithelial cells that lined the alveolar ducts were reactive with anti-αSMA antibody (Fig. 3B, arrows) but were only faintly stained by MAb58A (Fig. 3A, arrows). In dog No. 2, proliferative myoepithelial cells displaced the alveolar ducts and replaced some alveoli. These cells were reactive with MAb58A (Fig. 4A), but αSMA-positive cells were no longer observed in myoepithelial cell nodules (Fig. 4B). Some MAb58A-positive cells also expressed GFAP (Fig. 4C). The antibody to vimentin reacted intensely with proliferative myoepithelial cells and interstitial fibroblasts (Fig. 4D). The mammary mixed tumor in dog No. 3 was characterized by cartilaginous metaplasia and the accumulation of type II collagen (data not shown), indicating a late stage of tumor 4 Chondrocyte-like cells as well as proliferative myoepithelial cells surrounding the cartilaginous masses were positively stained with MAb58A (Fig. 5). In the normal mammary gland and “nonmixed” type mammary tumors or the malignant type of mixed tumors, there were no MAb58A-positive cells except for peripheral nerve fibers, vascular smooth muscle cells, and endothelia (data not shown).
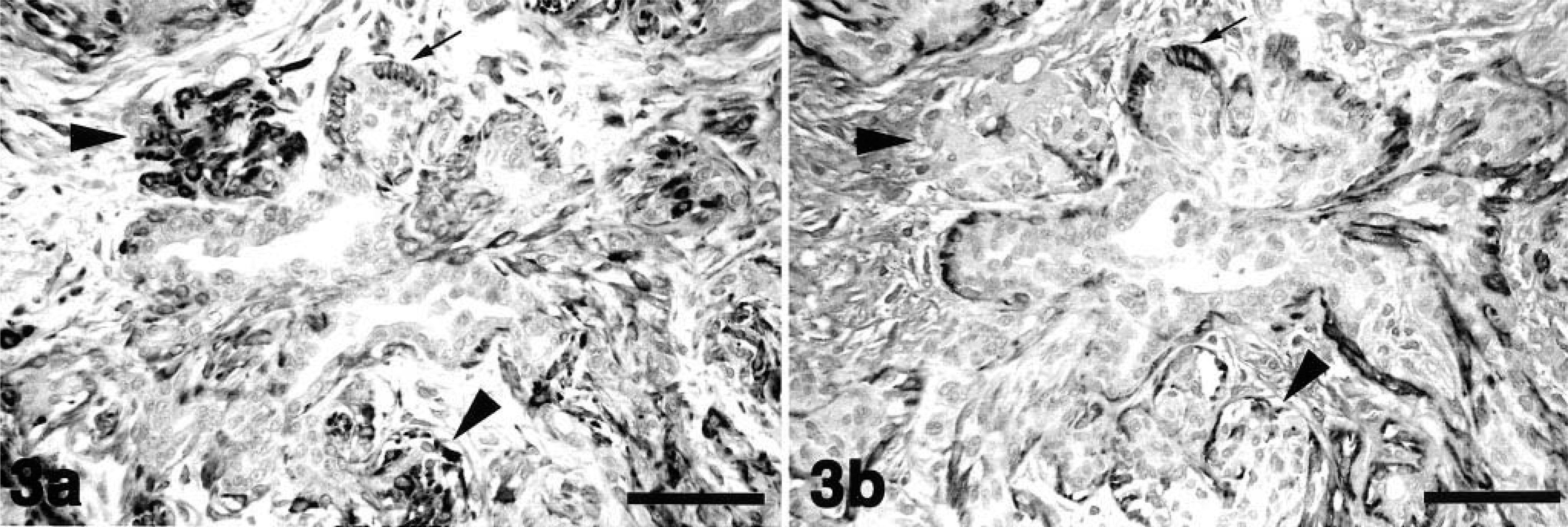
Mammary mixed tumor; dog No. 1.
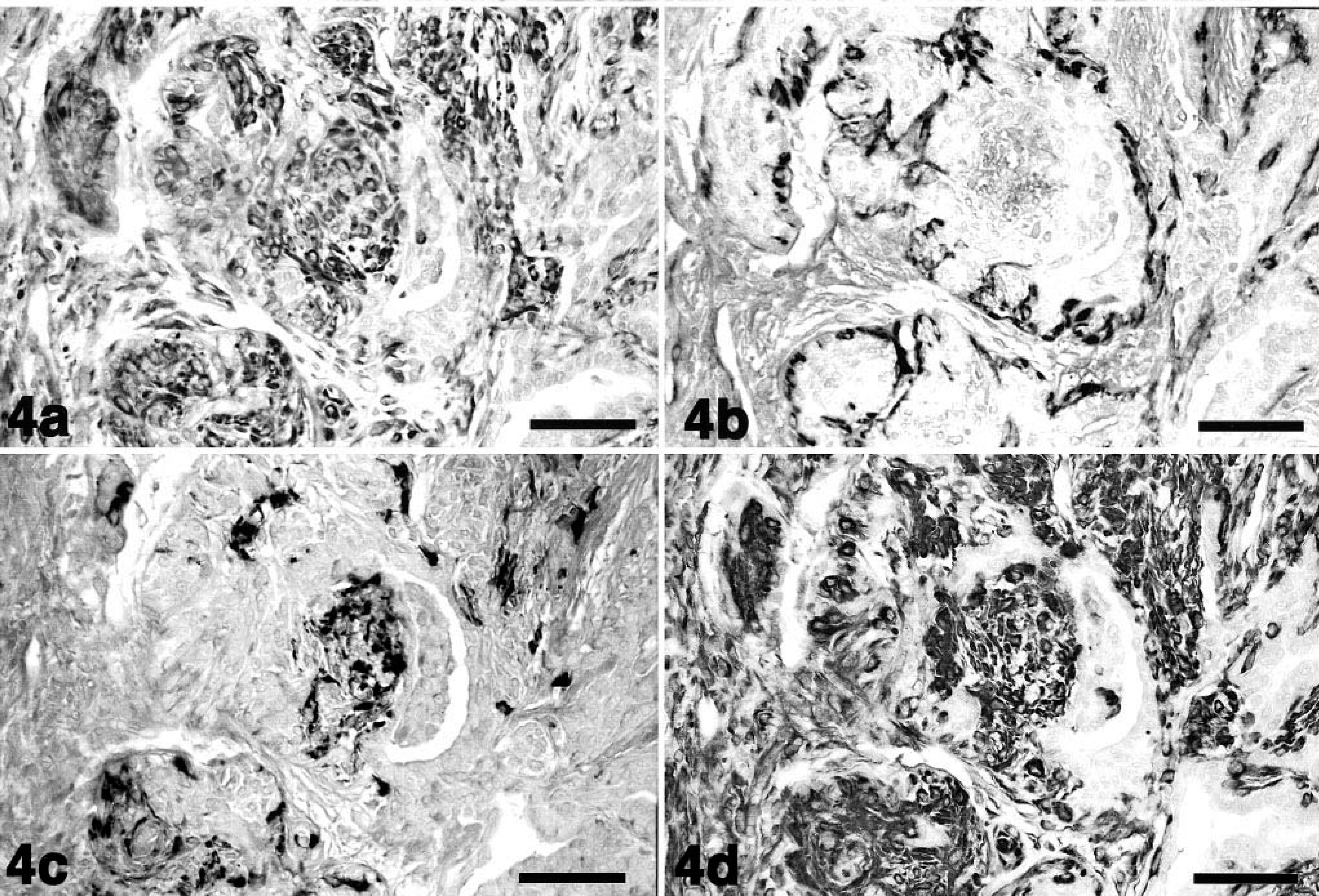
Mammary mixed tumor; dog No. 2.
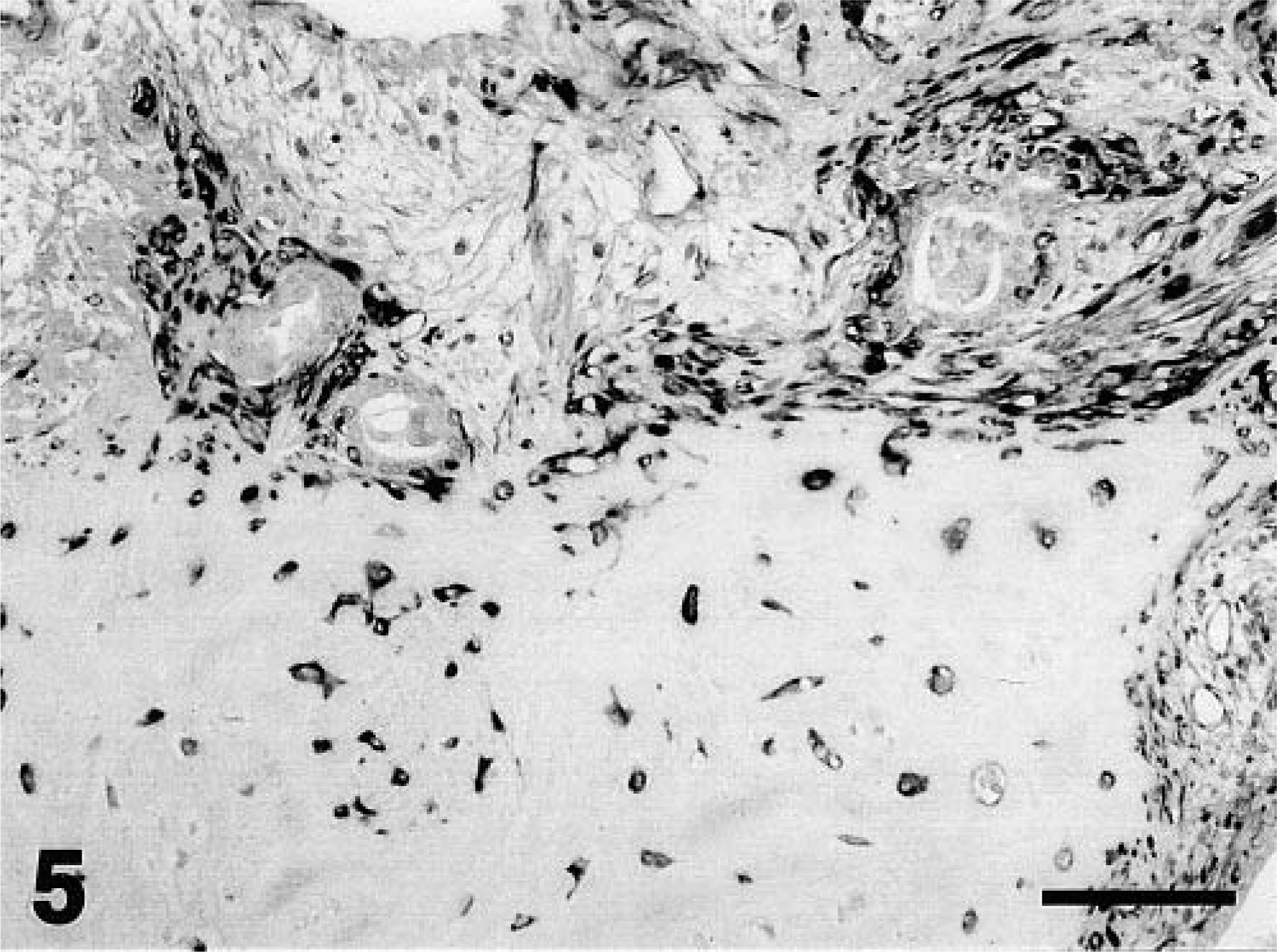
Mammary mixed tumor; dog No. 3. Immunostaining with class II β-tubulin. Chondrocyte-like cells in the metaplastic cartilage were also positive for class II β-tubulin. Mayer's hematoxylin counterstain. Bar = 100 µm.
Immunoblot studies
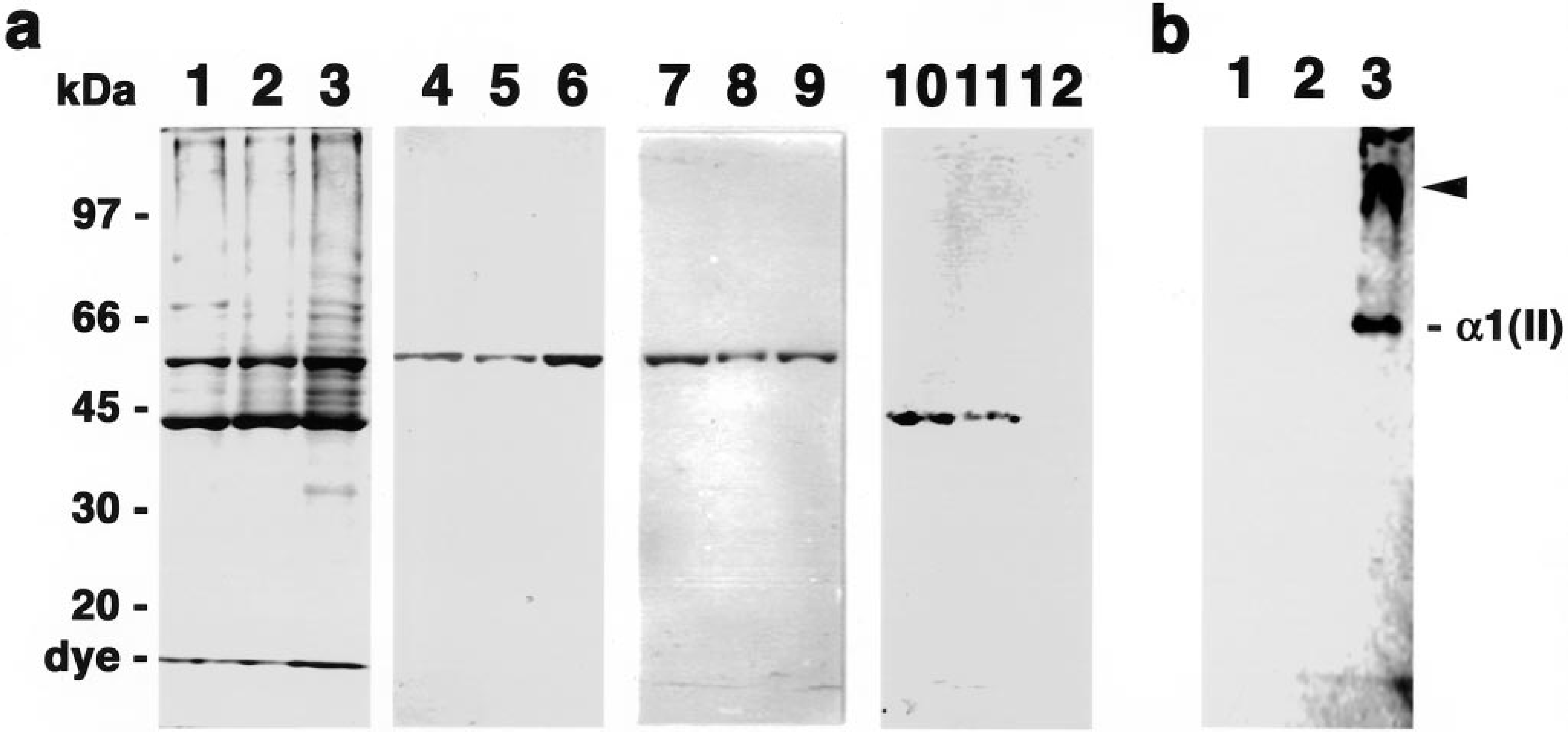
The expression of class II β-tubulin, vimentin, αSMA, and type II collagen in three cell lines derived from each of the mixed tumors (dog Nos. 1–3) was analyzed by immunoblot analysis. The cells of all three cell lines were spindle shaped and had chondrogenic ability in collagen gel as described previously. 3 There was no significant difference in the SDS-PAGE pattern of protein extracts from the three cell lines (Fig. 6A, lanes 1, 2, and 3) in Coomassie-blue-stained gels. The 55-kd class II β-tubulin isotype (Fig. 6A, lanes 4, 5, and 6) and 56-kd vimentin (Fig. 6A, lanes 7, 8, and 9) were detected in all three cell lines, although there were slight differences in the intensity of each component. On the other hand, the intensity of αSMA expression in the three cell lines differed. A positive band with a molecular weight of 43 kd corresponding to αSMA was clearly detected in MME-1 (derived from dog No. 1) and MME-2 (dog No. 2) (Fig. 6A, lanes 10 and 11) but was barely detectable in MME-3 (dog No. 3) (Fig. 6A, lane 12). On the other hand, α1 chain of type II collagen and its cross-linked dimer were detected in only MME-3 (Fig. 6B, lane 3); the two other cell lines did not produce detectable type II collagen (Fig. 6B, lanes 1 and 2).
Discussion
Despite their ectodermal origin, myoepithelial cells have both epithelial and mesenchymal characteristics. These cells are involved in the histopathogenesis of human pleomorphic adenomas that develop in several exocrine organs, including the salivary, sweat, and mammary glands 21,22,26,31 , and of canine mammary mixed tumors, 3,4,13,19 which are characterized by the development of heterotopic cartilage and bone. Recently, we reported production of the monoclonal antibody, MAb58A, which is specific for rat class II β-tubulin, and characterization of the epitope recognized. 2 Furthermore, the isotype-specific region in canine class II β-tubulin was shown to be identical to that in rat class II β-tubulin. 1 Therefore, MAb58A was thought to be class II specific also in dogs. In the present study, we confirmed this by immunoblotting and by immunochemical analysis with synthetic peptides.
Immunohistochemical analysis with MAb58A revealed that proliferative myoepithelial cells expressed class II β-tubulin at various stages of canine mammary mixed tumor development. Myoepithelial cells proliferating as a small mass adjacent to the alveolar ducts already expressed this tubulin isotype, whereas normal myoepithelial cells, alveolar cells, and interstitial fibroblasts did not. This isotype of β-tubulin was also detected in chondrocyte-like cells in areas of cartilaginous metaplasia that developed in the late stage of mixed tumor development.
These observations strongly suggest that induction of class II β-tubulin isotype expression was involved in the histogenesis of the metaplasia. During chick chondrocyte differentiation, β7-tubulin, the counterpart of mammalian class I β-tubulin, was upregulated in hypertrophic chondrocytes, suggesting that this tubulin isotype is involved in promotion of chondrocyte differentiation. 8 In mixed tumors, expression of the class II isotype may be involved in chondrogenic differentiation of myoepithelial cells. This speculation is also supported by the finding that malignant myoepitheliomas without chondroid metaplasia are negative for class II isotype (data not shown). Alpha SMA–positive microfilaments disappeared as proliferation of myoepithelial cells in mixed mammary tumors progressed. Zarbo et al. 31 noted that proliferative myoepithelial cells, which were observed in 50% of 26 cases of human salivary pleomorphic adenomas, lost αSMA expression. These results indicate that αSMA is not a marker of proliferative myoepithelial cells. Neuronal differentiation of proliferative myoepithelial cells in human salivary pleomorphic adenoma has been reported. 14 Nerve growth factor induced the formation of long cytoplasmic processes in these cells. In this study, we observed that some proliferative myoepithelial cells were positive for GFAP. We suggest that transient expression of GFAP might be the result of neuronal or glial differentiation in these cells.
Biochemical studies on the expression pattern of cytoskeletal proteins in mixed tumors will lead to improved understanding of the histopathogenesis of mixed tumors. However, studies using tissue homogenates of these tumors do not reflect the characteristics of particular cell types because the class II isotype of β-tubulin is also found in vascular endothelial and smooth muscle cells and in peripheral nerve fibers in the mammary gland. Thus, cultured neoplastic cells prepared from the mixed tumors were used for further analysis. Class II β-tubulin and vimentin were consistently detected in these cell cultures, in agreement with immunohistochemical observations on the mixed tumors. On the other hand, αSMA expression was variable, and GFAP expression was undetectable in the three cell lines examined (data not shown). Human and rat myoepithelial-like cell lines were established from neoplastic salivary glands 9,14,29 and mammary mixed tumor, 20 respectively, and these cell lines were positive for vimentin and αSMA. Among the three cell lines examined in this study, two cell lines expressed αSMA, manifesting a normal myoepithelial αSMA phenotype; however, one line, MME-3, had lost this phenotype. In canine mixed mammary tumors, the numbers of αSMA-positive proliferative myoepithelial cells decreased in the early stage of canine mammary mixed tumors. In addition, the cell line MME-3 produced type II collagen in monolayer culture. We showed that MME-1 and -2 had chondrogenic ability as described previously. 3 These cell lines produced cartilage-specific type II collagen when cultured in a collagen gel but not when cultured in monolayer. Thus, MME-3 was considered to be the most transformed type among the three cell lines, as evaluated by loss of αSMA expression and production of type II collagen. Although there is no direct evidence for the myoepithelial origin of these cell lines, a myoepithelial origin was suggested by the findings that these cells expressed αSMA and had chondrogenic ability.
We showed the distribution of the class II β-tubulin isotype in canine benign mammary mixed tumors immunohistochemically and demonstrated production of this tubulin isotype in vitro. These observations indicate that class II β-tubulin may be a new marker of proliferative myoepithelial cells. In addition, our results suggest that expression of class II β-tubulin in myoepithelial cells occurs at an early stage of proliferation and is associated with decreased αSMA expression and the production of cartilage-specific collagens. Although the critical roles of the class II isotype of β-tubulin in proliferative myoepithelial cells are still not known, the evidence suggests that its expression is the earliest event in chondroid metaplasia of proliferative myoepithelial cells in canine mixed mammary tumors.
Footnotes
Acknowledgements
This work was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Science, Sports, and Culture of Japan (grant nos. 106602809 and 12215042).
