Abstract
A female 8-month-old Simmental calf was presented with a history of a gradually enlarging mass in the ventral abdominal skin since 4 months of age. The mass was well circumscribed, lightly pigmented, and rough surfaced with many fine fissures and was attached to the skin by a relatively broad pedicle. On cut section, there was a border between the reddish-black stroma and overlying epithelium, including hemorrhagic foci of variable sizes. Histologically, the tumor was papillomatous with angiokeratomatous features and irregular hyperplasia with epidermal rete ridges and dilated vascular channels filled with blood in the superficial dermis. In the epidermis, orthokeratotic hyperkeratosis, variably sized keratohyalin granules, and many koilocytes, some of which had papillomavirus (PV) genus-specific structural antigen-positive nuclei, were also observed. Cells lining the dilated vascular spaces were positive for vimentin and alpha-smooth muscle actin but negative for factor VIII-related antigen, desmin, and PV. The lesion was regarded as an angiokeratomatous papilloma and was similar to other angiomatous lesions.
Benign vascular tumors originating in skin are very common in dogs, less frequent in cats, and only occasionally seen in other species. 16 Reports of bovine vascular tumors are few, 8,16 and benign angiomatous lesions in various tissues have been described as juvenile angiomatosis, 14 hemangioma, 7 or hamartoma. 15 Angiokeratoma, a rare wart-like vascular lesion, is well known in humans 9 and dogs. 16 Clinically, angiokeratomas appear as one or several dark red papules, mostly with verrucous surface and histologically defined as one or more dilated blood vessels lying in the superficial dermis and in most cases associated with an epidermal reaction such as acanthosis and/or hyperkeratosis. 9 The poorly defined and rare cutaneous vascular lesion, bovine cutaneous angiomatosis, characterized by irregular hyperplasia of overlying epidermis, is an exophytic, solitary or multiple hemangioma-like lesion. It has been reported in various countries and attributed to abnormal vascular repair after injury, highly vascular granulation tissue, congenital vascular anomalies, and bacterial or viral infections. 3,8,16
Papillomaviruses (PVs) are infectious deoxyribonucleic acid (DNA) viruses of the family Papovaviridae and associated with cutaneous papilloma or fibropapilloma in cattle. 8,11,16 The role of PVs in vascular tumors is not clear. An immunohistochemical study suggested that in human immature angiomas PV etiopathogenesis is the origin of the lesion. 1 Subcutaneous and intracerebral injection of bovine PV in hamsters has produced fibroma, fibrosarcoma, chondroma, and meningioma. 2,8,10 In goats, experimental inoculation of teat skin with a caprine papilloma extract, of which an infective agent was not defined even after electron microscopic and immunohistochemical and DNA-DNA hybridization studies, resulted in the formation of a focal cutaneous hemangioma without epidermal hyperplasia. 13 Here, we describe an unusual case of angiokeratomatous papilloma having PV-positive hyperplastic epithelium in a veal calf and compare immunohistochemically and morphologically this observation with similar lesions.
A female 8-month-old Simmental calf developed a solitary mass in ventral abdominal skin. The gradually enlarging mass was first noticed at 4 months of age. Complete surgical removal was curative, and routine clinical examinations revealed no recurrence of the tumor 6 months after excision. The mass was fixed in neutral buffered formalin, embedded in paraffin, sectioned at 4 µm, and stained with hematoxylin and eosin (HE), Masson trichrome, Verhoeff–van Gieson, and Gomori reticulin. Additional sections were immunostained with monoclonal antibodies: anti-PV genus-specific structural antigen (PVGA) (Dako), anti-human factor VIII–related antigen (FVIII) (Dako), antivimentin (Novocastra), anti-human alpha-smooth muscle actin (ASMA) (Novocastra), anti-desmin (Dako), using an avidin–biotin–peroxidase complex method (ABC) (Dako). The technique included enzyme (protease) pretreatment and heat-induced epitope retrieval steps. All deparaffinized sections were quenched for endogenous peroxidase with 3% H2O2 in phosphate-buffered saline (PBS) and blocked with 5% normal goat serum before exposure to primary antisera. The sections were incubated with the biotinylated secondary antibody, followed by streptavidin-peroxidase conjugate, and then observed with 3-amino-9-ethyl carbazole and counterstained with Mayer's hematoxylin. Negative controls were prepared both by omitting the primary antibodies and by replacing them with PBS, normal rabbit serum, or unrelated mouse monoclonal antibodies. Tissue sections known to express the corresponding antigens served as positive controls.
The mass was 9 × 5 × 3 cm (72 g), well circumscribed, lightly pigmented, and rough surfaced. It contained many fine fissures and was attached to the skin by a relatively broad pedicle. On cut section, there was an apparent border between the bluish black–red dermis and overlying fissured epidermis, with hemorrhagic foci of variable sizes (Fig. 1). Microscopically, the epidermal lesions included irregular acanthosis and orthokeratotic hyperkeratosis with rete ridges of variable length. Within the dermis there were discrete groups of variably sized, dilated, thin-walled, and well-differentiated vascular channels that were filled with blood and lined by a single layer of flattened endothelium. Clusters of ectatic vascular structures in the superficial dermis were partly enclosed by epidermal extensions (Fig. 2). Vacuolar changes in individual keratinocytes, irregular keratohyalin granules, and koilocytes, some of which had central nuclei surrounded by a clear halo and condensed eosinophilic intracytoplasmic inclusions, were observed in the epidermis, mainly within the granular layer (Fig. 3). The interstitial tissue consisted of hypocellular fibrous connective tissue with focal hemorrhage and occasional vascular thrombosis. The epidermis was focally ulcerated. The mass was not encapsulated. Inflammation, except in ulcerated areas, was absent or minimal. No mitoses or atypical features were observed in the endothelial cells. Positive immunostaining for PVGA was present only in nuclei of koilocytes in the stratum granulosum (Fig. 4). Cytoplasmic inclusions were negative. Vascular components and interstitial cells were also negative for PVGA. Scattered, infrequent spindle to plump cells with features of fibroblasts between groups of dilated vessels, and a few lumina-associated endothelial cells were positive for vimentin. ASMA was always expressed in pericytes associated with the lumina. All components of the mass were negative for FVIII and desmin. Fine reticulin fibers occurred around vascular spaces, obviously with periendothelial orientation. Verhoeff–van Gieson staining was negative for elastin in the dilated dermal vessels. Slight collagen strands were detected between the dermal vascular channels by trichrome stain, but collagen was more prevalent in the ulcerated areas.
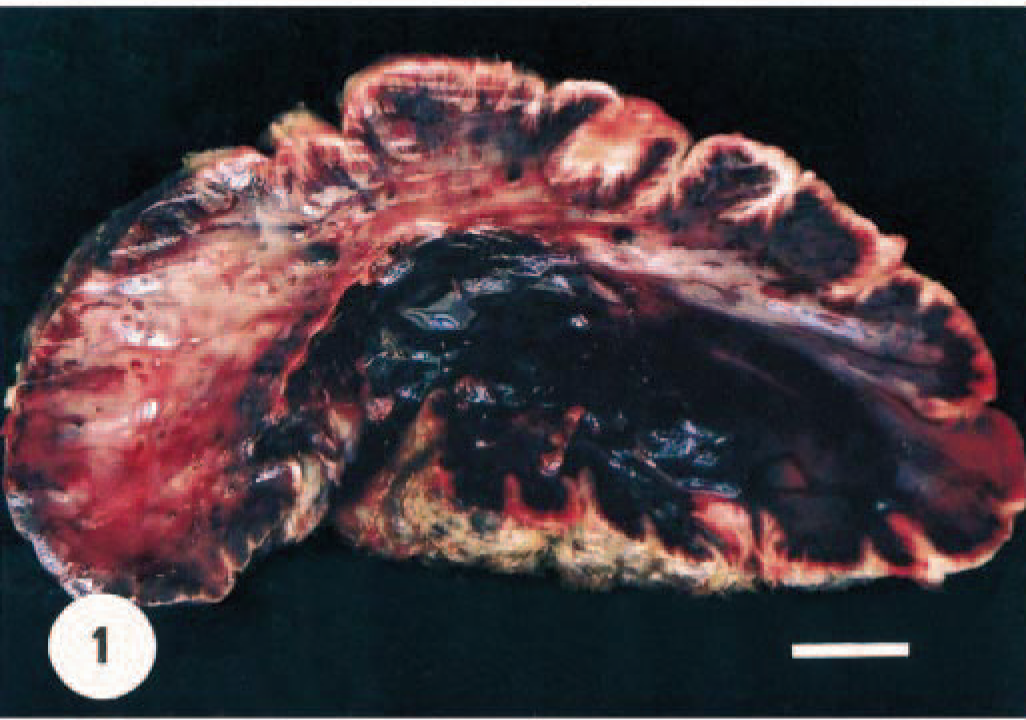
Angiokeratomatous papilloma; calf. Cut section of the mass including many hemorrhage foci and noticeable border between epidermis and vascular components of dermis. Bar = 1 cm.
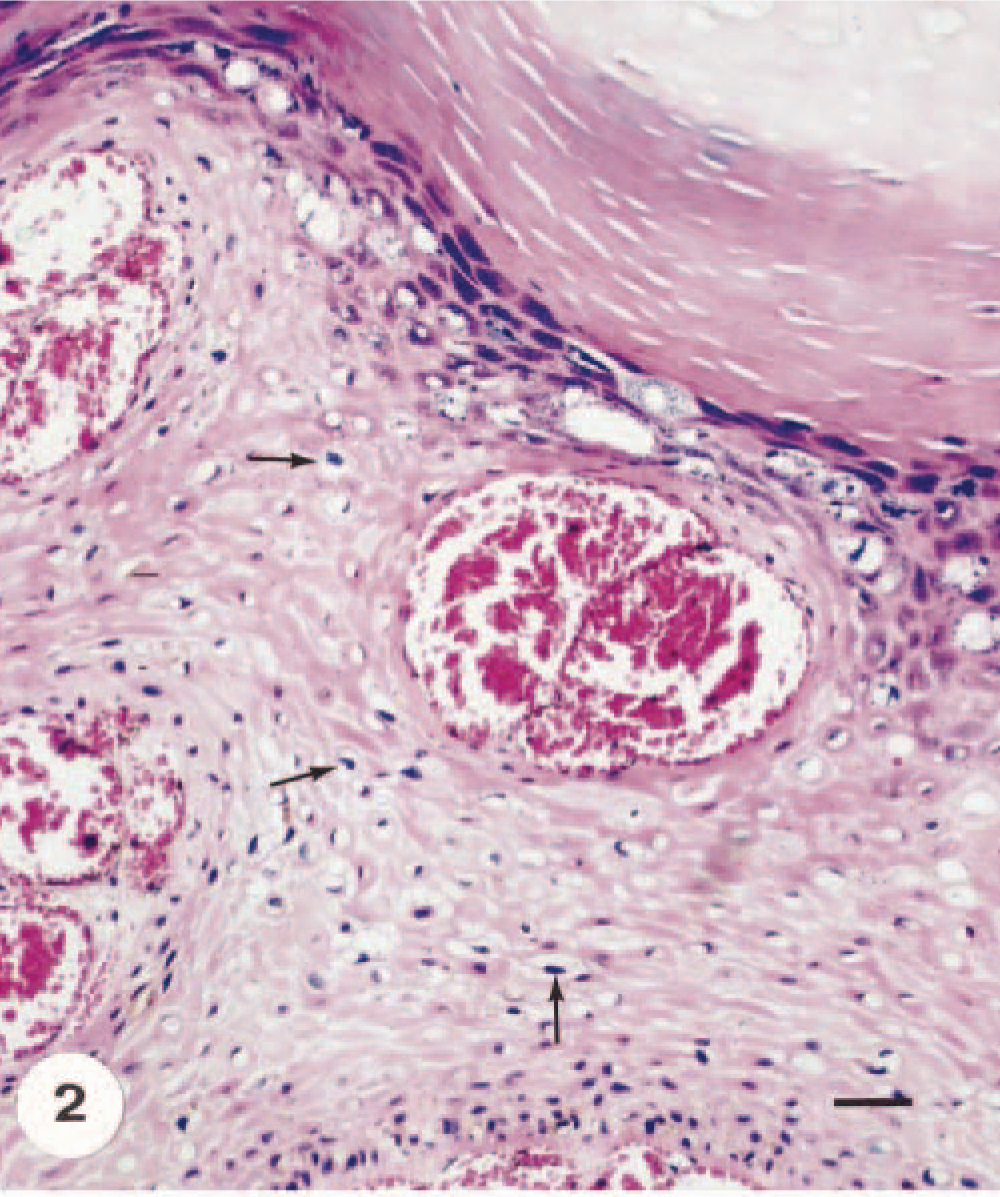
Angiokeratomatous papilloma; calf. Marked orthokeratotic hyperkeratosis, many koilocytes (arrows), and subepidermal clusters of thin-walled and dilated vascular spaces. HE. Bar = 80 µm.
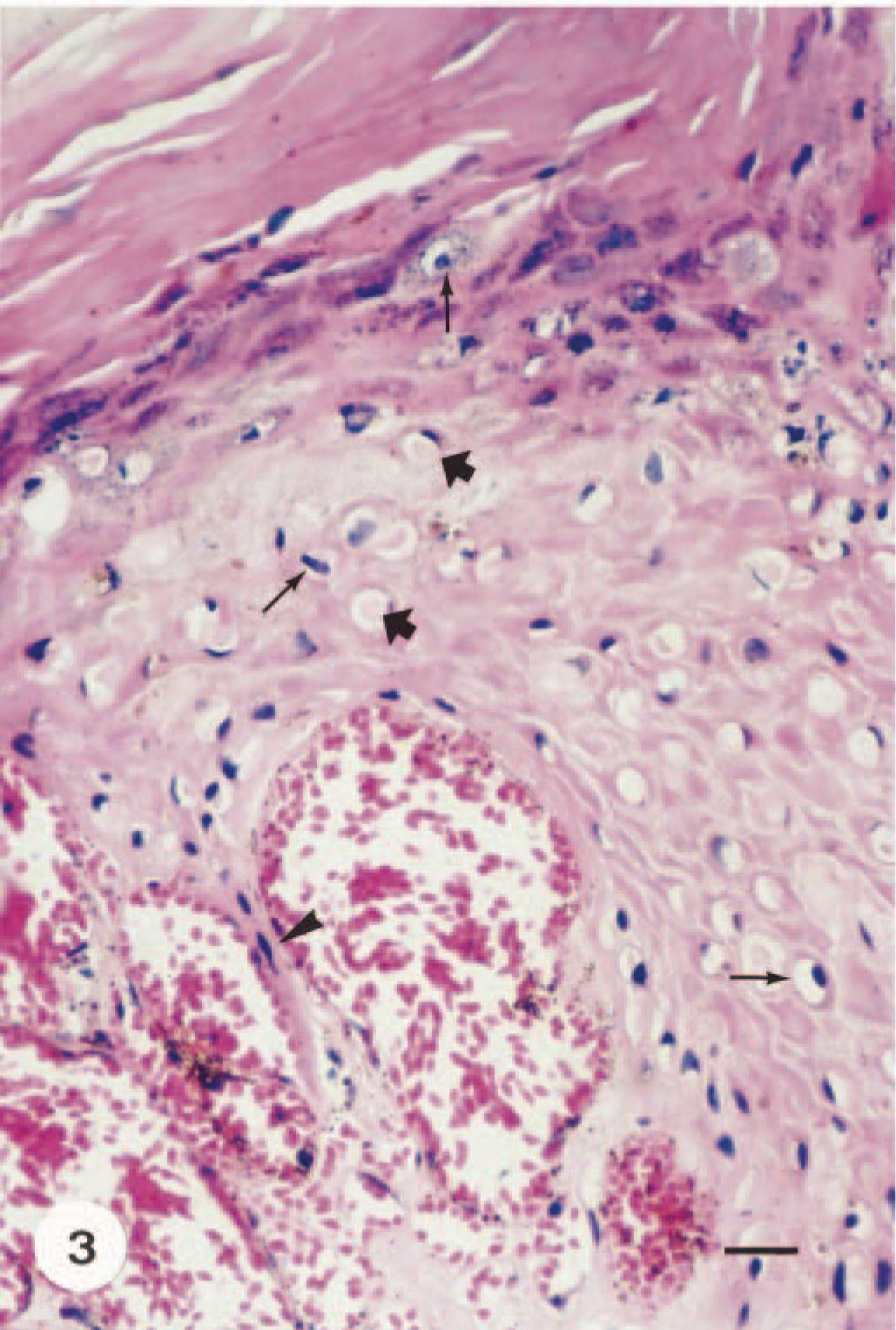
Angiokeratomatous papilloma; calf. Typical cytopathic effects of PVs in the epidermis with dilated vascular spaces in the superficial dermis. Koilocyte atypia (thin arrows), keratohyalin granules, and some intracytoplasmic inclusions (thick arrows). Note elongation of a rete ridge between dilated vessels (arrowhead). HE. Bar = 40 µm.
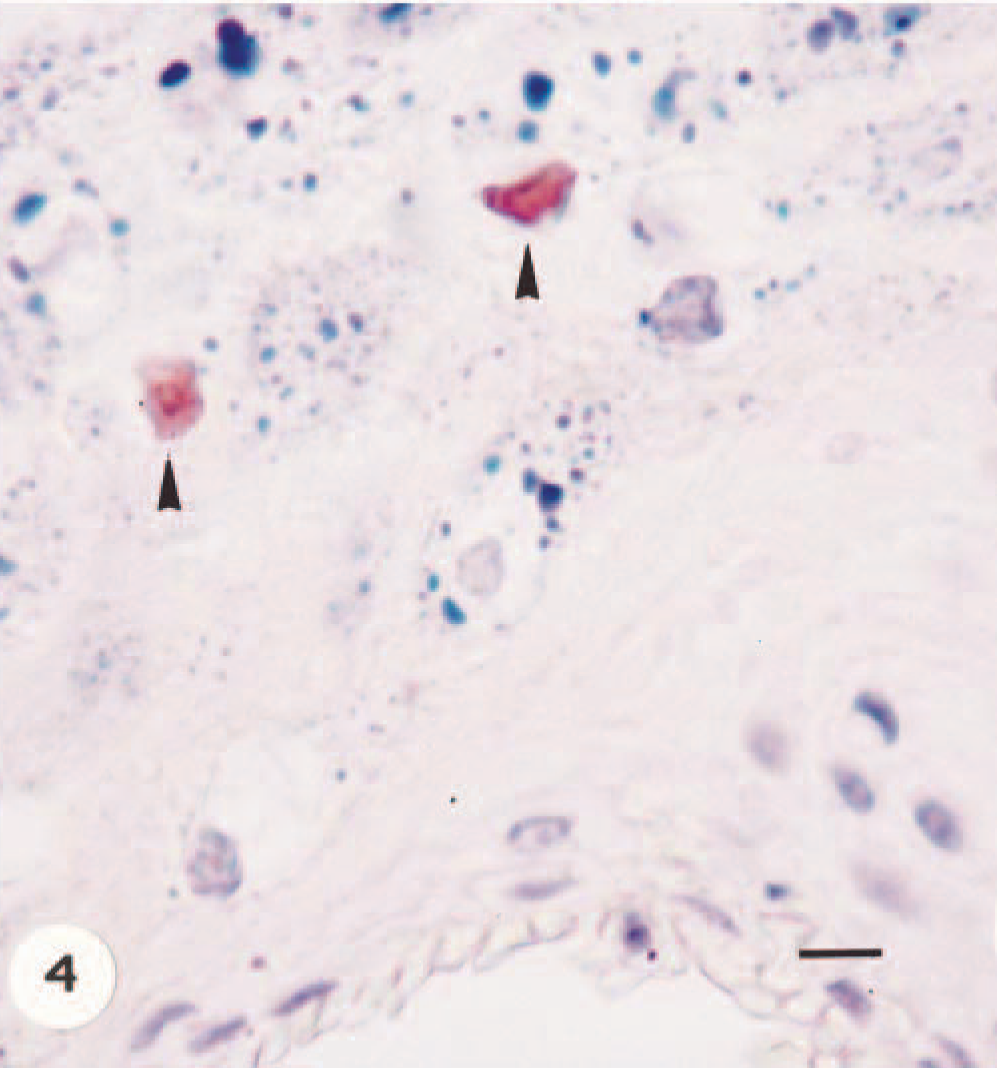
Angiokeratomatous papilloma; calf. Positive nuclear immunostaining (arrowheads) in koilocytes for PV genus-specific structural antigen. ABC method, Mayer's hematoxylin counterstain. Bar = 20 µm.
Bovine cutaneous angiomatosis has been compared to human pyogenic granuloma, hamartoma, or hemangioma 3,8,16 but is distinct from angiokeratoma. Human pyogenic granuloma is usually secondary to chronic localized infection and is an exuberant overgrowth of granulation tissue. 5 In the present case, there was localized inflammation below ulcerated epidermis, but granulation tissue or inflammatory infiltrations were not observed between the dilated vascular channels.
It is difficult to differentiate hamartomas and hemangiomas because of the considerable overlap in the use of the terms. Vessels in hamartomas are structurally normal, with all supporting elements, so that the proliferation is truly of blood vessels rather than just of endothelium. Vascular hamartomas are malformations, imply presence of the lesion at birth, and consist of localized dermal excesses of primitive to well-formed blood vessels, with no apparent site predilection. 16 Considering the age at which this lesion was first observed and the presence of overlying papillomatous changes associated with PV, the present case was not regarded as a hamartoma.
Hemangiomas are benign neoplasms of vascular endothelial cells. 8,16 Unequivocal differentiation is difficult between hemangiomas and other vascular lesions in animals. The critical feature for their diagnosis is proliferation of endothelial cells that form channels unaccompanied by the muscle or pericytes that support normal vessels of similar caliber. 16 The present case was not a hemangioma because the mass included pericytes that had a regular relationship with the dilated vascular channels and the cells were negative for FVIII. FVIII is a universally accepted marker for endothelial cells and is expressed in bovine endothelial cells; however, it may be undetectable even in some normal vessels. 6 Pericytes are associated with early capillary proliferation and express ASMA; and desmin staining may be useful to differentiate positive smooth muscle cells and negative pericytes. 4 In this case, however, desmin staining was negative, and only perivascular orientation of ASMA-positive cells and reticulin was evident.
A lesion with histologic appearance closely resembling that of angiokeratoma is verrucous hemangioma, recognized in humans 9 and in horses. 16 In cattle, no such lesion has been described. The term verrucous hemangioma has been applied to hyperkeratotic vascular malformations with superficial and deep involvement of the dermis. This designation is a misnomer; they are neither hemangiomas nor neoplasms but malformations. 9 For this reason, the present case was not considered a verrucous hemangioma because the cause was presumed to be PV.
Most of the lesions diagnosed as angiokeratoma in humans are not authentic vascular neoplasms but acquired vascular lesions that result from ectatic dilatation of preexisting blood vessels of the papillary dermis, accompanied by hyperkeratotic epidermis. 9 Subepidermal clusters of dilated thin-walled blood vessels, hyperkeratosis, and acanthotic epidermis with elongation of rete ridges are typical features of angiokeratomas. Our findings indicate morphologic features similar to those in angiokeratoma. Central to the pathogenesis is capillary ectasia within the papillary dermis, just beneath the basal membrane; epidermal acanthosis and hyperkeratosis seem to be reactive phenomena. It is not known why some ectasias elicit an epidermal reaction. Most human cases reported have a history of injury, trauma, or chronic irritation to the wall of a venule in the papillary dermis. 9 In this report, although there was no evidence of PV antigen in vascular components, a possible explanation for occurrence of this lesion was the repeated injury from PV infections of the epidermis.
Positive PVGA immunostaining and the presence of koilocyte atypia have been considered to be hallmarks of virus-induced papillomas. 5,11,12 In papillomas, true PV inclusions are seldom seen in paraffin-embedded sections of natural disease and they are intranuclear. 12,16 In the present case, intranuclear inclusions were not seen, and condensed cytoplasmic inclusions were negative for PVGA. At the ultrastructural level, these cytoplasmic inclusions in PV-induced lesions are considered aberrant intermediate filament assembly. 12 However, the association of the epidermal lesions with ectatic dermal vascular channels was consistent with a diagnosis of angiokeratoma as described in humans 9 and dogs. 16
Classification of vascular lesions remains confusing for lack of accepted terminology in veterinary literature. 16 In the present case, the unusual features of bovine papilloma in association with dilated dermal vascular channels warranted a diagnosis of angiokeratoma.
