Abstract
Subclinical gastritis was observed in 10 of 10 baboons (Papio spp.) from a toxicity study in a research facility. The lesions were similar in xenobiotic-treated and control animals, suggesting a spontaneous rather than chemical-induced disease. Histologic examination revealed lymphoplasmacytic gastritis in the antral mucosa. The fundic mucosa contained minor, scattered aggregates of lymphocytes and plasma cells. A Warthin-Starry silver stain and ultrastructural examination revealed numerous spiral-shaped bacteria morphologically resembling Helicobacter pylori in antral glands and numerous spiral-shaped bacteria morphologically consistent with H. heilmannii-like organisms in fundic glands. Polymerase chain reaction assay of paraffin-embedded antral and fundic tissue using primers for the urease gene and 16S ribosomal ribonucleic acid gene amplified deoxyribonucleic acid fragments with a high degree of sequence homology for H. pylori and H. heilmannii. This is the first report of gastritis associated with Helicobacter-like organisms in baboons.
Spiral-shaped bacteria of the genus Helicobacter have been associated with subclinical and clinical gastritis in humans and animals through both natural and experimental infection. 9,12,16 Persistent infection also is considered to be a risk factor in the development of gastric lymphoma and gastric adenocarcinoma in humans and ferrets. Two main groups of gastric Helicobacter species can be distinguished morphologically on silver-stained histologic sections and ultrastructurally. 9,12,16 The first group includes species similar to H. pylori, which are 2–5 µm long, with a comma, loose spiral, or gull-wing shape. The second group includes larger Helicobacter species, such as H. heilmannii–like organisms (HHLOs), which are 5–10 µm long, with a very tightly coiled spiral shape. H. pylori and HHLOs are the Helicobacter species most commonly identified in natural infections of the gastric mucosa of humans and nonhuman primates, including rhesus monkeys and other macaques, and dual infections with these organisms have been reported. 1,6,7,9,12,15,16 There are a few reports of natural infection of baboons (Papio spp.) with Helicobacter-like organisms that are not associated with gastritis. 4,5,10,14 This article describes subclinical gastritis associated with Helicobacter-like organisms in baboons from a toxicology study conducted at a research facility.
Adult male (n = 5) and female (n = 5) sacred baboons (P. hamadryas) were used in a safety-testing study of a pharmaceutical substance conducted during a 21-day period. These animals were from a colony of sacred baboons, all of which had been born in captivity either in Australia or in England. There had been no contact between any of the animals in this colony and other nonhuman primates for at least 5 years. Before the study, the animals were housed in groups in an indoor-outdoor complex that used a network of overhead races for ease of access and movement and to minimize handling by humans. 11 Animals were routinely subjected to a series of evaluations that included physical examinations, fecal examinations, clinical chemistry–hematology assessments, and tuberculin skin tests to monitor health status. During the study the animals were kept indoors in individual cages, and 4 people regularly worked with them. There was no contact with other nonhuman primates. Animals for this study included both controls and animals receiving a proprietary substance that revealed no drug-related clinical evidence of gastric disease or other disease before or during the study period. At study termination, animals were euthanatized. No gross lesions were reported at necropsy.
Samples of stomach (antral region from all 10 animals and fundic region from the five male animals) and other organs were removed from the carcass, fixed in 10% neutral buffered formalin, and submitted for histologic examination. Tissues were trimmed, processed, sectioned, and stained with hematoxylin and eosin (HE). Sections of the gastric fundus and antrum were treated with Warthin-Starry silver, periodic acid–Schiff, and Gram staining techniques. For the ultrastructural study, small pieces of the tissue were taken from the paraffin block from one animal, postfixed in osmium tetroxide, embedded in Spurr's resin, sectioned, stained with uranyl acetate and lead citrate, and examined using a Philips CM-10 transmission electron microscope.
To characterize the bacteria that were observed using light and electron microscopy, deoxyribonucleic acid (DNA) was extracted from paraffin-embedded antral tissue from one animal and fundic tissue from another animal by using the Puragene DNA Extraction Kit (Gentra Systems, Minneapolis, MN). Polymerase chain reaction (PCR) was then performed using previously described protocols to amplify specific regions of the 16S ribosomal ribonucleic acid (rRNA) gene for the genus Helicobacter. 8,17 Primers H676R and H276F were used to amplify a 400–base pair (bp) fragment, 17 and primers CO5 and H276F were used to amplify a 1,200-bp fragment. 8 To confirm the results of the 16S rRNA data, a nested PCR was performed by using primers that amplified a specific region of the urease gene of H. pylori. The outer primers U1735R (reverse 5′-CTTCGTGRATTTTAARRCCAAT-3′) and U430F (forward 5′-GCKGAWTTGATGCAAGAAGG-3′) were developed based on consensus regions of the urease gene of a number of gastric Helicobacter species, and they amplified a 1,224-bp fragment of the ureAB complex. After an initial denaturing step at 94 C for 3 minutes, amplification was performed for 35 cycles at 94 C for 10 seconds, at 52 C for 30 seconds, and at 72 C for 1.5 minutes, followed by an extension at 72 C for 5 minutes. The first-round product was then subjected to a second round of amplification based on a previously described protocol, 2 using primers HPU1 and HPU2 that amplified a 411-bp fragment of the ureA gene of H. pylori, with the annealing temperature adjusted to 47 C. After the PCR products were observed on a 1.5% agarose gel using ethidium bromide, they were purified for sequencing using the Wizard PCR Preps DNA Purification System (Promega, Madison, WI). Sequencing was performed using the ABI Prism Big Dye Terminator Cycle Sequencing Kit (Perkin–Elmer Applied Biosystems, Foster City, CA) according to the manufacturer's instructions. Sequencing products were separated on an ABI 377 DNA Sequencer (Perkin–Elmer Applied Biosystems), and sequences were assembled using Inherit software (Perkin–Elmer Applied Biosystems) to generate a consensus sequence. The consensus sequences were analyzed by using the BLASTN search from the National Center for Biotechnology Information (NCBI) GenBank database.
Histologically, there were no lesions of significance associated with the drug. Examination of the gastric mucosa revealed similar changes in all xenobiotic-treated and control monkeys, suggesting a spontaneous rather than chemical-induced disease. In the antral mucosa of 10 of 10 animals, there was lymphoplasmacytic gastritis characterized by marked, widespread infiltration of lymphocytes and numerous plasma cells into the lamina propria, with separation of gastric glands and prominent lymphoid follicles (Fig. 1). The inflammation was most pronounced in the superficial mucosa. Additional changes included gastric glandular epithelial hyperplasia, patchy necrosis and attenuation of glandular epithelium, and infrequent minor neutrophilic infiltrates in the superficial lamina propria and within the lumina of gastric glands. In the antral submucosa, tunica muscularis, or subserosa of most animals, there was minor multifocal accumulation of small lymphocytes with infrequent submucosal lymphoid follicles.
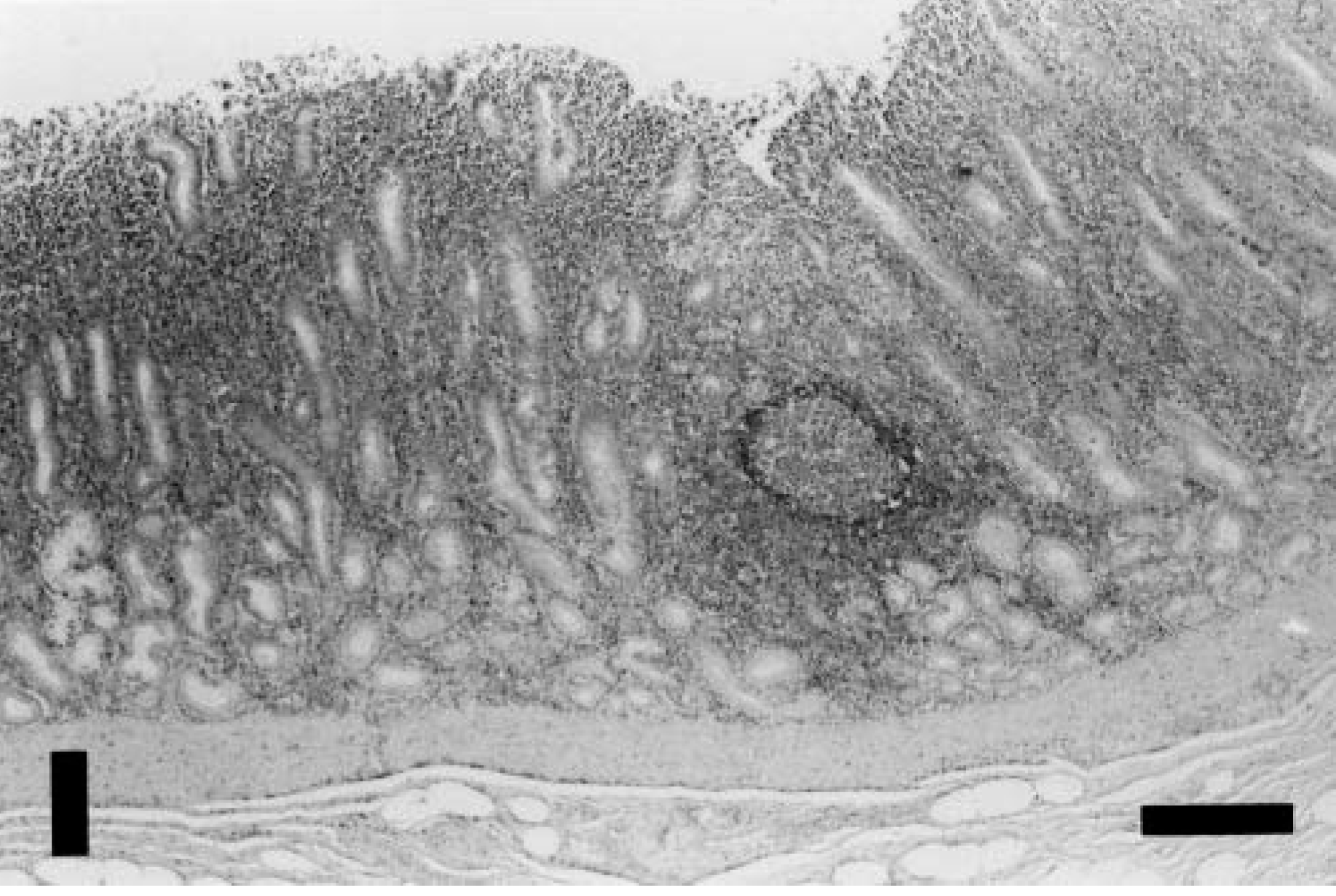
Gastric antral mucosa; baboon. Note marked lymphoplasmacytic inflammation that extends to the muscularis mucosae and lymphoid follicle. HE. Bar = 300 µm.
The fundic mucosa of all 5 animals examined contained relatively minor scattered aggregates of lymphocytes, with low numbers of plasma cells and occasional lymphoid follicles, which were most evident in the transition zone between the fundic and the antral mucosae. Glandular epithelial changes were not evident. In the fundic submucosa of two animals, there was minor multifocal accumulation of small lymphocytes.
The duodenal lamina propria of 10 of 10 animals contained a prominent plasmacytic infiltrate with low numbers of small lymphocytes, accompanied by occasional mucosal lymphoid follicles. The cellularity of the lamina propria of the jejunum, ileum, cecum, colon, and rectum was interpreted to be within normal limits.
Warthin-Starry–stained sections of the antral mucosa of all animals examined revealed numerous bacteria that were generally ∼ 2–5 µm long and had primarily loose spiral and “gull-wing” shapes (Fig. 2). These bacteria were located mainly in the gastric pits and the upper portions of gastric glands, where they were often in intimate association with the epithelial cell surface. Warthin-Starry–stained sections of the fundic mucosa revealed numerous tightly coiled helical bacteria that were generally ∼ 7–10 µm long (Fig. 3). These bacteria were located within the gland lumens, sometimes attached to the epithelial cell surface, and within the cytoplasm of parietal cells.
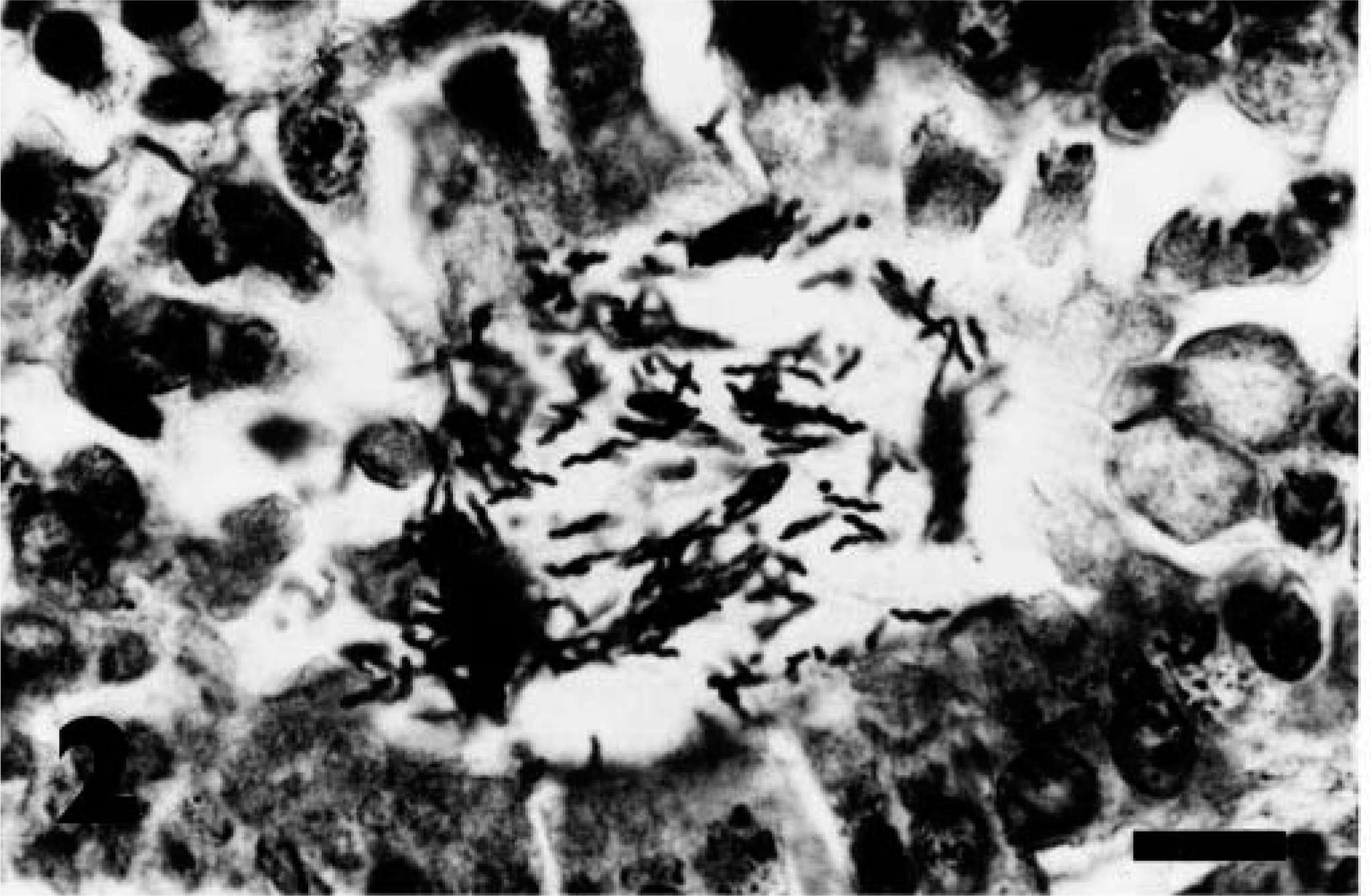
Antral mucosa; baboon. Numerous H. pylori–like organisms are located in the lumen of a gland. Note comma, loose spiral, or gull-wing shape and close association of some bacteria with epithelial surface. Warthin-Starry. Bar = 12 µm.
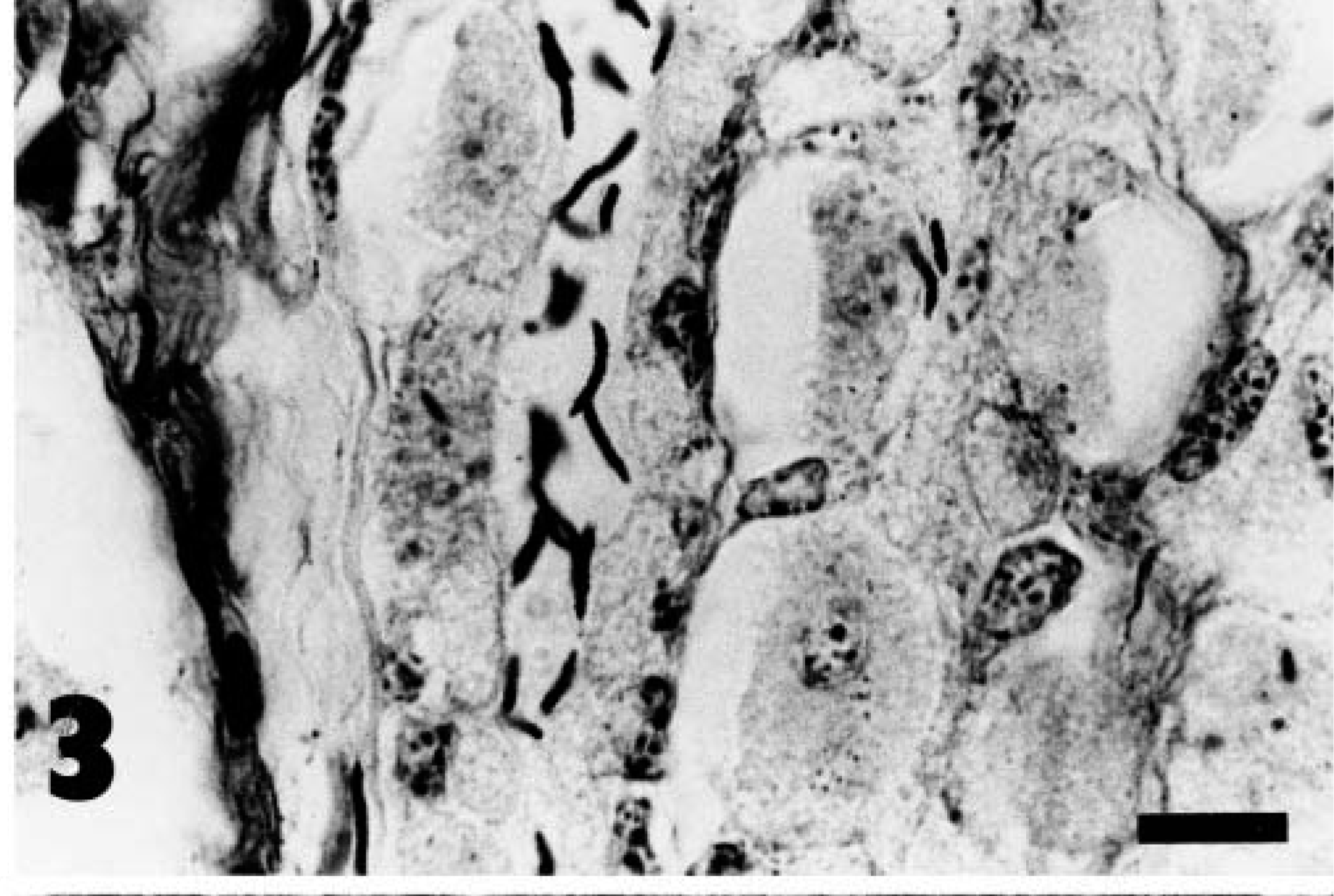
Fundic mucosa; baboon. Numerous H. heilmannii–like organisms are located in the lumen of a gland. Warthin-Starry. Bar = 12 µm.
Ultrastructural examination revealed that the organisms colonizing the antral mucosa were ∼ 0.4–0.5 µm wide (Fig. 4). The larger, tightly coiled organisms colonizing the fundic mucosa were similarly 0.4–0.5 µm wide, demonstrated one complete turn of their spiral per ∼ 0.6-µm length, and lacked periplasmic fibrils (Fig. 5). In these larger organisms, the outer membrane-like component of the cell wall did not closely follow the undulations of the body, creating the cigar-shaped profile similar to that reported previously in Helicobacter-like organisms in baboons. 4,5
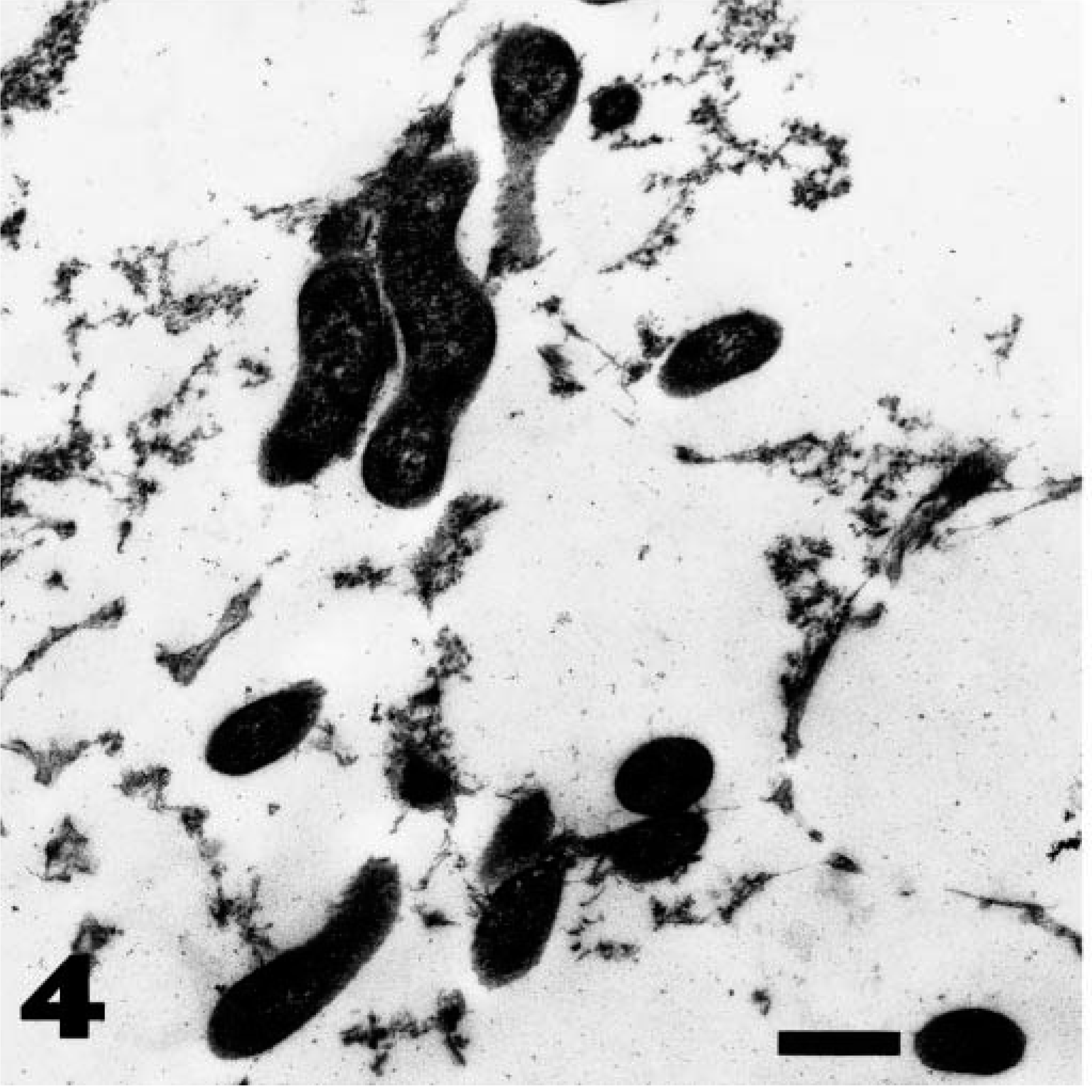
Transmission electron micrograph. Antral gland; baboon. H. pylori–like organisms within a disrupted gland lumen. Uranyl acetate and lead citrate. Bar = 0.64 µm.
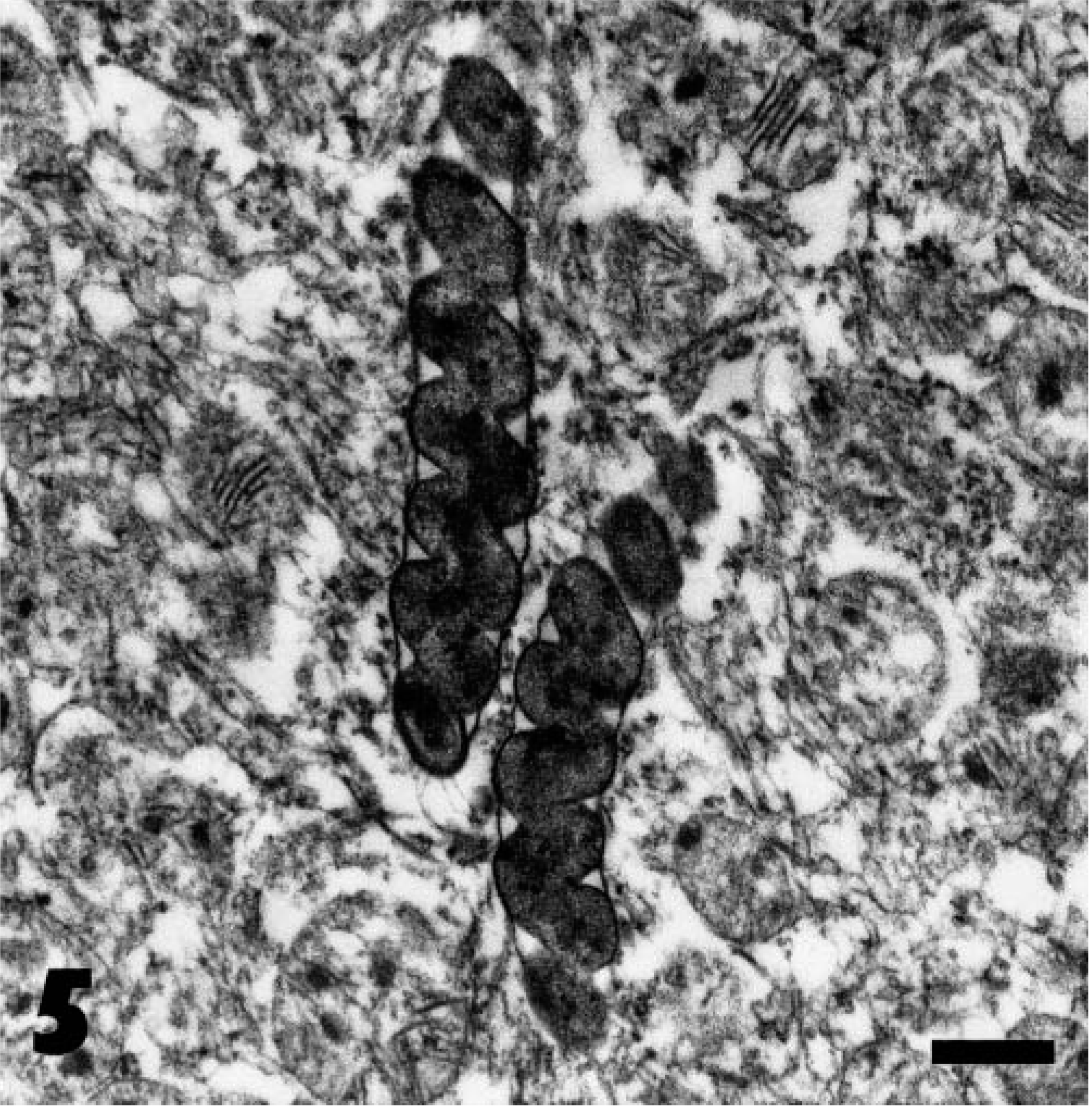
Transmission electron micrograph. Fundic gland; baboon. H. heilmannii–like organisms apparently within the cytoplasm of a parietal cell. Note tightly coiled shape, lack of periplasmic fibrils, and cigar-shaped profile. Uranyl acetate and lead citrate. Bar = 0.64 µm.
PCR assay of antral tissue amplified a 411-bp fragment of the ureA gene (nested PCR) and a 400-bp fragment of the 16S rRNA gene that demonstrated 99–100% and 100% similarity, respectively, to H. pylori. PCR assay of fundic tissue amplified a 400-bp fragment of the 16S rRNA gene, which demonstrated 98–99% similarity to H. heilmannii, and a 1,200-bp fragment of the 16S rRNA gene. The poor quality of the DNA recovered from the paraffin-embedded tissue restricted sequence analysis of this fragment to an 800-bp region that demonstrated 98–99% similarity to H. heilmannii.
Based on their size, morphology, location in tissue section, and partial sequences of their urease and 16S rRNA genes, the smaller argyrophilic bacteria colonizing the antral mucosa were most likely H. pylori. 1,9,16 The larger argyrophilic bacteria colonizing the fundic mucosa were best described as HHLOs. 9,12,16 Definitive identification would require isolation of the organisms and sequence analysis of 16S rRNA genes. Culture of the organisms at the time of necropsy was not attempted.
A survey of the literature has found no previous reports of gastritis associated with Helicobacter-like organisms in baboons. Previous reports of spiral bacteria in the baboon stomach have described both H. pylori–like organisms and tightly spiraled bacteria, which, based on their morphology, were most likely HHLOs. 4,5 H. pylori–like organisms have been isolated from the stomachs of baboons, 4,5,14 and the results of PCR amplification and partial 16S rRNA gene sequence analysis strongly support the contention that these organisms are H. pylori. 10
The close spatial association between the H. pylori–like organisms and the antral gastritis observed in the present study strongly suggests a causative role. Previous studies support a pathogenic role for H. pylori in naturally and experimentally infected humans, rhesus monkeys, and cynomolgus monkeys with histologic antral lesions similar to those reported in this study. 1,6,7,9,16 HHLOs, on the other hand, appear to be of relatively low pathogenicity in nonhuman primates, and the findings in the present study would support this contention. 6,9,16 In humans, HHLOs have been infrequently associated with mild gastritis, which is usually less severe than that associated with H. pylori. 9,12
Variation in gastric pathology may be influenced by the infecting Helicobacter species and strain. There is considerable genetic heterogeneity among H. pylori isolates from humans and rhesus monkeys. 3,7,9,13,16,18 Proposed pathogenic factors associated with H. pylori infection include bacterial production of adhesins, vacuolating cytotoxin, urease, protease, and phospholipase, inhibition of gastric acid secretion, and stimulation of cytokine production in the gastric mucosa. 3,9,13,16,18 Other factors that could influence gastric pathology include the number of organisms present, the host species and genotype, environmental factors such as diet, and concurrent infection with undetected organisms. 3,7,9,13,16,18
The source of infection of these monkeys was not established, though Helicobacter-like organisms may be naturally widespread in baboons. 4,5 Because most Helicobacter species do not have strict host specificity, humans, other nonhuman primates, or other mammalian species also are possible sources of infection. 7,16 There had been no contact between the animals in the present study and other nonhuman primates for at least 5 years.
These findings indicate that infection with H. pylori–like organisms has the potential to interfere with the interpretation of experimental and toxicity studies using baboons because subclinical disease could confound the interpretation of potential gastric toxicity. These findings also emphasize the pathogenicity of H. pylori relative to HHLOs and provide another nonhuman primate model in which to study Helicobacter species. 6,9 Further studies are required to determine how widespread infection with helicobacters is in baboons, to confirm that these bacteria are able to reproduce the gastric lesions and fulfill Koch's postulates, and to identify factors that may predispose one to subclinical or clinical disease.
Footnotes
Acknowledgements
We thank Dr. I. Meyer-Carrive and Ms. F. Brook of ICP Firefly, Dr. A. Birrell and Dr. S. Thompson of Royal Prince Alfred Hospital, and Mr. H. Prior of Queensland Department of Primary Industries for assistance with this study. We also thank Ms. J. Coombs and Ms. E. Baggs for excellent technical assistance.
