Abstract
Five young adult pet marmosets (Callithrix spp.) were presented with weight loss (5/5); fecal retention (3/5); diarrhea (2/5); impaired locomotion (3/5); anemia (4/4); hypoproteinemia or hypoalbuminemia (3/4); elevations of creatine phosphokinase, lactic dehydrogenase, and alanine aminotransferase (3/4); and renal failure with hypercholesterolemia (2/4). All anemic marmosets had low serum vitamin E levels. The anemia responded to vitamin E and selenium therapy in two marmosets. One of the five marmosets died before presentation, and two others died despite therapy. The two marmosets necropsied had degenerative myopathy, pyogranulomatous pansteatitis, and increased erythrophagocytosis and hemosiderosis. The striated muscle and adipose tissue of both marmosets were negative for coxsackievirus ribonucleic acid by in situ hybridization. These findings suggest that vitamin E deficiency may be involved in the development of anemia, myopathy, and steatitis in callitrichids; however, in some marmosets, underlying diseases such as chronic colitis may have influenced the development of anemia and impaired vitamin E status.
Keywords
Anemia, degenerative myopathy (DM), and steatitis have been described in laboratory-housed tamarins (Saguinus labiatus) and common marmosets (Callithrix jacchus) with variable vitamin E status and in association with wasting marmoset syndrome (WMS). 2,4 Coxsackieviruses were isolated from a few of the tamarins, but the viruses failed to transmit the disease. 2 Coxsackieviruses can cause DM and steatitis in mice, 7 and these lesions together are otherwise highly suggestive of vitamin E deficiency. Interestingly, both vitamin E and selenium deficiencies can induce the same six point mutations in the genome of coxsackieviruses isolated from humans with Keshan disease, a form of cardiomyopathy endemic in the selenium-deficient region in China, which occurs in selenium-deficient humans and can be prevented by supplementing selenium, and these mutations increase their pathogenicity in mouse experimental models. 14 Anemia and DM have been reported in another New World primate species, the owl monkey (Aotus trivirgatus), 3 and there are few descriptions of spontaneous DM, anemia, and cardiomyopathy in other vitamin E–deficient primates. 13,15 This article details clinical, hematologic, and pathologic findings and the response to treatment with vitamin E and selenium in pet common marmosets and a black tufted-eared marmoset (C. penicillata) with anemia, myopathy, and steatitis. The serum vitamin E status and coxsackievirus in situ hybridization (ISH) findings are reported for those marmosets for which serum and tissues are available.
Materials and Methods
Animals
All five callitrichids included in this study were young (1–2.5 years old), privately owned pet marmosets (Table 1); marmoset Nos. 2 and 3 were housed together. Their diet consisted mostly of fruits (marmoset Nos. 2–5) and cereal mixes (marmoset Nos. 2, 3, and 5); moderate (marmoset Nos. 2 and 3) or small amounts (marmoset No. 5) of insects; and, occasionally, callitrichid maintenance pellets (Zeigler Bros, Inc., Gardners, PA), eggs, yogurt, milk, and cheese (marmoset Nos. 2 and 3). The diet of marmoset No. 1 is not known.
Clinical findings in marmosets with anemia, myopathy, and/or pansteatitis. ∗
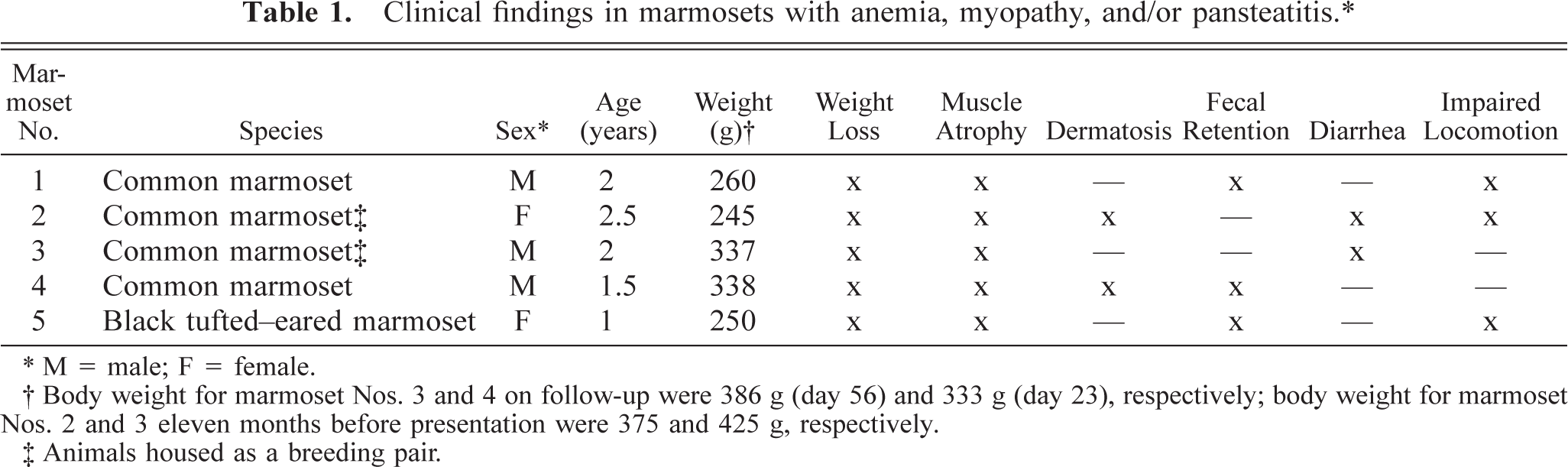
∗ M = male; F = female.
† Body weight for marmoset Nos. 3 and 4 on follow-up were 386 g (day 56) and 333 g (day 23), respectively; body weight for marmoset Nos. 2 and 3 eleven months before presentation were 375 and 425 g, respectively.
‡ Animals housed as a breeding pair.
Clinical pathology
Clinical pathology examination was performed in three common marmosets (marmoset Nos. 2–4) and the black tufted-eared marmoset (marmoset No. 5) and included complete blood counts (4/4), protein profile (4/4), and biochemistry (4/4; 17 or 18 parameters). Marmoset Nos. 2 and 3 had been tested similarly 11 months earlier when they suffered intermittent vomiting and glycosuria and apparent gestational diabetes in the female (marmoset No. 2). At that time, the animals did not show evidence of anemia or myopathy. Follow-up clinical pathology examination was performed twice in marmoset No. 3 and once in marmoset No. 4 to check for response to treatment with vitamin E and selenium. Animals were anesthetized with isofluorane (induction at 2–3% and maintenance at 0.5–1%). Blood was drawn from the femoral vein and placed in 1-ml lithium heparin tubes for hematology and in uncoated plastic tubes for serum. Serum was first frozen at −10 C and later at −80 C for variable periods of time. Reference values were obtained from the literature 16 and the International Species Information System (Apple Valley, MN); in-house values also were considered. Serum vitamin E levels were routinely determined in these four marmosets by high-pressure liquid chromatography at an accredited commercial laboratory. Serum from a black tufted-eared marmoset that died with bacterial fibrinopurulent pleuropneumonia and did not have DM or steatitis was used as a control for the vitamin E analysis.
Histopathology and ISH
Tissues from two of the three marmosets that died were preserved in 10% buffered formalin and processed routinely for histopathology. Marmoset No. 5 died within 24 hours of admission and was not submitted for necropsy. Stains included hematoxylin and eosin (HE), periodic acid–Schiff (PAS), Ziehl-Neelsen (ZN), Warthin-Starry (WS), and Perls' iron. ISH for coxsackievirus was performed on mesenteric adipose tissue (marmoset Nos. 1 and 2) and skeletal and cardiac muscle (marmoset No. 2) as described previously. 9 Skeletal and cardiac muscle from the aforementioned black tufted-eared marmoset was used as a control for vitamin E analysis.
Results
Clinical findings
Table 1 summarizes the clinical findings. All animals were presented with a history of weight loss and muscle atrophy. Only two of the five marmosets had the cutaneous component (hypotrichosis of the tail) of the so-called WMS; however, dermatosis was seen before presentation, although it was not temporally coincident with the diagnosis of anemia, DM, and vitamin E deficiency. Marmoset No. 1 was presented for necropsy with no previous clinical work. Fecal retention was marked in three marmosets (marmoset Nos. 1, 4, and 5) according to abdominal palpation and radiography. Watery diarrhea and vomiting were observed in marmoset Nos. 2 and 3; a rectal swab was negative for enteropathogenic bacteria, including Campylobacter, in marmoset No. 2. Impaired locomotion was due to hind limb paresis in marmoset No. 2. Cytologic screening for complicating oral infections revealed myriad chains of extracellular or intracellular cocci, within keratinocytes and neutrophils, in marmoset No. 5, whereas marmoset No. 2 had no evidence of fungal or bacterial stomatitis.
Clinical pathology findings
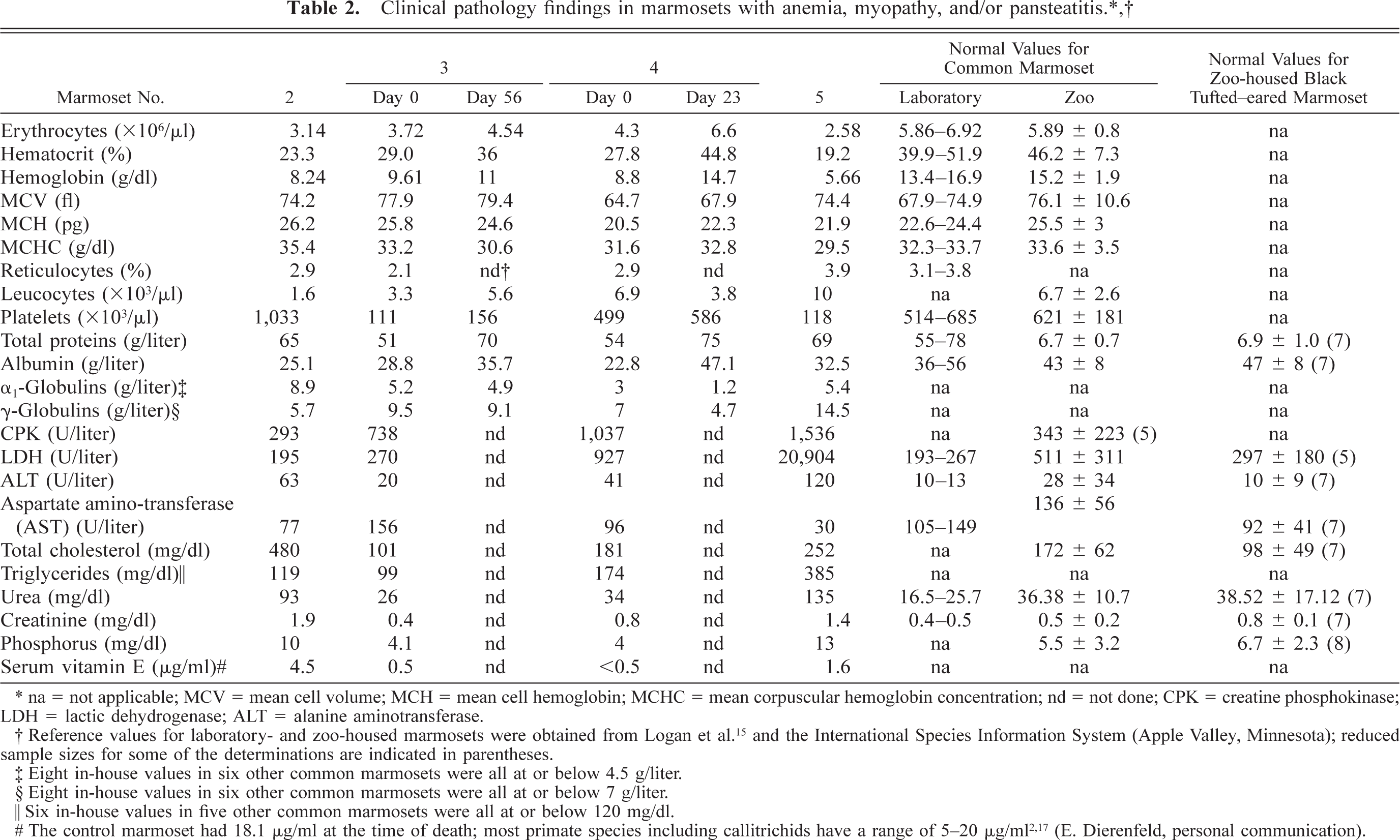
Table 2 details the most relevant clinical pathology findings, including serum vitamin E levels, in these marmosets. All the four marmosets had anemia, with less than 1% erythrocytes with Heinz bodies in the three marmosets tested (marmoset Nos. 2, 3, and 5). Two had leukopenia (marmoset Nos. 2 and 3), and marmoset No. 2 showed marked thrombocytosis. At the time of diagnosis of anemia, marmoset Nos. 3–5 had low serum vitamin E levels, and the level in marmoset No. 2 was slightly below the low normals reported for most primate species 2,18 (E. Dierenfeld, personal communication). When correlated with serum cholesterol, as reported for humans with hyperlipidemia, 17 marmoset No. 2 had values similar to that in the other two vitamin E–deficient marmosets. The control marmoset showed high normal serum vitamin E values. Marmoset Nos. 2–4 had hypoproteinemia or hypoalbuminemia or both. αI-Globulins were apparently elevated in marmoset Nos. 2 and 3, and marmoset Nos. 3 and 5 probably had hypergammaglobulinemia. There were mild elevations of creatine phosphokinase (CPK), lactic dehydrogenase (LDH), and alanine aminotransferase (ALT) in three marmosets (marmoset Nos. 2, 4, and 5); LDH was markedly elevated in marmoset No. 5. Two marmosets (marmoset Nos. 2 and 5) died with complicating renal failure characterized by azotemia, hyperphosphatemia, and a high calcium × phophorous product. Values for marmoset Nos. 2 and 5 were 94 and 102.7, respectively; the highest in-house value we have obtained in marmosets with normal calcium and phosphorous levels is 65.25. Marmoset No. 2 had polyuria, polydipsia, hematuria (250 red blood corpuscles/µl), proteinuria (30 mg/dl), and glycosuria (500 mg/dl). Urinalysis was performed a few hours before death using dry reagent dipsticks (Urispec 9-Way, Port Washington, NY) on reddish urine collected from the floor just after voiding. Marmoset Nos. 2 and 5 had hypercholesterolemia, and marmoset Nos. 4 and 5 also showed hypertriglyceridemia.
∗ na = not applicable; MCV = mean cell volume; MCH = mean cell hemoglobin; MCHC = mean corpuscular hemoglobin concentration; nd = not done; CPK = creatine phosphokinase; LDH = lactic dehydrogenase; ALT = alanine aminotransferase.
† Reference values for laboratory- and zoo-housed marmosets were obtained from Logan et al. 15 and the International Species Information System (Apple Valley, Minnesota); reduced sample sizes for some of the determinations are indicated in parentheses.
‡ Eight in-house values in six other common marmosets were all at or below 4.5 g/liter.
§ Eight in-house values in six other common marmosets were all at or below 7 g/liter.
∥ Six in-house values in five other common marmosets were all at or below 120 mg/dl.
# The control marmoset had 18.1 μg/ml at the time of death; most primate species including callitrichids have a range of 5–20 μg/ml2,17 (E. Dierenfeld, personal communication).
Treatment and follow-up
Marmoset No. 3 was treated with vitamin E (8 mg/kg i.m.) and selenium (0.06 mg/kg i.m.) (®Toco-Selenio, Calier, Barcelona, Spain) once a week for 6 weeks; diarrhea was treated with a protocol for chronic colitis (CC) in cotton-top tamarins (S. oedipus) 11 on the basis of the postmortem findings in its female cage mate. Three weeks later, anemia persisted (3.48 × 106 erythrocytes/µl; hemoglobin 9.84 g/dl; mean corpuscular volume, 80.5 fl), body weight and total proteins rose to 361 g and 60 g/liter, respectively, and γ-globulins were at 10.7 g/liter (17.83%). On day 56, there was considerable improvement of anemia (Table 2) in association with absence of diarrhea over the previous 2 weeks and increasing body weight (386 g), total proteins, and albumin. The owners refused further testing. Marmoset No. 5 was treated with vitamin E and selenium as described above for 4 weeks, a diet change to increase animal protein, animal fat, and lactobacilli. Anemia, hypoproteinemia, and hypoalbuminemia had resolved at the completion of treatment (Table 2), and feces returned to normal. At the time of writing this article, both marmosets were doing well.
Pathology and ISH
Gross findings consisted of generalized muscle atrophy (2/2), yellowish discoloration of internal fat (1/2), diffuse catarrhal enterocolitis, mesenteric lymphadenopathy, and whitish foci in the renal cortex (marmoset No. 2). Microscopically, the skeletal muscles of both marmosets had DM characterized by segmental necrosis, hyalinization, and loss of fiber striations; findings were particularly severe and disseminated in marmoset No. 2 (Figs. 1, 2). In both marmosets, DM affected the diaphragm. The mesenteric and perirenal adipose tissue of both animals had pyogranulomatous steatitis (Fig. 3), characterized by infiltrates of macrophages with an eosinophilic or brownish cytoplasm that stained positively with PAS and ZN, neutrophils, and fewer multinucleate giant cells, usually forming collarettes around adipocytes. Marmoset No. 1 also had increased fibrous tissue deposition intralesionally. Abundant PAS- or ZN-positive pigment was seen in the cytoplasm of esophageal skeletal fibers (Figs. 4, 5) and cardiacmyocytes (marmoset No. 2). Marmoset No. 2 also had severe CC, as commonly described in captive callitrichids, 11 and was negative for Helicobacter-like bacteria by WS. There was mesangioproliferative glomerulonephropathy, with prominent tubular reabsorption of proteins, and hyaline and giant-cell casts, with phagocytosis of hyaline casts. The pancreas had severe generalized islet hyperplasia, with insulin overexpression demonstrated by immunohistochemistry. 12 Marked hemosiderosis was seen in hepatocytes, phagocytic cells, and the reticuloendothelial system. There was also extramedullary hematopoiesis and increased erythrophagocytosis in lymph nodes; the latter was seen in marmoset No. 1 also. The duodenum had deep ulceration that was attributed to the stress of disease and hospitalization. Severe vacuolation and ballooning degeneration were seen in the superficial layer of the esophageal epithelium and were attributed to vomiting and DM. Both marmosets and the control were negative for coxsackievirus ribonucleic acid (RNA) by ISH.
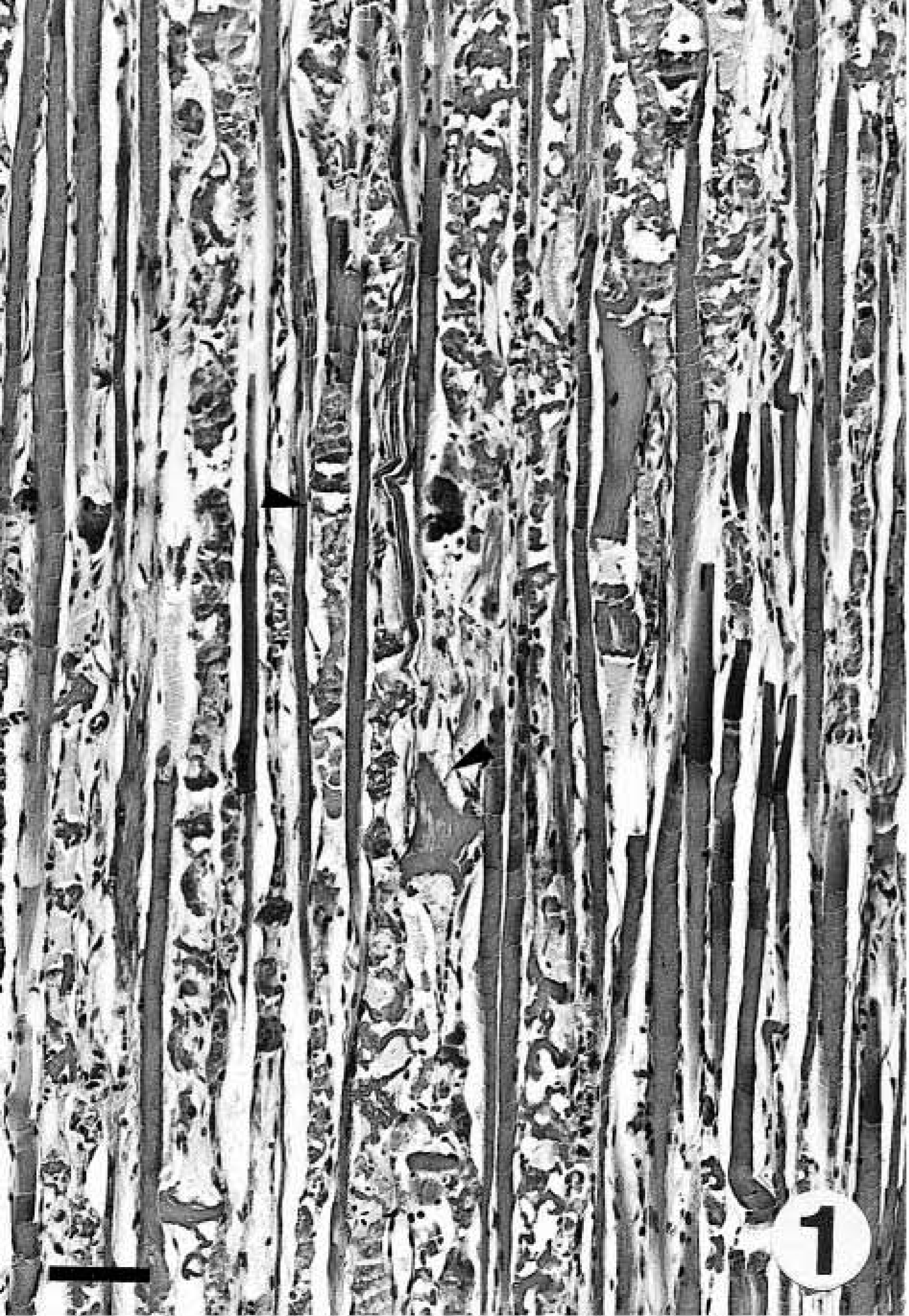
Skeletal muscle. Marmoset No. 2. Degenerative myopathy with hyalinization and necrosis of skeletal muscle fibers (arrowheads). HE. Bar = 79 µm.
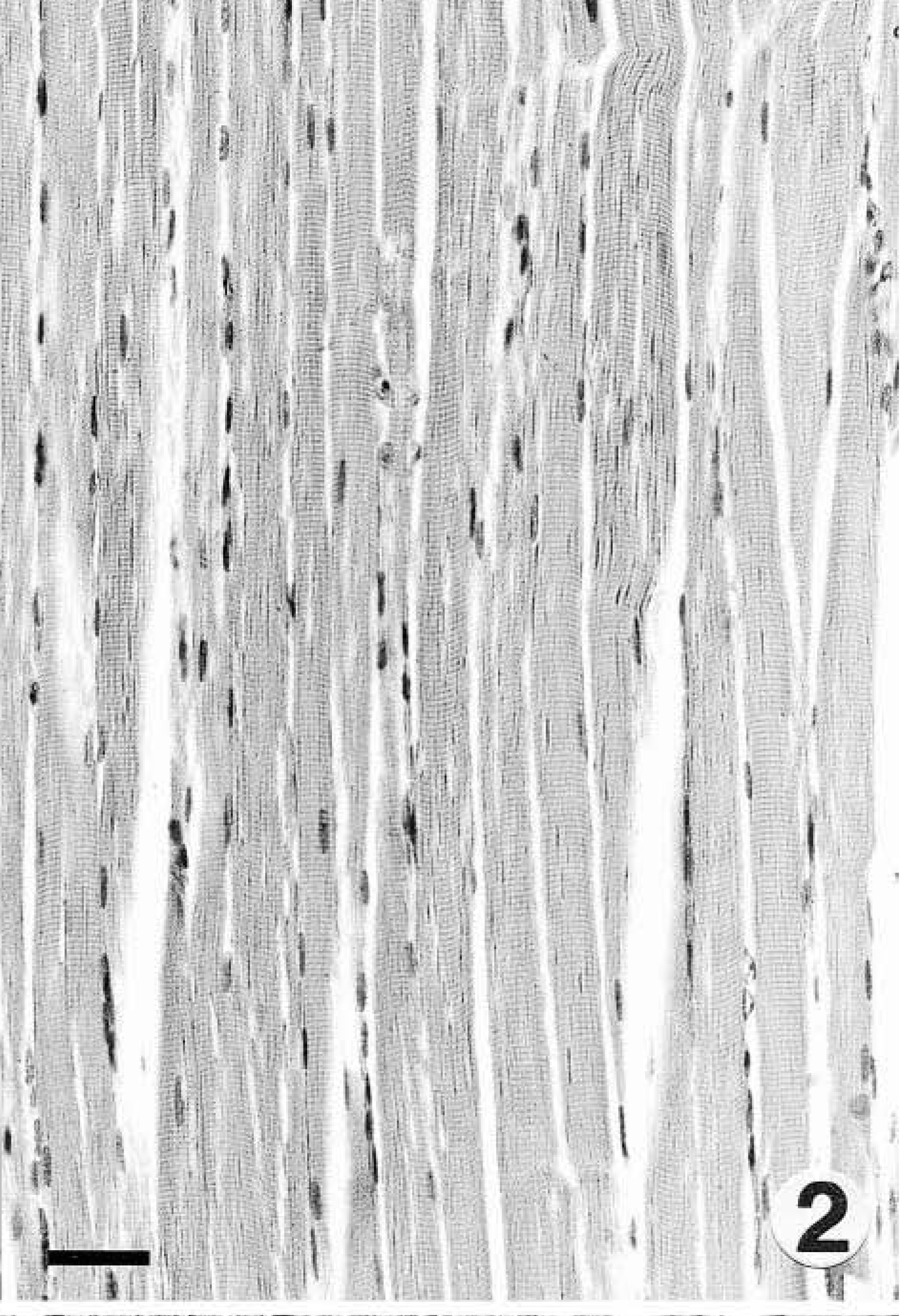
Control skeletal muscle; black-tufted–eared marmoset. HE. Bar = 39 µm.
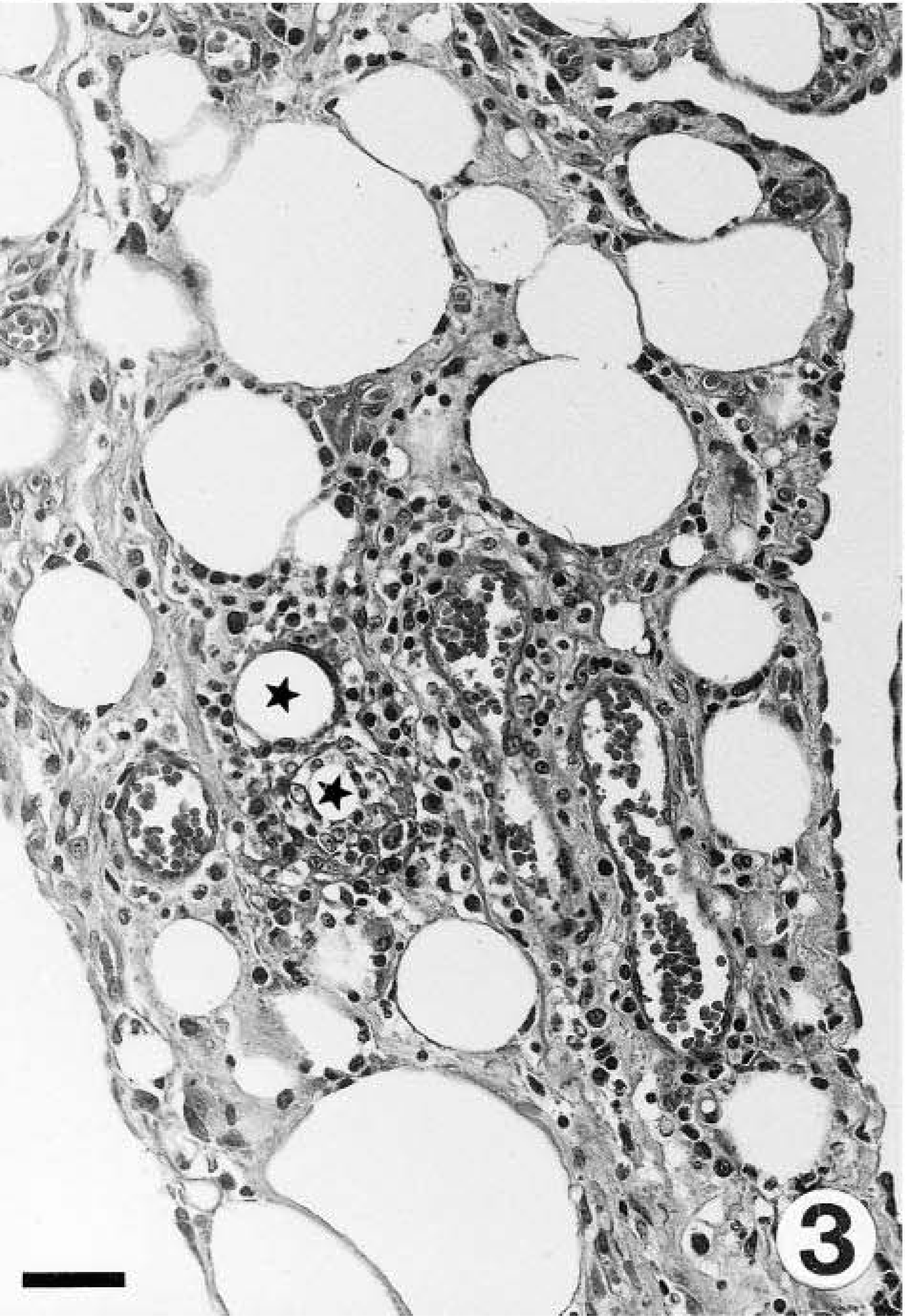
Mesenteric adipose tissue; marmoset No. 1. Steatitis with rings of inflammatory cells around degenerating adipocytes (stars) and increased interstitial fibrous tissue. HE. Bar = 39 µm.
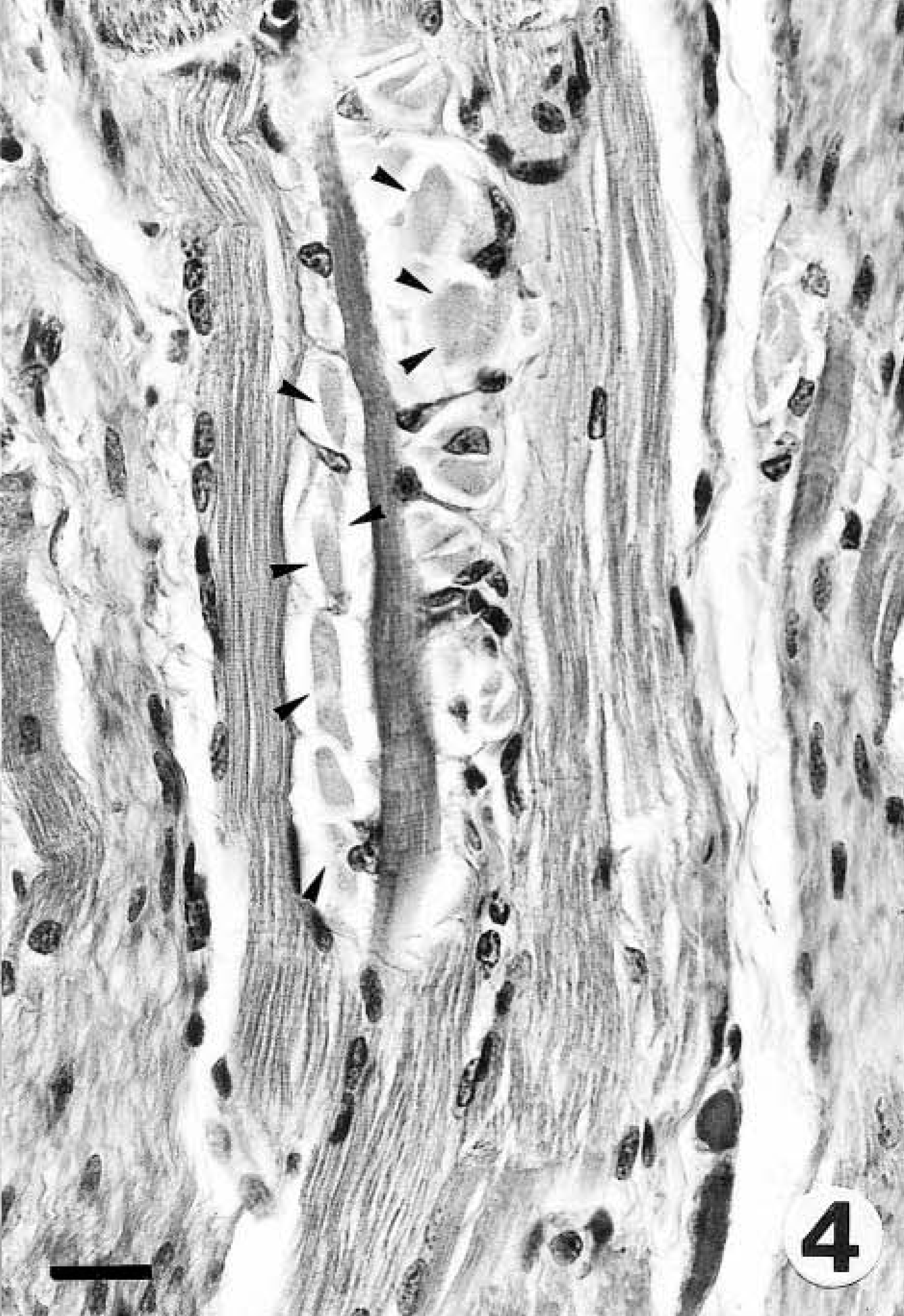
Esophagus; marmoset No. 2. Note the abundant intracytoplasmic basophilic material (arrowheads) in esophageal skeletal fibers. HE. Bar = 20 µm.
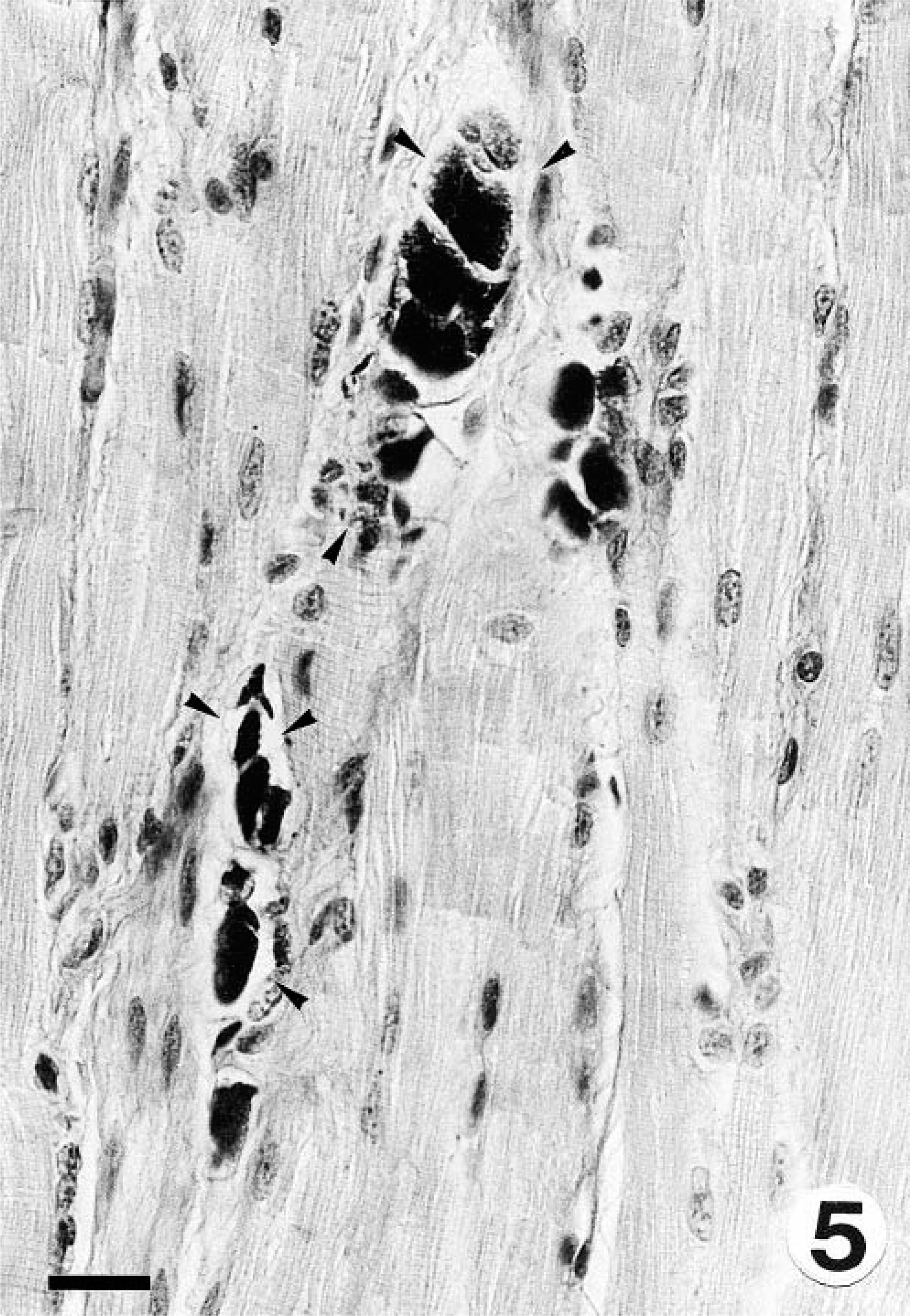
Esophagus; marmoset No. 2. Intracytoplasmic material shown in Fig. 4 stains positively with PAS (arrowheads). PAS. Bar = 20 µm.
Discussion
Anemia, DM, and steatitis with lipopigments in these marmosets were suggestive of vitamin E deficiency; marmoset Nos. 2–5 were vitamin E deficient. Additional support for this was provided by the presence of systemic lipopigment deposition, increased erythrophagocytosis, and marked hemosiderosis of the phagocytic and reticuloendothelial system in the necropsied marmosets and the response of anemia to treatment with vitamin E and selenium in marmoset Nos. 3 and 4. Tamarins previously reported with this condition had variable vitamin E status, 2 and a few common marmosets with hemolytic anemia and DM had vitamin E deficiency, 4 but the controls used had a mean serum vitamin E concentration slightly below the low normals for nonhuman primates.
Anemia could not be easily characterized because of the variability between the marmosets and in the reference values and the concurrent disease processes in some of the animals. In all marmosets, anemia was associated with vitamin E deficiency. Vitamin E deficiency may be a primary or secondary process in these marmosets. Some of the pathologic findings in marmoset Nos. 1 and 2 (increased erythrophagocytosis and hemosiderosis of the reticuloendothelial system) suggested the presence of an extravascular hemolytic component to the anemia. Chronic systemic disease with weight loss, CC, and renal insufficiency may have influenced anemia considerably in marmoset No. 2. Oxidative stress may play an important role in the pathogenesis of CC in humans, 1 and patients with inflammatory bowel disease may have low or normal vitamin E levels. 10 In a rat model of CC, antioxidant enzymes and vitamin E levels were decreased. 21 Therefore, vitamin E deficiency in this marmoset may have been, at least in part, secondary to CC. Marmoset No. 3 likely had CC on the basis of clinical signs, response of diarrhea to a treatment regimen previously used in cotton-top tamarins with CC, 11 and the diagnosis of CC in its female cage mate. Response of anemia to treatment with vitamin E and selenium in this marmoset was slow as compared with that of marmoset No. 4, and this may have been due to a more protracted course of disease that was complicated by other disease processes such as diarrhea. In the previous studies in tamarins and marmosets, vitamin E supplementation prevented the development of the disease, 2,4 but anemic tamarins did not respond to either oral or parenteral vitamin E and selenium. 2 However, our study involved a limited number of monkeys, and a common marmoset of unknown vitamin E status with WMS, hemolytic anemia, hind limb paresis, and myopathy did not respond to oral vitamin E.
In previous reports on tamarins and marmosets, WMS and pancreatic acinar atrophy were common concurrent findings 2,4 that may have influenced the vitamin E status of affected callitrichids. Owl monkeys with hemolytic anemia and DM had fat malabsorption of apparent intestinal origin. 18 Neither marmoset in this report had WMS at the time of diagnosis of anemia, DM, and steatitis, and marmoset Nos. 1 and 2 did not have pancreatic acinar atrophy. Pancreatic diseases and any other cause of fat malabsorption-maldigestion should be considered potential underlying factors for vitamin E deficiency in primates. 13
The reported association between coxsackieviruses and vitamin E and selenium deficiencies 14 and the isolation of these viruses from tamarins affected with anemia, steatites, and muscle necrosis 2 led us to test the muscular and adipose tissue lesions in marmoset Nos. 1 and 2 for the presence of coxsackievirus RNA by ISH. Although both marmosets were negative, preservation of RNA was not good in all tissues, and therefore infection with these viruses cannot be ruled out.
Some clinical pathology findings were interesting, especially the mild elevations of CPK, LDH, and aspartate amino-transferase (AST) in marmoset No. 2 despite severe DM demonstrated histologically a few hours later. Nutritional DM is usually episodic, and CPK is short-lived; thus DM cannot be ruled out on the basis of normal CPK. 20 Whenever DM is suspected, panels of CPK, LDH, ALT, and AST should be conducted for their higher sensitivity and specificity in the diagnosis of DM. 20 Callitrichids presenting with this syndrome should be evaluated for renal function. Hypercholesterolemia in marmoset Nos. 2 and 5 was attributed to renal insufficiency. Serum total cholesterol levels should be measured in callitrichids when presenting the clinical picture described to correct the vitamin E levels. The renal lesions in marmoset No. 2 may be related to CC because glomerulonephropathy is a rare complication of CC in humans, 5 probably because of the abnormal secretion of IgA by the chronically inflamed intestinal mucosa, with subsequent immune complex formation and deposition in the glomerular mesangium. Immune complexes from dietary antigens, such as gliadin from wheat, have been implicated in CC and glomerulonephropathy of callitrichids. 8,19 Primates with DM also can develop renal failure because of myoglobinemic nephrosis. 13 Thrombocytosis in marmoset No. 2 was considered reactive because of wasting and CC, 16 and the elevations of αI-globulins were attributed to an acute-phase response secondary to wasting and CC. 6 Hypoproteinemia and hypoalbuminemia in marmoset Nos. 2 and 3 were attributed to diarrhea and, in marmoset No. 4, to the inappropriate diet, which consisted of only fruits.
Further work is needed to better characterize the etiopathogenesis of this syndrome in callitrichids to determine the antioxidant status of affected animals, to test the association with underlying diseases, to explore the possibility of pancreatic and intestinal diseases causing malabsorption-maldigestion syndromes, and to determine the contribution of coxsackievirus infections.
Footnotes
Acknowledgements
We are grateful for assistance with histotechnology provided by Blanca Pérez and Pere Losada. This work is supported in part by Universitat Autònoma de Barcelona and Laboratorio de Diagnóstico General (Barcelona, Spain).