Abstract
Atrial natriuretic peptide (ANP) and brain natriuretic peptide (BNP) are cardiac hormones involved in electrolyte and fluid homeostasis. Our laboratory has investigated the use of ANP and BNP as diagnostic markers of cardiac disease in cats. We hypothesize that the cardiac distribution of ANP and BNP increases in cats with hypertrophic cardiomyopathy (HCM). Accordingly, we evaluated the immunohistochemical distribution of ANP and BNP in hearts of four cats with naturally occurring HCM relative to five healthy controls. Indirect immunoperoxidase was performed with polyclonal immunoglobulin G against feline ANP (1–28) and proBNP (43–56). In control cats, ANP and BNP immunoreactivity was restricted to the atria. Staining for both peptides was most intense adjacent to the endocardial surface. Auricles stained more diffusely than atria for both peptides. The interstitial capillaries and nerve fibers within the heart were positive only for BNP. Atrial immunoreactivity for ANP and BNP was more diffuse and had a less distinctly layered pattern in HCM than in control cats. Ventricular cardiomyocytes of HCM cats were negative for ANP but stained lightly and diffusely for BNP. The capillaries and nerve fibers remained positive for BNP. We conclude that in cats with HCM, the cardiac distribution of ANP and BNP is more diffuse in the atria and that novel expression of BNP in the ventricular cardiomyocytes occurs.
Introduction
Atrial natriuretic peptide (ANP) and brain natriuretic peptide (BNP) are hormones synthesized by cardiomyocytes released into the circulation as a result of cardiac wall stretching and play a role in electrolyte-fluid homeostasis by inducing diuresis, natriuresis, and peripheral vasodilation. 18 These members of the natriuretic peptide family, which also includes C-type natriuretic peptide, share similar amino acid sequences and a 17–amino acid ringlike structure resulting from the presence of an internal disulfide bond. 40 Because ANP and BNP messenger RNA (mRNA) are mainly produced by the heart tissue and correlate with increases in circulating peptide levels, 17,24,25,29 besides their hormonal function these peptides also have been indicated as potential markers for cardiac disease, such as congestive heart failure, 1,17,23 dilated cardiomyopathy, 37,41 and hypertrophic cardiomyopathy. 9,15,23,34
ANP in normal hearts of cats and several other species is reportedly restricted to the atria (including the auricles), with minimal or no presence in ventricular myocytes. 6,7,22,28,32 This peptide is normally stored in atrial myocardiocyte cytoplasmic granules, as observed in several species. 5–7,21,22,28,32 We have sequenced and shown previously that cardiac ANP gene expression in normal cats also is restricted to the atria. 3 We also have found that ANP gene expression in cats with naturally occurring hypertrophic cardiomyopathy (HCM) is maintained primarily in the atria but can be observed in variable amounts in the ventricles as well. 4 Similar findings were observed in dogs and humans, where variable levels of ANP were found in ventricles of hypertrophic hearts. 7,34
BNP has colocalization with ANP in the atrial cardiomyocyte granules, 27 but it is also detected in normal ventricular myocytes of mice and rats. 26,38 It is still controversial whether BNP is present in the ventricles of normal humans. 20 BNP gene expression is reportedly restricted to the atria of normal dogs and is also present in ventricles of dogs, rabbits, and humans with heart failure. 2,15,19
Although we have recently sequenced the feline BNP gene (Z. L. Liu, C. E. Wiedmeyer, D. D. Sisson, and P. F. Solter, Felis catus brain natriuretic peptide mRNA, GenBank Accession No. AF253495, 2001), the localization of its peptide product in hearts of either normal cats or cats with heart failure has not been done. Our laboratory investigated the potential use of natriuretic peptides as diagnostic tests of cardiovascular disease in cats. The purpose of this study was to determine whether a change in distribution of ANP and BNP occurs in the hearts of cats with naturally occurring HCM. We hypothesized that the histologic distribution of both hormones is more widespread within the hearts of cats with naturally occurring HCM. This could explain, at least in part, the increased plasma concentrations of ANP and BNP in cats with HCM.
Material and Methods
Animals and samples
Tissue samples were obtained from five cats free of heart disease and four cats with confirmed HCM. The cats were euthanatized for purposes unrelated to this study, by using an overdose of barbiturate and following approved guidelines for care and use. Clinical evaluation included physical examination, electrocardiography, thoracic radiography, echocardiography, blood pressure measurement, and serum T4 concentration. The diagnosis of HCM was based on the findings of a hypertrophied, nondilated left ventricle by echocardiographic evaluation and the absence of other cardiac diseases or systemic disorders (e.g., systemic hypertension or hyperthyroidism) capable of producing left ventricular hypertrophy and was confirmed on necropsy and histology. 10 Full-thickness sections were removed from the free walls (middle third from apex to base) of left ventricle, right ventricle, and interventricular septum of each heart and cut into 1-cm3 pieces for fixation. The right atria and left atria, as well as the respective auricles, were separated and removed completely. All samples were placed in 4% paraformaldehyde overnight at 4 C, processed, and embedded in paraffin until immunohistochemistry was performed.
Immunohistochemistry
An avidin–biotin–peroxidase complex commercial method (DAKO Co., Carpinteria, CA) was used for immunohistochemistry. Briefly, 4-mm-thick paraffin wax sections were mounted on slides, which were dried for 30 minutes in an oven (60–70 C) and deparaffinized in xylene. The slides were then placed in changes of ethanol for 2 minutes each. Washing in buffer solution was performed between steps. The slides were then placed in two changes of 3% hydrogen peroxide and incubated in a casein-based block (Powerblock, Biogenex, San Ramon, CA). The slides were subsequently incubated in avidin block for 15 minutes, biotin block for 15 minutes, primary antibody (ANP or BNP) for 30 minutes, and biotinylated secondary antibody for 15 minutes. The reagent incubation was performed with streptavidin peroxidase for 15 minutes. A 5-minute Mayer's hematoxylin counterstain was used. The slides were dehydrated, cleared with xylene, and mounted with permanent mounting medium.
Because feline and human ANP sequences are identical, 3 commercial polyclonal rabbit antibodies designed for immunohistochemistry and directed against the C-terminal human ANP (C-ANP 1–28) were obtained and used as a primary antibody (Phoenix Pharmaceuticals Inc., Belmont, CA). Specific feline BNP polyclonal rabbit antibodies directed against the N-terminal portion of feline BNP precursor peptide sequence (proBNP 43–56) (Z. L. Liu, C. E. Wiedmeyer, D. D. Sisson, and P. F. Solter, Felis catus brain natriuretic peptide mRNA, GenBank Accession number AF253495, 2001) were obtained commercially (Quality Bioresources Inc., Austin, TX). Titrational studies were conducted for both antibodies, and the optimum working concentration obtained was 1 : 1,000 for both.
Results
In all control cats, ANP and BNP immunoreactivity was restricted to the atria (Figs. 1–12). Atrial cardiomyocyte staining for ANP and BNP was more intense and diffuse adjacent to the endocardial surface as compared with the epicardial surface (Figs. 1, 4) for all cats. The auricles stained more diffusely than the atria for both peptides. None of the ventricular cardiomyocytes of control hearts stained for either ANP or BNP (Figs. 7, 10). The interstitial capillaries and nerve fibers within the heart were strongly positive for BNP (Figs. 2, 6, 10) and negative for ANP (Fig. 1)
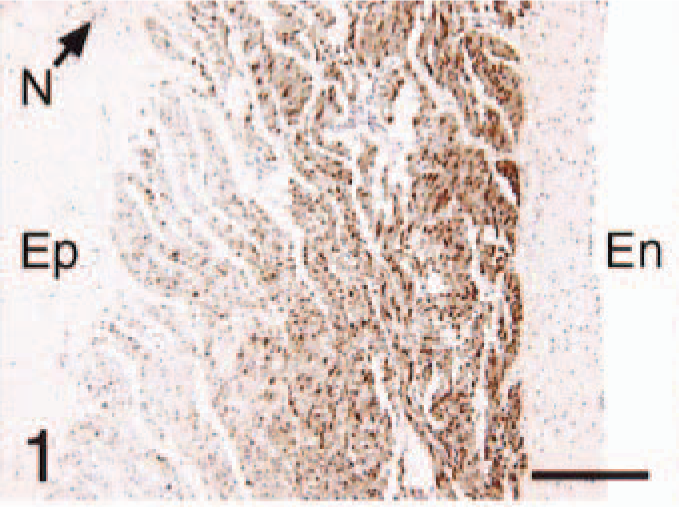
Hearts; control cats and cats with HCM. En, endocardial surface; Ep, epicardial surface; N, nerve bundle. Avidin–biotin–peroxidase complex method.
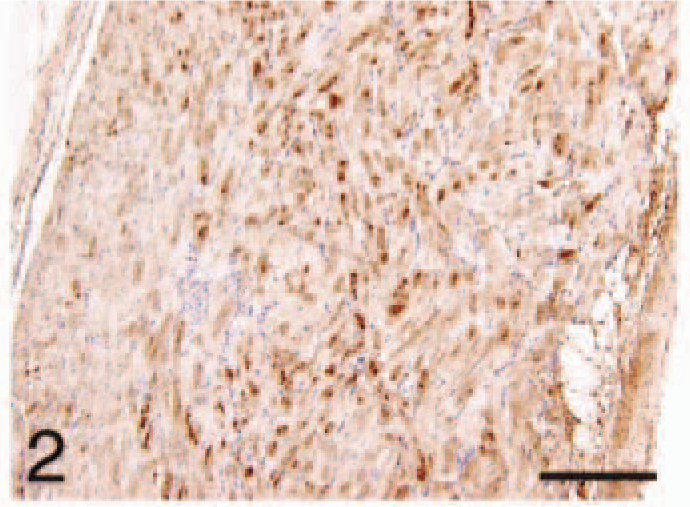
Atrium; HCM, ANP. Immunoreactivity more diffuse and the layered pattern less distinct. Bar = 220 µm.
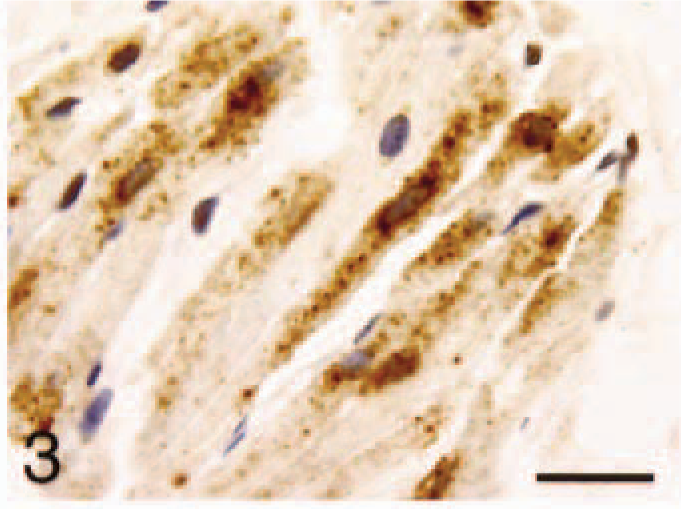
Atrium; control, ANP. Granular distribution in the cytoplasm of cardiomyocytes. Bar = 22 µm.
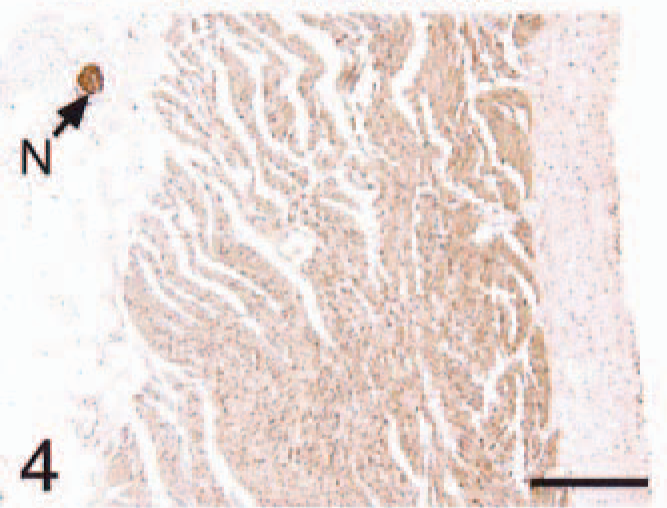
Atrium (same tissue block as
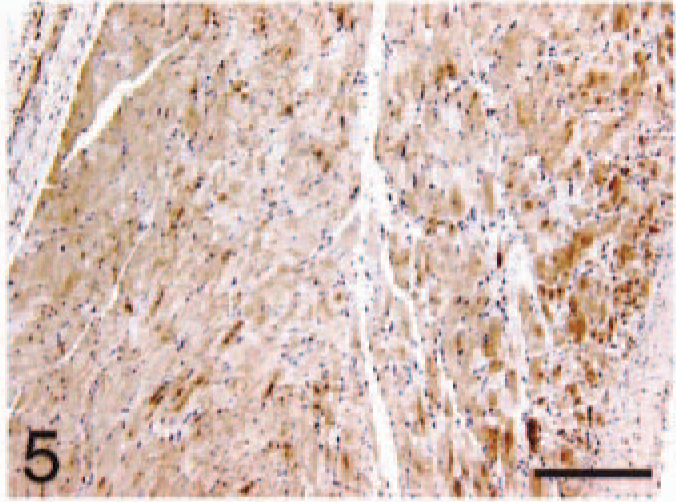
Atrium; HCM, BNP. Diffuse immunoreactivity with similar pattern of
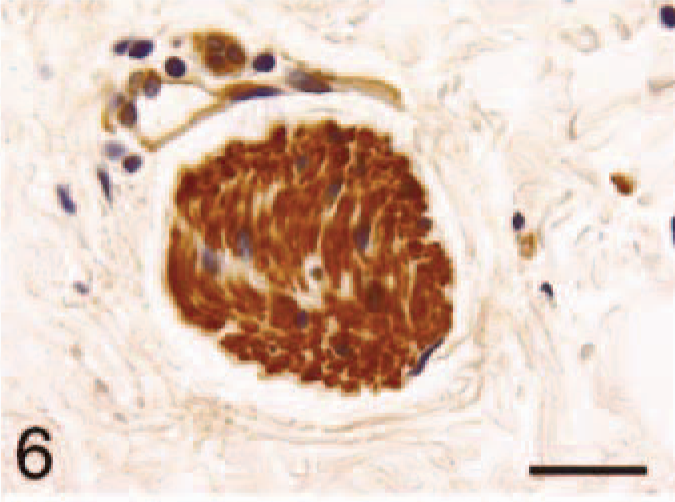
Atrium; control, BNP. Immunoreactivity of nerve fibers within the cardiac muscle. Bar = 22 µm.
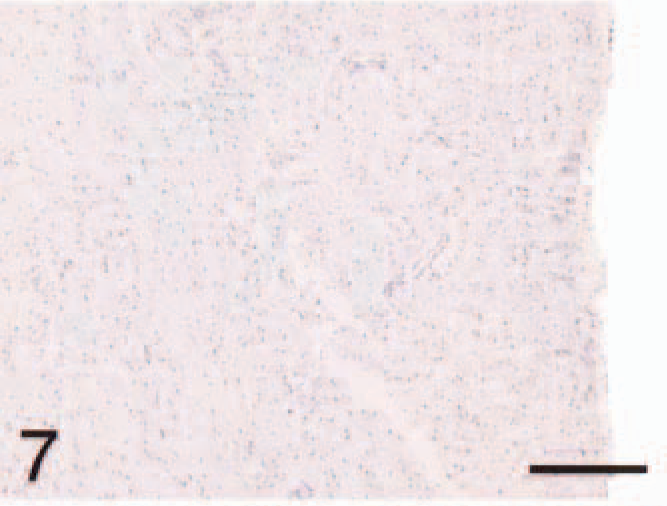
Ventricle; control, ANP. Cardiomyocytes lack staining. Bar = 220 µm.
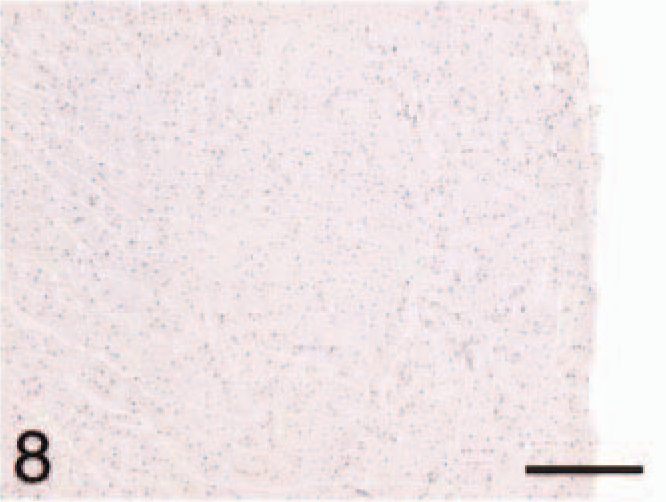
Ventricle; HCM, ANP. Cardiomyocytes lack staining. Bar = 220 µm.
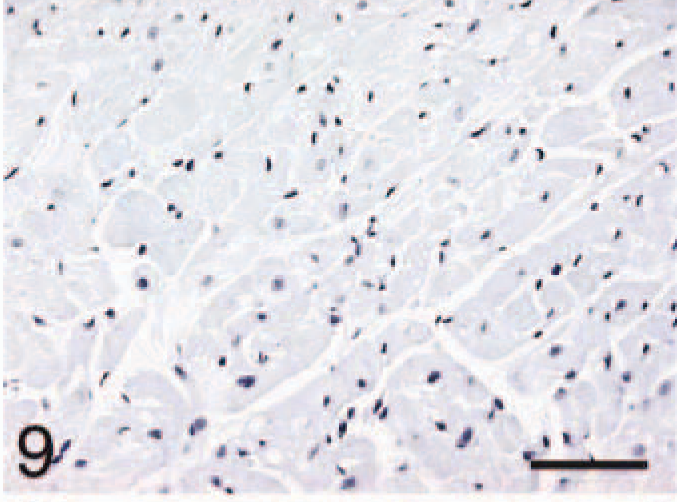
Ventricle; control, BNP. Negative stain control for 10–12 slides. Bar = 55 µm.
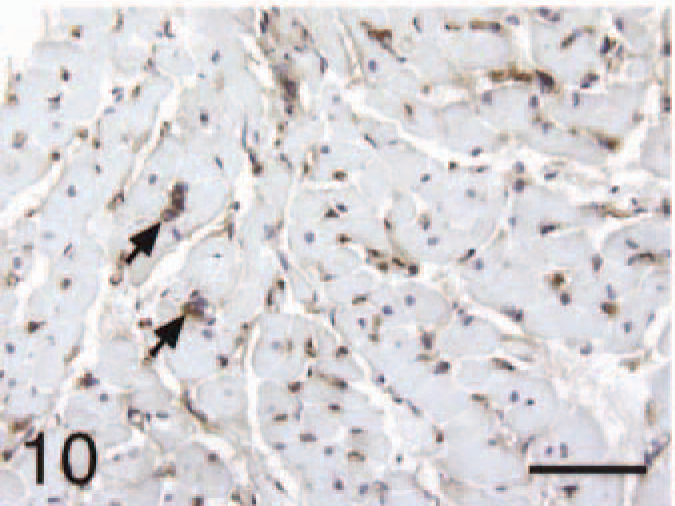
Ventricle; control, BNP. Cardiomyocytes staining negative and interstitial capillaries positive (arrows). Bar = 55 µm.
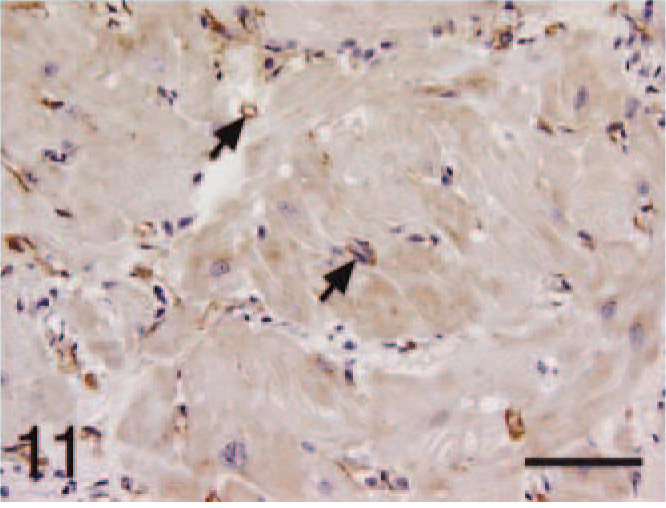
Ventricle; HCM, BNP. Capillaries staining positive (arrows) and cardiomyocytes with light and diffuse staining for BNP. Bar = 55 µm.
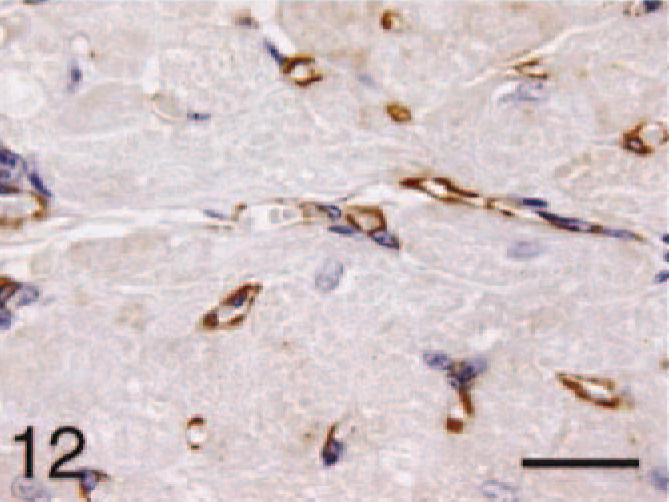
Ventricle; HCM, BNP. Higher magnification of the capillary and cardiomyocyte staining. Bar = 22 µm.
In cats with HCM, the atrial immunoreactivity for ANP and BNP was more diffuse and the layered pattern less distinct than in controls for both (Figs. 2, 5). The HCM ventricular cardiomyocytes were negative for ANP (Fig. 8), staining lightly and diffusely for BNP (Figs. 11, 12). The capillaries and nerve fibers remained positive for BNP in HCM (Figs. 11, 12).
Discussion
In this study, we have found ANP immunoreactivity in atrial cardiomyocytes of control cats as reported previously in other species. 5–7,21,22,28,32 Similar to what occurs in dogs, 7 rabbits, 19 and humans, 33,37 this reactivity was restricted to atrial cardiomyocytes with no ventricular staining. This contrasts with the distribution of ANP in rats, wherein ANP is also present in healthy ventricular cardiomyocytes. 30,38 We have previously shown that feline ANP gene expression also is restricted to the atria in normal cats. 3 Normal production, storage, and release to circulation of ANP in cats are likely restricted to atrial myocardiocytes, with no ventricular involvement.
We have shown previously that gene expression of ANP in cats with naturally occurring HCM is primarily in the atria but can be observed in variable amounts in the ventricles as well. 4 Similar results were reported in dogs and humans, where variable amounts of ANP were found in hypertrophic hearts. 7,34
Similar to what occurs in humans 20 and rats 31,39 but not in dogs, 7 the most intense staining for ANP was found in the auricles when compared with the body of the atria. The preferential location of ANP in the auricular appendage suggests a difference in tissue production. The significance of this finding is still uncertain, maybe because of a more sensitive response to stretching stress of the auricles than to the atrial body.
In cats, atrial cardiomyocyte staining for ANP was more intense and diffuse adjacent to the endocardial surface as compared with the epicardial surface. Similar findings are reported in healthy pig and human atria, 16,35,37 and in abnormal ventricles in dogs. 7 Presumably, this pattern of distribution developed to facilitate endocardial release of the peptide after myocardial production.
Immunohistochemical staining of BNP was restricted to the atria in control cats. With the exception of ventricular myocytes of mice 26 and rats, 27 BNP was not found in the ventricle of most species that have been studied. 5,18,21,40 Minute amounts of BNP were found in ventricle of dogs, 2 and its localization in humans is still controversial. 15,20,37 These inconsistencies in many species may result from differences in sensitivity of immunohistochemistry to the uneven tissue distribution of the peptide (demonstrated as a layered pattern with higher concentration at the endocardial side), to different affinities of the antibodies used, or to a variability in distribution of BNP in those species.
As in dogs, 2 the atrial staining for BNP in control cats was more intense at the auricles. We also observed a layered pattern of atrial cardiomyocyte staining for BNP that was more intense and diffuse adjacent to the endocardial surface than at the epicardial surface. A similar pattern of distribution for BNP has been observed in humans 8 and pigs. 16 An analogous mechanism of peptide production and release in different myocardial layers could explain the codistribution of ANP and BNP in cat atria.
Interestingly, the nerve fibers within the hearts of the control cats stained positive for BNP. Studies performed on other species, such as cows, sheep, rats, pigs, and humans, also have shown localization of natriuretic peptides to nervous tissue in the heart or peripheral conduction system. 12–14,42 The role of natriuretic peptides in cardiac conduction cells is still unclear. It is speculated that the natriuretic peptides function in an autocrine or paracrine manner to balance the cardiac conduction rate. 12 The interstitial capillaries stained positive for BNP. Some investigators suggest that BNP and ANP bind to common receptors in brain capillary and aortic endothelial cells. 11 Capillaries and nerve fibers remained positive for BNP in feline hearts with HCM.
In cats with HCM, the atrial walls were thicker than those of the control cats, and atrial immunoreactivity for ANP and BNP was distributed more prominently and more diffusely throughout the atria. This change may represent an increase in peptide production throughout the atrial cardiomyocytes, with increased release of natriuretic peptides. Increased ANP and BNP immunoreactivity in the atria of cats with HCM could reflect a physiologic stimulation, e.g., chronic atrial stretch caused by elevated filling pressures resulting from reduced ventricular compliance or valvular insufficiency. Alternatively, these changes could be an integral part of the underlying disease process producing myocardial hypertrophy.
In the present study we have not found ANP immunostaining in ventricles of cat hearts with HCM. However, we have previously shown variable ventricular ANP gene expression in feline cardiac tissue with HCM. 4 This ventricular expression of ANP also has been reported in human dilated cardiomyopathy and cardiac infarction. 33,34 In our present study, we believe that the negative immunostaining was a result of tissue ANP levels below the detectable limit or may be caused by an immediate release of ANP after transcription.
Although ventricular cardiomyocytes with HCM were negative for ANP, they stained for BNP lightly and diffusely. The presence of ventricular BNP has been observed previously in several cardiac diseases, including myocarditis, 35 myocardial infarction, 5 amyloidosis, 36 chronic heart failure, 17 dilated cardiomyopathy, 37 and HCM. 20,34
Tissue expression of ANP and BNP can correlate to their plasma levels, as reported previously. 17 If found to be true in cats, the novel production of BNP to include the ventricles suggests that BNP could be a sensitive plasma marker of feline HCM. Moreover, the clear differences in the patterns of ANP and BNP immunostaining between the control hearts and the hearts with HCM suggest a potential use of these peptides as auxiliary markers for the histologic diagnosis of HCM. Further studies would be necessary to establish the diagnostic accuracy of these as tissue markers, particularly in their ability to differentiate HCM from other causes of cardiac hypertrophy.
In summary, cardiac ANP and BNP production/storage in healthy cats appears to be restricted to atrial cardiomyocytes, particularly those lining the endocardial surface. In addition, BNP also can be found in the interstitial capillaries and nerve fibers within the normal cat heart. In cats with spontaneously occurring HCM, ANP and BNP are more prominently and more diffusely distributed within the atria, and BNP has novel expression within the ventricle.
Parts of this article were presented at the 2002 Meeting of the American College of Veterinary Pathologists and American Society of Veterinary Clinical Pathology in New Orleans, LA.
Footnotes
Acknowledgements
We thank Jane Chladny for help with the immunohistochemical processing. A. W. Biondo was sponsored through a research fellowship from CNPq—Brazilian National Council of Scientific and Technologic Development. This project was funded in part by a grant from the Winn Feline foundation.
