Abstract
The mechanisms of Salmonella serovar-host specificity are not well defined. Pig ileal loops were used to compare phenotypic differences in early cellular invasion between non-host-adapted Salmonella serovar Typhimurium (SsT) and host-adapted Salmonella serovar Choleraesuis (SsC). By 10 minutes postinoculation, both serovars invaded a small number of M cells, enterocytes, and goblet cells. Multiple SsC organisms (up to 6 per cell) simultaneously invaded M cells, whereas SsT often invaded as one to two organisms per M cell. Internalization of both serovars resulted in vacuoles containing a single bacterium. The follicle-associated epithelium (FAE) of SsC-inoculated loops responded with more filopodia and lamellipodia although exhibiting less cell swelling than SsT. Additionally, SsT showed an enhanced affinity for sites of cell extrusion compared with SsC at 60 minutes. These results suggest: 1) both SsC and SsT exhibit non-cell-specific invasion as early as 10 minutes postinoculation, 2) Salmonella serovars exhibit differences in early invasion of FAE and M cells, and 3) cells undergoing extrusion may provide a site for preferential adherence by SsT and SsC.
Introduction
The pig is a good model for studying the effect of host adaptation and pathogenesis in systemic and enteric salmonellosis of humans. 22 Salmonella enterica serovars Choleraesuis (SsC) and Typhimurium (SsT) are the two most common etiologic agents of swine salmonellosis. 17 , 18 SsC is host adapted to swine and leads to a septicemic, typhoid-like disease. SsT is not host adapted to swine and leads to localized enterocolitis. Because SsC-infected animals experience more severe clinical disease, the case mortality rate is higher than in SsT-infected animals. Salmonella-host adaptation and host immune status are critical components that influence the manifestation of localized or systemic salmonellosis. 1 Attempts have been made, in many species, to examine and elucidate the mechanisms of host adaptation in Salmonella. 2 , 17 , 20–22 Phenotypic differences between Salmonella serovars during epithelial invasion could help illuminate mechanisms of serovar-host adaptation. Recent work done in our laboratory suggests that there may be differences between SsC and SsT in early epithelial invasion. 14 The purpose of this experiment was to compare early cellular invasion by SsC and SsT.
Materials and Methods
Bacteria
Swine isolates included Salmonella enterica serovar Typhimurium phagetype DT104 (NVSL 98–745), a clinical swine isolate obtained from the National Veterinary Services Laboratories (NVSL, Ames, IA), and SsC var. kunzendorf strain χ3246 (courtesy of Dr. Roy Curtiss III, Washington University, St. Louis, MO). Bacteria were grown as previously described 14 and to final inoculum concentrations of 8 × 108 CFU/ml for SsT and 6 × 108 CFU/ml for SsC (CFU refers to colony-forming unit).
Animals
Four mixed-breed pigs were obtained at ∼10 days of age and grown in isolation facilities at the National Animal Disease Center (NADC, Ames, IA). The pigs were rectal-culture negative by enrichment broths for Salmonella in the weeks before the experiment. For the experiment, 4- to 6-week-old pigs were allowed to fast for 18 hours before surgery.
Surgical procedures
Anesthesia for the terminal surgery was induced by intramuscular injection of Telazol (tiletamine/zolazopam, 18 mg/kg, Fort Dodge Labs, Ft. Dodge, IA), Ketaset (ketamine, 24 mg/kg, Fort Dodge Labs), and Rompun (xylazine, 12 mg/kg, Bayer Animal Health, Shawnee Mission, KS). Surgical gut-loop procedure and tissue collection were performed as previously described. 14 Briefly, the ileum was exteriorized and ligated to form 6-cm test loops, each separated by 2-cm spacer loops. The test loops were randomly injected with 6 ml of SsC, SsT, or sterile Luria-Bertani broth (Table 1). The ileum was then replaced and the abdomen closed. At the appropriate time point, the abdomen was reopened and each loop was injected to moderate distension with fixative (2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer [pH 7.2]). The loop was harvested and placed in a fixative bath. The pig was then euthanatized with intravenous sodium pentobarbital.
Pig number, time point, and loop inoculum for comparison of epithelial invasion by Salmonella serovars.


∗ Time points are minutes postinoculation.
† Describes the number of loops formed and the inoculum used.
‡ SsC = Salmonella serovar Choleraesuis, SsT = Salmonella serovar Typhimurium, LB = Luria-Bertani broth.
Electron microscopy
After 1–2 hours in fixative, loops were opened, tissue sections cut, and blocks placed in fresh fixative. Preparation of samples for transmission and scanning electron microscopy (TEM and SEM, respectively) were performed as previously described. 14 Briefly, for TEM, 1-mm blocks were stained in 1% osmium tetroxide, dehydrated in alcohols, cleared in propylene oxide, and embedded in epoxy resin. The 1-µm sections were cut with an ultramicrotome, stained with toluidine blue, and examined with a light microscope. Thin sections were cut from blocks, placed on nickel grids, and viewed on a Philips 410 transmission electron microscope (NADC).
Briefly, for SEM, the tissues were postfixed with 1% osmium tetroxide and 1% thiocarbohydrazide. The samples were dehydrated through a series of graded ethanol baths and then critical point dried. Tissues were mounted on aluminum stubs with colloidal silver and sputter coated with gold-palladium. Samples were viewed at the Bessey Microscopy Facility (Iowa State University, Ames, IA) on a JSM-5800LV (Japanese Electronic Optical Laboratories) scanning electron microscope.
Results
SEM examination
At 10 minutes, both SsC and SsT were associated with a small number of M cells and a few enterocytes. The M cells reacted with minor filopodia, blebbing, and cell swelling, whereas enterocytes exhibited focal, minor microvillar elongation. Sites of cell extrusion were identified, and a small number of bacteria were observed preferentially adhered near the base of the extruded cells in both SsC- and SsT-inoculated loops (Table 2).
Characterization of bacteria-associated cell extrusions over time.
∗ Minutes postinoculation.
† SsC = Salmonella serovar Choleraesuis, SsT = Salmonella serovar Typhimurium.
‡ Cellular extrusions associated with bacteria were divided by the total number of cell extrusions (n = 20).
§ Total number of bacteria associated with cell extrusions was divided by the total number of cell extrusions associated with bacteria.
At 20–60 minutes, the bacterial interaction with epithelial cells increased through the course of infection. In the SsC-inoculated loops, the follicle-associated epithelium (FAE) expressed more filopodia and lamellipodia formation and less cell swelling than did the SsT-inoculated loops (Figs. 1, 2). On the absorptive epithelium, both serovars induced similar focal disruptions and filopodia formation in the apical cell membrane of enterocytes and goblet cells. The frequency of bacterial association at sites of cell extrusion (Fig. 3) was similar for most time points, except for the 60-minute loop, where SsT associated with 60% of extrusions and SsC with 10% of the extrusions (Table 2). Furthermore, the average number of bacteria per extrusion site increased to 6.3 in 60-minute SsT loops.

Ileum; pig 4. Scanning electron micrograph of FAE lamellipodia (L) and filopodia (arrows) formation 60 minutes after Salmonella serovar Choleraesuis loop inoculation. Gold-palladium. Bar = 4 µm.
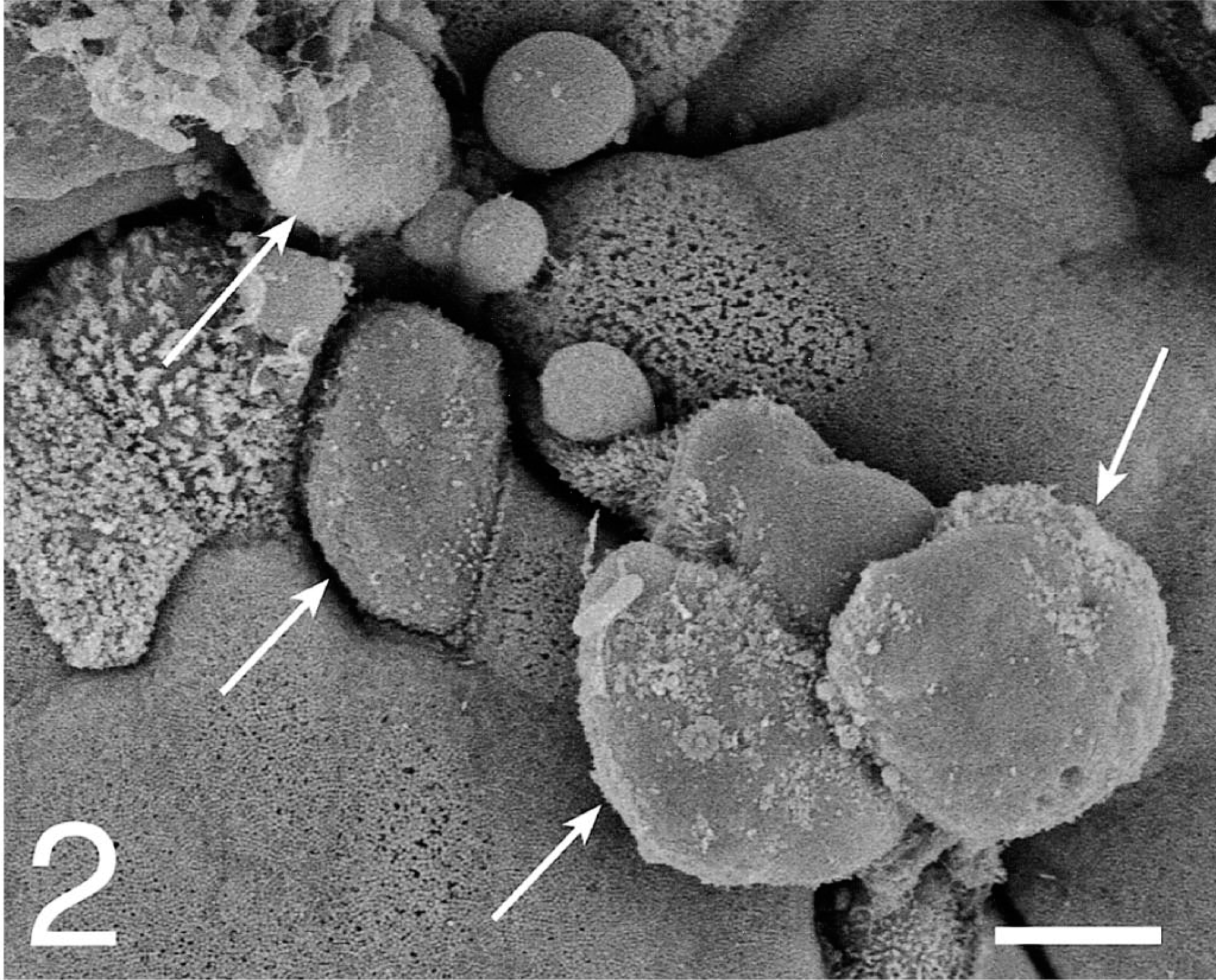
Ileum; pig 4. Scanning electron micrograph of FAE cell swelling (arrows) 60 minutes after SsT loop inoculation. Gold-palladium. Bar = 5 µm.
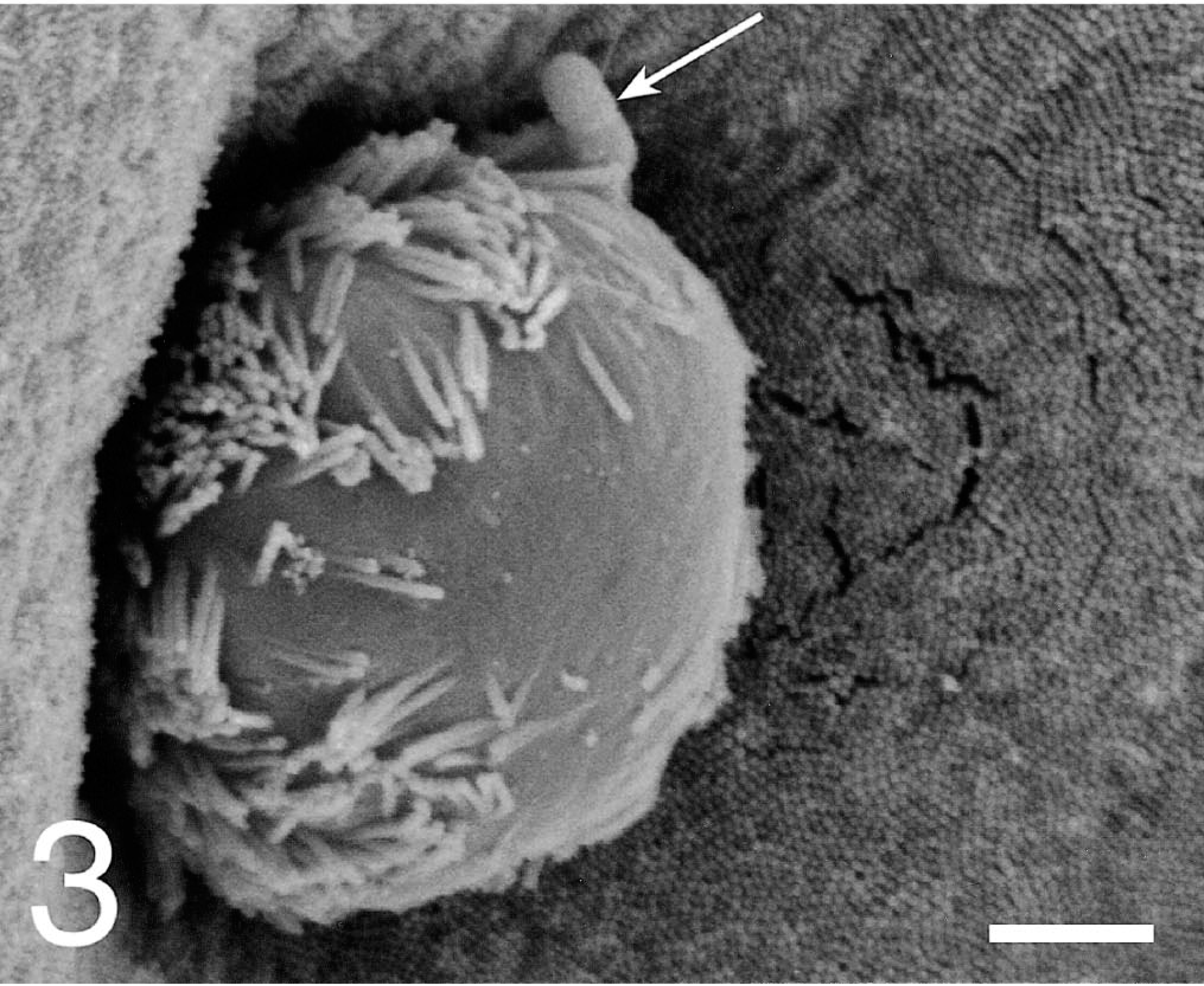
Ileum; pig 4. Scanning electron micrograph of absorptive epithelium cell extrusion with associated bacteria (arrow) 60 minutes after SsC loop inoculation. Gold-palladium. Bar = 3 µm.
TEM examination
At 10 minutes, both SsC and SsT organisms interacted with a small number of M cells, enterocytes, and goblet cells. Internalized bacteria were seen in all the three cell types.
At 20–60 minutes, the invasion of M cells, enterocytes, and goblet cells was more readily observed through the chronologic course of infection. In the SsC-inoculated loops, infected M cells contained one to six internalized bacteria (Fig. 4). This contrasted with the SsT loops in which infected M cells contained only one to two internalized bacteria. Inoculation with either serovar usually resulted in vacuoles containing a single bacterium.
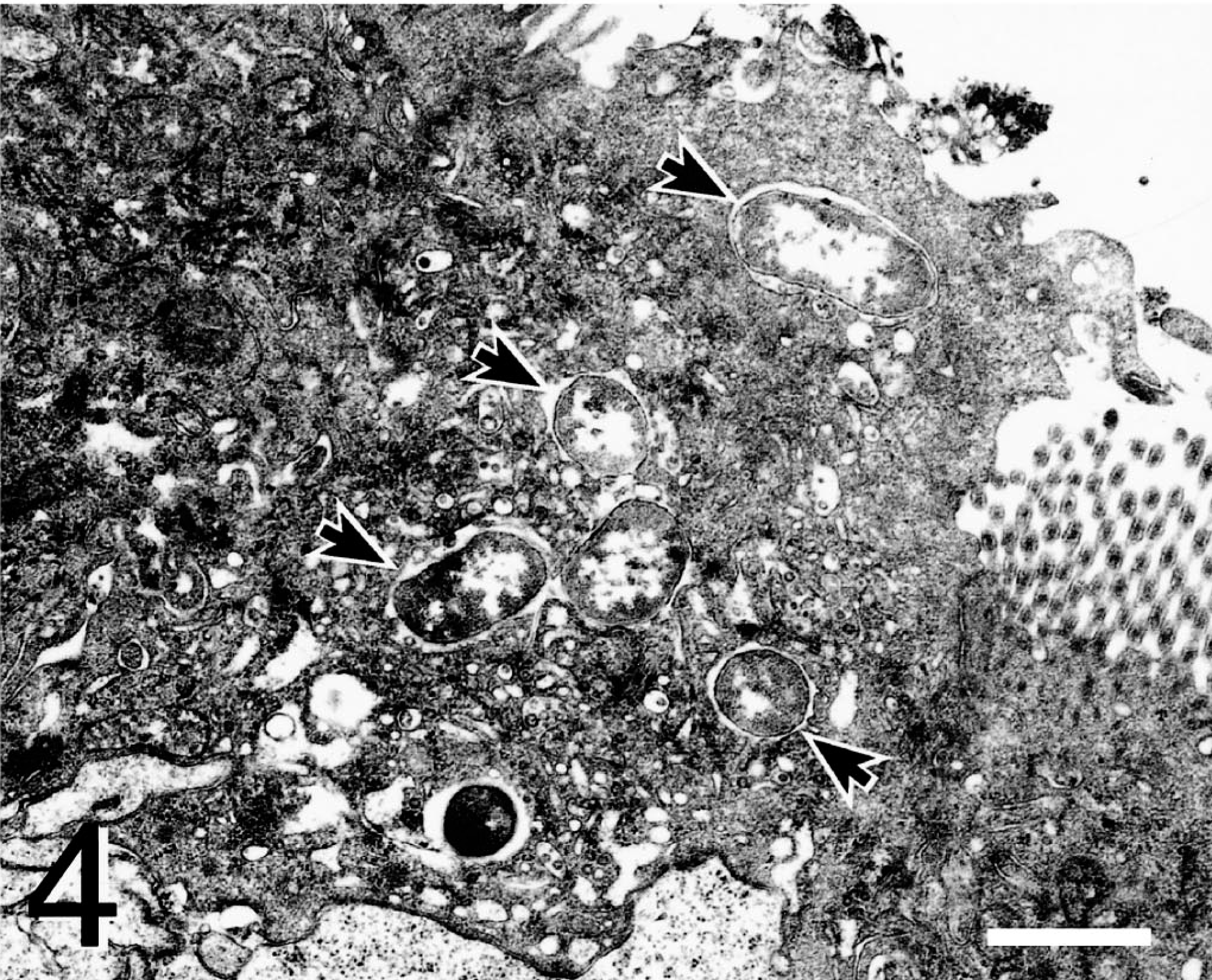
Ileum; pig 3. Transmission electron micrograph of FAE M cell with multiple internalized bacteria (arrows) 30 minutes after SsC loop inoculation. Uranyl acetate and Reynolds lead acetate. Bar = 2 µm
Discussion
The purpose of this study was to compare early cellular invasion of SsC and SsT. Examination of tissues at 10 minutes by electron microscopy showed that both SsC and SsT invade a small number of M cells, enterocytes, and goblet cells in a non–cell-specific manner. Through the course of early infection, the number of active invasion sites increased, with the most activity occurring at 60 minutes. The rapid, non–cell-specific invasion is consistent with recent work done with SsT in our laboratory. 14 In vitro comparison of SsC and SsT early invasion in Caco-2 cells produced no significant difference in cellular interactions as assessed by TEM and SEM. 5 SsC and SsT exhibit similar nonspecific cellular invasion in the swine ileum.
In our TEM studies, differences in M cell invasion were noted between serovars. SsC invaded as multiple organisms (1–6 per M cell), whereas SsT invaded as one or two organisms per M cell. This is in contrast to previous ileal loop studies in which SsC was shown to primarily invade M cells as a single organism. 2 , 17 However, these SsC studies used time points that ranged from 90 minutes to 24 hours postinoculation, whereas our observations were made earlier (10, 20, 30, and 60 minutes) in the course of infection. Furthermore, evidence suggests that the in vivo kinetics after early Salmonella invasion and epithelial interaction may rapidly diminish as early as 60 minutes postinoculation. 6 Previous studies concerning SsT have shown that cell invasion may occur by multiple organisms. 6 , 23 Our work focused on early cell invasion by SsT phagetype DT104. The significance of SsT phagetype DT104 is its increasing isolation frequency and association with multiple antibiotic resistance. 7 A recent in vitro study of SsT phagetype DT104 invasion suggests that the associated multiple antibiotic resistance may attenuate cellular invasion. 3 Although this attenuation has not been defined at the ultrastructural level, we speculate that the observed M cell invasion by individual rather than multiple organisms may be attributed to the multiple antibiotic resistance phenotype. The time postinoculation and phagetype differences could account for contrasts in M cell invasion.
Scanning electron micrographs of SsC-inoculated loops showed a greater proportion of filopodia and lamellipodia formation over the FAE as compared with SsT-inoculated loops. We speculate that this cellular rearrangement may be related to the increased numbers of organisms invading the cell. Salmonella can induce filopodia and lamellipodia formation in host cell by injection of bacterial proteins through a type-III secretion system. 10 Some of these injected proteins have been shown to induce the apical lamellipodia and filopodia by the Rho family of G-proteins. 9 , 11 , 19 The SsC may induce more filopodia and lamellipodia by the increased numbers of organisms that enter the cell, secreting more of these invasion proteins. Interestingly, the SsT produced proportionally fewer filopodia and lamellipodia but more cell swelling. Cell swelling is indicative of acute cell injury and can lead to an inflammatory response. 4 , 12 SsT and other non–host-adapted serovars have been implicated to cause many of their early intestinal lesions by induction of inflammation and neutrophil migration into the intestinal lumen. 8 , 13 Some have suggested that avoidance in the induction of intestinal inflammation by host-adapted serovars may actually promote systemic spread of the organisms. 20 In this study, the comparative lack of evidence of epithelial cell injury from SsC-inoculated loops during early invasion may reflect a host-adapted mechanism that enhances systemic spread.
In addition, sites of cell extrusion were examined for preferential adherence by both serovars. In SsT- and SsC-inoculated loops, bacteria were seen adherent to a small portion of the cell extrusions at all time points. However, in the 60-minute SsT-inoculated loop, the percentage of extrusions associated with bacteria increased to 60%, whereas in the SsC-inoculated loop, it remained at 10%. Furthermore, in the SsT loop, the average number of bacteria per extrusion increased to 6.3 per site. Differences in bacterial affinity to sites of cell extrusion at 60 minutes have been suggested in the previous work done in our laboratory. 14 These could be the result of different kinetics of invasion between the serovars or a true specific preference by SsT to these sites. In vitro comparison of invasion by SsT and Salmonella enterica serovar Typhi on HeLa cells suggested similar mechanisms of invasion and intracellular trafficking, although the more efficient invasion by SsT was suggested to be due to differential adherence to sites of apical membrane rearrangements. 15 In contrast, in vitro examination of invasion in mucosal explants showed that SsC organisms do have the capacity to adhere and swarm at sites of epithelial loss. 16 Additional in vivo work is needed to determine the differences in affinity to sites of extrusion by various serovars and their relevance to natural disease.
In this study, we show that SsC and SsT can invade as early as 10 minutes postinoculation and in a non–cell-specific manner. The mechanism of early M cell invasion differs between the serovars. SsT organisms enter the M cell in small numbers, whereas SsC can enter as multiple organisms. SsC loops induce proportionally more filopodia and lamellipodia with less cell swelling than can the SsT loops. Additionally, preliminary comparison of bacterial affinity to sites of cell extrusion suggests serovar differences at 60 minutes. Evaluation of extended time points with statistic comparisons is warranted.
Footnotes
Acknowledgements
We thank Judy Stasko, Mike Marti, and John Mattila for technical assistance and Dr. Mark R. Ackermann for assistance in interpretation of ultrastructural pathology.
