Abstract
The host response to Salmonella plays a major role in the outcome of infection. The present study was undertaken to further characterize Salmonella typhimurium infection in neonatal calves at both the morphologic and the molecular level using the ligated ileal loop model. Eight 4–5-week-old male Holstein calves underwent laparotomy, and loops were prepared in the ileum. The loops were either inoculated with an S. typhimurium strain pathogenic for cattle or injected with sterile LB broth as control. Samples for histology, transmission and scanning electron microscopy, and RNA extraction were collected at various time points between 5 minutes and 12 hours postinfection. Invasion of both M cells and enterocytes began at 15 minutes postinfection. No specific cell type was the main target for invasion. Intracellular bacteria were observed in the lamina propria after 1 hour postinfection. A severe acute neutrophilic response was associated with invasion of the Peyer's patches. Upregulated expression of CXC chemokines (interleukin [IL]-8, growth-related oncogenes, [GRO] α and γ, and granulocyte chemotactic protein [GCP]2) was detected by reverse transcription polymerase chain reaction beginning at 1 hour postinfection. Expression of proinflammatory (IL-1β, IL-18, and tumor necrosis factor [TNF]α) and anti-inflammatory (IL-10, IL-1Ra, and IL-4) cytokines was also assessed. A marked increase in expression of IL-1β was observed, whereas the profile of expression of IL-18 and TNFα did not change after infection. Upregulation of IL-1Ra and IL-4 but not of IL-10 was observed. These findings indicate that infection of bovine ligated ileal loops with S. typhimurium results in an acute neutrophilic inflammatory response that is associated with the upregulation of CXC chemokines (IL-8, GROα and γ, and GCP2), IL-1β, IL-1Ra, and IL-4.
Keywords
Salmonellosis has a significant impact on animal health. Although cattle may be infected with many different serotypes,45,46,61 bovine salmonellosis is caused mostly by two of them, Salmonella enterica serovar Typhimurium (hereinafter referred to as S. typhimurium) and serovar Dublin (S. dublin).27,53 S. typhimurium is the serotype most frequently isolated from ill cattle in the USA, thereby resulting in significant economic losses.61 S. typhimurium affects mostly young cattle and may cause various clinical manifestations that can range from a peracute course with death within 24 hours of infection to a chronic asymptomatic infection. However, the most common clinical manifestation of the infection is an acute diarrheal disease.47,63 In addition to its impact on animal health, S. typhimurium is the second most prevalent food-borne disease of bacterial origin in the USA, with an estimated annual 1.4 million cases36 resulting in 0.6–3.5 billion dollars of annual medical and productivity costs.61 From an experimental standpoint, cattle are a useful model for Salmonella-induced diarrhea in humans because the clinical manifestation of the infection is similar in both species.51,57 The usefulness of cattle as a model for studying human enteritis is highlighted by the fact that laboratory animals, such as the mouse, do not develop diarrhea as a consequence of S. typhimurium infection and are thus not well suited for use in the study of this disease.51,57
Our understanding of Salmonella virulence factors has increased greatly during recent years.24 Many Salmonella genes required for invasion of epithelial cells,9,18 intracellular survival within macrophages,6,40 and enteropathogenicity57,59 have been identified. Salmonella pathogenicity island 1 (SPI-1) encodes a type III secretion system that translocates effector proteins into the cytoplasm of host cells, resulting in invasion of epithelial cell lines in vitro8,19,42 and enteropathogenicity in the calf.56 SPI-5-encoded genes, whose products are secreted via the SPI-1 type III secretion system, contribute to the pathogenesis of Salmonella-induced enteritis in calves.62 SPI-2 encodes a second type III secretion system that is involved in intracellular survival in macrophages in vitro.6,14,40,52 However, the role of intracellular survival in the pathogenesis of diarrhea is not clear. Although mutations in SPI-2 cause more that 10,000-fold attenuation during S. typhimurium infection in mice,40,52 they cause less than 15-fold attenuation during oral infection in the calf model of enterocolitis.56,58
In contrast to what is known about bacterial virulence factors, the role played by the host in the pathogenesis of Salmonella-induced diarrhea is still poorly understood. Although M cells in the follicle-associated epithelium have been identified as the primary target for Salmonella invasion in mice,7,28 S. typhimurium invades both enterocytes and M cells in calves; however, invasion of enterocytes seems to be delayed when compared with that of M cells.17 Shortly after invasion, the organisms are phagocytosed by macrophages, where they localize within membrane-bound vacuoles.17 The massive infiltration of neutrophils that occurs during the first hours after infection is thought to be a key event in the pathogenesis of diarrhea because the neutrophilic response precedes and is correlated with fluid accumulation into the intestinal lumen, and both increase over time.50 Furthermore, a sopB mutant of S. typhimurium, which is attenuated for induction of fluid secretion, triggers a delayed inflammatory response when compared with the wild type.50
To develop a better understanding of the bovine host response to Salmonella infection, we employed a bovine ligated ileal loop model, and here we describe the histologic and ultrastructural changes and the profile of expression of chemokines and cytokines in bovine Peyer's patches infected with S. typhimurium.
Material and Methods
Bacterial strain and growth conditions
The S. typhimurium strain IR715,54 which is a spontaneous nalidixic acid–resistant derivative of the strain ATCC14028 (American Type Culture Collection, Rockville, MD), was used in this study. Bacteria were grown in LB broth for 20 hours at 37 C under agitation (230 rpm), and 50 µl of the culture was inoculated into 5 ml of fresh LB broth and cultured for additional 6 hours before inoculation.
Animals, surgical procedure, and sampling
Eight male Holstein calves, 4–5 weeks of age and weighing 45–55 kg, were used in these experiments. They were fed milk replacer twice daily and water ad libitum. The calves were clinically healthy before the experiment and were culture negative for fecal excretion of Salmonella. Examination for Salmonella serotypes in fecal swabs was performed twice, 2 weeks before and immediately before the experiment, by enrichment in tetrathionate broth (Difco, Detroit, MI) and by streaking on brilliant green agar (BBL, Sparks, MD).
The surgical procedures employed in this study have been described previously.50 The calves were fasted for 24 hours prior to the surgery. Anesthesia was induced with Propofol (Abbot Laboratories, Chicago, IL) followed by placement of an endotracheal tube and maintenance with isofluorane (Isoflo, Abbot Laboratories) for the duration of the experiment. A laparotomy was performed, the ileum was exposed, and 13 loops from 6 to 9 cm in length were ligated, leaving 1-cm loops between them. The loops were inoculated by intralumenal injection of 3 ml of a suspension of S. typhimurium strain IR715 in LB broth containing approximately 0.75 × 109 Colony-forming units (CFU) ml. Sterile LB broth was injected into the control loops. The loops were replaced into the abdominal cavity. Samples for bacteriologic culture, histopathology, and ultrastructural studies were collected at 5, 15, and 30 minutes and at 1, 2, 3, 4, 5, 6, 8, 10, and 12 hours. Tissue samples from Peyer's patches were weighed, homogenized in phosphate-buffered saline, serially diluted, and plated onto LB agar plates containing nalidixic acid (50 µg/ml) for counting of the CFU.
Fragments from the Peyer's patches were fixed by immersion in formalin, processed according to the standard procedures for paraffin embedment, sectioned at 5 μm, and stained with hematoxylin and eosin (HE).
Scanning and transmission electron microscopy
Small fragments from Peyer's patches were fixed by immersion overnight at 4 C in a solution of 5% glutaraldehyde and 4% paraformaldehyde in 0.1 M sodium cacodylate buffer. Tissues were then washed three times with 0.1 M sodium cacodylate buffer and postfixed for 2 hours at 4 C in 1% osmium tetroxide in 0.1 M sodium cacodylate buffer. For transmission electron microscopy, the samples were stained overnight at 4 C in a saturated uranyl acetate solution. Tissues were dehydrated in a graduated series of ethanol solutions and propylene oxide and embedded in Epon Araldite. Sections (0.5 μm) were stained with toluidine blue and examined under a light microscope for selection of the microscopic fields. The blocks were trimmed, and thin sections (60–90 nm) were cut, mounted onto copper grids, stained with uranyl acetate and lead citrate, and examined with a Zeiss 10C transmission electron microscope. For scanning electron microscopy, the samples were dehydrated, critical point dried, coated with a thin layer (approximately 500 Å) of AuPd, and examined with a JEOL JSM-6400 scanning electron microscope at an accelerating voltage of 15 kV.
Reverse transcription polymerase chain reaction
Fragments of infected and uninfected Peyer's patches were stored at −80 C for RNA isolation. Total RNA was extracted after dissection of the mucosa from samples obtained at 5 and 30 minutes and at 1, 3, and 6 hours postinfection using Tri Reagent (Molecular Research Center, Cincinnati, OH) according to the manufacturer's instructions. RNA samples were treated with RNase-free DNase I and purified by phenol–chloroform extraction prior to reverse transcription (RT) polymerase chain reaction (PCR) amplification to avoid amplification of genomic DNA template. A one-step RT-PCR was carried out using a commercial kit (Qiagen, Valencia, CA) and 1 µg of total RNA as template. RT was performed at 50 C for 30 minutes followed by PCR amplification. The PCR conditions, primers, and product size for each target are described in Table 1. The RT-PCR products were resolved in a 1.2% agarose gel, stained with ethidium bromide, and photographed. The intensity of the specific bands were measured using the NIH Image software.
RT-PCR parameters for amplification of bovine chemokines and cytokines.
Results
Morphologic findings
All the lesions described below were found in either the ileal mucosa or the submucosa. Changes in the muscular layer and serosa were not taken into consideration because of the responses to surgical manipulation. Except for the above changes, sections from uninfected control loops showed no other significant histologic lesions.
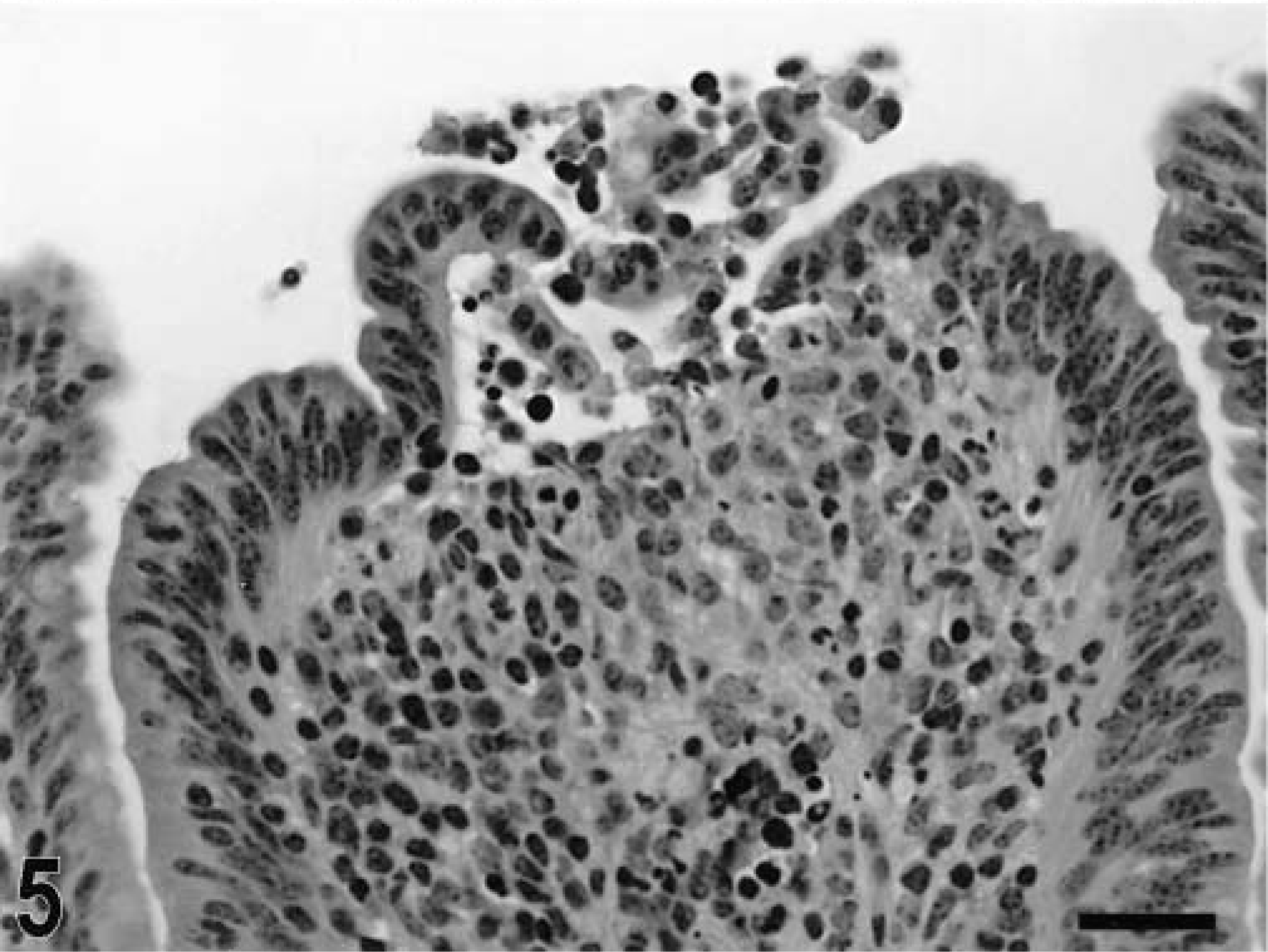
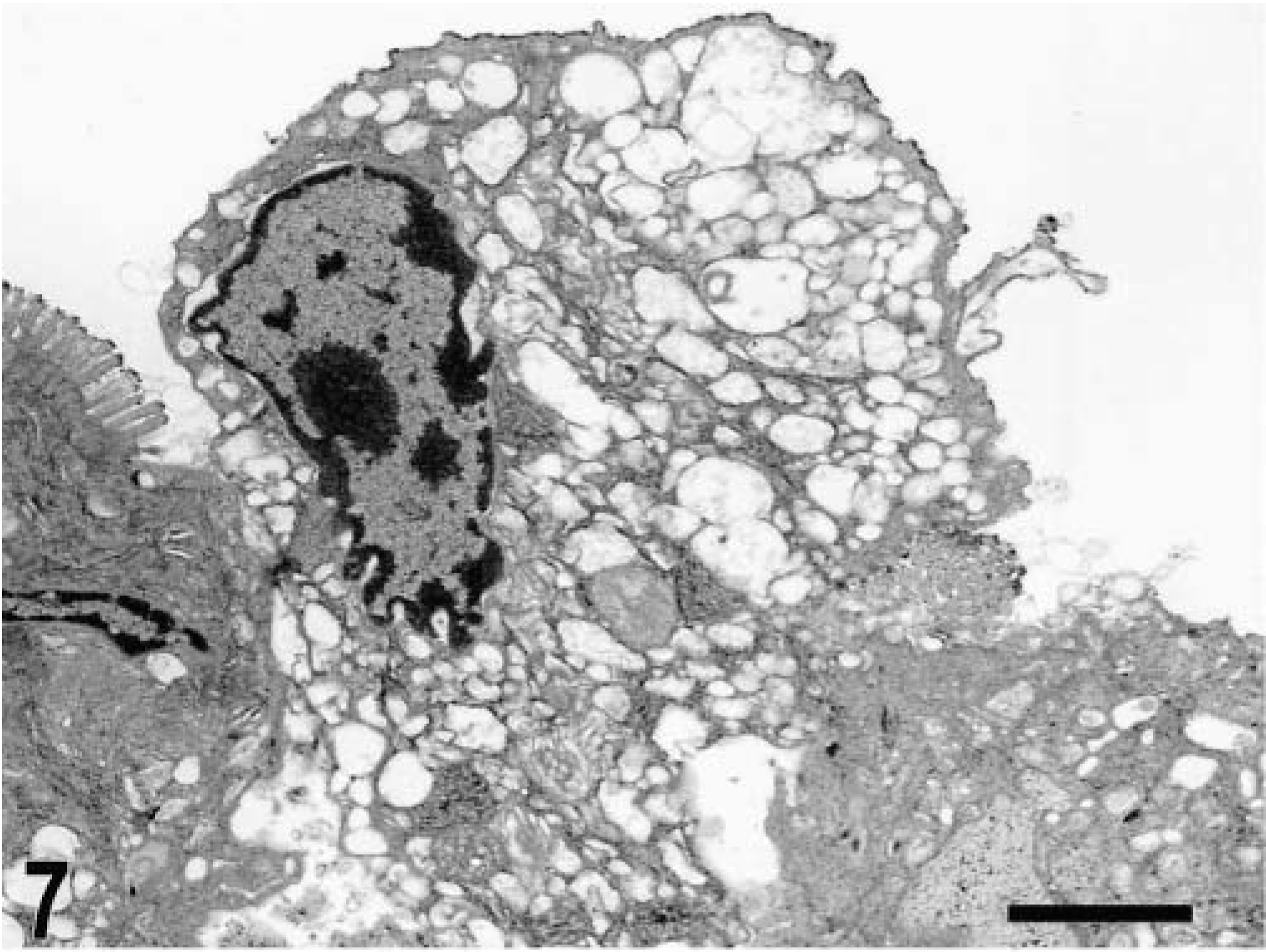
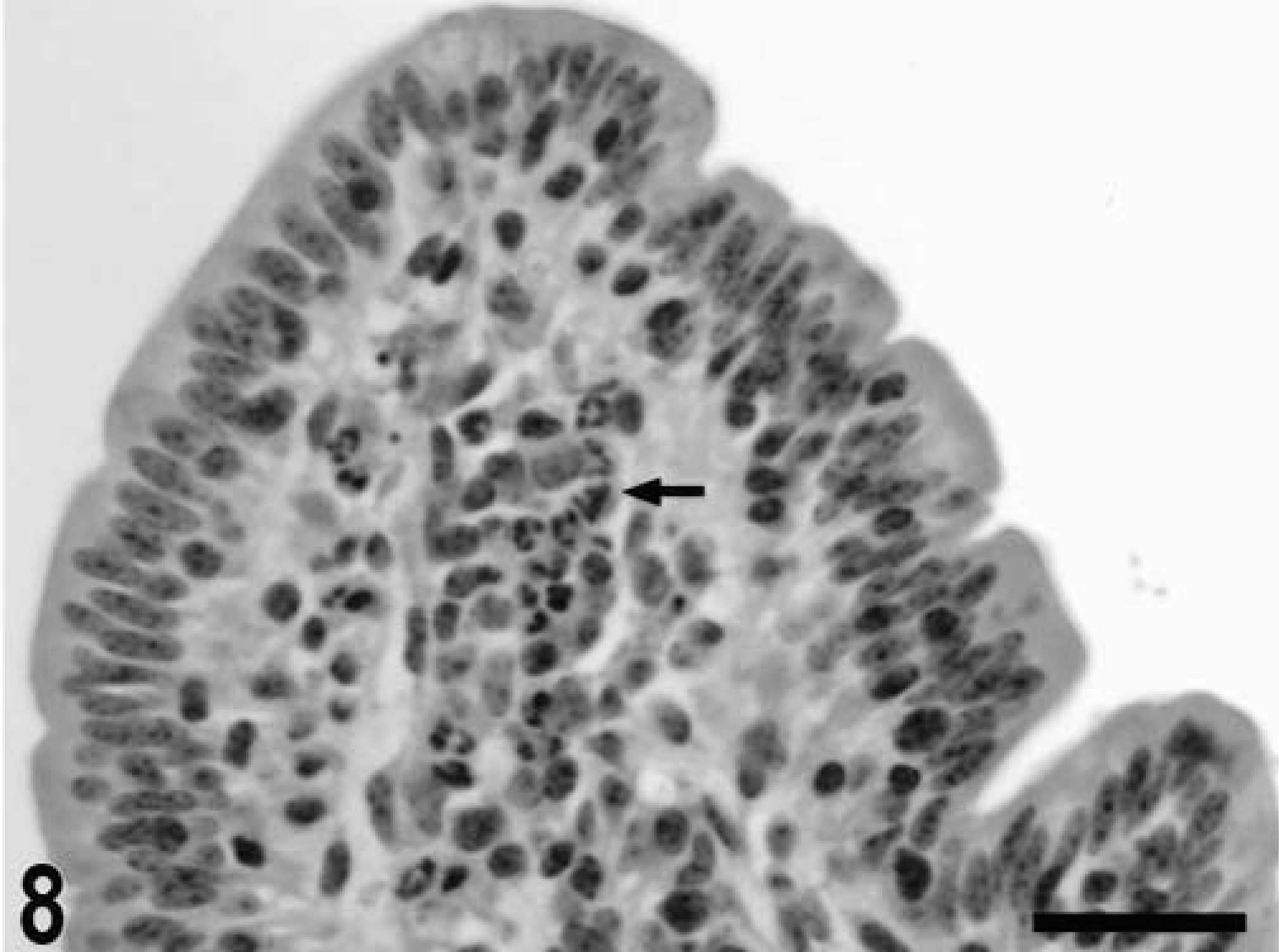
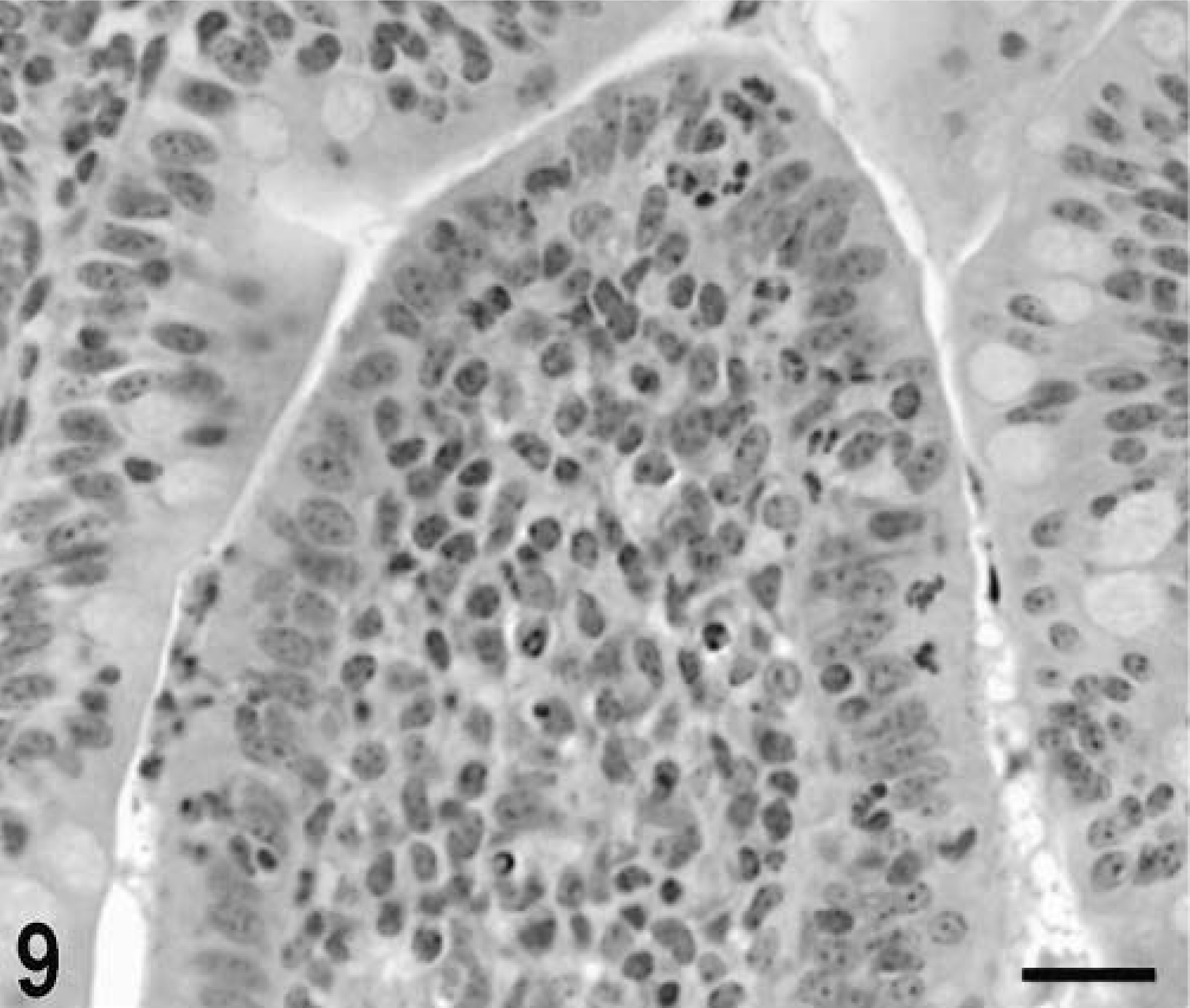
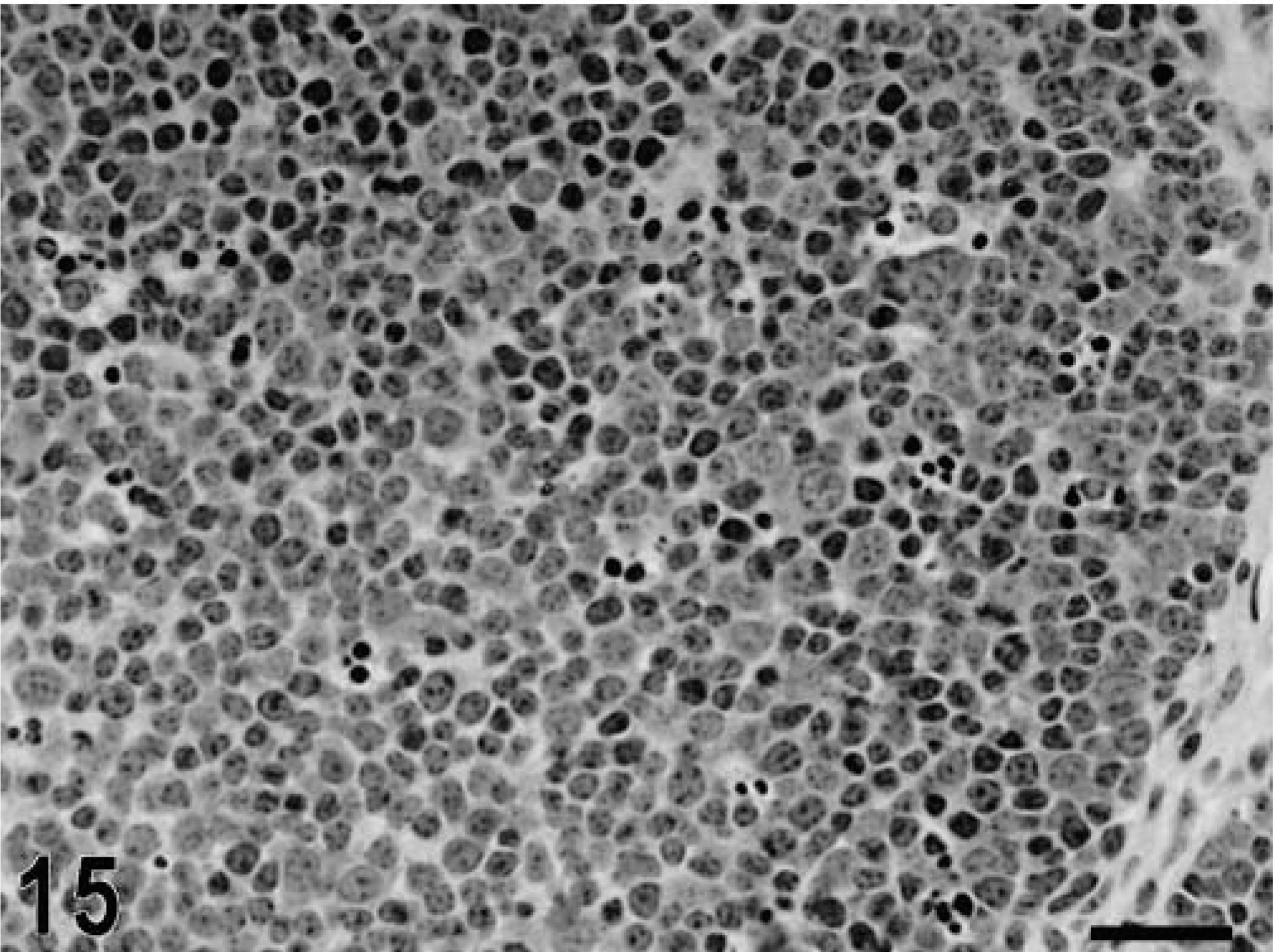
Blunting of the absorptive villi was detected in HE-stained sections as early as 15 minutes postinfection in one of the infected loops and at later time points in all infected loops (Figs. 1, 2). Blunting of absorptive villi was also evident by scanning electron microscopy (Figs. 3, 4). Detachment of epithelial cells from the tip of the absorptive villi (Figs. 5, 6) was first detected at 3 hours postinfection and progressed throughout the time course. At 10–12 hours postinfection, the epithelium covering the absorptive villi and the upper portion of the crypts was completely lost. Cytoplasmic vacuolization was observed in many enterocytes prior to detachment from the basal membrane (Fig. 7). All infected loops had mild inflammatory changes at 1 hour postinfection, which were characterized by intravascular margination and mild perivascular infiltration of neutrophils or mild diffuse infiltration of neutrophils in the lamina propria (Fig. 8). A large number of neutrophils migrating through the epithelium in the domed villi was observed as early as 15 minutes postinfection in some loops (Fig. 9) and became more prominent at later time points, with accumulation of neutrophils into the lumen adjacent to the domed villi (Fig. 10). Although transepithelial migration of neutrophils was detected in both absorptive and follicle-associated epithelium, this phenomenon was observed more frequently in the follicle-associated epithelium at the domed villi. The inflammatory changes were progressive throughout the time course, and a marked suppurative inflammatory reaction was present in all infected loops at 5 hours postinfection (Fig. 11). Progression of the inflammatory reaction was associated with massive transepithelial migration of neutrophils and accumulation of neutrophils and fibrin in the crypts and intestinal lumen (Figs. 12, 13). At 10 and 12 hours postinfection, some lymphoid follicles had a prominent intrafollicular accumulation of neutrophils, which replaced most of the lymphoid cells in the center of the follicle (Fig. 14). A moderate depletion of lymphoid cells in the lymphoid follicles was observed at 10 and 12 hours postinfection (Figs. 15, 16).
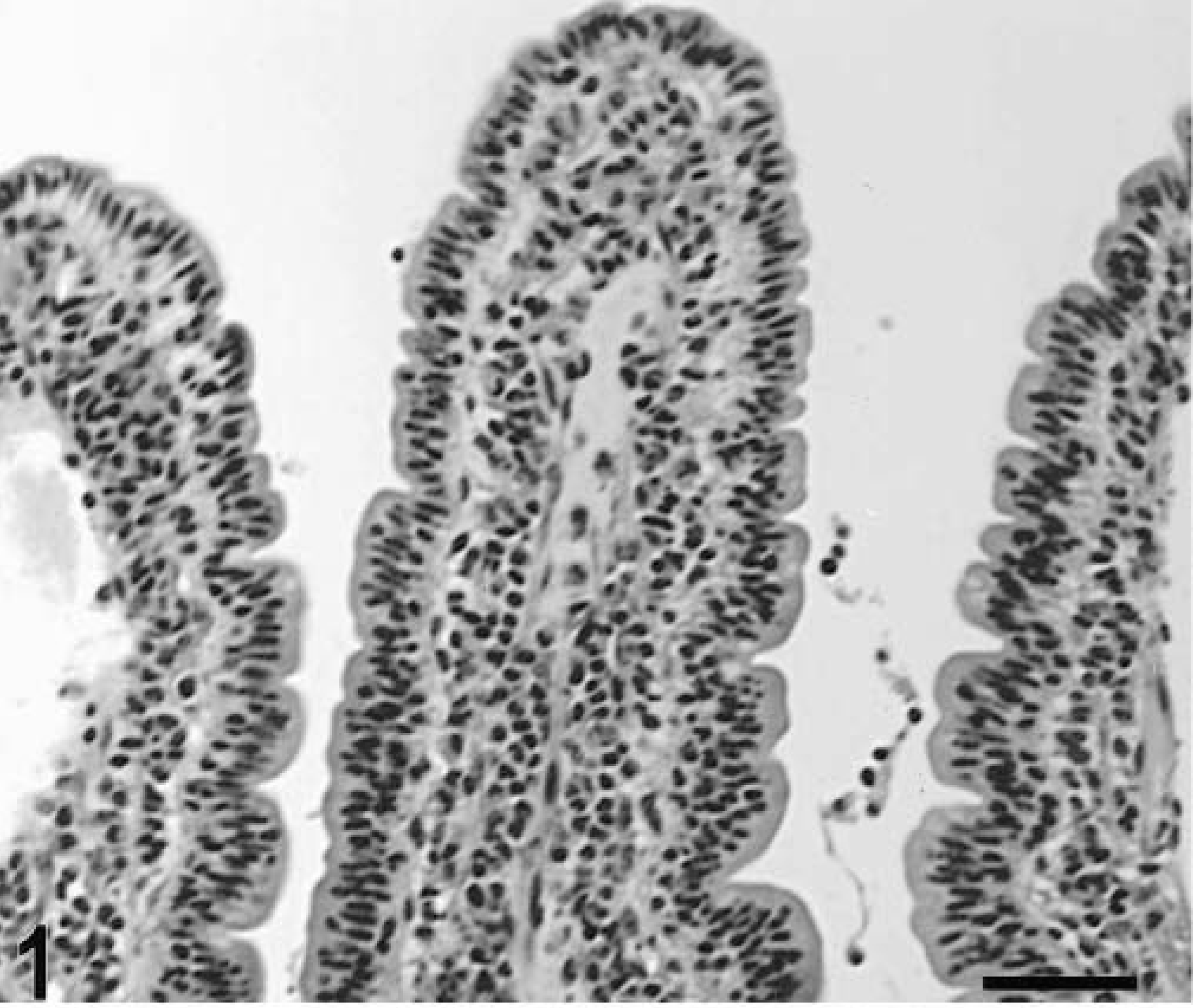
Intestine; calf, uninfected control. Normal histologic appearance of an absorptive villus in the Peyer's patches. HE. Bar = 50 μm.
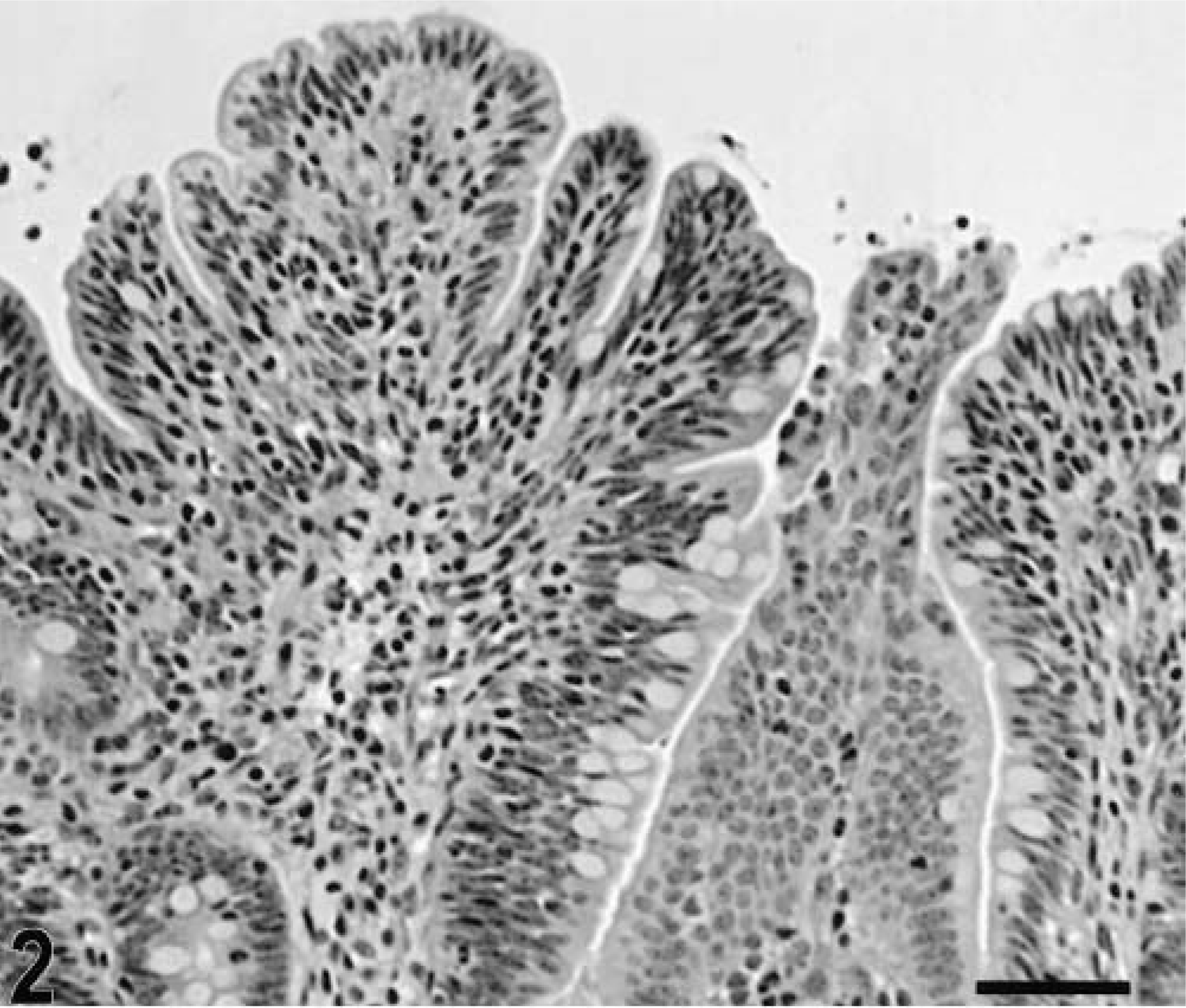
Intestine; calf, 2 hours postinfection. Blunting of the absorptive villi. HE. Bar = 50 μm.

Scanning electron micrograph. Intestine; calf, uninfected control. Normal villi over the Peyer's patches. Bar = 100 μm.
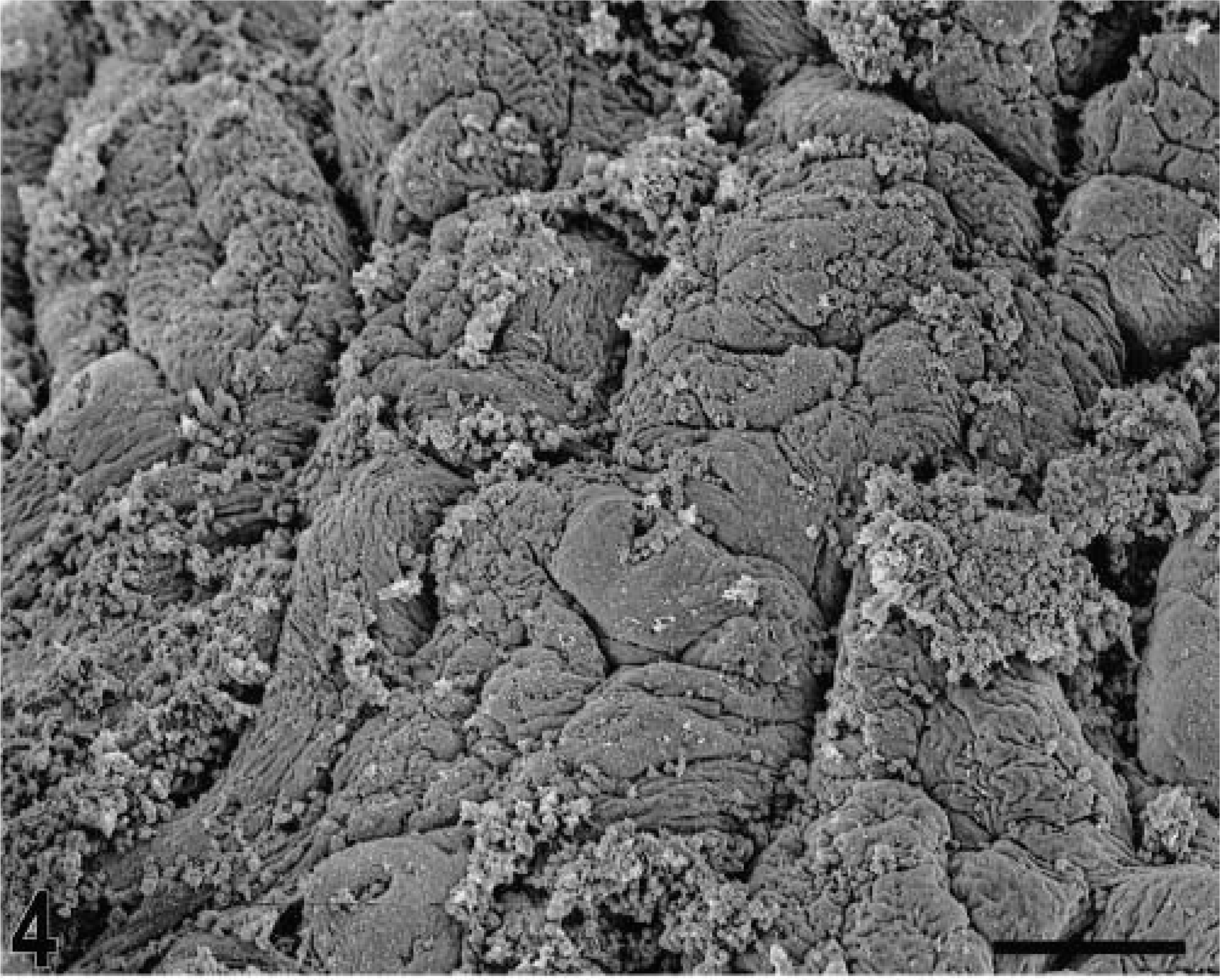
Scanning electron micrograph. Intestine; calf, 3 hours postinfection. Blunting of the absorptive villi. Bar = 100 μm.
Intestine; calf, 3 hours postinfection. Detachment of epithelial cells at the tip of an absorptive villus. HE. Bar = 30 μm.

Scanning electron micrograph. Intestine; calf, 3 hours postinfection. Detachment of epithelial cells at the tip of an absorptive villus (arrow). Bar = 30 μm.
Transmission electron micrograph. Intestine; calf, 2 hours postinfection. Severe cytoplasmic vacuolization of enterocytes. Bar = 2 μm.
Intestine; calf, 1 hour postinfection. Mild focal infiltration of neutrophils (arrow) in the lamina propria of an absorptive villus. HE. Bar = 30 μm.
Intestine; calf, 15 minutes postinfection. Migration of neutrophils through the follicle-associated epithelium in the domed villi. HE. Bar = 30 μm.
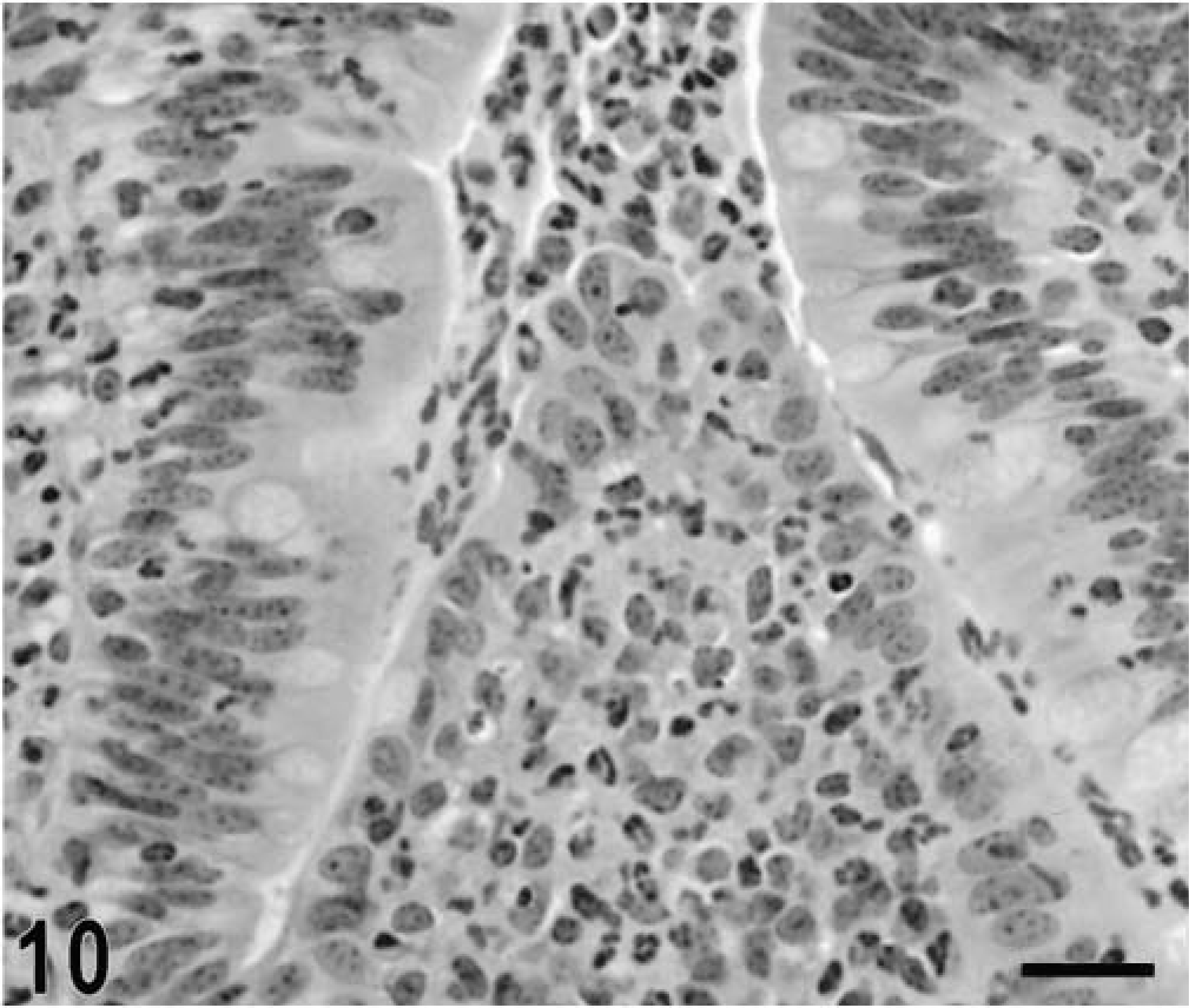
Intestine; calf, 8 hours postinfection. Intense migration of neutrophils through the follicle-associated epithelium in the domed villi, with accumulation of neutrophils into the intestinal lumen. HE. Bar = 30 μm.
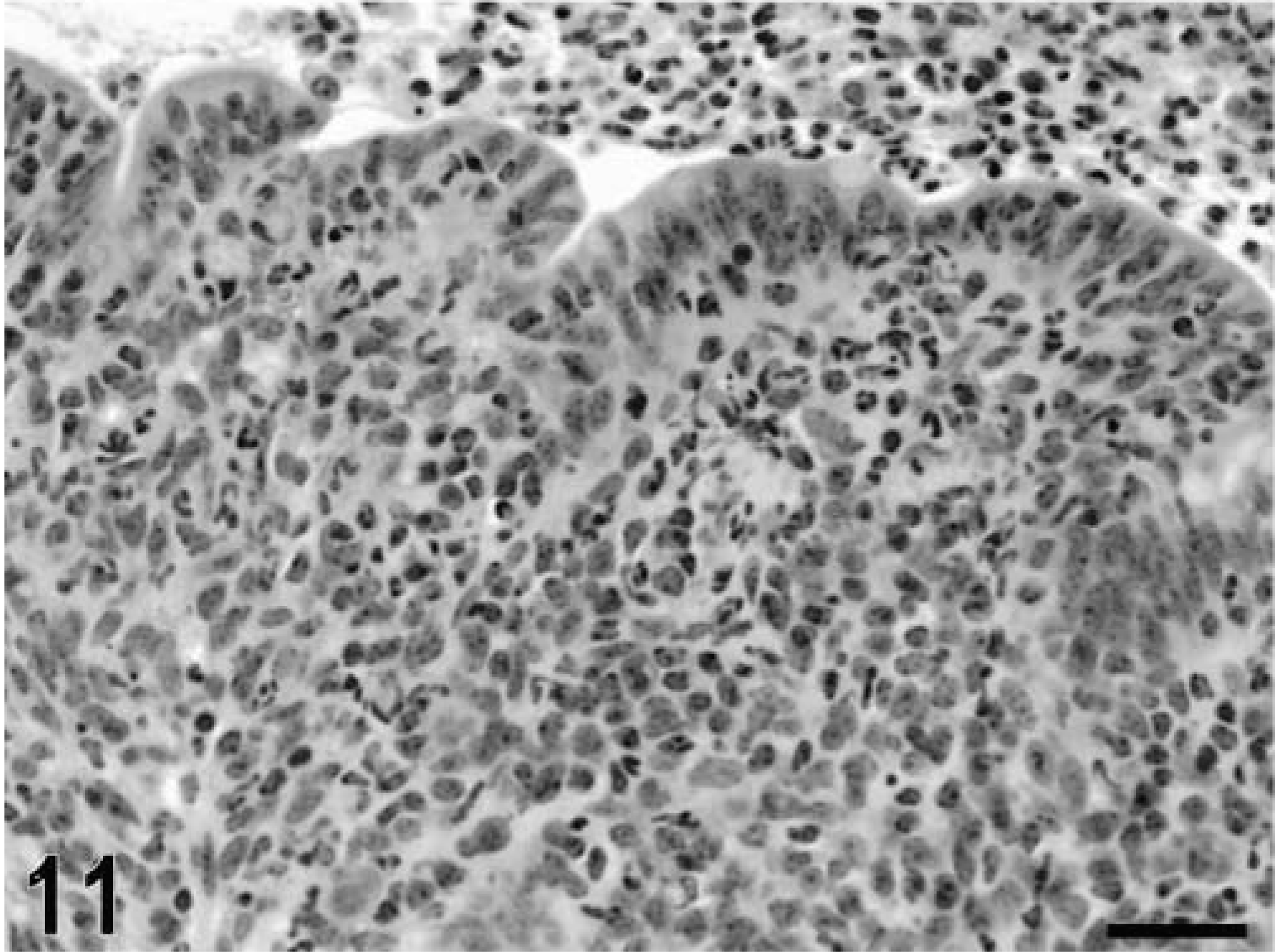
Intestine; calf, 5 hours postinfection. Diffuse infiltration of neutrophils in the mucosa and accumulation of neutrophils in the lumen. HE. Bar = 30 μm.
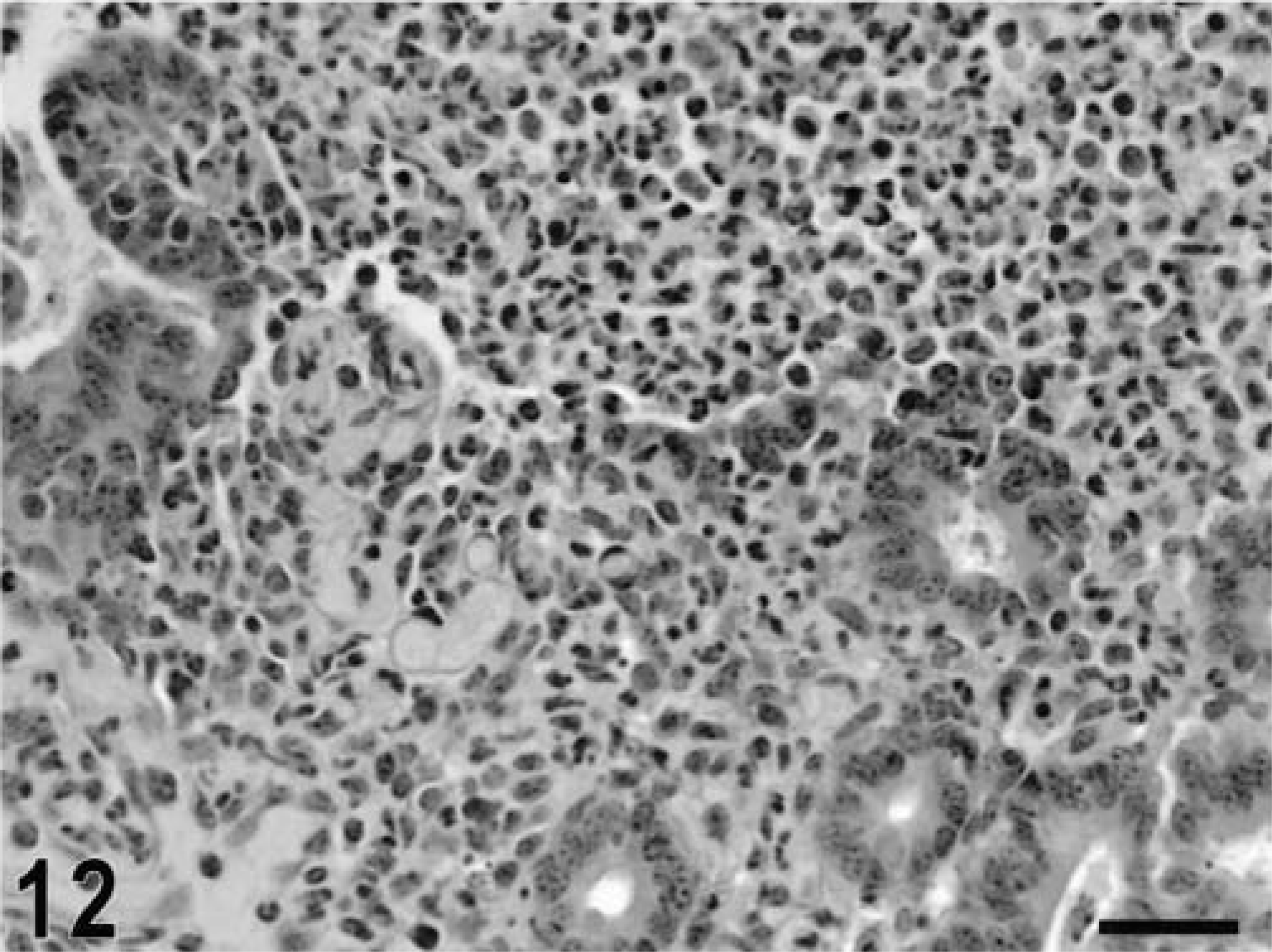
Intestine; calf, 12 hours postinfection. Severe diffuse infiltration of neutrophils in the mucosa, loss of epithelium, and accumulation of a fibrinopurulent exudate in the intestinal lumen. HE. Bar = 30 μm.

Scanning electron micrograph. Intestine; calf, 12 hours postinfection. Fibrinopurulent exudate completely covers the mucosal surface. Bar = 50 μm.
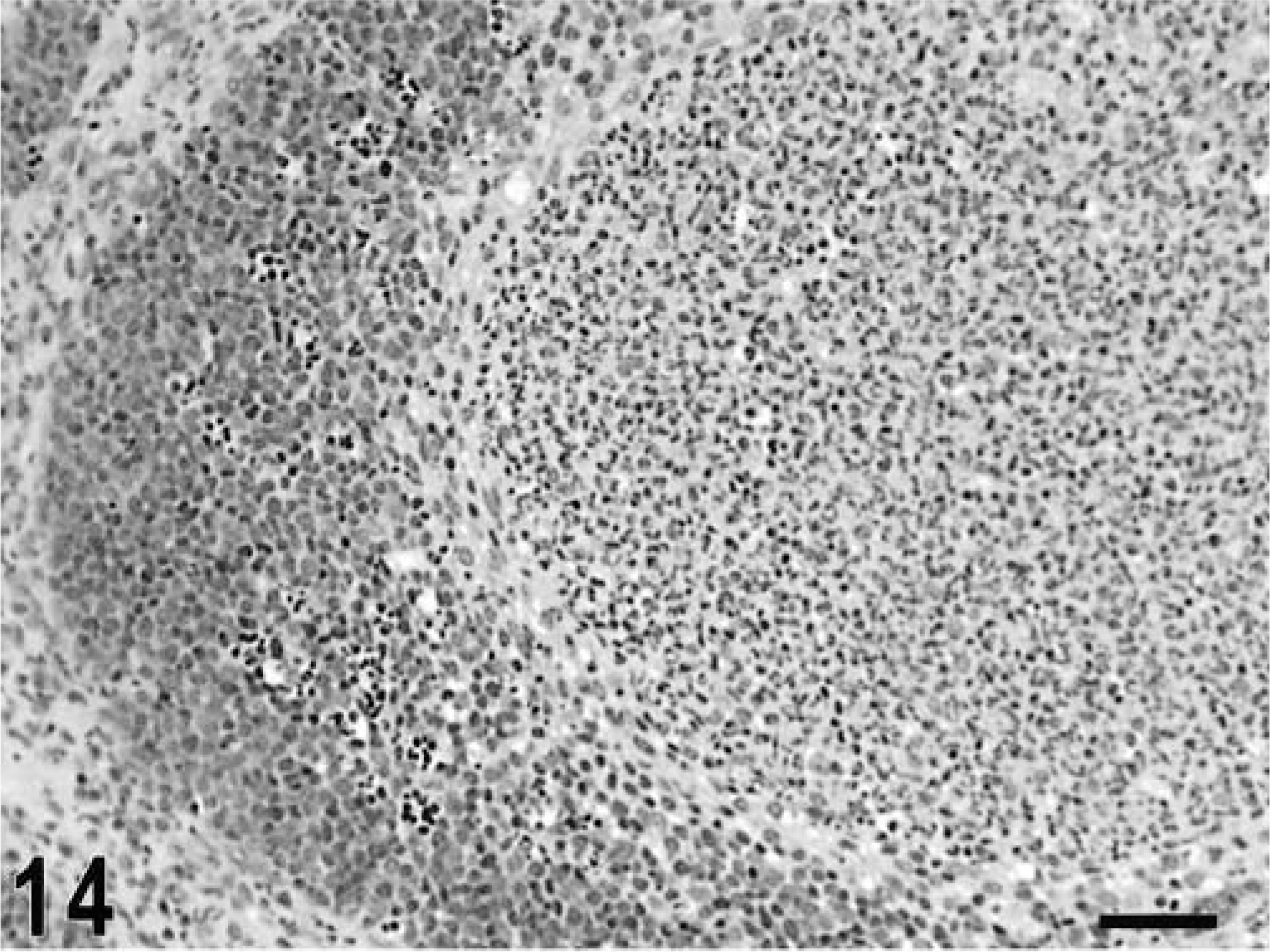
Intestine; calf, 10 hours postinfection. Intrafollicular accumulation of neutrophils replacing most of the lymphoid cells at the center of a lymphoid follicle in the Peyer's patches. HE. Bar = 50 μm.
Intestine; calf, uninfected control. Lymphoid follicle in the Peyer's patch with a dense population of lymphoid cells. HE. Bar = 30 μm.
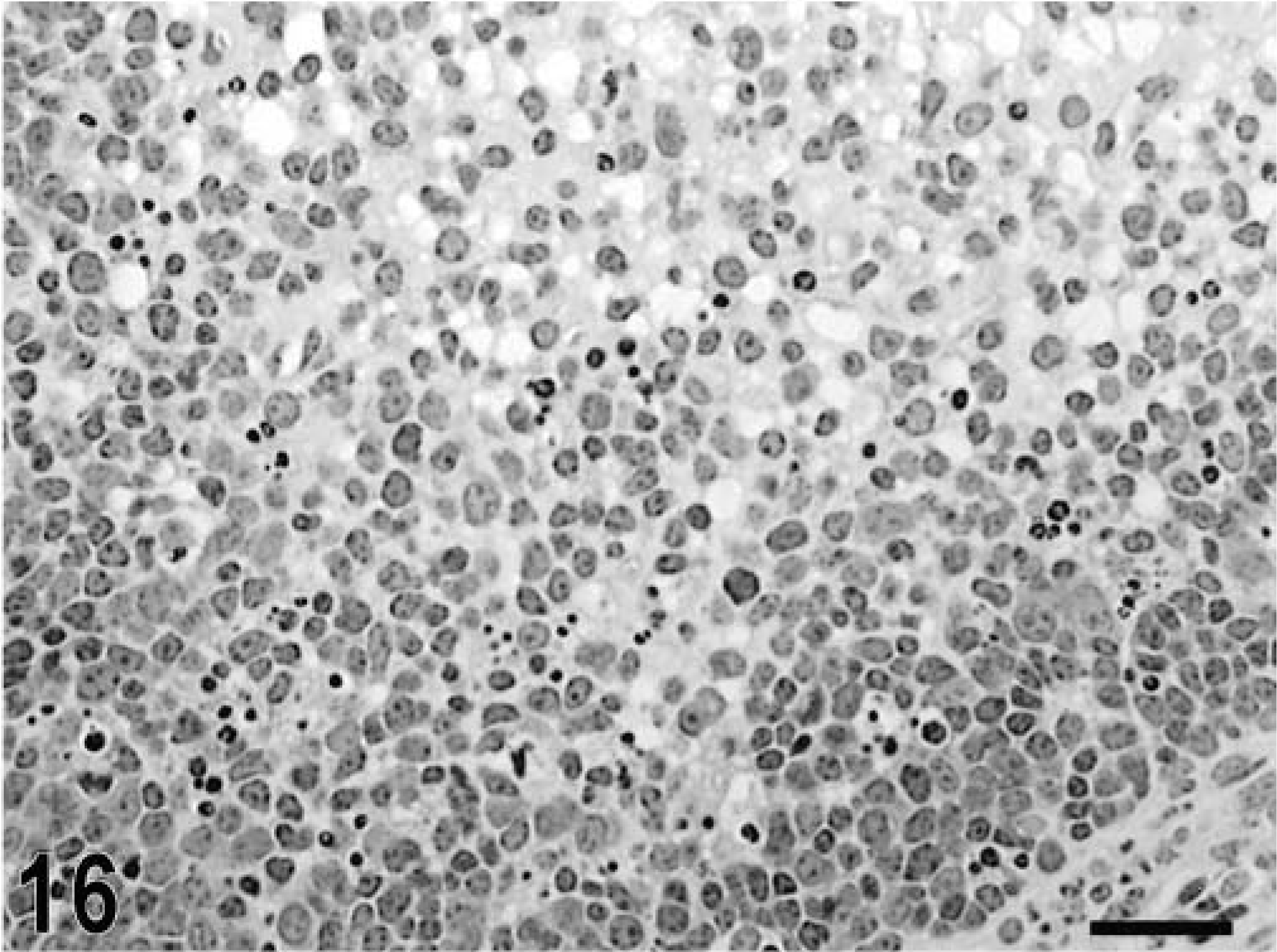
Intestine; calf, 12 hours postinfection. Depletion of lymphoid cells in a Peyer's patch lymphoid nodule. HE. Bar = 30 μm.
Ultrastructurally, invasion of epithelial cells by S. typhimurium was evident between 15 minutes and 1 hour postinfection in most of the studied sections. Both enterocytes and M cells were equally targeted for invasion and no predilection for invasion of a given cell type was detected (Figs. 17, 18). In a few preparations, invading bacteria were observed within goblet cells (Fig. 19). Bacterial attachment and invasion were often associated with the loss of microvilli and ruffling of the apical cell surface (Fig. 17). Invading bacteria were contained in membrane-bound vacuoles within epithelial cells, and eventually multiple organisms were located within a single large vacuole (Fig. 17). Intracellular bacteria were often detected within phagocytic cells in the intestinal lumen adjacent to the epithelial surface. After 1 hour postinfection, most of the bacteria within epithelial cells were located at the basal side of the cells as opposed to the apical location seen at earlier time points (Fig. 20). An increasing number of neutrophils migrating through the epithelial layer was also observed at that stage of the infection (Fig. 20). After 1 hour postinfection, bacteria were detected in the lamina propria. Almost all bacteria observed within the Peyer's patches had an intracellular location, either within mononuclear phagocytic cells or neutrophils (Fig. 21). At later time points, particularly at 10 and 12 hours postinfection, a massive infiltration of neutrophils, which was associated with breakdown of collagen fibers, was observed (Fig. 22). Some of the bacteria phagocytized by neutrophils were not morphologically intact (Fig. 23). Large numbers of cells with features of apoptosis or phagocytic cells containing apoptotic bodies were detected in both infected and control loops, mostly in the lymphoid follicles, but no bacteria were associated with these cells (Fig. 24).
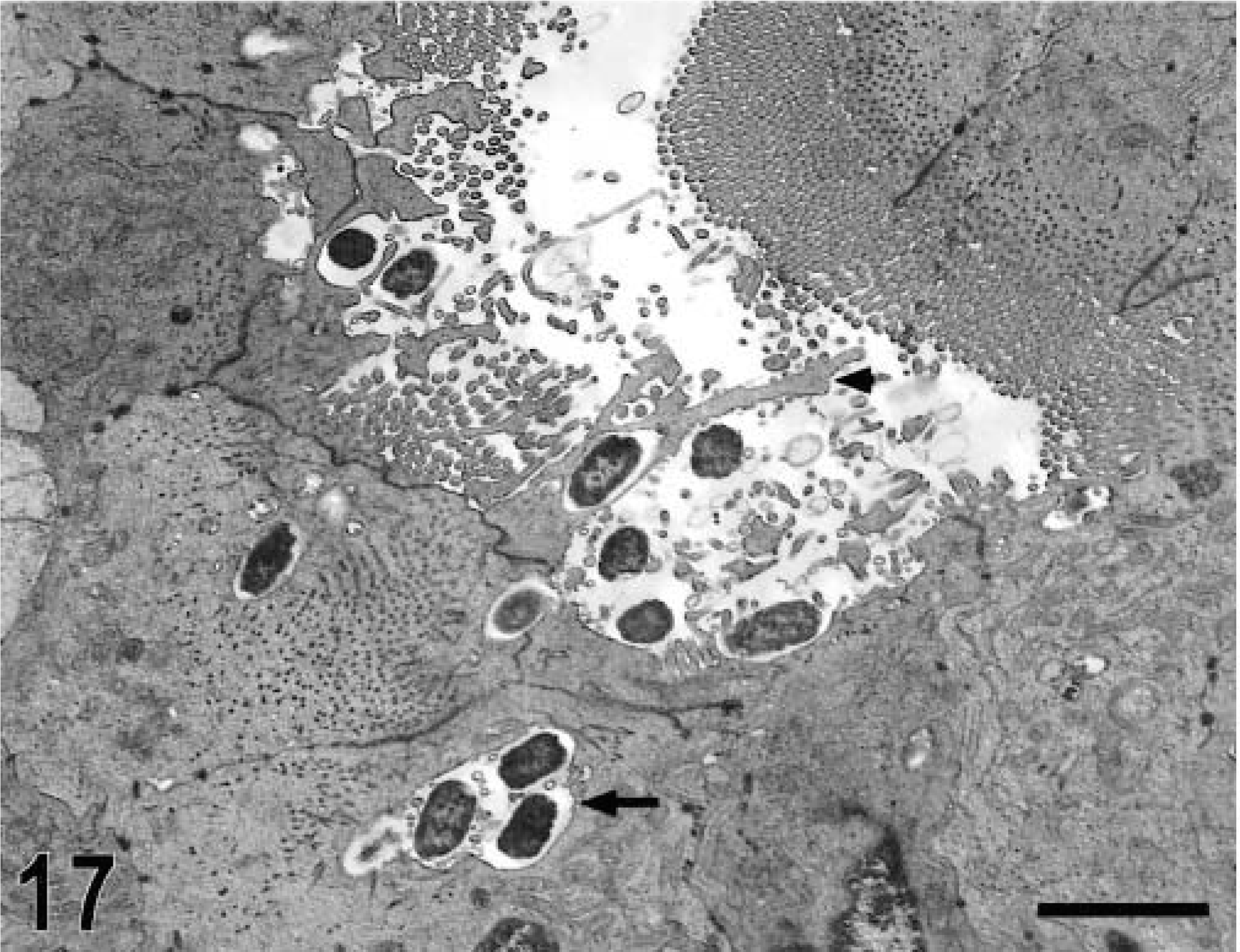
Transmission electron micrograph. Intestine; calf, 15 minutes postinfection. Bacteria in contact with brush border of an enterocyte with ruffling formation (arrow head) and internalized bacteria within membrane-bound vacuoles that may contain multiple organisms (arrow). Bar = 2.4 μm.
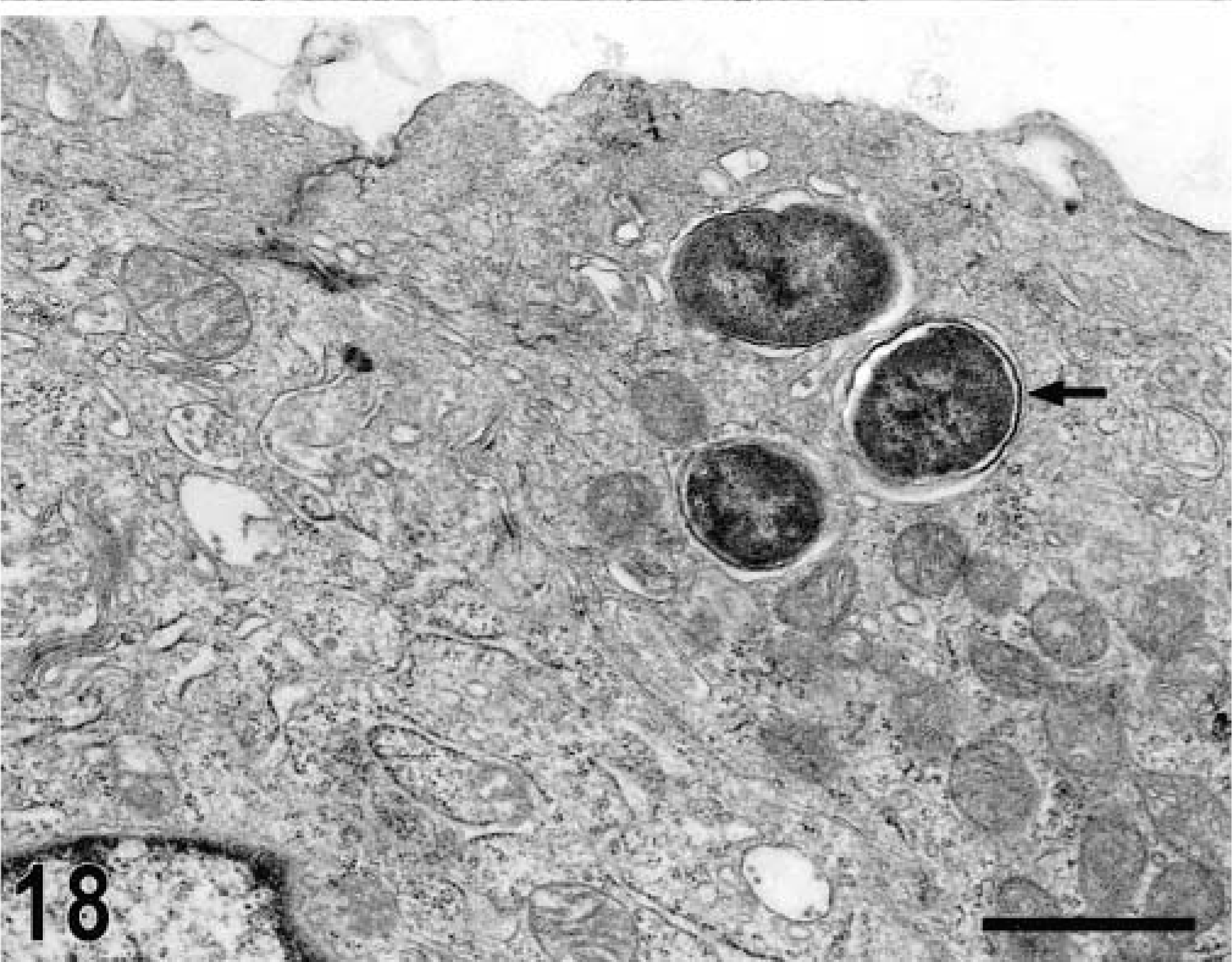
Transmission electron micrograph. Intestine; calf, 15 minutes postinfection. M cell in the follicle-associated epithelium containing internalized bacteria (arrow). Bar = 1 μm.
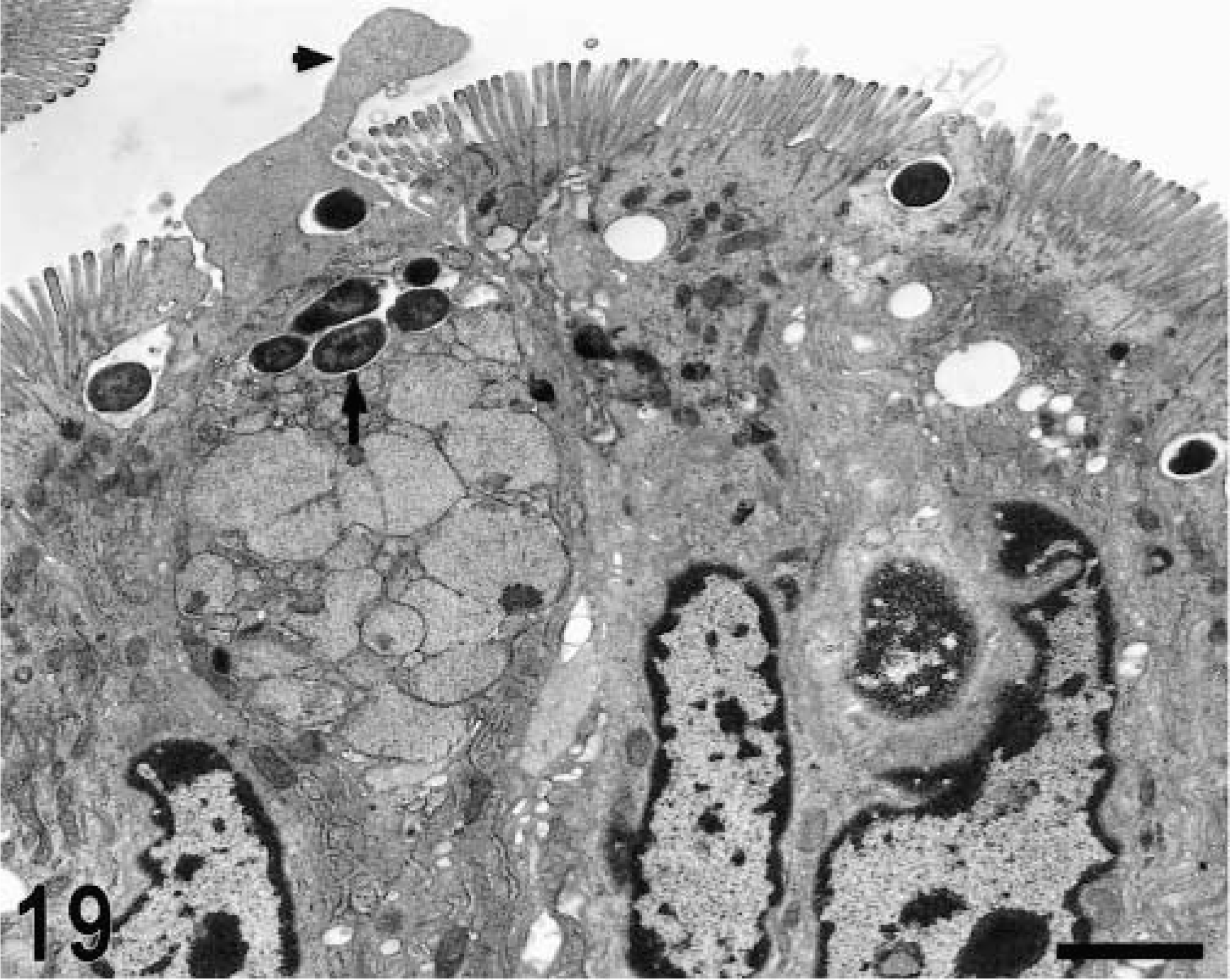
Transmission electron micrograph. Intestine; calf, 1 hour postinfection. Goblet cell in the absorptive villi with intracellular invading bacteria (arrow) and ruffling formation (arrowhead). Bar = 2 μm.
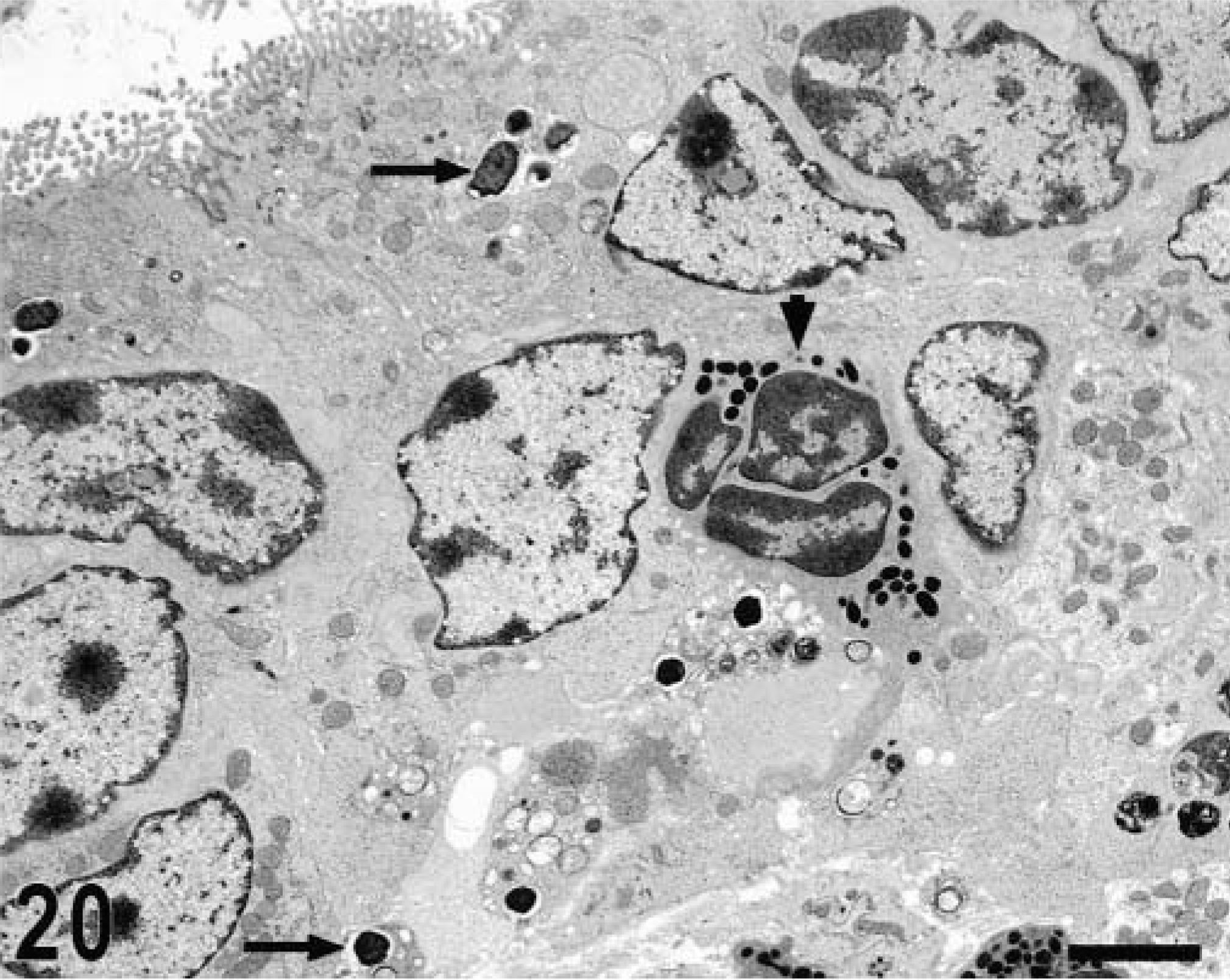
Transmission electron micrograph. Intestine; calf, 4 hours postinfection. Apical and basal location of bacteria within enterocytes (arrows) and neutrophil in the epithelial layer (arrowhead). Bar = 3 μm.
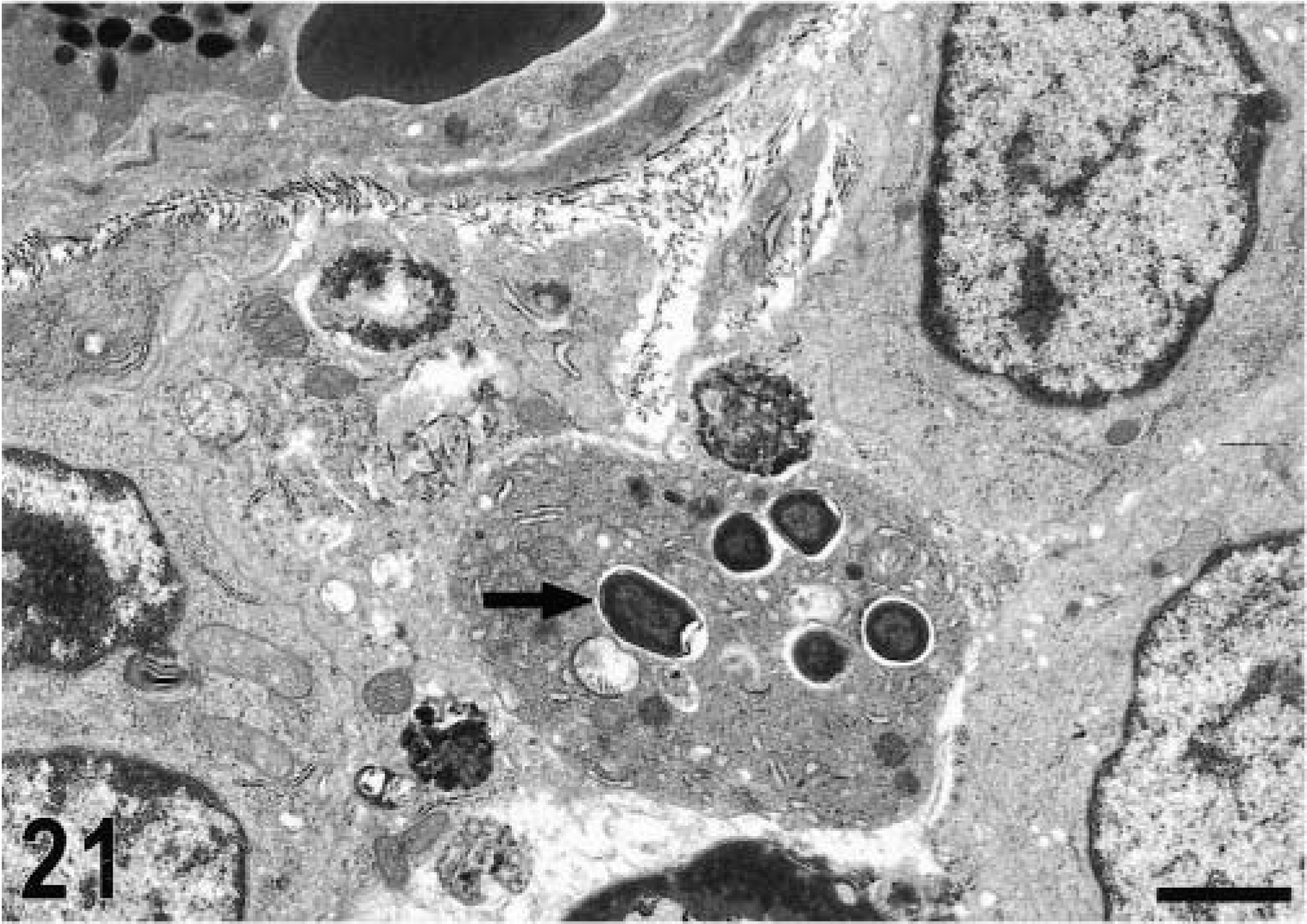
Transmission electron micrograph. Intestine; calf, 4 hours postinfection. Intracellular location of bacteria in the lamina propria (arrow). Bar = 1.5 μm.
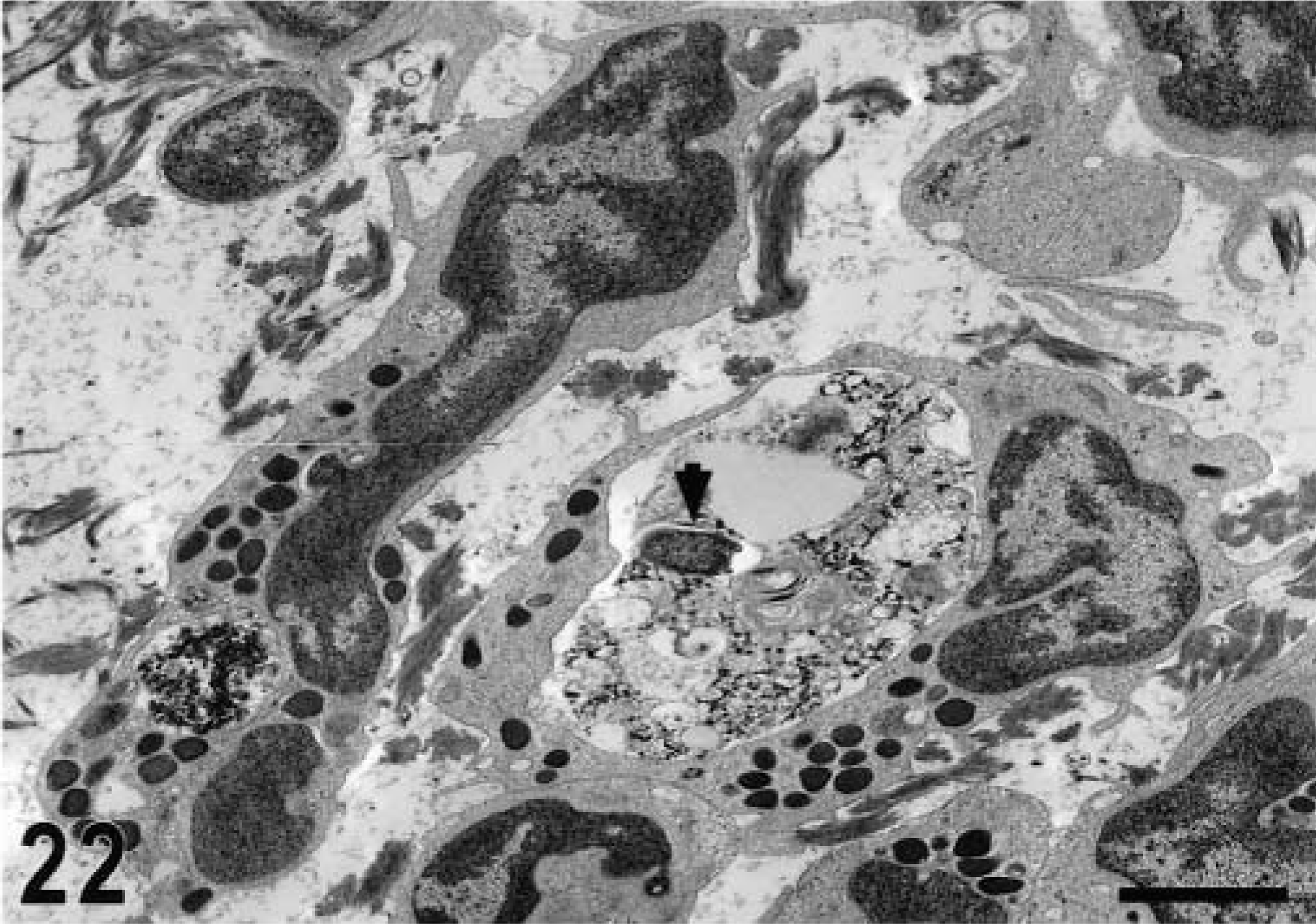
Transmission electron micrograph. Intestine; calf, 12 hours postinfection. Accumulation of neutrophils in the mucosa with a large phagocytic vacuoles containing bacteria (arrowhead). Fragmented collagen fibers are in the interstitial space. Bar = 2 μm.
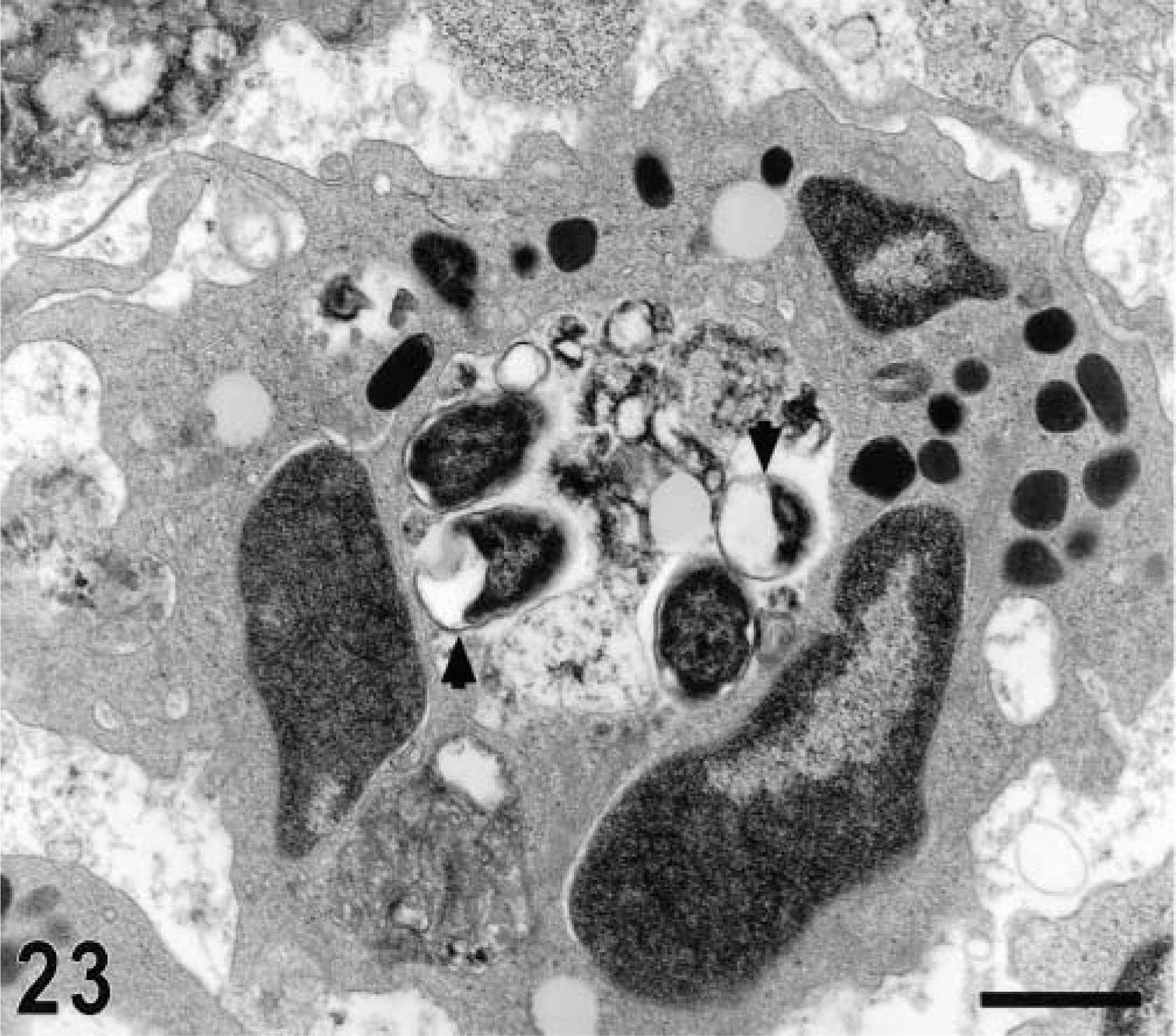
Transmission electron micrograph. Intestine; calf, 12 hours postinfection. Neutrophil containing multiple morphologically intact and fragmented organisms (arrow heads) in a large phagosome. Bar = 1 μm.
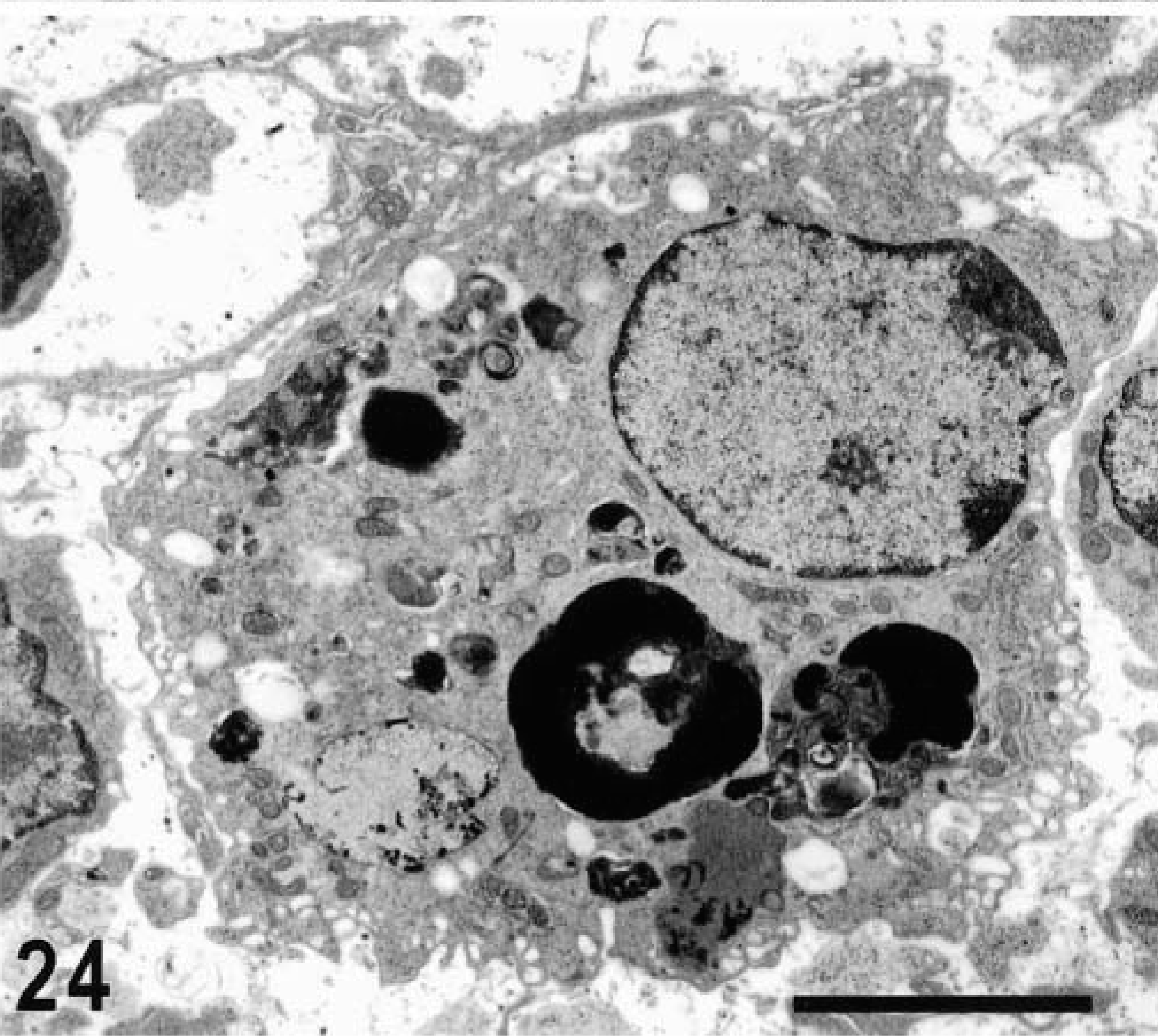
Transmission electron micrograph. Intestine; calf, 30 minutes postinfection. Phagocytic cell within a lymphoid nodule containing multiple apoptotic bodies. No bacteria were detected in association with these apoptotic cells or with phagocytic cells containing apoptotic bodies. Bar = 5 μm.
Chemokine and cytokine profile
Infection of bovine Peyer's patches with S. typhimurium resulted in a severe acute neutrophilic response, which is thought to play a significant role in the pathogenesis of Salmonella-induced diarrhea. To further characterize the host inflammatory response to S. typhimurium infection, we determined the profile of expression of chemokines attractant to neutrophils (interleukin [IL]-8, growth-related oncogene [GRO] α and γ, and granulocyte chemotactic protein [GCP]2) and that of pro-inflammatory (IL-1β, tumor necrosis factor [TNF]α, and IL-18) and anti-inflammatory (IL-10, IL-1 receptor antagonist [IL-1Ra], and IL-4) cytokines.
Expression of all the chemokines studied was increased after infection (Fig. 25). A clear difference between samples from infected tissue and those from controls was detected at 1 hour postinfection for GROγ and at 3 hours postinfection for the other chemokines (Fig. 25). Among the proinflammatory cytokines, expression of IL-1β was markedly increased at 3 and 6 hours postinfection (Fig. 26). No significant changes after infection, compared with uninfected loops, were detected in the profile of TNFα expression. IL-18 was constitutively expressed and its expression was not affected by infection (Fig. 26). With the exception of IL-10, the expression of the other anti-inflammatory cytokines (IL-1Ra and IL-4) was increased in a manner similar to that of the chemokines and of IL-1 (Fig. 27).
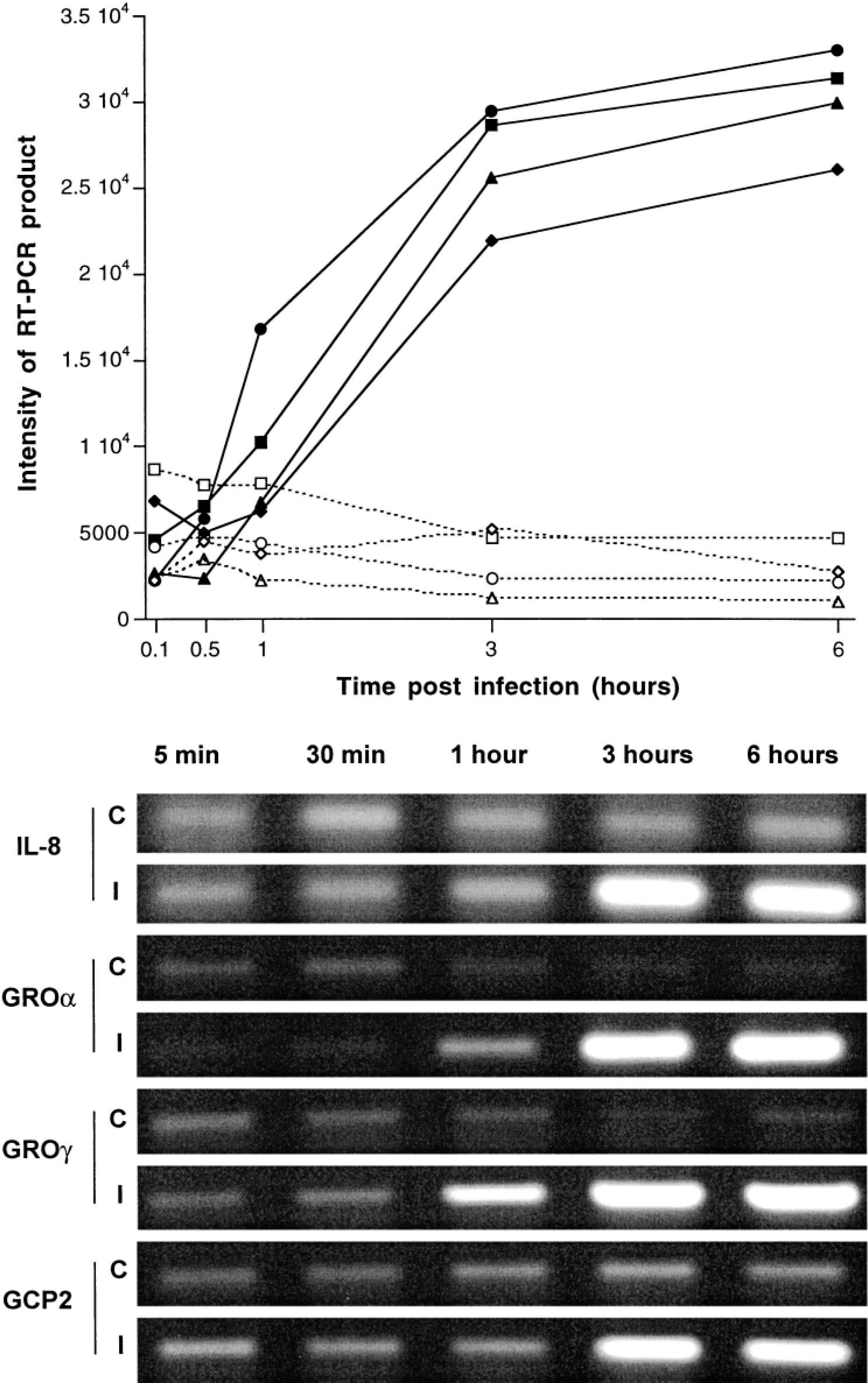
Expression of IL-8, GROα and γ, and GCP2 in bovine Peyer's patches infected with S. typhimurium and in uninfected controls at 5 and 30 minutes and 1, 3, and 6 hours postinfection. Top. Representation of expression of IL-8 (▪———▪/□— — —□ for infected and control loops, respectively), GROα (▴———▴/▵— — —▵ for infected and control loops, respectively), GROγ (••/○○ for infected and control loops, respectively), and GCP2 (♦———♦/⋄— — —⋄ for infected and control loops, respectively). Each data point represents the average of loops from 3 different calves. Intensity of RT-PCR products was determined with NIH Image software. Bottom. Agarose gel of a representative RT-PCR for detection of IL-8, GROα and γ, and GCP2 expression in control (C) and infected (I) loops.
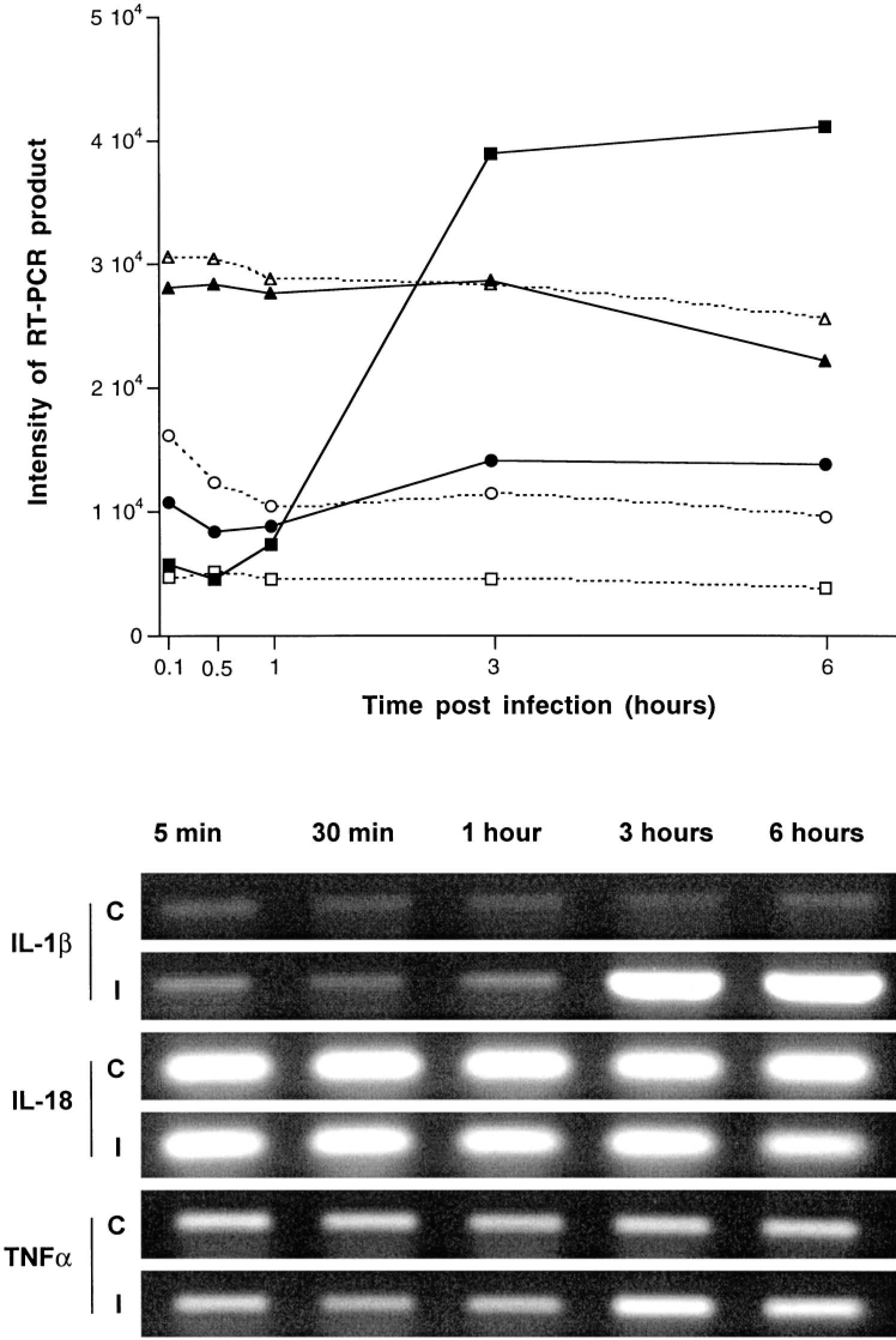
Expression of the proinflammatory cytokines IL-1β, IL-18, and TNFα in bovine Peyer's patches infected with S. typhimurium and in uninfected controls at 5 and 30 minutes and 1, 3, and 6 hours postinfection. Top. Representation of expression of IL-1β (▪———▪/□— — —□ for infected and control loops, respectively), IL-18 (▴———▴/▴— — —▵ for infected and control loops, respectively), and TNFα (•———•/○— — —○ for infected and control loops, respectively). Each data point represents the average of loops from 3 different calves. Intensity of RT-PCR products was determined with NIH Image software, Bottom. Agarose gel of a representative RT-PCR for detection of IL-1β, GROα and γ, and GCP2 expression in control (C) and infected (I) loops.
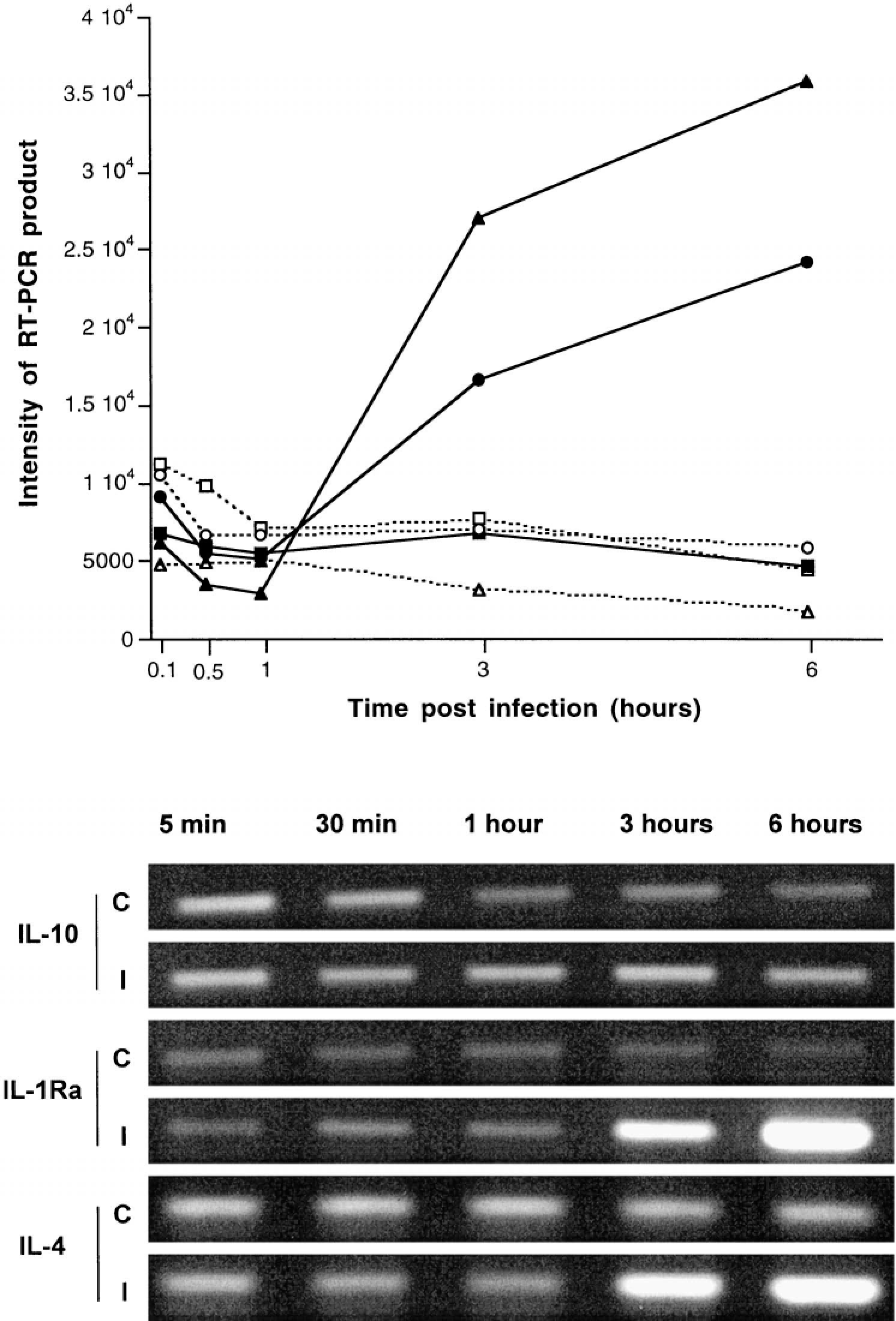
Expression of the anti-inflammatory cytokines IL-10, IL-1Ra, and IL-4 in bovine Peyer's patches infected with S. typhimurium and in uninfected controls at 5 and 30 minutes and 1, 3 and 6 hours postinfection. Top. Representation of expression of IL-10 (▪———▪/□— — —□ for infected and control loops, respectively), IL-1Ra (▴———▴/▵— — —▵ for infected and control loops, respectively), and IL-4 (•———•/○— — —○ for infected and control loops, respectively). Each data point represents the average of loops from 3 different calves. Intensity of RT-PCR products was determined with NIH Image software. Bottom. Agarose gel of a representative RT-PCR for detection of IL-10, IL-1Ra, and IL-4 expression in control (C) and infected (I) loops.
Discussion
The continuous Peyer's patches in the bovine ileum have a follicle-associated epithelium, composed mostly of M cells, that covers the domed villi. These domed villi are surrounded by absorptive villi in which the epithelium is composed of enterocytes and goblet cells.43 M cells have been demonstrated to be the target for Salmonella invasion in the mouse,7,28 and an ultrastructural study indicated that invasion of bovine M cells by S. typhimurium starts prior to the invasion of enterocytes.17 However, a subsequent study indicated that M cells are not the major route of entry in cattle.2 Our data do not support any specific cell type as main target for invasion of the intestinal epithelium in cattle; S. typhimurium invaded both M cells and enterocytes equally. Further indication for a lack of cell type specificity of S. typhimurium invasion was the finding that bacteria invaded goblet cells. The morphologic features of S. typhimurium invasion of epithelial cells were similar to those previously described for invasion of epithelial cell lines in vitro15,16 and for experimental infections in vivo.2,17,55
Our findings indicate that infection of bovine Peyer's patches by S. typhimurium results in an acute severe infiltration of neutrophils. These morphologic changes are associated with increased expression of CXC chemokines and of some pro- and anti-inflammatory cytokines. Interaction of S. typhimurium with epithelial cells in vitro results in NF-κB-dependent expression and secretion of IL-811,20,26 and other chemoattractant factors for neutrophils.21,35 Salmonella-induced IL-8 expression by epithelial cells requires an increase in cytosolic levels of calcium,20 which may result from an increase in inositol phosphate,48 and I-kB degradation through the ubiquitin proteasome pathway.38 Recently, expression of GROβ was detected in epithelial cell lines in vitro after infection with S. typhimurium 12 and increased expression of GROα and γ and IL-8 was detected in human epithelial cell lines within 2–3 hours after exposure to S. dublin.64 The present study is the first comprehensive assessment of expression of chemokines in vivo in a diarrhea model.
The marked upregulation of IL-8 expression detected in bovine Peyer's patches infected with S. typhimurium in vivo was consistent with previous data obtained with epithelial cell lines in vitro.11,20,26 Furthermore, other CXC chemokines (GROα, GROγ, and GCP2) with neutrophil chemoattractant activity29 were also upregulated after S. typhimurium infection (Fig. 21). IL-8 is thought to play a crucial role in the recruitment of neutrophils to the site of infection.4,21 We found a temporal correlation between expression of these chemokines and the infiltration of neutrophils in the infected ligated ileal loops. Although a correlation does not prove a causal relationship, this observation strongly suggests a significant role for these chemokines in the in vivo pathogenesis of Salmonella-induced enteritis. A positive correlation between neutrophilic inflammatory response and fluid secretion into the intestinal lumen has also been noted in a previous study, indicating that the inflammatory response precedes fluid secretion in bovine ligated ileal loops.50
The finding that depletion of the polymorphonuclear pool by nitrogen mustard inhibits Salmonella-induced fluid secretion in rabbits further implicates the neutrophilic inflammatory response in the pathogenesis of Salmonella-induced enteritis.23 The available data are thus consistent with a possible role of the neutrophilic reaction in the pathogenesis of Salmonella-induced diarrhea.
Among the proinflammatory cytokines studied, IL-1β had a marked increase in expression after infection with a profile similar to that of the chemokines discussed above (Fig. 22). The requirement of posttranslational cleavage for maturation of the IL-1β precursor into the active cytokine is a drawback of evaluation of IL-1β based on mRNA levels, because the precise mechanism of IL-1β secretion is not completely understood.25 A mechanism for IL-1β secretion from murine macrophages infected with S. typhimurium has been described in a cell culture system.25 According to in vitro studies, the Salmonella protein SipB is secreted into the cytoplasm of the macrophage, where it activates caspase-1 (=IL-1-converting enzyme), which cleaves and activates the IL-1β precursor. Secretion of the mature cytokine in this case is possibly linked to cell death triggered by caspase-1 activation.25 Although it remains controversial whether the Salmonella-mediated cell death is apoptotic or necrotic in nature,3,5,33,37,60 a similar mechanism of cell death, which requires both SipB and caspase-1 activation, has been described in bovine macrophages.49,60 However, in a previous study we determined that Salmonella-induced cell death is not required for the acute neutrophilic response elicited by S. typhimurium in calves.50
IL-18 neutralization inhibits neutrophil infiltration in the liver of mice treated with S. typhimurium lipopolysaccharide (LPS),39 and IL-18 has a protective effect for mice infected with virulent S. typhimurium,34 but the significance of this cytokine in a diarrhea model is still obscure. IL-18, which is also substrate for caspase-1 and has to undergo posttranslational cleavage for activation,22 was constitutively expressed, and no significant changes in its expression were observed upon infection. However, because IL-18 requires maturation, the possibility that an increase in maturation and secretion of this proinflammatory cytokine is triggered during S. typhimurium infection can currently not be excluded.
Unlike IL-1β, TNFα did not show a marked increase in expression after infection. IL-1 and TNF are potent proinflammatory mediators that act synergistically.10 Our results are in agreement with those of a previous report that a positive blood culture after oral infection of calves with S. typhimurium does not result in increased TNFα levels in the peripheral circulation.44 In contrast, TNFα has been detected in stools of children with diarrhea caused by S. typhimurium.31
In addition to their immunomodulatory functions, IL-4 and IL-10 exert anti-inflammatory effects by inhibiting expression and release of proinflammatory cytokines such as TNF and IL-1, whereas IL-1Ra blocks the action of IL-1 through competition for the receptor.41 No significant increase in IL-10 expression was detected in the present study, but IL-4 showed an increased expression in the infected loops at 3 and 6 hours postinfection. IL-4 expression at these early stages of infection may function as a modulator of the inflammatory response; mice harboring mutations in the IL-4 gene (IL-4(−/−)) have a tendency to develop microabscesses.13 In the present study, the profile of IL-1Ra expression was similar to that of IL-1β. Although expression of IL-1β and IL-1Ra may be differentially regulated, LPS stimulates expression of both.41 This result suggests that S. typhimurium does not induce downregulation of IL-Ra expression in bovine ileal ligated loops, as reported for Shigella flexneri infection in ligated loops in rabbits.1
The inflammatory response in calves is characterized by a rapid neutrophilic infiltrate, whereas mice infected with S. typhimurium develop a slow inflammatory response composed predominantly of mononuclear cells.51 Our data suggest that these striking differences in the host response to infection are a reflection of the marked differences in the cytokine profile elicited during S. typhimurium infection of mice and calves. A previous study on the cytokine response elicited in murine ligated ileal loops infected with S. typhimurium showed upregulation of TNFα and IL-10 mRNA expression, but expression of IL-4 was not altered.30 In contrast, expression of IL-4 was upregulated in bovine ligated ileal loops infected with S. typhimurium, but no changes in expression of TNFα and IL-10 mRNA were observed (Figs. 26, 27). These findings underscore the importance of using an animal for the study of Salmonella-induced enterocolitis, which develops similar pathology and signs of disease as the human host infected with S. typhimurium.
Infection of bovine ligated ileal loops with S. typhimurium results in a profound acute neutrophilic inflammatory response that is associated with upregulation of CXC chemokines IL-1β, IL-1Ra, and IL-4. Considering that some of the proteins secreted by S. typhimurium play an active role in eliciting an inflammatory response,32,50,56,58,62 the present study provides the background information to evaluate the effect of these proteins on chemokine/cytokine expression, which may explain the mechanisms by which S. typhimurium actively stimulates the host inflammatory response. Such knowledge would be valuable for understanding the pathogenesis of diarrhea and eventually for identification of new targets for therapeutic intervention.
Footnotes
Acknowledgements
This work was supported by grant DHHS/PHS/NIH-1 RO1 A144170 from the National Institutes of Health and Texas Agricultural Experiment Station project 8409. R. L. Santos was supported by CAPES, Brasília, Brazil, and Universidade Federal de Minas Gerais, Belo Horizonte, Brazil. We thank Thomas Stephens of the Texas A&M Electron Microscopy Center, Miles Frey, Rosemary Vollmar, Melissa Kahl, and Denise Santos for technical assistance and Colin Tanksley and Alan Patranella for care of animals.
