Abstract
Salmonella enterica serovar Typhimurium is an important intestinal pathogen in swine. This study was performed to document the early cellular invasion of Salmonella serovar Typhimurium in swine ileum. Ileal gut-loops were surgically prepared in ten 4- to 5-week-old mixed-breed pigs and inoculated for 0-60 minutes. Loops were harvested and prepared for both scanning and transmission electron microscopy (SEM and TEM, respectively). Preferential bacterial adherence to microfold cells (M cells) was seen within 5 minutes, and by 10 minutes bacterial invasion of the apical membrane was seen in M cells, goblet cells, and enterocytes. This multicellular invasion was observed throughout the course of infection. In addition, SEM revealed a specific affinity of Salmonella serovar Typhimurium to sites of cell extrusion. Using TEM, bacteria in these areas were focused in the crevices formed by the extruding cell and the adjacent cells and in the cytoplasm immediately beneath the extruding cell. Our results suggest that early cellular invasion by Salmonella serovar Typhimurium is nonspecific and rapid in swine. Furthermore, the combination of SEM and TEM data suggests that Salmonella serovar Typhimurium may use sites of cell extrusion as an additional mechanism for early invasion.
The pig is a good model for studying the pathogenesis of systemic and enteric human salmonellosis because it is susceptible to both host-adapted and non–host-adapted serovars that clinically manifest as distinctly different diseases.22,29 In swine, most clinical cases of salmonellosis are caused by Salmonella enterica serovars Choleraesuis or Typhimurium (hereafter referred to as Salmonella serovar Choleraesuis and Typhimurium, respectively).22,23 Salmonella serovar Choleraesuis is host adapted to swine, causing a systemic, typhoid-like disease. Salmonella serovar Typhimurium is not host adapted to swine, and infection leads to a localized enterocolitis.
Salmonella serovar Typhimurium phagetype DT104 is isolated with increasing frequency among clinical cases of Salmonella serovar Typhimurium. It is associated with multiple antibiotic resistance and is a major public health concern.2
Sites of early Salmonella cellular invasion in pig ileum are not well defined. Previous work has shown that Salmonella serovar Typhimurium can be seen 24 hours after oral inoculation in phagocytic vacuoles of ileal enterocytes and in the interepithelial space. No bacteria were seen within or adherent to microfold cells (M cells).19 In inoculated swine ileal gut-loops, Salmonella enterica serovars Heidelberg and Typhimurium organisms were found intracellularly within 2 hours after inoculation. The intracellular organisms were in the apical cytoplasm of enterocytes, but penetration by the organisms was not demonstrated. Examination of the 2-, 4-, and 6-hour Salmonella serovar Choleraesuis loops revealed no intracellular bacteria in ileal epithelium or phagocytes.21,22 In another study conducted 90 minutes after inoculation, Salmonella serovar Choleraesuis organisms were documented in M cells, absorptive enterocytes, and goblet cells. The authors concluded that Salmonella serovar Choleraesuis could be internalized by enterocytes removed from active M-cell foci; however, definitive statements concerning apical invasion or cellular kinetics of infection could not be made.4
Previous studies assessing Salmonella invasion in swine have used time points of 90 minutes or more. But a bovine gut-loop model suggests that invasion can occur within minutes.7 In addition, it has been suggested that after apical invasion, Salmonella have the capacity for basolateral invasion of adjacent cells.10,25 In pig, lack of early time points has prevented definitive statements from being made concerning Salmonella cellular invasion. The purpose of this study was to identify sites of early cellular invasion by Salmonella serovar Typhimurium in ileal epithelium of the swine gut-loop model.
Materials and Methods
Bacteria preparation
Swine isolates included Salmonella serovar Typhimurium phagetype DT104 (NVSL 98-745), a clinical swine isolate obtained from the National Veterinary Services Laboratories (Ames, IA), and Salmonella serovar Choleraesuis var. kunzendorf strain χ3246 (courtesy of Dr. Roy Curtiss III, Washington University, St. Louis, MO). Luria-Bertani (LB) cultures (5 ml) were inoculated from 20% glycerol stocks stored at −70 C. Cultures were grown at 37 C, with shaking at 220 rpm for 7 hours. Next, 10 µl of the 7-hour growth was inoculated into 5 ml of LB broth and grown statically for 16 hours at 37 C. At that time, the cultures were placed on ice, and a sample was taken for serial dilutions and subsequent plating on tryptose agar. The final inoculum concentration for Salmonella serovar Typhimurium was 8 × 108 colony-forming units (CFU)/ml, and for Salmonella serovar Choleraesuis it was 6 × 108 CFU/ml.
Animals
Ten mixed-breed pigs were obtained at approximately 10 days of age and grown in isolation facilities at the National Animal Disease Center (NADC), Ames, Iowa. The experiments were also performed at this center. The pigs were rectal-culture negative for Salmonella in the weeks before the experiment. For the experiment, 4- to 5-week-old pigs were starved for 18 hours before the surgery.
Surgical procedures
Anesthesia for terminal surgery was induced by intramuscular injection of Telazol™ (tiletamine/zolazopam, 18 mg/kg, Fort Dodge Labs, Ft. Dodge, IA), Ketaset™ (ketamine, 24 mg/kg, Fort Dodge Labs), and Rompun™ (xylazine, 12 mg/kg, Bayer Animal Health, Shawnee Mission, KS). After the surgical plane of anesthesia was reached and the abdomen prepared, a midline abdominal incision was made and the ileocecal junction exteriorized. Surgical loop procedures were adapted from previous descriptions.21,28 In brief, within 10–15 cm of the ileocecal junction and working cranially, the ileum was ligated (#2 silk) to form 6-cm test loops, each separated by 2-cm spacer loops. Care was taken not to compromise the blood supply and to ensure the presence of Peyer's patch. The test loops were injected with 6 ml of either sterile LB or bacterial inoculum (Table 1). The ileum was then replaced and the abdomen closed. At the appropriate time point the abdomen was reopened and the loops exteriorized. Each loop was injected to moderate distension with fixative (2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer, pH 7.2). The loop was harvested and placed in a fixative bath. The pig was then euthanatized with intravenous sodium pentobarbital.
Pig number, time point, and loop procedure for evaluation of early invasion by Salmonella serovar Typhimurium.

∗ Time points are minutes after inoculation.
† Describes the number of loops formed and inocula used. ST = Salmonella serovar Typhimurium; LB = Luria-Bertani broth; SC = Salmonella serovar Choleraesuis.
For the modified-loop procedure the ileum was exteriorized and the ileum gently occluded by digital pressure near the ileocecal junction and ∼ 60 cm cranially. To facilitate adequate cellular exposure, the Salmonella serovar Typhimurium DT104 inoculum was increased to 20 ml and injected into the lumen, and the ileum was gently rocked. The functional occlusions were removed and the ileum replaced into the abdomen. After 30 minutes the ileum was exteriorized, and at 30 cm cranial to the ileocecal junction a 6-cm portion of the ileum was formed into a loop and immediately injected with the fixative. The loop was harvested and placed in the fixative.
Transmission electron microscopy procedures
After 1–2 hours in the fixative, the loops were opened, and a 3- to 4-mm-wide cross section of the loop was removed and placed into fresh fixative. The samples were refrigerated (4 C) until further processing, usually within 48 hours. Sample preparation was performed as previously described.1 In brief, 1-mm blocks were stained in 1% osmium tetroxide, dehydrated in alcohols, cleared in propylene oxide, and embedded in epoxy resin. Then, 1-μm sections were cut with an ultramicrotome, stained with toluidine blue, and examined under a light microscope. Thin sections were cut from blocks and placed on nickel grids. This was then stained with 5% uranyl acetate and Reynolds lead acetate for contrasting and viewed under a Philips 410 transmission electron microscope (NADC, Ames, IA).
Scanning electron microscopy procedures
After 1–2 hours in the fixative, the loops were removed and opened. A 1- × 1-cm section each of both Peyer's patch and non–Peyer's patch ileum was removed and lightly agitated in the fixative to wash off the debris. This was kept cool (4 C) until further processing, usually within 48 hours.
Scanning electron microscopy (SEM) sample preparation was modified from previous descriptions.11,13 In brief, the tissues were postfixed with 1% osmium tetroxide and further treated with 1% thiocarbohydrazide. These steps were repeated twice. The samples were dehydrated through a series of graded ethanol baths and then critical point dried. Tissues were mounted on aluminum stubs with colloidal silver and sputter-coated with gold-palladium for a final coating of approximately 300 Å. Samples were viewed at the Bessey Microscopy Facility (Iowa State University, Ames, IA) on a JSM-5800LV (Japanese Electronic Optical Laboratories, Akishima, Japan) scanning electron microscope at 10 kV.
Experimental design
Ten pigs (Table 1) were randomly assigned to a time point (0, 5, 10, 15, 20, 25, 30 [n = 2], 45 and 60 minutes after inoculation). Four 6-cm loops were prepared in each pig (except the 0- and 30-minute modified gut-loop) to accommodate both SEM and transmission electron microscopy (TEM) processing. Two of the loops received sterile LB, and the other two received Salmonella serovar Typhimurium DT104. A time “0-minute” point was defined as a loop being injected with the fixative immediately after formation. An additional 30-minute time point was allotted for the modified–gut-loop procedure described above. For the 60-minute time point, two additional loops were made for Salmonella serovar Choleraesuis for SEM and TEM processing.
Results
SEM examination
In pig, the follicle-associated epithelium (FAE) villi were shorter than the adjacent absorptive epithelium (AE) villi. FAE villi were seen in the gaps between AE villi and also on the cut edge of the sample. On FAE, M cells and enterocytes were the predominant cell types, with a noted absence of goblet cells. The M cells were distinguished by microvilli, which often appear thicker and more irregular compared with adjacent enterocyte microvilli (Fig. 1).

Ileum, sterile media–inoculated loop, 5 minutes after infection; pig No. 2. Scanning electron micrograph, apical surface of M cells (m) and enterocytes (e) on follicle-associated epithelium. Gold-palladium. Bar = 10 μm.
Bacteria were seen in the intervillus space at all time points (5–60 minutes) of the infected loops and rarely in the controls. Cellular extrusion was seen at the luminal tip of some villi. Additionally, scattered individual cellular extrusion sites marked by cell swelling were noted at various heights of the villi and could be seen in both FAE and AE. This type of cellular extrusion was found with similar frequency in both sterile LB and Salmonella serovar Typhimurium loops. For the 0-minute time point, special emphasis was placed on the evaluation of sites of cellular extrusion. Sites that are similar in frequency and character to the 5-minute time point were seen (Table 2). The extrusion rate over 0–60 minutes increased from 0.4 to 1.5 extrusions per villus for the LB loops, whereas the Salmonella enterica serovar Typhimurium loops increased from 0.4 to 1.7 extrusions per villus.
Observed number of cell extrusions and associated bacteria over time (minutes).
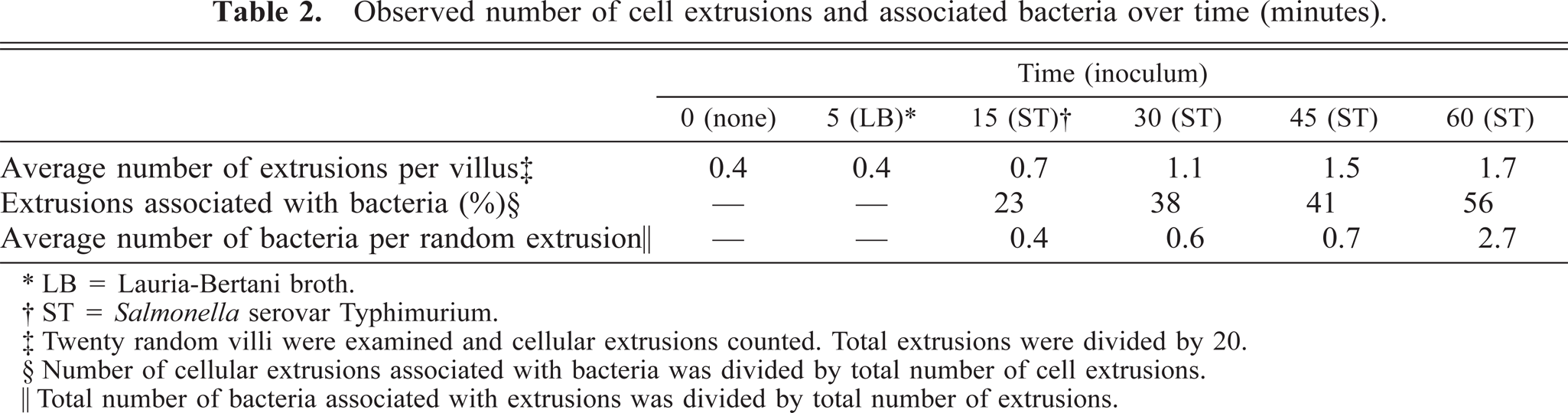
∗ LB = Lauria-Bertani broth.
† ST = Salmonella serovar Typhimurium.
‡ Twenty random villi were examined and cellular extrusions counted. Total extrusions were divided by 20.
§ Number of cellular extrusions associated with bacteria was divided by total number of cell extrusions.
‖ Total number of bacteria associated with extrusions was divided by total number of extrusions.
At 5 minutes, bacteria were preferentially adhered to a small number of M cells (Fig. 2). Bacteria were seen on a few FAE enterocytes adjacent to M cells with adherent bacteria but not on AE enterocytes. At sites of cell extrusion, bacteria were preferentially located at the base of the extruding cell (Fig. 3).

Ileum, Salmonella serovar Typhimurium–inoculated loop, 5 minutes after infection; pig No. 2. Scanning electron micrograph, M cell (m) with adherent bacteria (arrows). Gold-palladium. Bar = 5 μm.
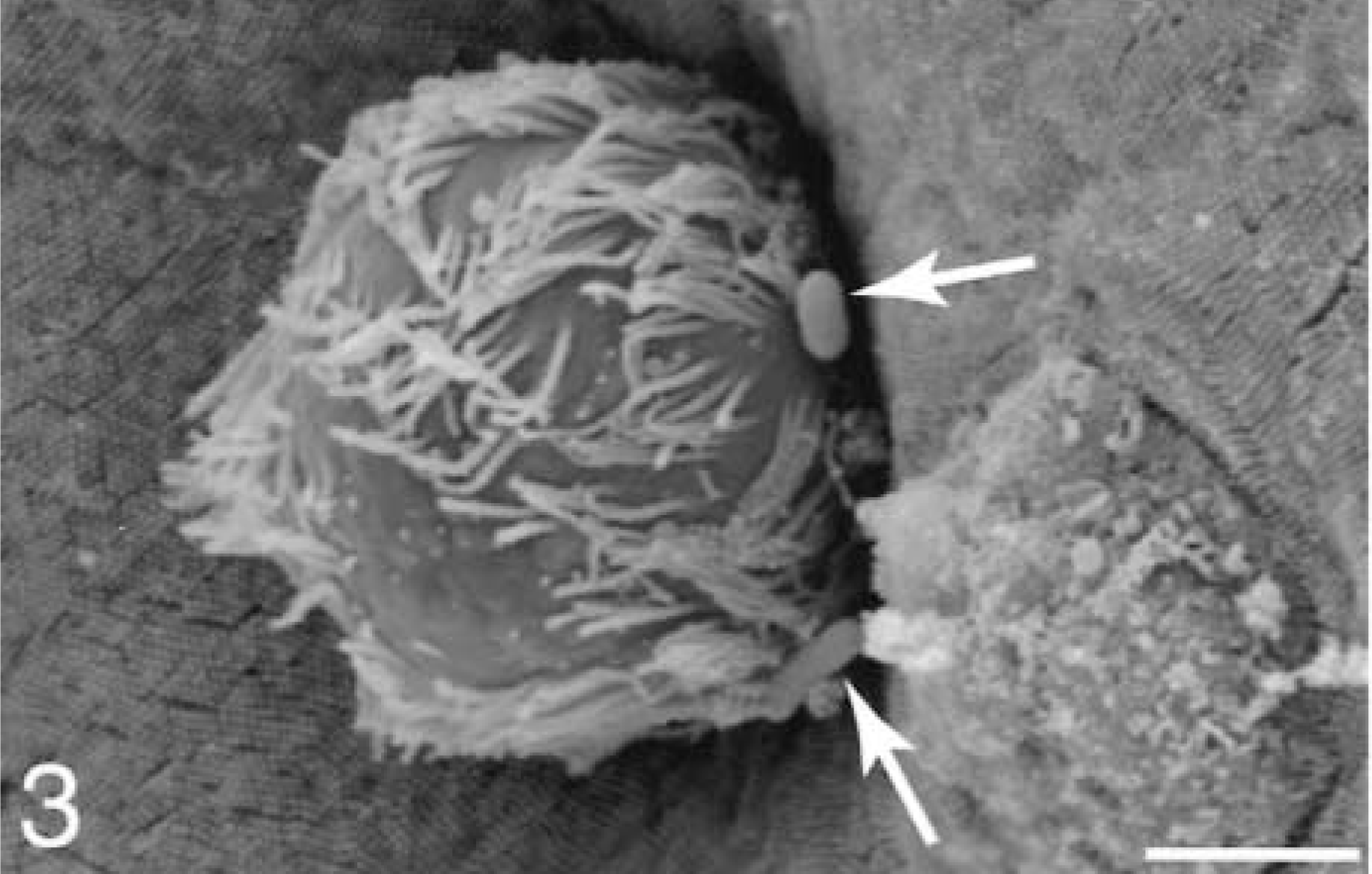
Ileum, Salmonella serovar Typhimurium–inoculated loop, 5 minutes after infection; pig No. 2. Scanning electron micrograph, cell extrusion with bacteria (arrows) near its base. Gold-palladium. Bar = 2 μm.
At 10 minutes, M cells with adherent bacteria had filopodia and early bleb formation. Rarely, an isolated patch of AE enterocytes with adherent bacteria was seen. Each patch contained 5–10 bacteria, with some filopodia being manifested only at the site of attachment.
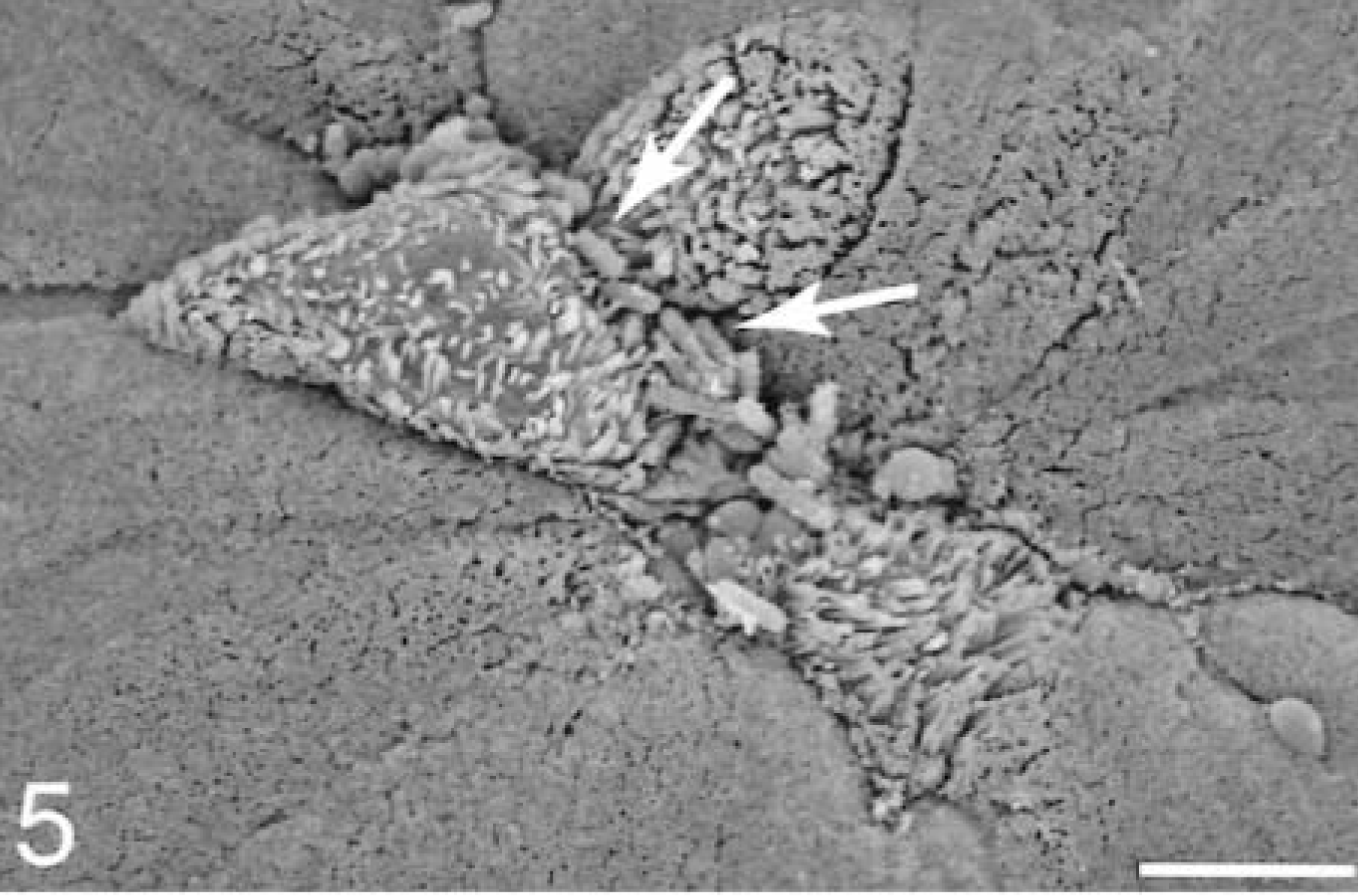
At 15–30 minutes, M cells with attached bacteria had surface rearrangements, including filopodia, lamellipodia, surface blebbing, and cell swelling; however, most M cells had no adherent bacteria. Enterocytes with attached bacteria exhibited filopodia and occasional lamellipodia formation at the point of contact with the bacterium (Fig. 4). Apical membrane reaction sites, with no adherent bacteria, exhibited elongated and degenerated microvilli. Bacteria were present in extrusion zones near the AE villi tips (Fig. 5). The bacteria were adherent at the lateral margins of cells that were beginning to degenerate and slough. At sites of individual cellular extrusion, the bacteria were most evident near the base of the extruding cell and appeared to be migrating through the crevices (Fig. 6).
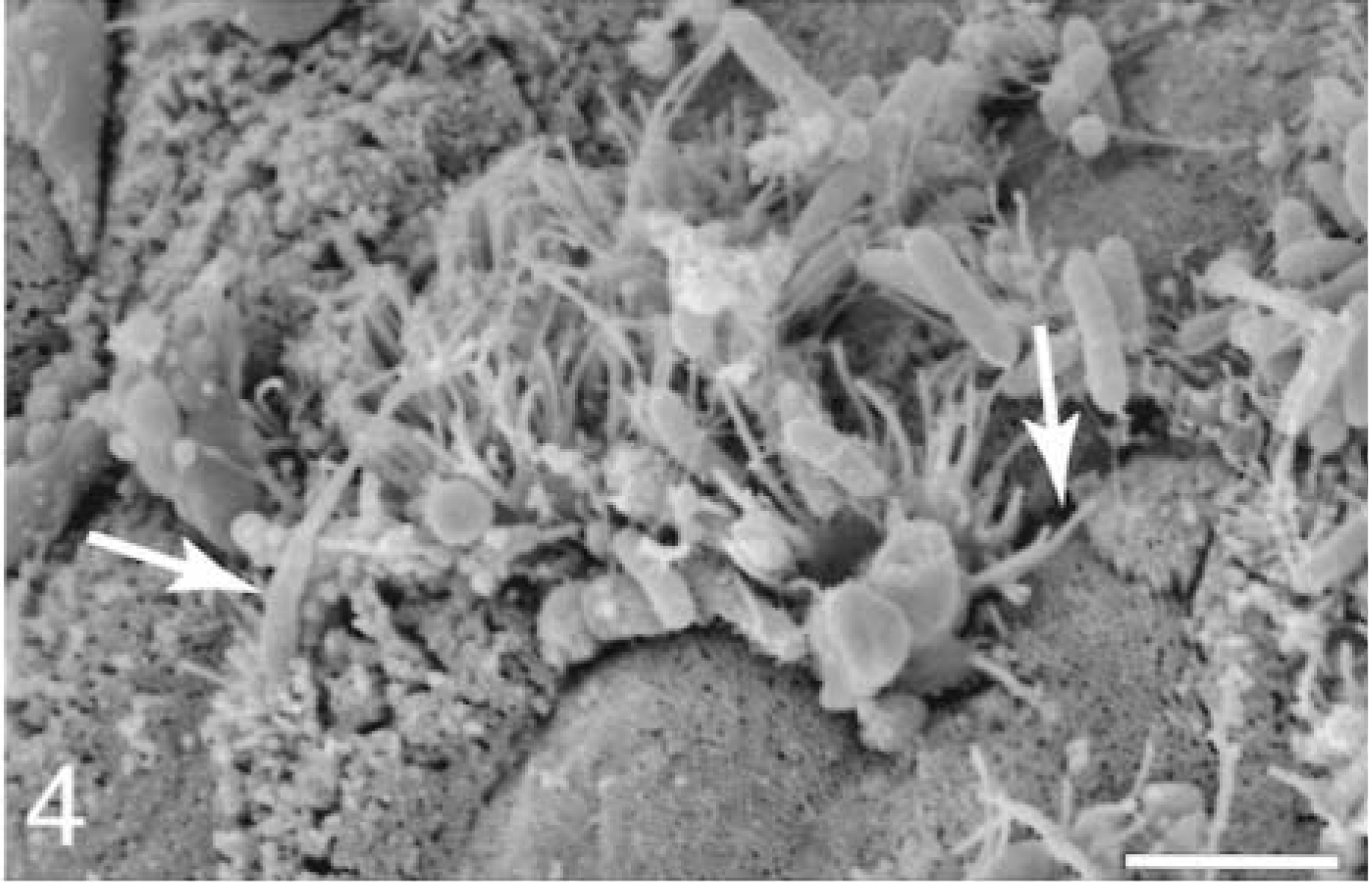
Ileum, Salmonella serovar Typhimurium–inoculated loop, 25 minutes after infection; pig No. 6. Scanning electron micrograph, absorptive epithelium enterocytes associated with bacteria exhibit filopodia (arrows) formation. Gold-palladium. Bar = 3 μm.
Ileum, Salmonella serovar Typhimurium–inoculated loop, 25 minutes after infection; pig No. 6. Scanning electron micrograph, villus tip with adherent bacteria (arrows) preferentially located at the lateral border of the sloughing cells. Gold-palladium. Bar = 5 μm.
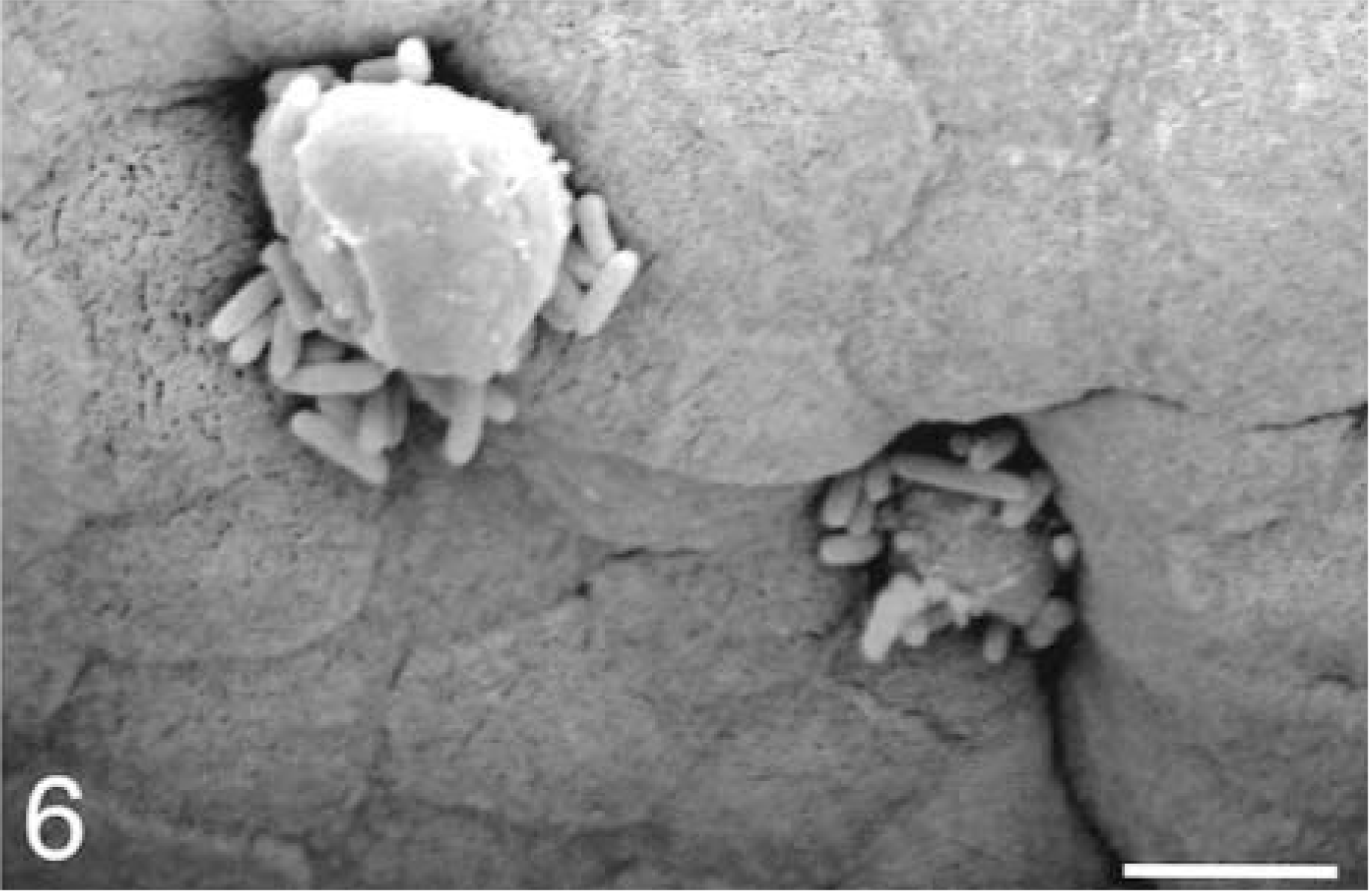
Ileum, Salmonella serovar Typhimurium–inoculated loop, 15 minutes after infection; pig No. 4. Scanning electron micrograph, bacteria at the crevices formed by the extruded cells. Gold-palladium. Bar = 5 μm.
At 45 and 60 minutes the reacting FAE villi exhibited severe cellular swelling and blebbing near the tip, whereas lower on the villi normal M cells could be seen with no visible reaction or bacterial adherence. The frequency of extrusion sites with adherent bacteria increased to 56%, and the bacterial count averaged 2.7 bacteria per site (Table 2). For comparison, a Salmonella serovar Choleraesuis–inoculated loop was examined, and after detailed examination, only two extrusion sites could be found with adherent bacteria.
In the 30-minute modified–gut-loop experiment, emphasis was placed on searching for sites of cellular extrusion. The sites found were similar in character to standard loops, and bacteria were preferentially adhered to many of these extrusion sites.
TEM examination
In FAE, scattered among enterocytes were M cells associated with intraepithelial lymphocytes. M cells were distinguished from enterocytes by thicker, more irregular microvilli and a less electron-dense cytoplasm (Fig. 7). The AE contained enterocytes with intermittent goblet cells.
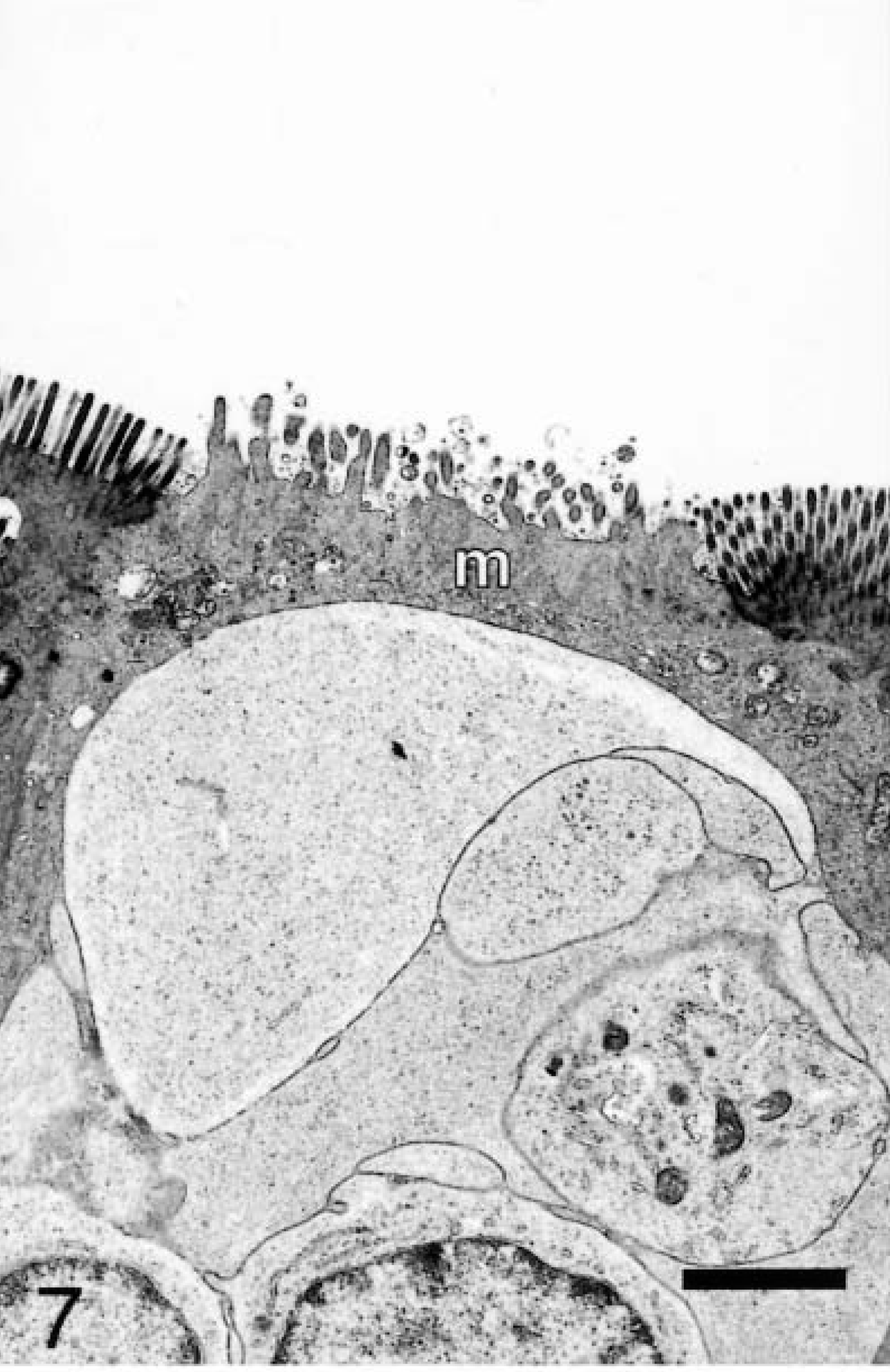
Ileum, sterile media–inoculated loop, 20 minutes after infection; pig No. 5. Transmission electron micrograph, M cell (m) with thicker and more irregular microvilli than adjacent enterocytes. Uranyl acetate and Reynolds lead acetate. Bar = 2 μm.
At 5 minutes, bacteria could rarely be seen in the lumen. A few M cells were seen initiating filopodia development, however, no bacteria were associated in close proximity to those reacting sites.
At 10 minutes, a small number of internalized bacteria were observed apically in M cells, enterocytes, and goblet cells (Fig. 8). M cells expressed filopodia and surface blebs, and luminal bacteria were often associated with these sites. Affected enterocytes and goblet cells had focal apical disruptions unlike the M cells in which the entire apical membrane was rearranged.
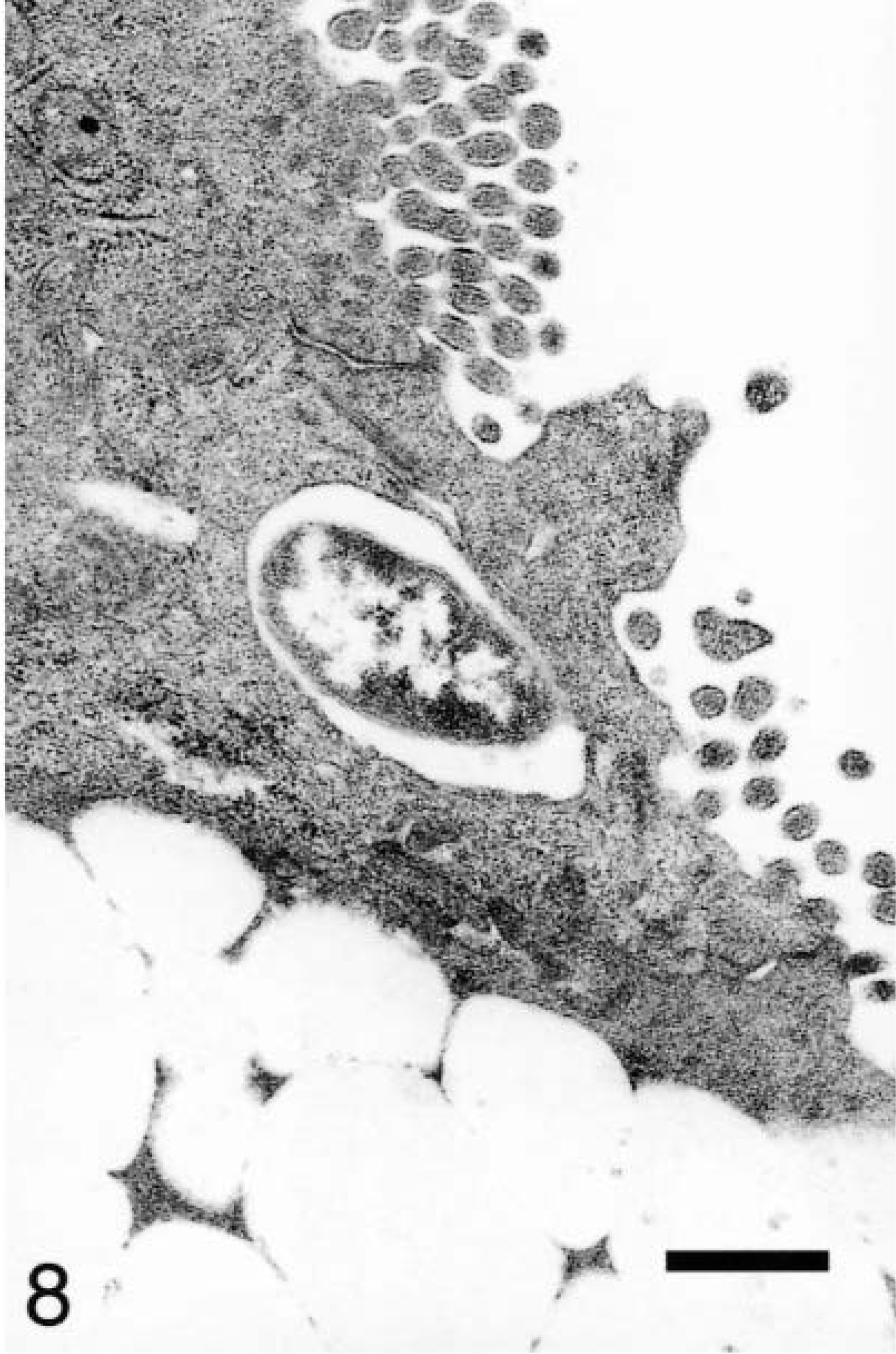
Ileum, Salmonella serovar Typhimurium–inoculated loop, 10 minutes after infection; pig No. 3. Transmission electron micrograph, a bacterium engulfed by an enterocyte. Uranyl acetate and Reynolds lead acetate. Bar = 1 μm.
At 15–30 minutes, internalized and vacuolated bacteria (usually 1–2 organisms) were observed within the affected M cells. The bacteria were located predominantly in the apical cytoplasm, with a few having progressed to the basolateral space (Fig. 9). Granulocytes could be seen near the basement membrane. On AE, goblet cells and enterocytes containing bacteria occurred in patches on the villi. Bacteria could be seen at individual extrusion sites especially along the base of the exiting cell and in the crevices formed by the extruding cell and the adjacent enterocytes (Fig. 10). Some bacteria were located in the cytoplasm beneath the exiting cells (Fig. 11). The swollen cells extended into the lumen and contained large vacuoles with degenerate mitochondria.
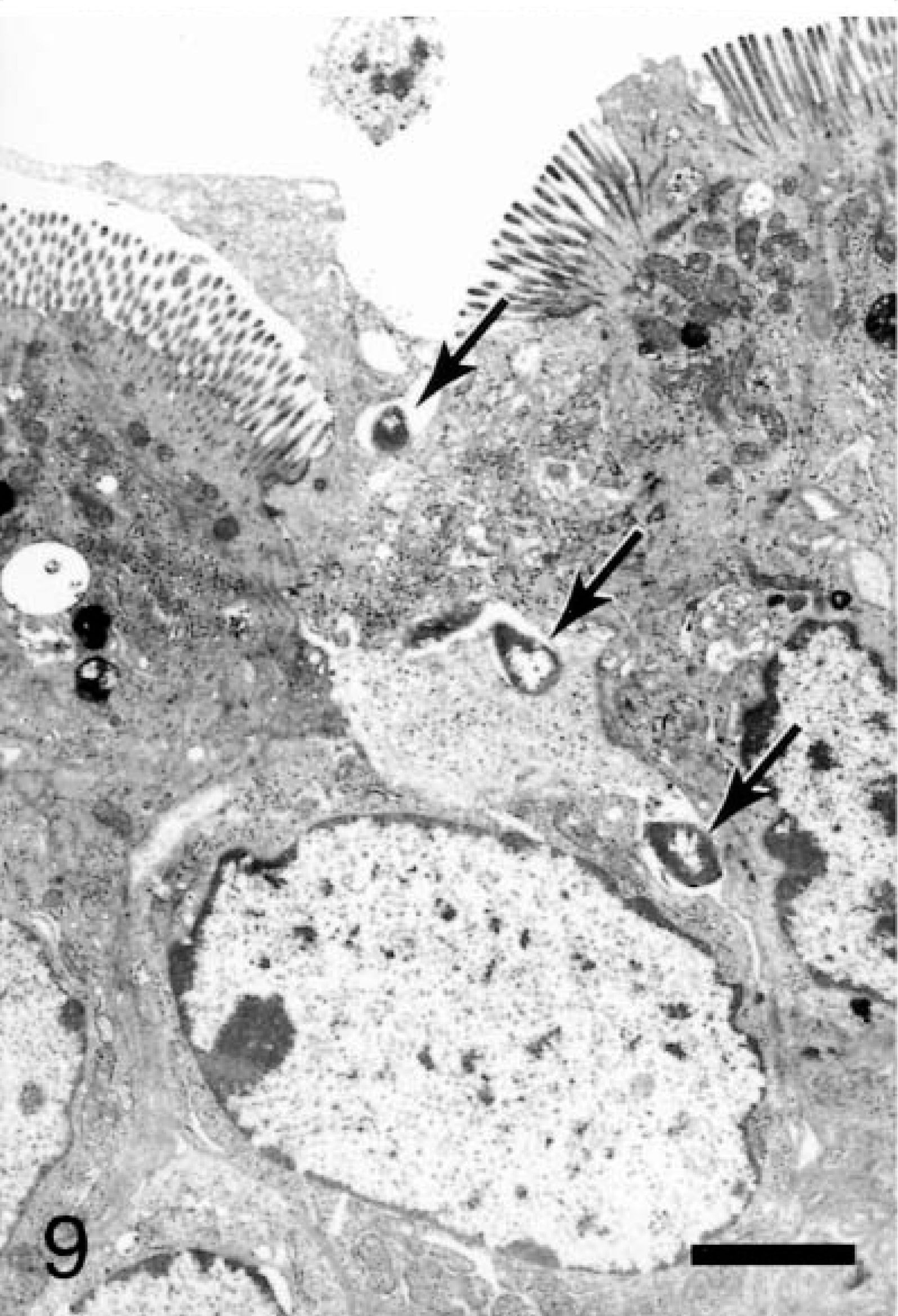
Ileum, Salmonella serovar Typhimurium–inoculated loop, 25 minutes after infection; pig No. 6. Transmission electron micrograph, bacteria (arrows) traffic through an M cell to cross the basolateral membrane and enter the paracellular space. Uranyl acetate and Reynolds lead acetate. Bar = 2 μm.
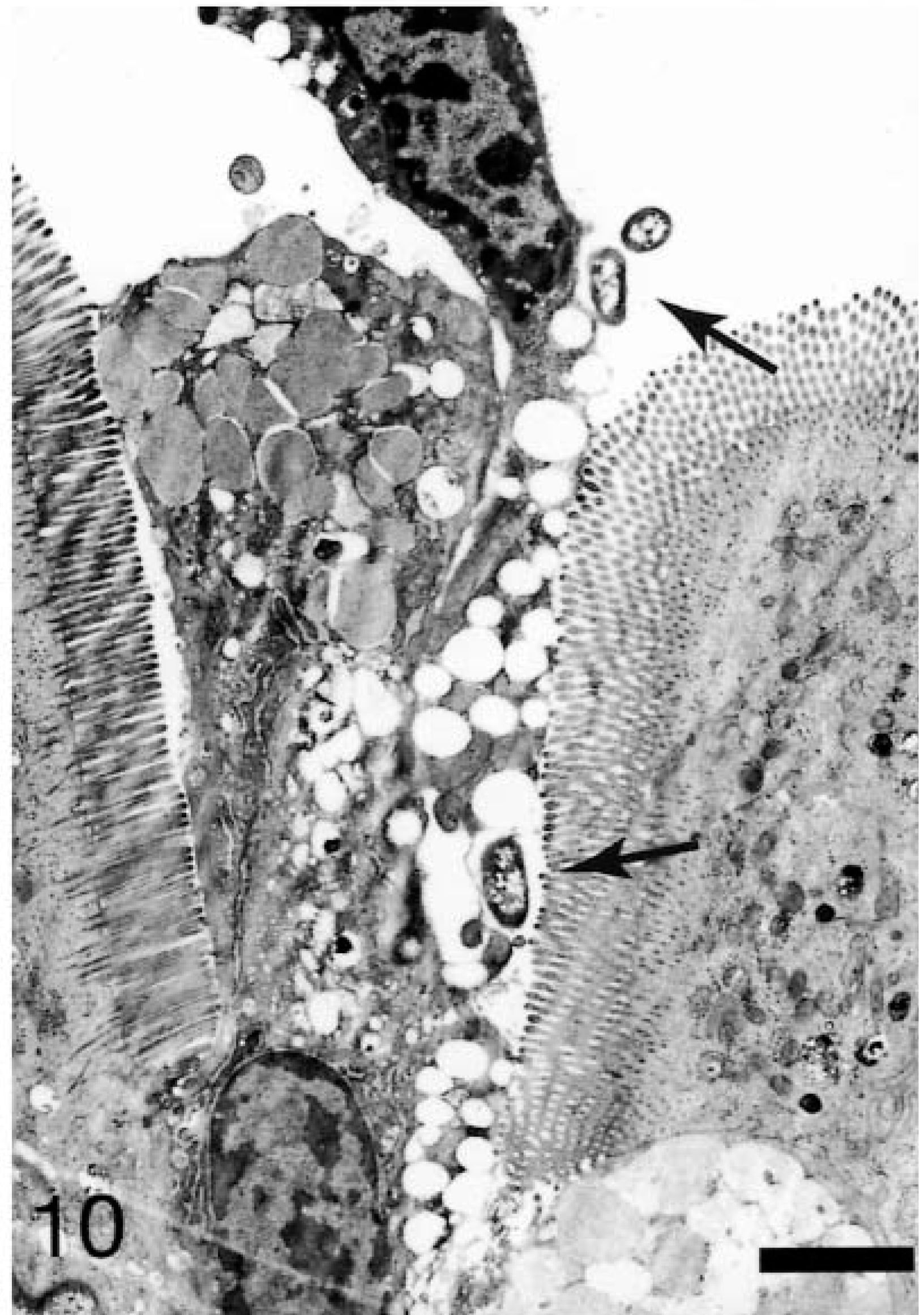
Ileum, Salmonella serovar Typhimurium–inoculated loop, 20 minutes after infection; pig No. 5. Transmission electron micrograph, bacteria (arrows) in the lumen and in the crevices formed by extruded cells. Uranyl acetate and Reynolds lead acetate. Bar = 4 μm.
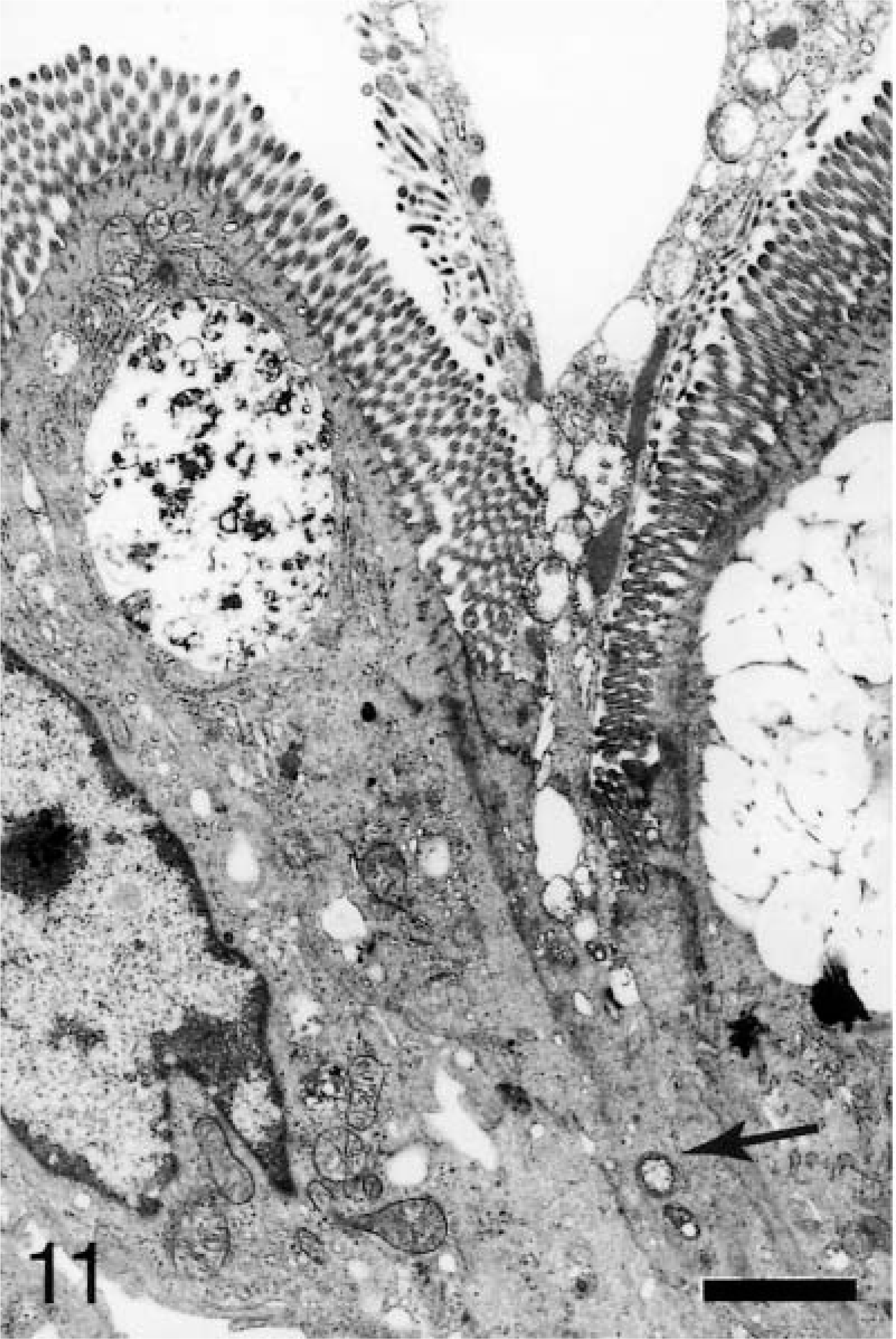
Ileum, Salmonella serovar Typhimurium–inoculated loop, 25 minutes after infection; pig No. 6. Transmission electron micrograph, bacteria (arrow) in the cytoplasm beneath a site of extrusion. Uranyl acetate and Reynolds lead acetate. Bar = 4 μm.
At 45–60 minutes, bacterial invasion continued through the apical membranes of M cells, enterocytes, and goblet cells. A few bacteria were present within apical membrane invaginations of enterocytes that exhibited little microvilli disruption (Fig. 12). Enterocytes with internalized bacteria exhibited complete resolution of the apical membrane rearrangements and resulted in normal microvilli morphology. Bacteria were also observed in the paracellular space after invasion near the tight junction. Bacteria were rarely evident in the lamina propria immediately below the basement membrane.
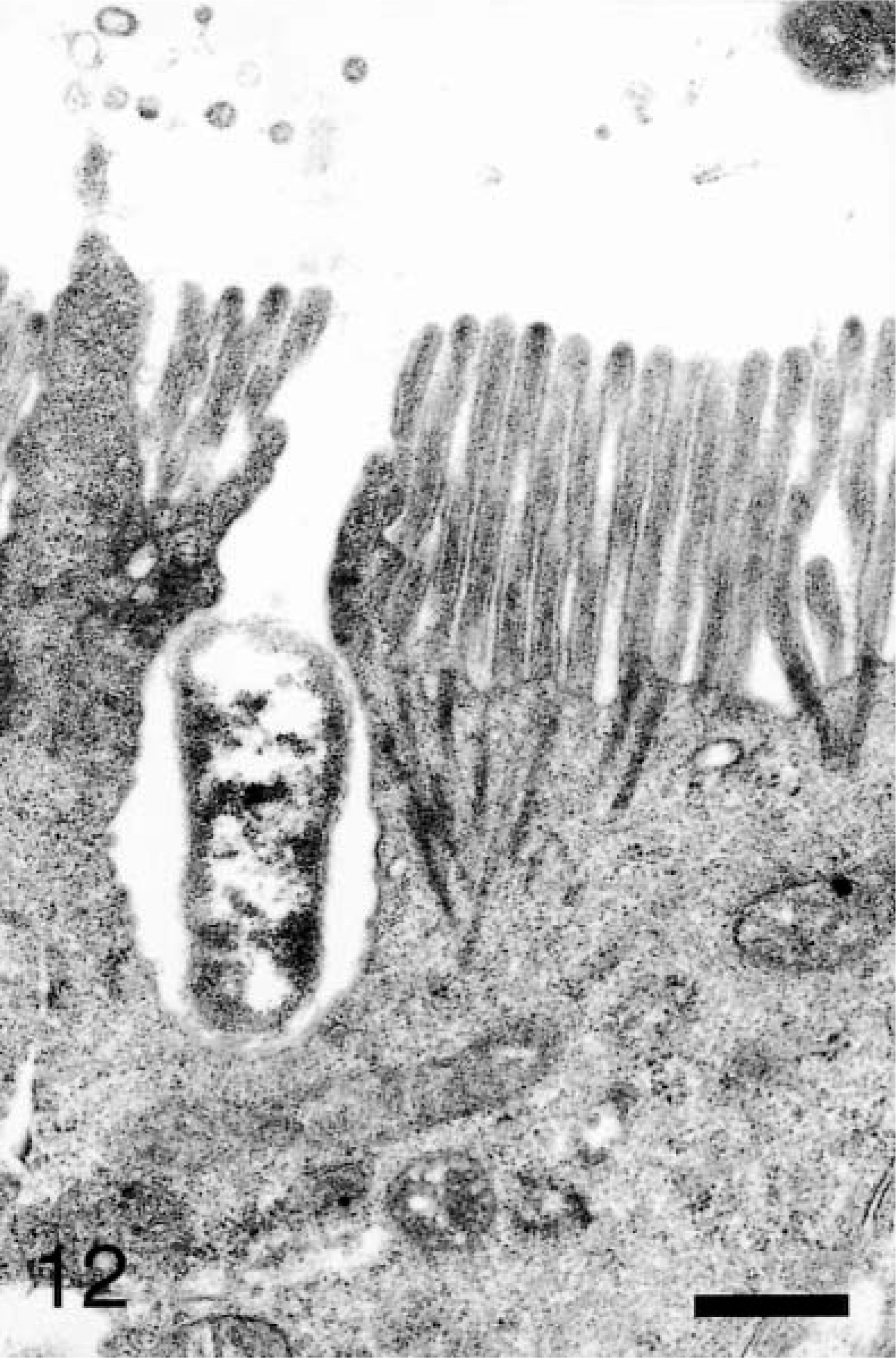
Ileum, Salmonella serovar Typhimurium–inoculated loop, 60 minutes after infection; pig No. 10. Transmission electron micrograph, bacterium within an apical membrane invagination with minimal apical membrane reaction. Uranyl acetate and Reynolds lead acetate. Bar = 1 μm.
Discussion
Identification of the definitive sites of early cellular invasion by Salmonella serovar Typhimurium is the major goal of this experiment. In previous swine studies definitive statements concerning the cellular kinetics of invasion could not be made because of lack of short time points.4,19,21,22 This work demonstrates early preferential adherence to M cells within 5 minutes, and by 10 minutes apical invasion occurs in M cells, enterocytes, and goblet cells. These observations are similar to observations in a bovine model, which showed that Salmonella invasion occurs within minutes of inoculation.7 But the bovine study suggested a halt of bacterial interaction with FAE by 60 minutes. We saw continued invasion of FAE and AE cells at 60 minutes. The difference between the bovine and swine models might represent differences in protocols between the two experiments or a difference in the kinetics of infection between the species. The early Salmonella invasion is also consistent with recent evidence on natural infection that naive pigs placed in a Salmonella-contaminated environment can become Salmonella positive within 2 hours.9 Our study suggests that early Salmonella serovar Typhimurium invasion is rapid and not limited by cell type in the swine ileum.
Early preferential adherence to the M cell may be due to lack of a physical barrier compared with other cells.17 The filamentous brush-border glycocalyx of rabbit enterocytes can be a barrier up to 500 nm thick, whereas the variable M-cell glycocalyx barrier may be as small as 20 nm.6 In addition, goblet cells are abundant on AE and absent on FAE, potentially reducing the mucus covering on FAE.3,8 The short time difference between invasion of M cells and other cell types may be a reflection of the absence of these barriers on M-cell apical membranes, thereby increasing the efficiency of invasion.
In addition to finding a lack of cell specificity for invasion, our observations also suggest another potential mechanism for entry. Scanning electron micrographs show that Salmonella serovar Typhimurium preferentially adheres at sites of cell extrusion. Bacteria appear to migrate into the crevices formed by the extruding cell and the adjacent enterocytes. We characterize this affinity at 15-minute-interval time points in Table 2. Transmission electron micrographs confirm bacterial migration through the crevices and that bacteria are located in the cytoplasm beneath the extruding cells. To evaluate the specificity of this preferential adherence, SEM examination of the Salmonella serovar Choleraesuis–inoculated loop shows minimal bacterial affinity to these sites. The culmination of these observations suggests that Salmonella serovar Typhimurium may use these sites of cell extrusion as an additional mechanism for host invasion.
Bacteria associated with cell extrusion sites have been previously observed by SEM in Salmonella serovar Typhimurium–infected bovine gut-loops, but the significance of this affinity was not known.7,28 Similar effects with enhanced invasion are observed in vitro. Clostridium difficile toxins A and B applied to HT-29 cells result in apical cell rounding and increased permeability of tight junctions. This is associated with preferential adherence by Salmonella serovar Typhimurium to these sites and an increase in paracellular invasion.5 In comparing Salmonella enterica serovars Typhimurium and Typhi invasion through HeLa cells, Salmonella serovar Typhimurium is believed to be more efficient at invasion because of increased adherence near cells with apical membrane rearrangements.15 Other in vitro studies show that Salmonella serovar Typhimurium preferentially adheres to and invades epithelium when exposed to the lateral surface of enterocytes.30,31 Our findings are consistent with in vitro studies that document Salmonella invasion at sites of cell loss.
The exposed lateral membrane may contain surface markers that can facilitate paracellular invasion. In polarized Madin-Darby canine kidney cell line 1 cells, apical infection with Yersinia pseudotuberculosis leads to tight junction permeability and attachment to basolateral βl-integrins for subsequent paracellular translocation of the bacteria.24 The increased permeability of the tight junctions and the subsequent exposure of basolateral markers may play a role in this specific affinity. The increased affinity to these sites may also be, in part, a result of chemotaxis. Increased Salmonella serovar Typhimurium attachment to acid-damaged HeLa cells was proposed to be the result of chemotaxis by components from the damaged cell. The authors suggested that factors increasing cell permeability could act as an attractant to Salmonella serovar Typhimurium in vivo.26
Salmonella serovar Typhimurium invasion occurring at sites of enterocyte loss may be especially relevant because swine and other species have been suggested to have a type-3 cell loss mechanism.14,16 This type of cell loss tends to occur in isolated individual enterocytes, allowing for breeches of integrity in the tight junction. These cells exhibit morphologic changes similar to oncotic necrosis and are often associated with intraepithelial lymphocytes and possibly neutrophils.12,16,18,27 In the mouse, individual cell loss can occur over the entire villus and may exhibit a circadian variation.20 In this experiment the extrusion frequency increases over time similarly in both treatment and control loops. Concern that the extrusions are induced by the loop procedure was addressed by a time zero loop. Scanning electron micrographs demonstrate extrusion sites similar in frequency and character to those at the 5-minute level. This suggests that extrusion sites are not solely surgery induced and may play a normal role in the physiology of the swine ileum. In addition, SEM examination of the 30-minute modified-loop procedure shows that bacteria preferentially accumulate at sites of extrusion. This procedure minimizes potential issues such as inadequate blood flow, increased lymphatic hydrostatic pressure, or tissue damage. These results suggest that Salmonella serovar Typhimurium adherence to sites of extrusion is an event independent of the gut-loop procedure. Although anesthesia and the associated drugs could potentially play a role in the extrusions, eliminating their effects in a humane manner is almost impossible.
SEM highlights FAE as an active area of infection by the obvious lesions of cell swelling and blebbing compared with the less noticeable AE lesions of minor filopodia and lamellipodia formation. SEM is useful to assess a wide area of tissue and discern the differences in adherence to the sites of extrusion. With TEM, these sites of invasion could be interpreted as bacterial-induced lesions rather than a preexistent entity that preferentially adheres bacteria. SEM and TEM are complementary in assessing intestinal epithelium invasion by Salmonella.
The swine gut-loop model demonstrates preferential adherence of Salmonella serovar Typhimurium to M cells after 5 minutes and apical invasion of M cells, enterocytes, and goblets cells after 10 minutes. Our work suggests Salmonella serovar Typhimurium invasion to be non–cell-specific in pig ileum. This work also provides the first in vivo evidence of invasion by Salmonella serovar Typhimurium at sites of cell extrusion. This finding is consistent with in vitro studies. The significance of this mechanism in natural infection is unknown.
Footnotes
Acknowledgements
We thank Judy Stasko, John Mattila, and Tracy Pepper for electron microscopy sample preparation and examination. We also thank Missy Yungclas, Ruth Willson, and Chuck Greiner for technical assistance.
