Abstract
Feline ocular sarcomas are malignant intraocular neoplasms that are frequently associated with a history of ocular trauma. They usually present as fibrosarcomas, but some have both epithelial and mesenchymal features. The purpose of this study was to determine the cell of origin of a subset of feline intraocular sarcomas that display a mixed epithelial-mesenchymal phenotype, with elaboration of basement membrane—type matrix. We examined the morphology and histochemical and immunohistochemical phenotypes of nine feline intraocular sarcomas. Immunohistochemistry and in situ hybridization were performed to detect expression of crystallin alpha A. In addition, tumors were examined for expression of vimentin, cytokeratin, smooth muscle actin, desmin, melan A, neural cell adhesion molecule, S-100, glial fibrillary acidic protein, nerve growth factor receptor, and collagen type IV. Animals ranged from 7 to 17 years of age—no breed or sex predilection for tumor occurrence was present. Tumors were characterized by mixed epithelial and mesenchymal phenotypes, both of which elaborated basement membrane-type material and expressed vimentin highly. On the basis of collagen type IV and crystallin alpha A immunopositivity, we established that three of nine tumors were of lens epithelial origin. Expression of desmin and smooth muscle actin identified one tumor as a leiomyosarcoma. The remainder were undifferentiated sarcomas of myofibroblastic origin. This is the first report of lens epithelial neoplasia in clinical material from any species. The history and morphologic features of feline ocular sarcomas are reminiscent of feline vaccine-induced sarcomas. These tumors may share pathophysiologic similarities unique to this species.
Feline ocular sarcomas are malignant intraocular neoplasms that are often associated with a history of ocular trauma. 1 , 4 , 5 In general, they are locally invasive, potentially metastasize, and usually necessitate enucleation of the affected eye. Their cellular origin is unclear. They typically assume a mesenchymal phenotype and are usually classified as fibrosarcomas. 4 , 5 However, they may contain cells with an epithelial phenotype that elaborate basement membrane–like material, 5 thus prompting speculation that they can originate from cells other than fibroblasts.
In those cases where the eye is removed before the globe has been completely invaded by the tumor, a typical distribution pattern is present. Tumors occupy the posterior iris and expand diffusely into the posterior chamber, retina, and choroid. Extraorbital invasion is common and may begin at the limbus or occur through the optic nerve. The lens is invariably destroyed. 4 , 5 Whether this occurs because of the initial traumatic event or is secondary to ocular neoplasia is unclear. The distribution of early tumors suggests that the cell of origin resides in the ciliary body (ciliary muscle, pars ciliaris retinae), iris (constrictor-dilator muscles, melanocytes, fibroblasts), or lens (lens epithelial cells).
Spontaneous tumors of the lens (i.e., those arising from conditions other than an experimental intervention or another clearly defined cause) have not been described in any species. However, lens epithelium is not refractory to proliferation or transformation. In cataractous lenses, the lens epithelium will proliferate and transdifferentiate to assume a myofibroblastic phenotype, 2 , 3 and in the mouse, lens tumors can be induced in vivo by genetic manipulation. 15 , 16 Given these data, it is reasonable to suspect that some ocular sarcomas in cats may be of lens epithelial origin. The purpose of this study is to determine the cell of origin of a subset of feline intraocular sarcomas, specifically those that display a mixed epithelial-mesenchymal phenotype and elaborate a basement membrane–type matrix.
Methods
Animals and tissues
Nine cases were selected from an archived collection, which includes 110 cases of feline ocular sarcoma in the Comparative Ocular Pathology Laboratory of Wisconsin. Osteosarcomas, chondrosarcomas, and fibrosarcomas, as well as round cell variants, were excluded. Two additional normal cat eyes were used in the study. After enucleation, eyes were fixed in 10% formalin and embedded in paraffin. Sections were cut at 5 µm and stained with hematoxylin and eosin, periodic acid–Schiff (PAS), and Masson's trichrome stain.
Immunohistochemistry
Sections were stained with antibodies to crystallin alpha A (a gift from Joseph Horwitz), a specific marker for lens epithelial cells. In addition, a panel of commercial antibodies were applied to determine whether tumors displayed mesenchymal, epithelial, melanocytic, or neural differentiation. Antibodies were used to identify vimentin (Biogenix, cat No. AM074-5M), collagen type IV (Dako, cat No. M0785), muscle-specific actin (MSA) HHF35 (Dako, cat No. M0635), pancytokeratin AE1-AE3 (Chemicon, cat No. MAB-3412), glial fibrillary acidic protein (GFAP; Dako, cat No. N1506), desmin (Dako, cat No. M0760), neural cell adhesion molecule (NCAM; Novacastra, cat No. NCL-CD56-1B6), melan A (Dako, cat No. M7196), S-100 (Dako, cat No. Z0311), and nerve growth factor receptor p75 (Novacastra, cat No. NCL-NGFR). Commercial antibodies were obtained from Dako (Carpenteria, CA), Novacastra (Newcastle upon Tyne, UK), Biogenex (San Ramon, CA), and Chemicon (Temecula, CA) and were used in the dilutions indicated by the manufacturers. Crystallin alpha A was used at a dilution of 1 : 400. Goat anti-rabbit or rabbit anti-mouse secondary antibodies were used. Negative controls were incubated with normal goat or rabbit serum instead of the primary antibody. Positive controls were either internal (pancytokeratin, collagen type IV—corneal epithelium and Descemet's membrane, respectively; desmin and MSA—vessels) or were included in the staining panel (vimentin—liver; GFAP—brain; S100 and nerve growth factor—peripheral nerves in skin; melan A—melanoma; crystallin alpha A—normal cat and mouse lens). Staining was performed using avidin–biotin–peroxidase complex with diaminobenzidine as the chromagen (Dako).
In situ hybridization
Crystallin alpha A mRNA sequences from human (GenBank accession: U05569.1), cow (GenBank accession: M26142.1), and mouse (GenBank accession: J00376.1) were aligned and consensus primers designed using the Primer 3 program (http://www-genome.wi.mit.edu/cgi-bin/primer/primer3_www.cgi). Primer sequences are CRY AA forward, CAC CAT CAG CCC CTA CTA CC; CRY AA reverse, CAG AGC TGG GTT TCT CCT C. Ten lenses from 6-week-old C3H mice were extracted and snap frozen in liquid nitrogen. Total RNA was extracted using the RNEasy kit (Quiagen, Valencia, CA), reverse transcribed, and 1µg of cDNA used in an reverse transcriptase–polymerase chain reaction (RT-PCR) reaction. Reactions conditions were 95 C, 30 minutes; 54 C, 1 minute; and 72 C, 1 minute for 35 cycles. Reaction volume was 50 µl. One hundred microliters of RT-PCR product was gel-purified using a Qiaquick kit (Qiagen). Polymerase chain reaction products were used as a template to generate a random-primed 32Pd cytidine triphosphate–labeled probe (NEB, Beverly, MA). Using a standard protocol, 5 × 106 counts per slide were applied to paraffin-embedded sections (Current Protocols in Toxicology, Unit 2.7). Slides were hybridized for 2 days at 42 C, then washed, and dipped in KODAK NTB2 emulsion before being developed for 1 week. Emulsion was removed in the dark and slides counterstained with hematoxylin. Control tissues included normal feline and mouse eyes—hybridization was limited to the lens in both species.
Results
Nine globes containing intraocular tumors and two normal eyes were included in the study. Of the animals with tumors, five had a history of ocular trauma of the affected eye. In all cases, tumors involved the ciliary body and iris with variable extension to the anterior chamber, posterior segment, and extraorbital regions through the limbus. In all cases, the lens was destroyed, with residual fragments of lens capsule and lens fiber material embedded within the tumor. Neoplastic tissue was poorly circumscribed, unencapsulated, and highly infiltrative. Most tumors were accompanied by significant inflammatory infiltrates. These were most commonly lymphohistiocytic but were fibrinosuppurative in regions where tumor mass had undergone necrosis. Neoplastic tissue was usually intimately associated with normal cellular elements—these were most commonly melanocytes, melanophages, smooth muscle cells, fibroblasts, and mixed inflammatory cell infiltrates (Figs. 1, 2).
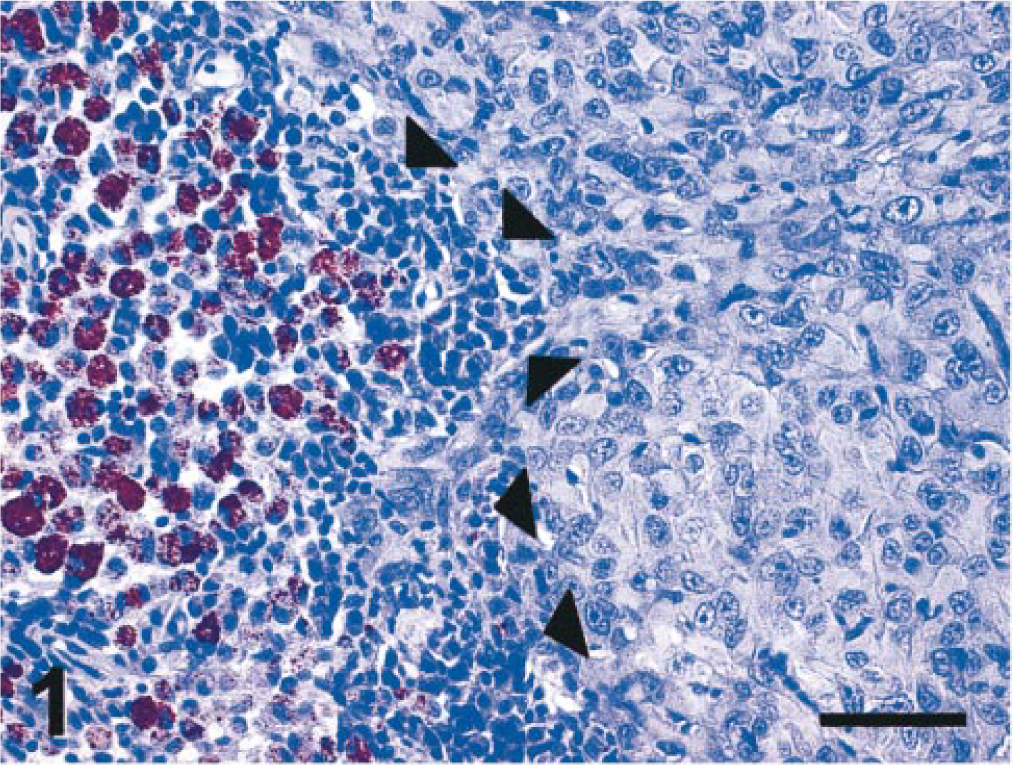
Intraocular tumor; cat. The tumor mass is intimately associated with a lymphoplasmacytic infiltrate (arrowheads) admixed with melanophages containing brown intracytoplasmic granules. Hematoxylin stain. Bar = 50 µm.
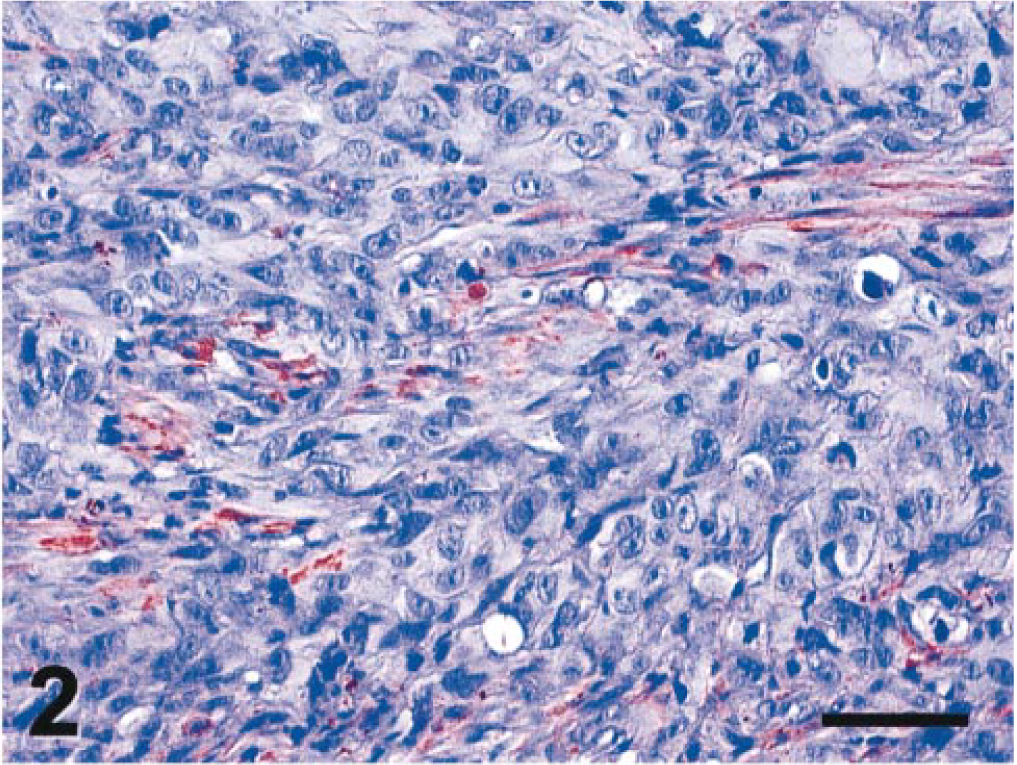
Intraocular tumor; cat. Neoplastic cells infiltrate preexisting structures, in this case, the iris constrictor (immunopositive for MSA), to create a heterogenous mixture of normal and neoplastic tissue. MSA immunohistochemistry. Bar = 50 µm.
Tumor cells were arranged in one of two major patterns—interlacing bundles of spindle cells (mesenchymal phenotype) or irregular sheets of variably sized polygonal cells (epithelial phenotype). Often, both patterns were present in the same tumor; however, in regions where the tumor infiltrated the globe, the mesenchymal phenotype tended to prevail. In these regions, tumor cells dissected preexisting structures, creating a heterogenous pattern of normal and neoplastic tissue. Neoplastic cells with an epithelial phenotype were polygonal, with well-defined cytoplasmic borders and variable anaplasia. In general, the degree of anaplasia was modest. Similarly, cells with a mesenchymal phenotype presented the appearance of moderately anaplastic fibrosarcomas. Neoplastic cells elaborated variable amounts of basement membrane–like material, which stained positively with both PAS and Masson's trichrome stain. This material was produced by cells irrespective of whether they demonstrated epithelial or mesenchymal morphology and varied greatly in amount and thickness (Figs. 3–6).
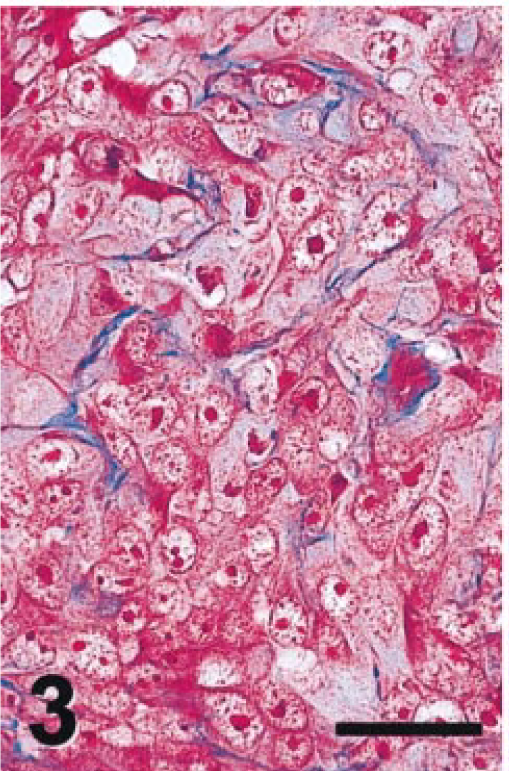
Intraocular tumor; cat. Epithelial phenotype, Masson's trichrome stain. Bar = 25 µm.
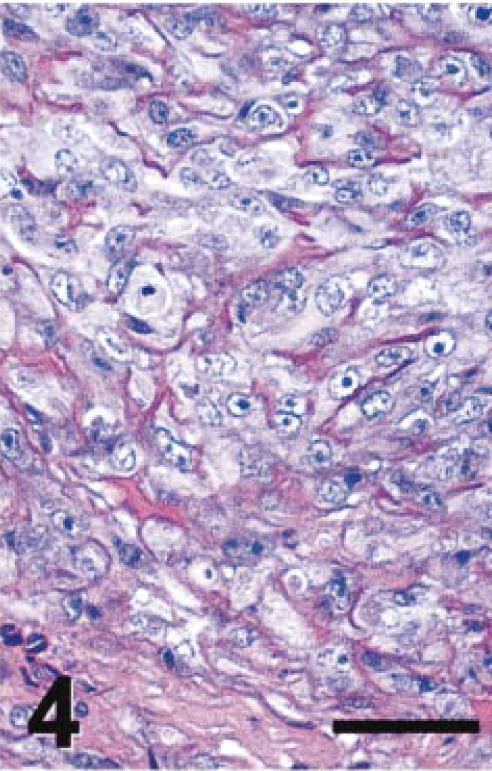
Intraocular tumor; cat. Epithelial phenotype, PAS stain. Bar = 25 µm.
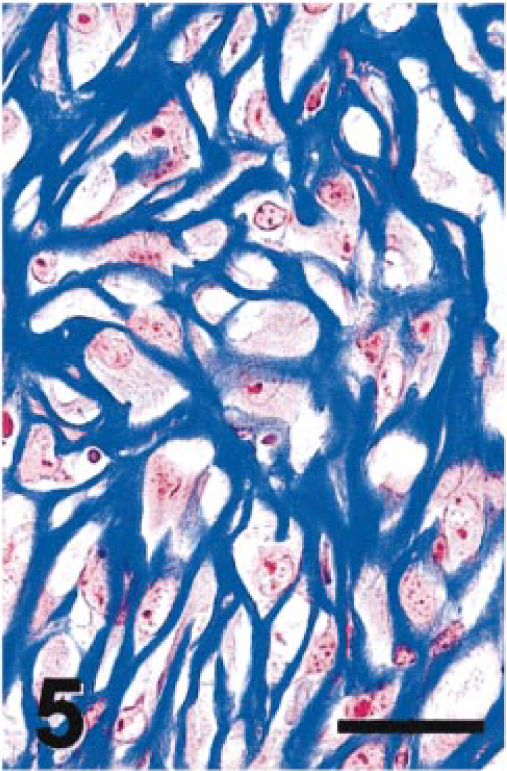
Intraocular tumor; cat. Mesenchymal phenotype, Masson's trichrome stain. Bar = 25 µm.
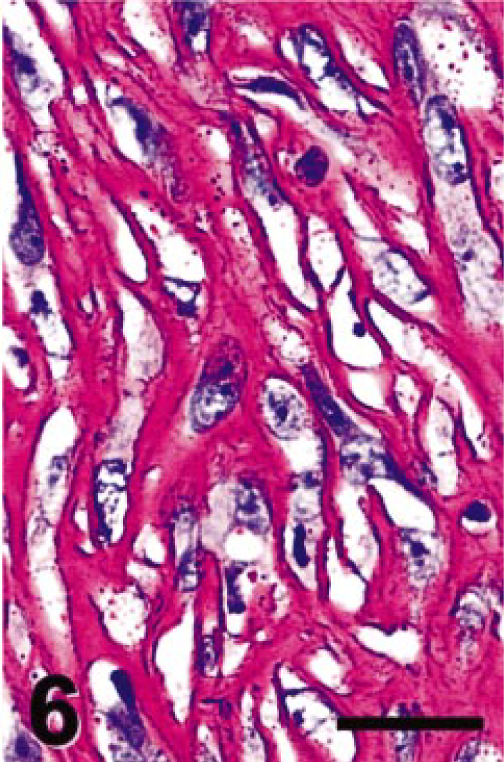
Intraocular tumor; cat. Mesenchymal phenotype, PAS stain. Neoplastic cells of both epithelial and mesenchymal phenotypes elaborate a matrix, which like the anterior lens capsule, stains with both PAS and Masson's trichrome stain. Bar = 25 µm.
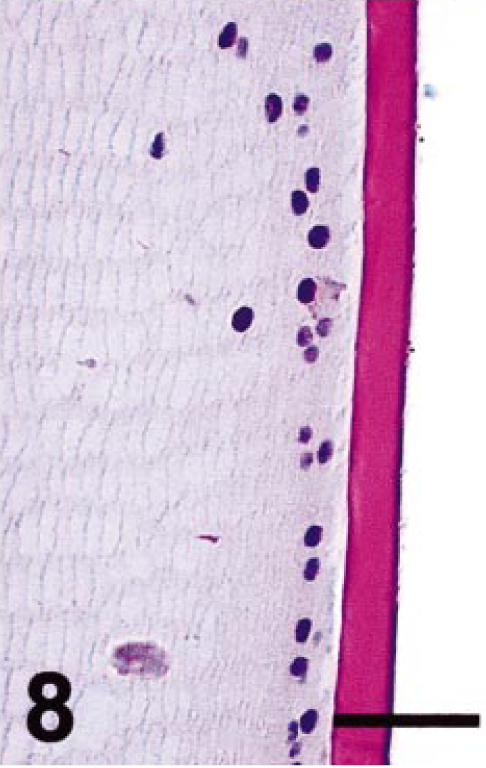
Immunohistochemical and special staining of normal adult feline eyes revealed a characteristic spectrum of staining for each structure (Table 1). In the lens, Masson's trichrome identified collagen throughout the entire thickness of the posterior lens capsule. At the lens bow, trichrome staining assumed a bilaminar appearance because only the most superficial portion of the anterior lens capsule was stained (Fig. 7). The lens capsule was PAS positive throughout its thickness (Fig. 8). Collagen type IV immunopositivity in the lens capsule replicated the staining pattern identified with Masson's trichrome stain. Immunohistochemical staining for crystallin alpha A protein was noted in lens epithelial cells through their differentiation into fiber cells and persisted in fiber cells. Crystallin alpha A mRNA expression was strongest at the lens bow and diminished as fiber cells matured (Fig. 9, Fig. 14, inset). No extralenticular expression of crystallin alpha A was present. The anterior lens epithelium stained strongly for vimentin—this diminished at the lens bow as epithelial cells differentiated into fiber cells (Fig. 10). Lens epithelium was faintly immunopositive for MSA. Normal adult feline lens epithelium was immunonegative for all other markers tested.
Immunohistochemical and staining features of the normal feline eye.


Normal cat lens (equator). The anterior lens capsule assumes a bilaminar appearance resulting from failure of the inner portion to stain. The posterior capsule is indicated with an arrow. Masson's trichrome stain. Bar = 40 µm.
Normal cat lens (equator). The posterior and anterior lens capsule stains homogeneously for PAS. PAS stain. Bar = 40 µm.
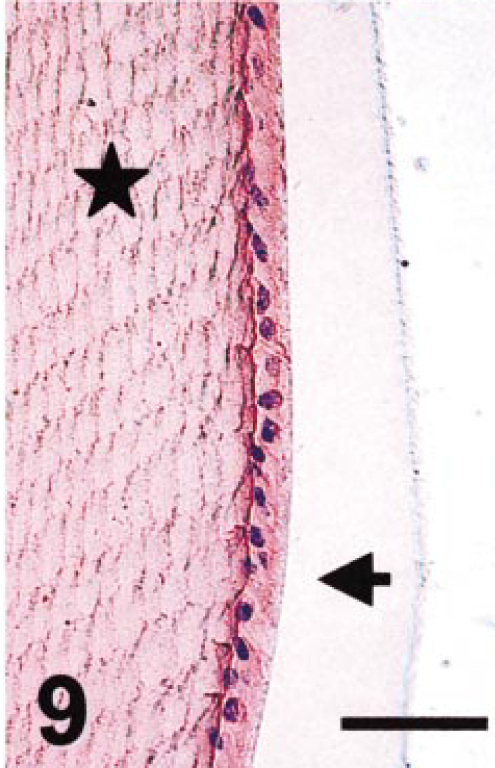
Normal cat lens (equator). Both anterior lens epithelium (arrow) and secondary lens fibers (star) are immunopositive. Crystallin alpha A immunostain. Bar = 40 µm.
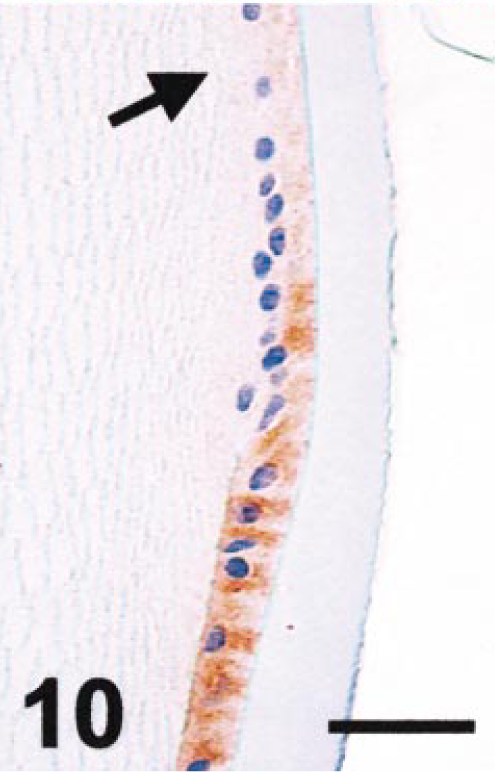
Normal cat lens (equator). The anterior lens epithelium contains vimentin until fiber differentiation begins (arrow). Vimentin immunostain. Bar = 40 µm
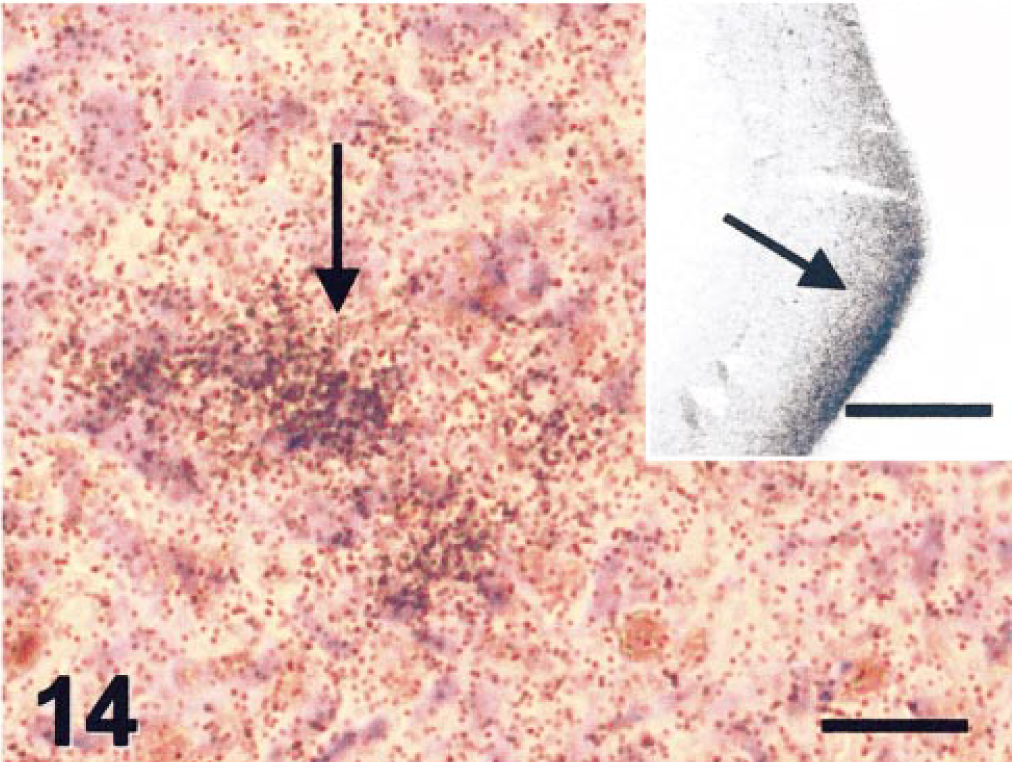
Intraocular tumor; cat. There was patchy labeling of tumor cells by crystallin alpha A immunostaining within epithelial regions (arrow). Inset: In situ hybridization for crystallin alpha A in normal feline lens. Crystallin alpha A expression is strongest at the lens bow (arrow), where lens epithelial cells differentiate into fiber cells. In situ hybridization for crystallin alpha A. Bar = 20 µm. Inset: Bar = 200 µm.
Ciliary body and iridal musculature were immunopositive for MSA, desmin and vimentin. Ciliary musculature stained most strongly for desmin, whereas iridal musculature stained more strongly for MSA. Collagen type IV was present in the lens capsule, ciliary zonules, and weakly between myocytes of the ciliary body. The nonpigmented neural epithelium stained most strongly for NCAM and weakly for vimentin and desmin. Melanocytes were identified with melan A, Muller cells with GFAP, and corneal epithelium with pancytokeratin.
All neoplasms (Table 2) were consistently and strongly vimentin immunopositive (Fig. 11). PAS-Masson's trichrome–stained basement membrane–like material was usually immunopositive for collagen type IV (Fig. 12). Three of nine tumors demonstrated variable immunopositivity for staining with crystallin alpha A (Fig. 13) and variable in situ hybridization to the crystallin alpha A probe (Fig. 14). Apart from vimentin staining, immunohistochemical staining was often clonal and weak. In general, areas with an epithelial phenotype were crystallin alpha A positive, whereas MSA tended to stain mesenchymal areas. In these areas, most tumors were immunonegative for all markers except vimentin. One of nine tumors demonstrated patchy immunopositivity for desmin and MSA. Rarely, isolated areas were immunopositive for GFAP. Pancytokeratin, melan A, and NCAM staining were not present in any of the tumors. In one animal, localized spindle cell proliferation was associated with the ruptured lens capsule (Figs. 15–17). Cells in these regions produced PAS-positive and Masson's trichrome–positive matrix and were immunopositive for MSA (Figs. 15–17). They were immunonegative for pancytokeratin and crystallin alpha A. These areas had features of localized lens epithelial proliferation associated with cataract extraction in dogs. 2 , 3
Immunohistochemical and staining features of feline ocular sarcomas.
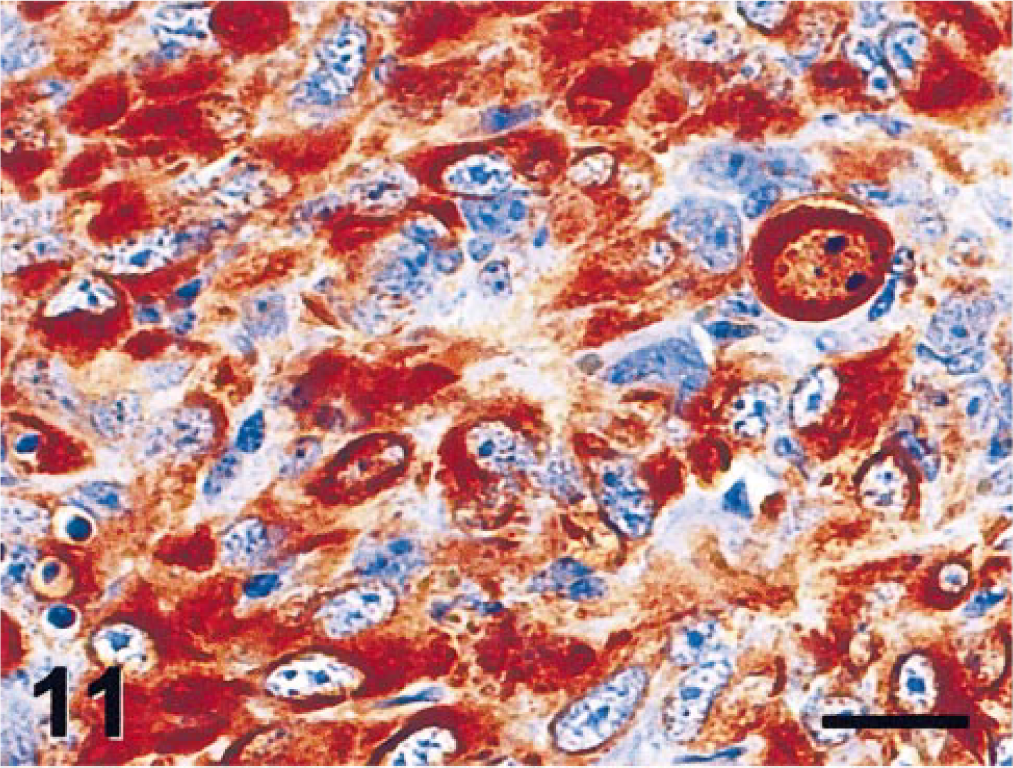
Intraocular tumor; cat. Neoplastic cells are strongly vimentin positive. Vimentin immunostain. Bar = 20 µm.
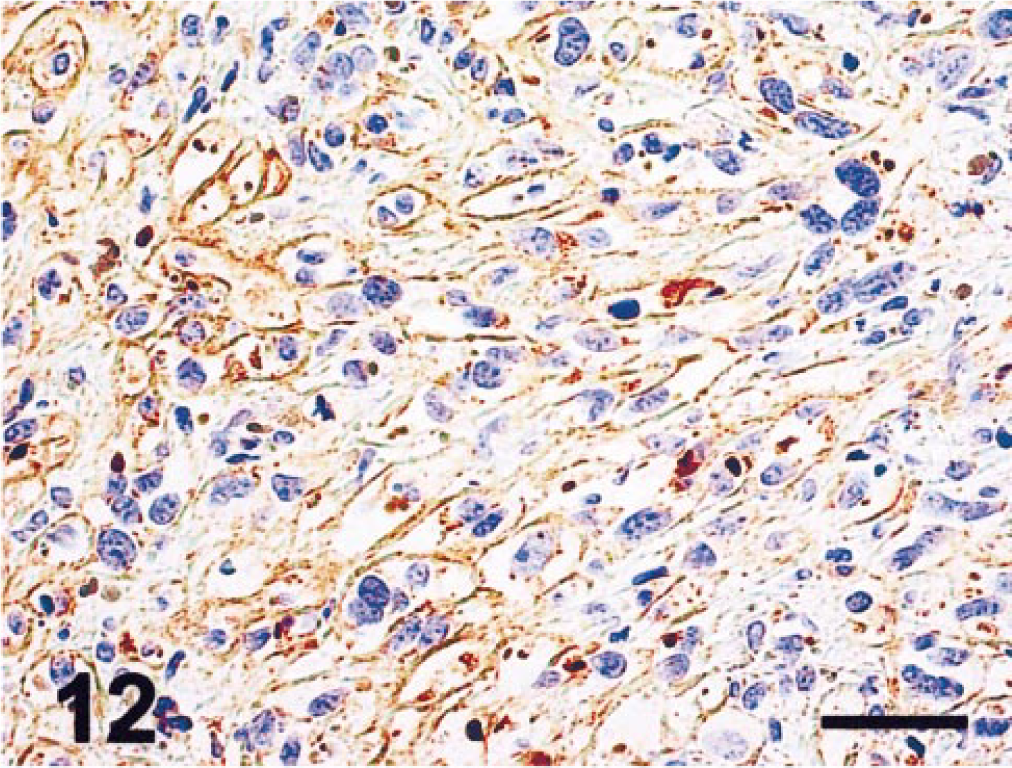
Intraocular tumor; cat. Extracellular matrix was variably immunopositive for collagen type IV in both epithelial and mesenchymal regions. Collagen type IV immunostain. Bar = 20 µm.
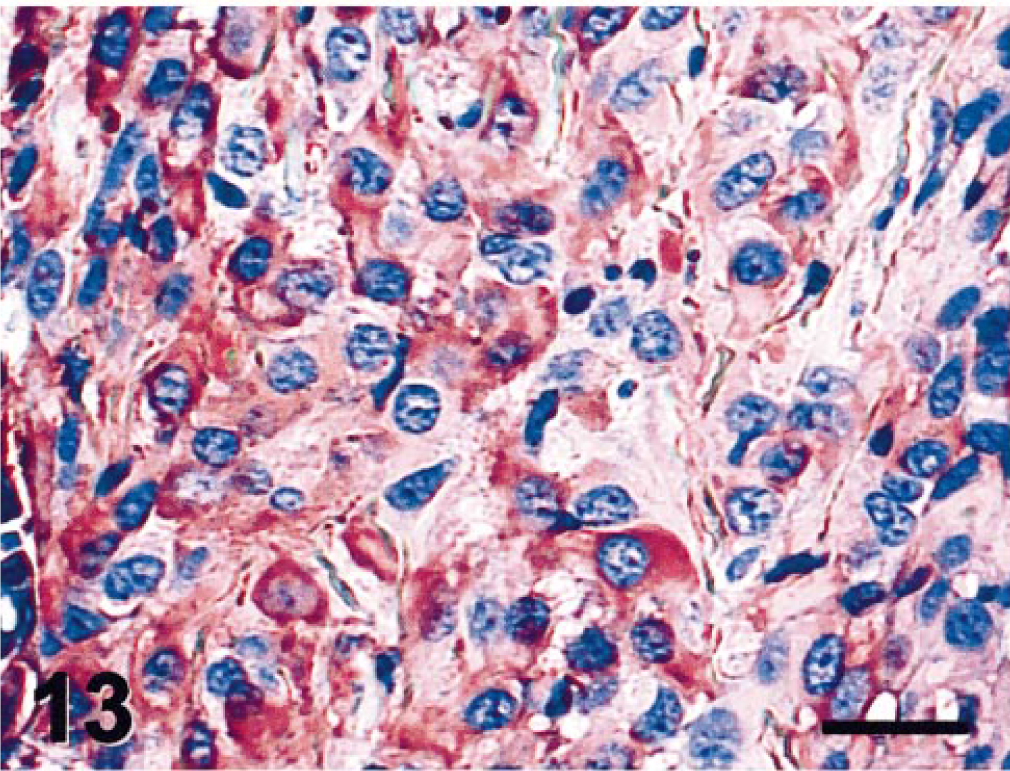
Intraocular tumor; cat. Regions of the tumor with an epithelial phenotype were most consistently positive for crystallin alpha A. Crystallin alpha A immunostain. Bar = 20 µm.
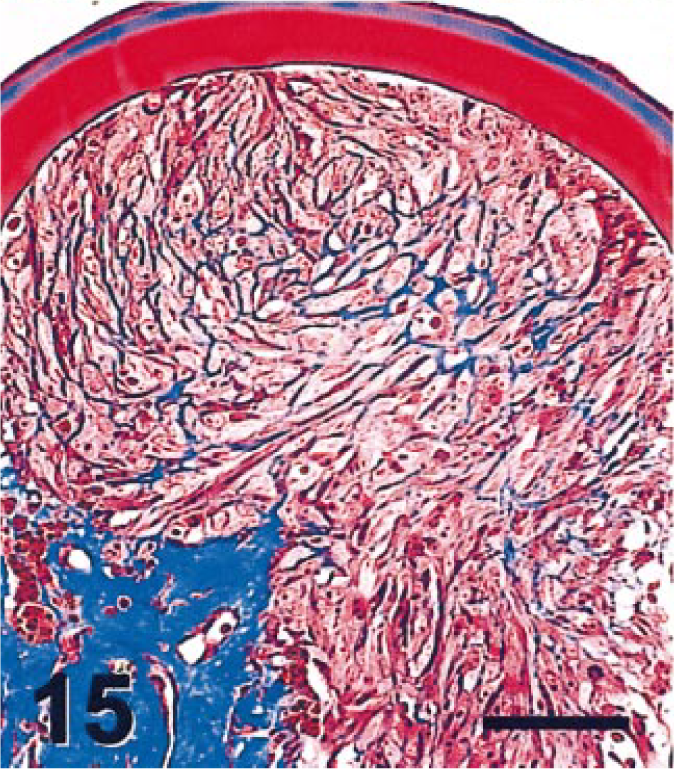
Eye; cat. Region of lens epithelial proliferation. Masson's trichrome stain. The external aspect of the lens capsule and intercellular matrix of proliferating lens epithelial cells stain positively for Masson's trichrome stain. Bar = 50 µm.
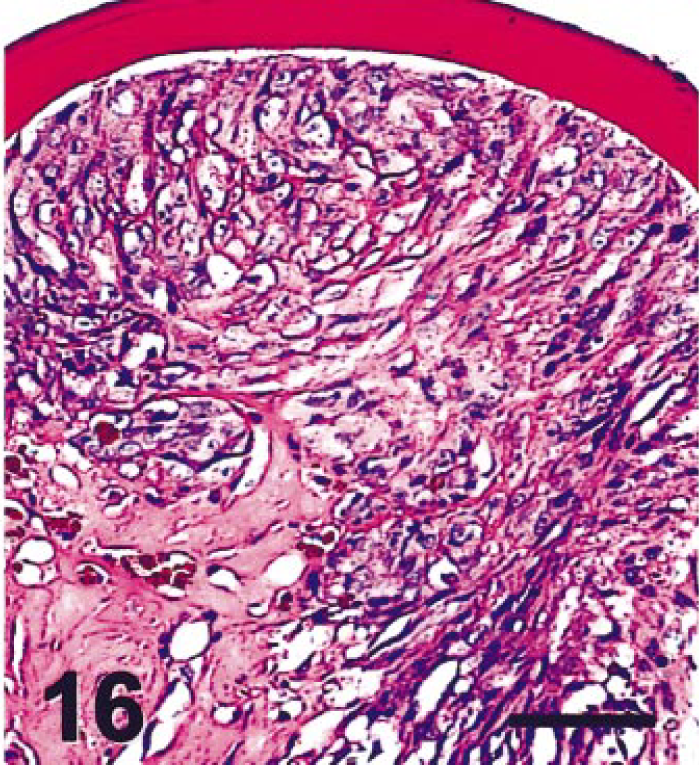
Eye; cat. Region of lens epithelial proliferation. PAS stain. The full thickness of the lens capsule and intercellular matrix of proliferating lens epithelial cells stain positively for PAS. Bar = 50 µm.
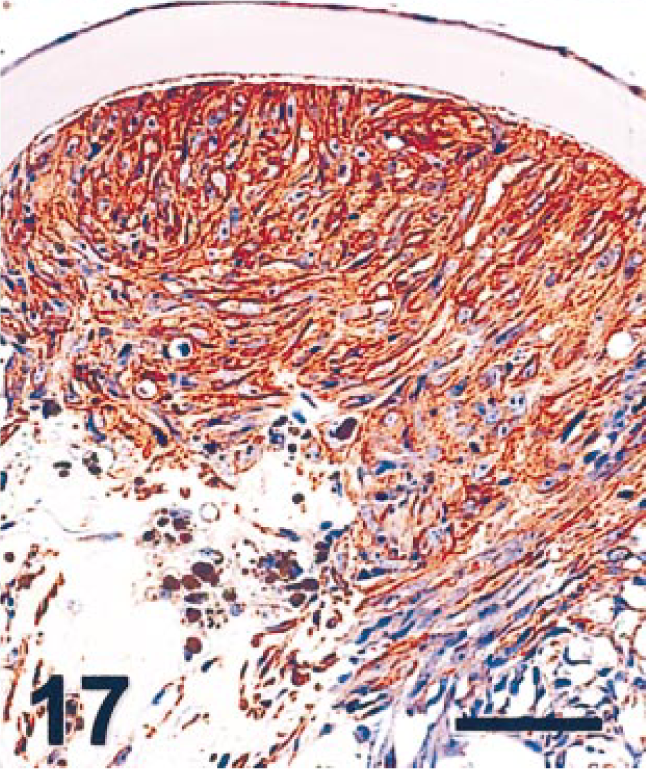
Eye; cat. Region of lens epithelial proliferation. Proliferating lens epithelial cells are strongly immunopositive for MSA. MSA immunostain. Bar = 50 µm
Discussion
Feline intraocular sarcomas are characterized by the following features. They are locally invasive malignant neoplasms that occupy the iris, ciliary body, and posterior chamber and are often associated with a history of trauma.
4
,
5
Invariably the lens is destroyed, and significant inflammation accompanies tumor growth. Although the majority are pure mesenchymal tumors,
4
,
5
a subset of feline ocular sarcomas present with a mixed epithelial-mesenchymal phenotype. In these cases, neoplastic cells elaborate a matrix that has the electron microscopic and histochemical features of basement membrane material.
5
On the basis of these morphologic criteria, three possible differential diagnoses appear reasonable:
Leiomyosarcoma of ciliary or iridal musculature. Leiomyosarcomas are characterized by interlacing fascicles of neoplastic mesenchymal cells but can present with blastic or epithelioid morphology. Each myofiber is invested in a basal lamina containing collagen type IV and elaborates a reticulin- and collagen-rich matrix.
18
Anaplastic carcinoma of the ciliary body epithelium. In extremely aggressive tumors, these will present as anaplastic epithelial cells admixed with regions of mesenchymal dedifferentiation. Both intraocular leiomyosarcomas and ciliary carcinomas are described in humans.
19
The latter are extremely rare. Malignant transformation of anterior lens epithelium. Apart from the mouse, lens epithelial tumors have not been described in any species. However, lens epithelia will proliferate in response to trauma or cataract and will elaborate the basement membrane material typical of their native state.
2
,
6
,
8
On the basis of morphologic features of the intraocular tumors examined in this study, we were unable to differentiate between these three possible diagnoses. Further classification was dependent on immunohistochemical typing. Both lens capsule and the matrix elaborated by the tumor cells in this study were PAS positive. Similarly, both were also immunopositive for collagen type IV, indicating that the tumor matrices, like the lens capsule, are of basement membrane–type composition. Three markers of the 11 examined were immunopositive in anterior lens epithelium—vimentin, MSA, and crystallin alpha A. In normal eyes, the anterior lens epithelium expresses vimentin strongly—this diminishes with secondary lens fiber differentiation at the lens bow. 13 Vimentin downregulation is accompanied by appearance of the intermediate filaments phakinin and filensin, which typify the lens fiber phenotype. 12 These proteins are lens fiber–specific and are thus of little use in determining lens epithelial differentiation in our subset of ocular sarcomas. Cytokeratins (8/18) are present in lens epithelium during embryonal development but are not expressed in the adult lens epithelium. 13 Lens epithelial cells fall into the category of vimentin-cytokeratin–coexpressing cell types, with the caveat that cytokeratin expression is developmentally downregulated. Consequently, cytokeratin staining cannot be used as a marker of epithelial origin in lens epithelial tumors. In this study, demonstration of lens epithelial differentiation in neoplastic tissue was made on the basis of crystallin alpha A immunopositivity. Crystallins comprise the major structural component of the lens. 7
They are divided into alpha, beta, gamma, and delta crystallins—of these, alpha crystallins predominate in the lens. Alpha crystallins are of two subtypes. Crystallin alpha A is the only lens-specific protein and is expressed in both lens epithelium and lens fibers. Crystallin alpha B, in contrast, occurs in multiple extraocular tissues. 7
On the basis of immunohistochemical staining, we were able to determine the cell of origin in four of nine tumors. In general, regions with an epithelial phenotype provided the most diverse immunohistochemical pattern, whereas mesenchymal areas of the same tumor were less informative. In all cases, both epithelial and mesenchymal phenotypes expressed vimentin. Those tumors that expressed crystallin alpha A in addition could be identified as originating from the lens epithelium (three of nine tumors). In mesenchymal areas, immunopositivity for crystallin alpha A declined, whereas expression of MSA and vimentin prevailed. Tumors that expressed MSA and desmin were classified as leiomyosarcomas (one of nine tumors). Over half of the tumors examined expressed only vimentin and a variable amount of smooth muscle actin and collagen type IV. These could be classified only as undifferentiated sarcomas of myofibroblastic origin. The origin of the tumors from a pluripotent stem cell is a theoretical possibility; however, this scenario is generally considered most likely in tumors with a primitive neuroctodermal phenotype. 19
Crystallin expression will decline in experimentally induced murine lens epithelial tumors 15 and cultured lens cells, 17 and it is probable that this phenomenon occurs in spontaneously occurring lens epithelial tumors as well. Like lens epithelial cells in culture, transformed intraocular lens epithelia appear to express an immunohistochemical spectrum, which reflects their morphology and behavior rather than their origin. Thus, it appears that dedifferentiation of neoplasms derived from lens epithelium can reduce them to vimentin-expressing mesenchymal tumors that resist classification.
Lens epithelial tumors have not been reported in clinical material from any species. In mice, they can be induced by expression of SV40 Tag under the control of the crystallin alpha A promoter. 15 , 16 In this model, Tag expression is induced in the lens during early embryonal development, when posterior lens epithelial cells differentiate into primary lens fiber cells. These tumors will express crystallins, but this becomes downregulated and patchy with tumor expansion. 15 The murine model of lens neoplasia demonstrates that lens cells are not refractory to neoplastic transformation as previously thought. However, the primary lens fiber origin of these tumors differs fundamentally from the anterior lens epithelial tumors seen in cats. A more comparable phenomenon is seen in traumatized or cataractous human and canine lenses, which demonstrate proliferation and fibrous metaplasia of anterior lens epithelial cells. 2 , 3 , 6 In this case, lens epithelial cells assume a mesenchymal phenotype, produce collagen type IV, and express MSA, all features that characterize proliferating lens epithelial cells in culture. Expression of tumor growth factor beta appears to be the primary event promoting transdifferentiation and proliferation of lens epithelia in humans. 14 In one eye that was examined in this study, proliferating lens epithelial cells coexisted with an intraocular sarcoma, prompting speculation that proliferating lens epithelial cells induced by trauma may, in some cases, undergo neoplastic transformation. This may be enhanced by concurrent inflammation, presumably induced by release of lens proteins into the intraocular space.
In dogs and humans, lens epithelial tumors do not occur, despite regular appearance of proliferative lens epithelial foci associated with trauma or cataracts. Thus, it appears that the transition from lens epithelial proliferation to transformation is limited to the cat. This phenomenon is reminiscent of vaccine-induced sarcomas in cats. 9–11 These tumors arise after persistent inflammation and attendant fibroplasia induced by components of rabies and other vaccines. A variety of mesenchymal tumors can occur, including fibrosarcomas, leiomyosarcomas, rhabdomyosarcomas, malignant fibrous histiocytomas, and osteosarcomas. 11 Vaccine-induced sarcomas and ocular sarcomas in cats share the common features of an initiating insult, persistent inflammation accompanied by mesenchymal proliferation, and, finally, neoplastic transformation to produce a spectrum of tumor types.
