Abstract
Intraocular neoplasms are described in 2 adult rabbits. The left globe of an 8-year-old male rabbit was enucleated after chronic inflammatory disease resulted in a nonvisual eye. The left globe of a 5-year-old female rabbit also was enucleated after a history of lens-induced uveitis, cataract formation, and resultant glaucoma. In both rabbits, histopathology revealed a variably pleomorphic, poorly differentiated, invasive, intraocular spindle cell neoplasm closely associated with lens and lens capsular fragments. Gram stains failed to detect bacterial organisms or Encephalitozoon cuniculi. Polymerase chain reaction assays, used to amplify the 16S RNA gene of numerous bacteria and E. cuniculi, were also negative. Immunohistochemical staining demonstrated strong, diffuse expression for vimentin; however, staining for smooth muscle actin, cytokeratin, S100, and desmin were negative. Long-standing intraocular inflammation and/or traumatic insults to the eyes were considered as causes of these neoplasms. The histologic features of these intraocular neoplasms closely resemble post-traumatic ocular sarcomas in cats.
Post-traumatic sarcomas are neoplasms that arise secondary to ocular trauma involving the lens and have been previously described only in cats. 6 These feline neoplasms consist of a proliferation of spindle to polyhedral cells that often surround a ruptured lens. In more advanced tumors, the neoplastic infiltrate can extend along the posterior portion of the globe and the optic nerve. Metastasis also has been reported. 1 The neoplastic cells are consistently vimentin positive, and a subset of cells may express variable positivity for crystallin alpha A and collagen type IV. Immunohistochemical staining for pancytokeratin and Melan A are negative in these feline neoplasms. Most feline tumors are of myofibroblastic origin, but a small percentage are of lens epithelial cell origin. 11 The present report describes the gross, histologic, and immunohistochemical findings ofintraocular sarcomas in 2 rabbits that were similar to feline post-traumatic sarcomas.
An 8-year-old, 3.68 kg, male rabbit of unspecified breed (case 1) was evaluated by the University of Tennessee College of Veterinary Medicine (UTCVM, Knoxville, TN) for an abnormal left eye. The owner had obtained the rabbit 2 months prior to the ocular evaluation. The medical history indicated that the rabbit had been treated for eye infections by the previous owner. Upon obtaining the rabbit, the present owner noted ocular discharge from the left eye and a black spot on the cornea that progressively enlarged for 4 weeks prior to presentation at UTCVM.
The physical examination was unremarkable with the exception of the left eye. The direct pupillary light reflex was absent in the left eye but normal in the right eye. In the left eye, the ventral third of the cornea appeared to have a perforation through which a 10 mm × 7 mm × 5 mm pink to tan mass extended from the anterior chamber (Fig. 1). The mass contained large, tortuous blood vessels on the anterior surface, and the enlarged conjunctival space was filled with a suppurative exudate (Fig. 1). Ocular examination of the right eye was unremarkable. The left globe was subsequently removed by a routine transconjunctival enucleation and submitted for histopathologic examination (Fig. 2). The rabbit was re-examined 14 days postoperatively, at which time the skin incision had healed and the enucleation site appeared unremarkable.
A 5-year-old, female, domestic rabbit of unspecified breed (case 2) was presented to the referring veterinarian with a 4-year history of lens-induced uveitis, cataract formation, and iris bombe of the left eye. A fibrosarcoma had been excised from the dorsal limbus 2 months prior to referral. Due to diffuse enlargement of the globe and the suspicion of an intraocular neoplasm, the affected eye was subsequently enucleated by the referring veterinarian and submitted to the pathology service at the University of Wisconsin College of Veterinary Medicine (Madison, WI).
The globes from cases 1 and 2 were submitted in 10% buffered formalin and Zenker solution, respectively. The specimens were transected, routinely processed, embedded in paraffin, and sectioned at 5 μm. Sections were routinely stained with hematoxylin and eosin, tissue Gram stain, and periodic acid-Schiff (PAS) stain; coverslipped; and examined by light microscopy. Additionally, immunohistochemical staining was performed at the UTCVM Immunohisto-chemistry Laboratory using anti-cytokeratin (AE1/AE3), vimentin, desmin, smooth muscle actin (SMA), Melan A, and S100 antibodies. Additional 5-μm tissue sections were placed on positively charged slides for immunohistochemical staining. All slides were deparaffinized and rehydrated through graded alcohols to water. For cytokeratin staining, antigen sites were unmasked using proteinase K a treatment at room temperature for 5 min. Antigen retrieval for vimentin, b desmin, c SMA, d and Melan A e was performed by heating the tissue sections in buffered ethylenediamine tetra-acetic acid (EDTA; pH 9) for 25 min at 95°C and cooling for 20 min at room temperature. An antigen retrieval step was not required for S100. f
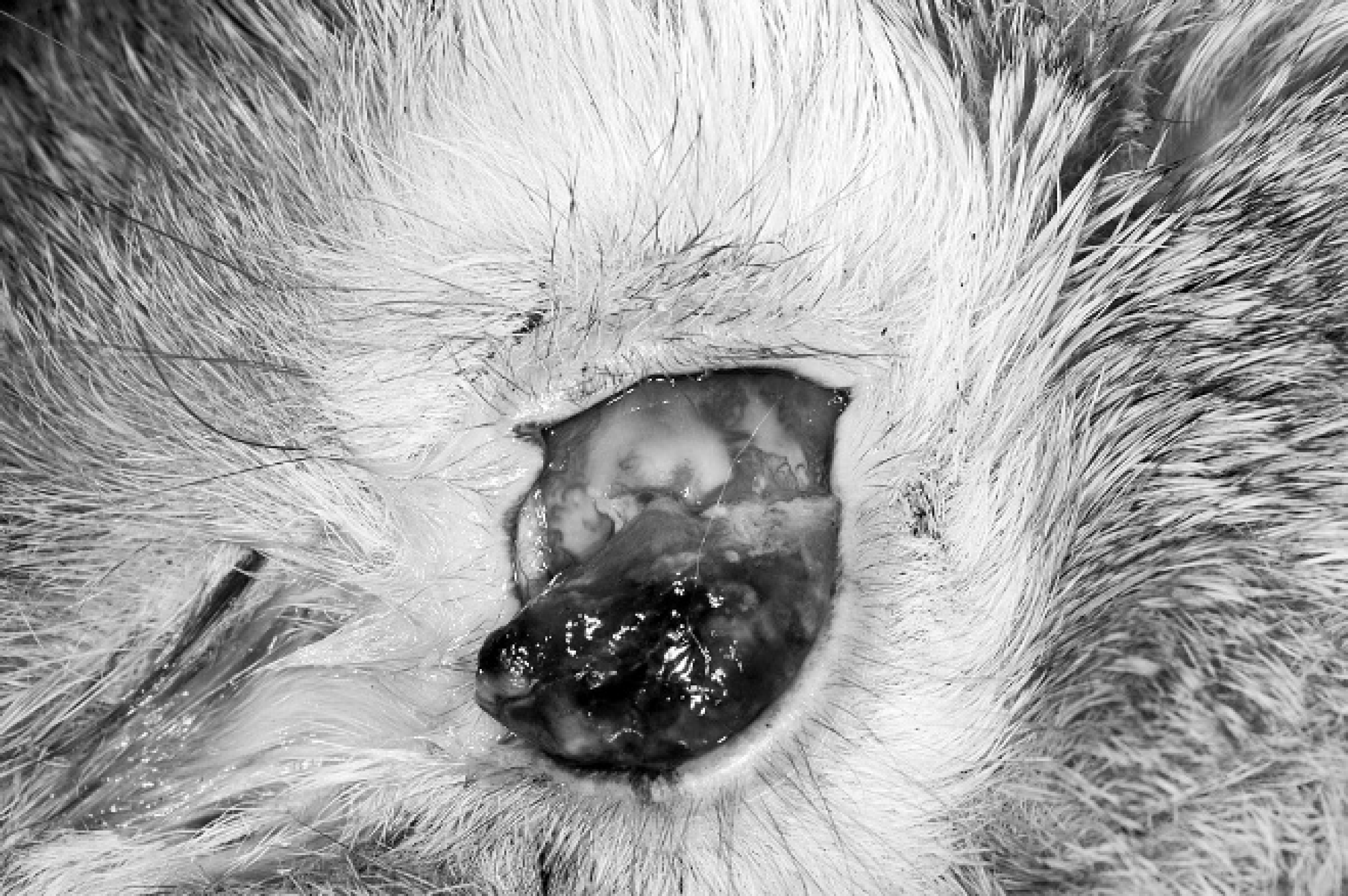
Photograph of an intraocular mass protruding through a ventral corneal perforation. Rabbit, intraocular sarcoma, case 1.
All slides were rinsed in Tris-buffered saline (pH 7.6) for 5 min before being loaded into an automated stainer. g For cytokeratin, vimentin, desmin, S100, and SMA, tissue sections were initially treated with 3% hydrogen peroxide for 5 min followed by serum-free protein blocking a for 5 min. For Melan A, an initial dual endo enzyme block a was performed for 10 min followed by serum-free protein blocking a for 5 min. For immunohistochemical staining, the primary antibodies were applied as follows: mouse AE1/AE3 cytokeratin (1:800), mouse V9 vimentin (1:8,000), mouse D33 desmin (1:800), mouse 1A4 SMA (1:1,000), mouse A103 Melan A (1:50), and mouse 4C4.9 S100 (1:1,000). All primary antibodies were incubated for 30 min. For cytokeratin, vimentin, desmin, S100, and SMA, a horseradish peroxidase-labeled polymer system h was applied for 30 min. Slides were then rinsed in Tris-buffered saline, and 3,3′-diaminobenzidine chromogen was applied for 10 min. For Melan A, a streptavidin and biotin system a was applied for 30 min followed by permanent red chromogen a for 10 min. After rinsing, all slides were counterstained with hematoxylin, i dehydrated, and cover-slipped. Negative control slides were processed without primary antibody and with substitution of Universal Negative Control+ mouse serum. a Vimentin validity was evaluated using both canine master control blocks composed of skeletal muscle, smooth muscle of urinary bladder, small intestine, and large intestine and rabbit tissues including extraocular skeletal muscle and smooth muscle of blood vessels that acted as internal controls within the slides examined. Vimentin expression was detected within periocular fat, skeletal muscle, and vascular smooth muscle. Cytokeratin (AE1/AE3) was also validated using canine master control blocks composed of epithelium of the liver, thyroid, pancreas, kidney, and small and large intestine and rabbit tissues including conjunctival epithelium that acted as an internal control within the slides examined. A control for S100 included sections of brain from a juvenile intact female rabbit, which strongly expressed S100 within neurons. Desmin was validated using both canine master control blocks composed of skeletal muscle and rabbit tissues including extraocular muscles that acted as an internal control within the slide examined. Desmin expression was detected in the periocular skeletal muscle.
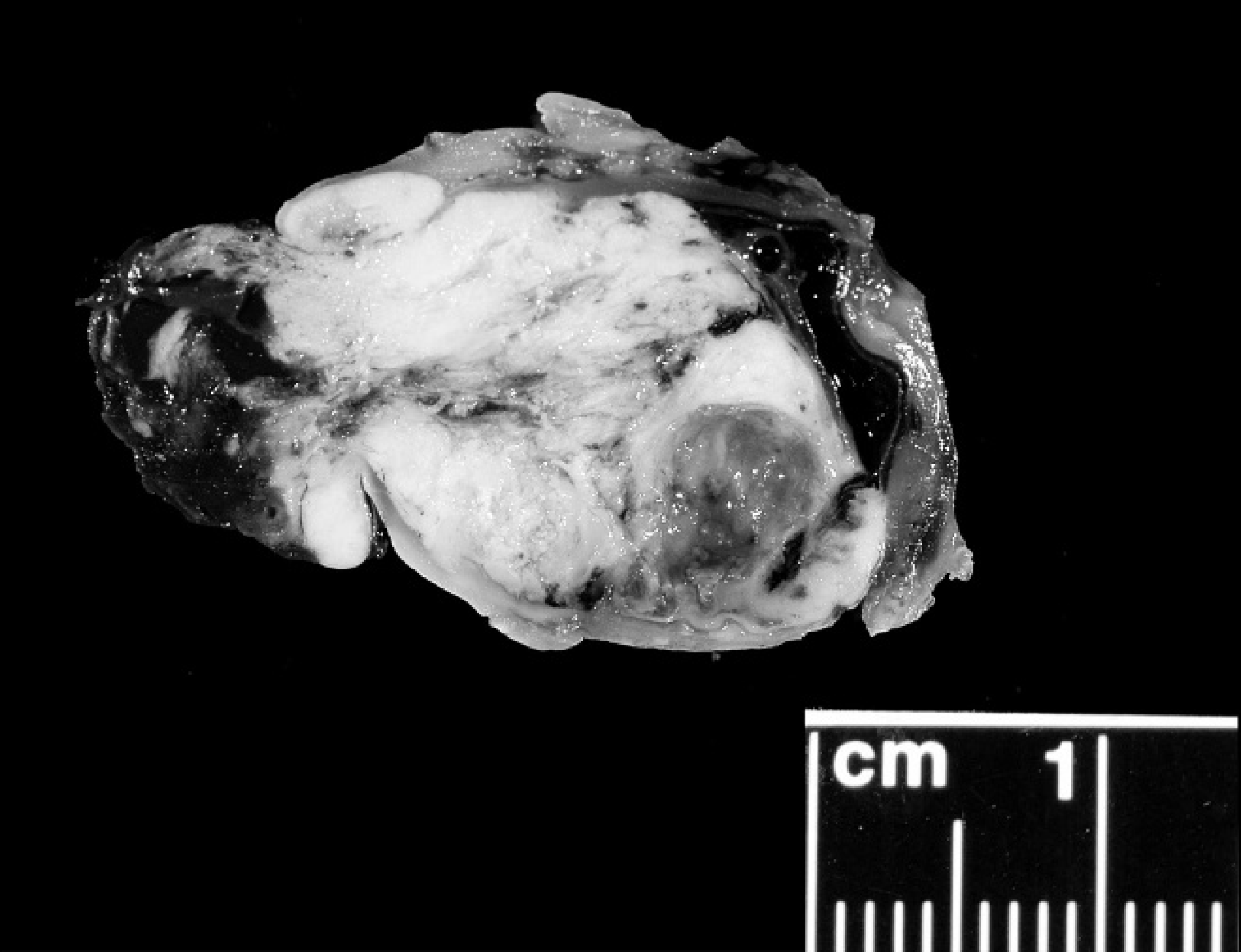
Cross-section of formalin-fixed enucleated left globe. Normal intraocular architecture is obliterated by a multinodular, tan mass that extends through the corneal perforation. Rabbit, intraocular sarcoma, case 1.
DNA was extracted from paraffin-embedded tissue using a DNA extraction kit j and then subjected to polymerase chain reaction (PCR) amplification using universal primer sets against 16S ribosomal RNA (rRNA) as reported previously. 5,8,10 For detection of Encephalitozoon cuniculi DNA, previously reported conventional and real-time PCR assays were employed. 3,9 Alpaca tissues with confirmed E. cuniculi were used as a positive control. 9
The normal architecture of the globe from case 1 was effaced by a tan to pink infiltrative mass that extended throughout the vitreous, posterior, and anterior chambers (Fig. 3). The mass was composed of a population of spindle cells forming streams and sheets that infiltrated the iris leaflets and cornea. The neoplasm extended through the sclera into the adjacent extraocular musculature. The nuclei were round to irregular with coarse chromatin and showed marked anisokaryosis (up to a 20-fold variation in nuclear size). Prominent, multiple nucleoli were present, and up to 4 mitotic figures were observed per high-power field. Multinucleated neoplastic cells contained up to 7 nuclei. The cytoplasm of the neoplastic cells was moderate in amount, fibrillar, and eosinophilic with indistinct cell borders. The lens had ruptured, and multiple lens capsule fragments were embedded within the mass. The retina was detached, and the retinal pigmented epithelium was hypertrophic. The remaining retinal layers were absent (atrophy). Extensive necrosis, hemorrhage, and occasional mineralization were present throughout the mass. The histologic diagnosis was intraocular anaplastic spindle cell sarcoma, most consistent with post-traumatic ocular sarcoma.
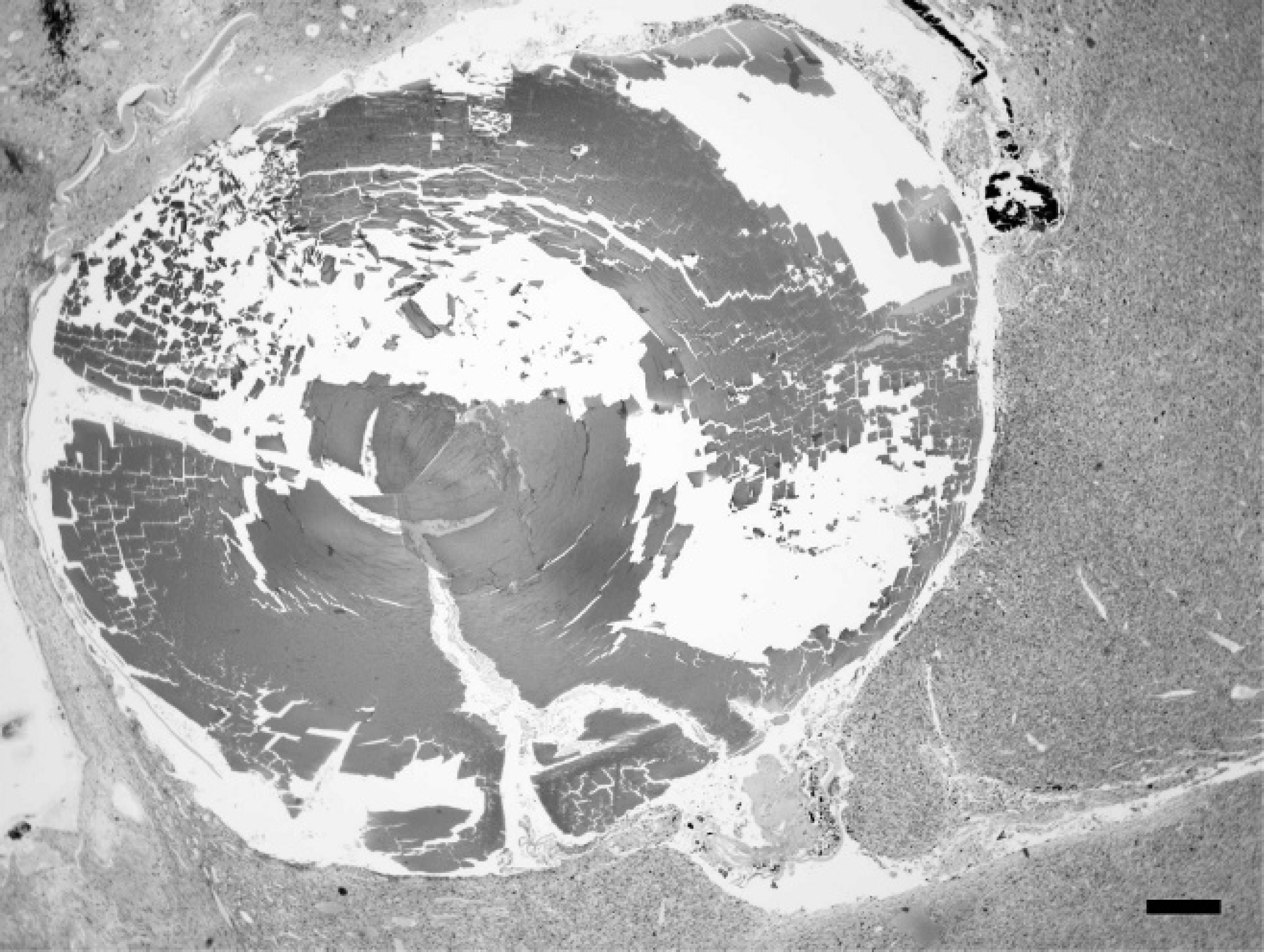
Photomicrograph of the left globe. Much of the globe is filled with a neoplasm surrounding remnants of the lens. Rabbit, intraocular sarcoma, case 1. Hematoxylin and eosin. Bar 5 200 μm.
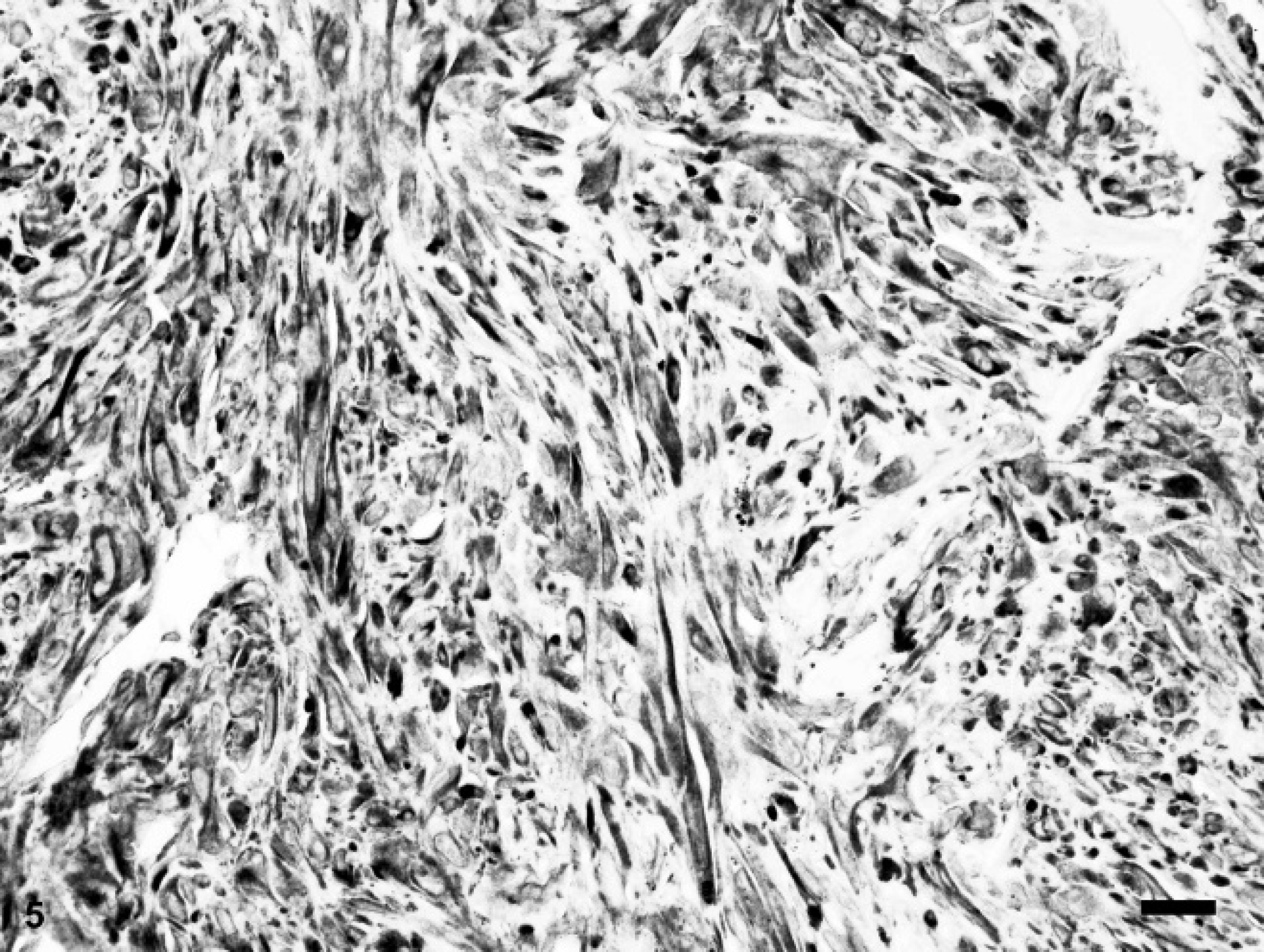
The globe from case 2 bulged outwards circumferentially at the limbal margin. The ocular contents posterior to the iris were disorganized and displaced by an infiltrative mass consisting of highly pleomorphic and anaplastic spindle cells. Occasional multinucleated giant cells had a strap-like configuration. The nuclei were elongate to irregularly shaped with coarse chromatin and prominent single to infrequent multiple nucleoli (Fig. 4). Marked anisokaryosis was present with up to an 8-fold variation in nuclear size. Multinucleated cells contained up to 5 nuclei. Mitotic figures were rare. The cytoplasm was moderate in amount, lightly eosinophilic, and fibrillar with indistinct cell borders. Neoplastic cells infiltrated the ciliary body, iris, and limbal to equatorial sclera, extending into the orbital and subconjunctival tissue. A wrinkled lens capsule surrounding residual globular lens material was entrapped within the neoplastic mass. Complete retinal detachment was accompanied by retinal atrophy, fibrous metaplasia, and hypertrophy of the retinal pigmented epithelium. The histologic diagnosis was intraocular spindle cell sarcoma, possibly post-traumatic ocular sarcoma.
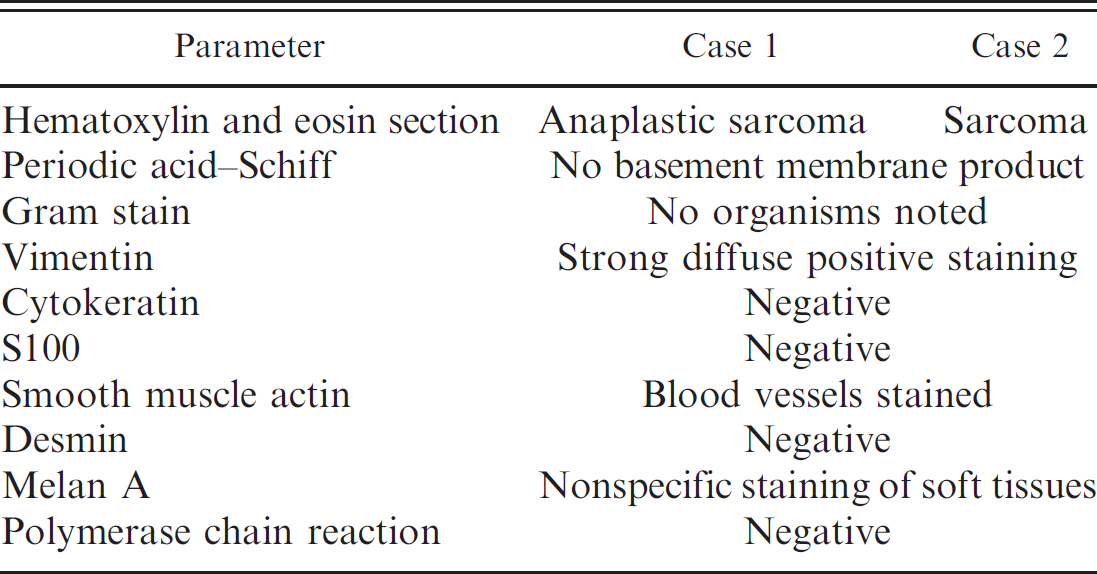
The results of the various diagnostic tests were similar in both neoplasms and are summarized in Table 1. In both neoplasms, PAS stains failed to identify matrix production by the neoplastic spindle cells, and tissue Gram staining failed to identify bacteria or E. cuniculi organisms. Immunohistochemical staining demonstrated strong diffuse positive expression for vimentin (Fig. 5). Cytokeratin, SMA, S100, and desmin were not expressed by the neoplastic cells. Polymerase chain reaction, using primers specific for bacterial 16S rRNA, failed to amplify any DNA fragment. No E. cuniculi-specific DNA was detected in either conventional or real-time PCR assays.
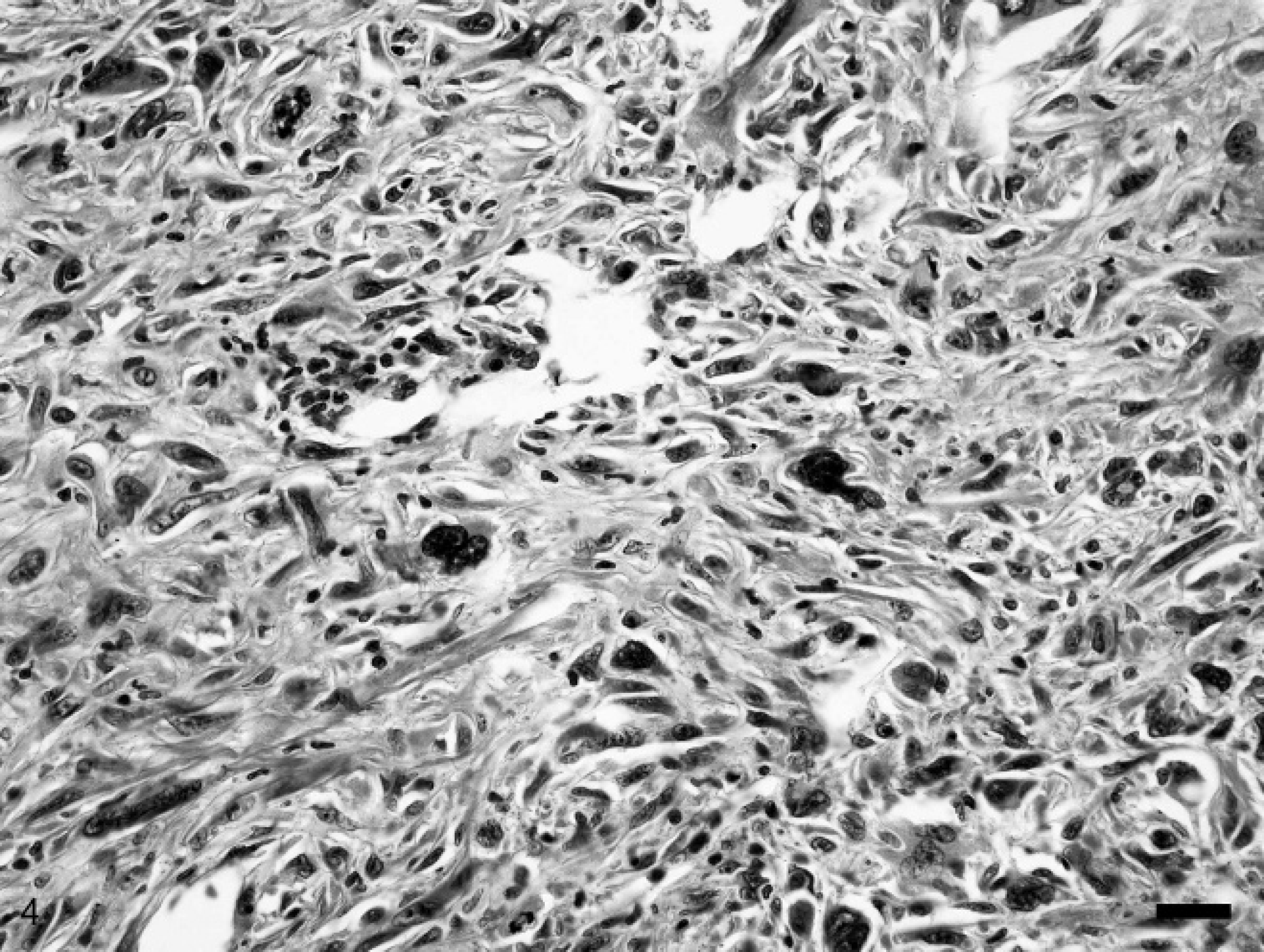
Photomicrograph of an intraocular sarcoma composed of pleomorphic spindle cells with marked anisokaryosis, karyomegaly, and frequent mitoses. Rabbit, intraocular sarcoma, case 2. Hematoxylin and eosin. Bar = 50 μm.
Naturally occurring, post-traumatic sarcomas have previously been reported only in cats. 6 The current report characterizes 2 intraocular sarcomas in rabbits. The feline neoplasms were associated with history of ocular trauma or a protracted uveitis, which ultimately resulted in leakage of lens protein and rupture of the lens. 1 Trauma was not documented for either rabbit. However, the 2 intraocular sarcomas in the rabbits of the present report were similar to feline intraocular sarcomas in that the lenticular region seemed preferentially involved in tumor development, aggressive infiltration of the vitreous chamber and anterior chambers was present, and subsequent perforation of the cornea occurred in each neoplasm. Additionally, neoplastic cells in both tumors extended through the sclera into the surrounding extraocular tissues by the time of enucleation, resulting in incomplete surgical resection. Because both rabbits were lost to follow-up examination after surgery, recurrence of the neoplasms could not be determined. Extraocular tissue invasion in cats is reported to occur via the optic nerve 1 ; however, the optic nerves were not evaluated in these 2 rabbits. In contrast, the intraocular sarcomas in the rabbits extended through the sclera into the extraocular tissues.
Histologically, both the feline and rabbit tumors were composed of a proliferation of anaplastic mesenchymal cells, often with multinucleated neoplastic cells. Although cartilage and bone production have been reported in feline intraocular sarcomas, this was not observed in the rabbit neoplasms. 6 Lens fragments were present within both rabbit intraocular sarcomas, but this was not a feature of most feline intraocular sarcomas.
Results of routine, special, and immunohistochemical stains and polymerase chain reaction analysis for intraocular sarcomas in 2 rabbits.
The cellular origin of feline intraocular sarcomas is controversial, but some neoplasms are clearly sarcomatous and thought to originate from metaplastic myofibroblasts, which express vimentin. Further attempts to identify the neoplastic lineage have excluded smooth muscle, skeletal muscle, nerve, and epithelial cells. In addition, a subset of feline neoplasms has expressed crystallin alpha A, a marker for lens epithelium. Production of PAS-positive basement membrane material also has been reported in neoplasms that originate from lens capsule epithelium. 11 The rabbit neoplasms in the current report were strongly positive for vimentin but negative for PAS-positive matrix. Cytokeratin, desmin, S100, and Melan A reactivity is not typically observed in the feline tumors. 11 Immunohistochemical staining was negative for cytokeratin, desmin, S100, and SMA in both rabbit intraocular sarcomas. However, Melan A staining was nondiagnostic because of diffuse nonspecific staining of all ocular structures. Immunohistochemical staining for crystallin alpha A was not done, because the primary antibody is not available commercially. Lens epithelial neoplasms have been experimentally induced in mice by gene manipulation. 7
The cause of the prolonged uveitis with lenticular destruction in both rabbits is unknown. Encephalitozoon cuniculi has been implicated in lenticular rupture in rabbits and is thought to colonize the lens after in utero infection. 2,4 Tests for E. cuniculi were carried out because infection with this organism could be responsible for eventual development of lens-induced ocular sarcomas in rabbits. However, the absence of infection based on histologic examination and PCR analysis is not unexpected based on the long duration of clinical signs prior to the development of neoplasia. No other bacteria could be documented in either neoplasm.
In conclusion, rabbits with ocular disease associated with rupture of the lens may develop intraocular sarcomas with a cellular morphology and aggressive behavior similar to feline post-traumatic sarcomas. Vimentin expression is the only consistent immunohistochemical marker. More cases of intraocular sarcoma in rabbits should be evaluated to determine the cellular lineage, pathogenesis, and prognosis of this particular neoplasm.
Photomicrograph of an intraocular sarcoma with strong, diffuse expression of vimentin. Rabbit, intraocular sarcoma, case 2. Bar = 50 μm.
Acknowledgements. The authors thank LaDonna Mrkonjich for performing the immunohistochemical procedures, Anik Vasington for providing graphical assistance, and Misty Bailey for technical editing.
Footnotes
a.
Dako North America Inc., Carpinteria, CA.
b.
V9 clone, Dako North America Inc., Carpinteria, CA.
c.
D33 clone, Dako North America Inc., Carpinteria, CA.
d.
1A4 clone, Dako North America Inc., Carpinteria, CA.
e.
A103 clone, Dako North America Inc., Carpinteria, CA.
f.
4C4.9 clone, Novus Biologicals LLC, Littleton, CO.
g.
Model S3400, Dako North America Inc., Carpinteria, CA.
h.
EnVision+ Anti-Mouse, Dako North America Inc., Carpinteria, CA.
i.
Richard-Allan Scientific, Kalamazoo, MI.
j.
QIAamp® minikit, Qiagen Inc., Valencia, CA.
