Abstract
We attempt to determine and compare the localization of apolipoproteins (apo) apoA-I and B-100 in atherosclerotic lesions of canine aortas, coronary arteries, and the peripheral arteries, using immunohistochemical techniques. Histopathologically, atherosclerotic lesions were characterized by deposition of lipids and infiltration of lipid-laden foamy cells in the tunica intima and tunica media, sometimes forming fibrofatty plaques containing abundant sudanophilic and mineralized material. Canine apoA (CapoA)-I and canine apoB (CapoB)-100 immunopositive signals were simultaneously observed in mild and severe atherosclerotic lesions of the aorta, coronary arteries, splenic arteries, and renal arteries in the double-immunolabeled sections. Both CapoAI and CapoB-100 positive signals were seen in the cytoplasm of endothelial cells, smooth muscle cells, and macrophages. The subendothelial space and extracellular matrix in the tunica intima and media were also positive. Neither CapoA-I nor CapoB-100 positive signals were seen in normal arteries. These findings closely resemble those of the localization of apoA-I and apoB-100 in human atherosclerotic lesions.
Apolipoproteins A (apoA-I) and B (apoB) are the major protein components of very low-density lipoproteins (VLDL), intermediate-density lipoproteins (IDL), low-density lipoproteins (LDL), and high-density lipoproteins (HDL), and their association with atherosclerosis and other forms of cardiovascular disease in humans has been studied. 9 , 14 Many investigators have measured plasma levels of apoB and apoA-I in atherosclerotic patients. An elevated plasma level of apoB is the most important risk factor in patients with atherosclerosis and other forms of cardiovascular disease. Conversely, lowered plasma levels of apoA-I is considered to be correlated to an increased risk of such diseases. 1 Little information concerning plasma levels of apoB and apoA-I in normal dogs has been gathered. The distribution of plasma lipids and lipoproteins in dogs is quite unlike that in humans, with dogs having approximately five to six times as much HDL as LDL. 10 Although a few reports referred to the distribution of apoB in canine atherosclerotic lesions, 7 , 8 there is no information about the distribution of apoA-I. In this study, using immunohistochemical techniques, we attempt to determine and compare localization of apoA-I and apoB-100 in normal and atherosclerotic lesions of canine aortas, coronary arteries, and peripheral arteries.
The tissues used in this study were collected from aortas, coronary arteries, spleen, and kidney of dogs (n = 10) over 10 years of age with systemic atherosclerosis (including four dogs noted in an earlier report) 7 as well as normal dogs (n = 3) aged 3 years. Sections for histopathologic examination were cut at 4 μm from paraffin-embedded blocks and stained with hematoxylin and eosin (HE). Frozen sections were cut at 10 μm in a cryostat at −20 C and stained with Sudan black B. Histopathologically, mild atheromatous lesions were observed in the aorta. The subendothelial space was dilated by an extracellular network containing small amounts of sudanophilic material in sections stained with Sudan black B. A small number of macrophages and occasionally neutrophil infiltrates were observed in the subendothelial space with lipid deposition. The intimal and medial smooth muscle cells usually had swollen nuclei and abundant cytoplasm. Severe atheromatous lesions containing abundant sudanophilic material were frequently observed in the tunica media and adventitia of coronary arteries. Numerous macrophages with foamy cytoplasm infiltrated the tunica intima and media (Fig. 1). Smooth muscle cells frequently had vacuolated cytoplasm. Subendothelial space of central and penicillar arteries of the spleen was dilated by an extracellular network with a small number of macrophage infiltrates. Some macrophages were enlarged and had foamy cytoplasm. In severe lesions, the tunica media and cystic space contained enlarged macrophages with foamy cytoplasm, hyaline material, and mineralized material (Fig. 2). Severe or mild atheromatous lesions containing abundant sudanophilic material were frequently observed in the interlobar, arcuate, and interlobular arteries of the kidney. No significant changes were observed in walls of aortas, coronary arteries, splenic arteries, renal arteries, and glomeruli in any samples from normal dogs.
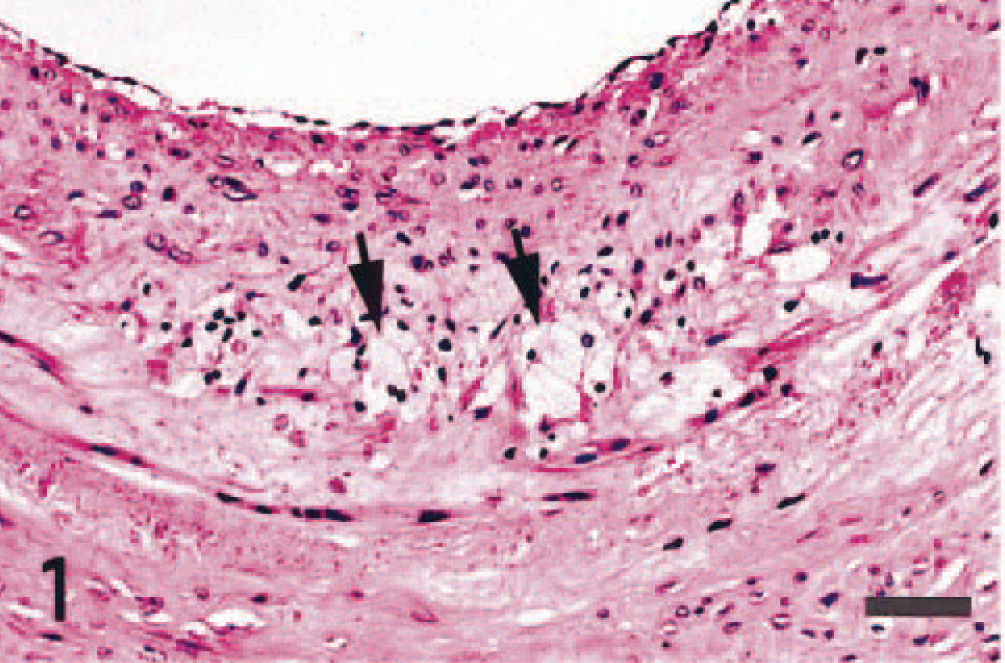
Atherosclerosis; dog. Atheromatous lesion in the coronary artery. Many macrophages with foamy cytoplasm (arrows) infiltrate in the tunica media. HE. Bar = 20 μm.
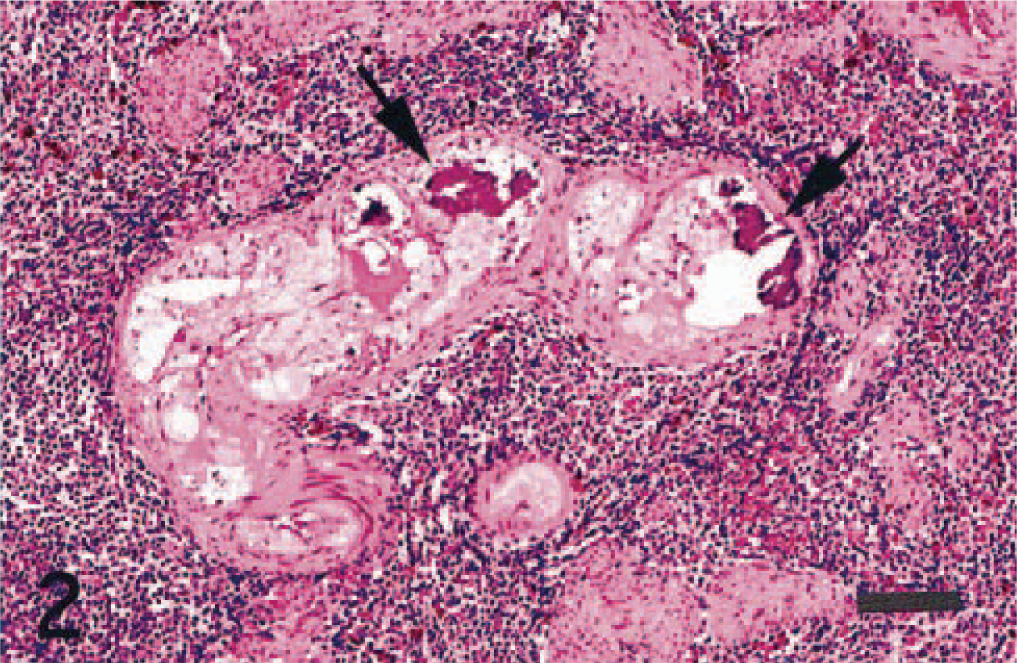
Atherosclerosis; dog. Atheromatous lesion in the central artery of the spleen. Enlarged macrophages with foamy cytoplasm, hyaline material, and mineralized material (arrows) are seen in the tunica media. HE. Bar = 50 μm.
Distribution of canine apoB-100 (CapoB-100) immunopositive signals in atherosclerotic lesions has been reported previously 7 , 8 . Immunopositive reactions against anti-CapoB-100 polyclonal antibody 12 were seen in endothelial cells and foamy cytoplasm of macrophages (Fig. 3a) by the avidin–biotin–peroxidase complex (ABC) procedure with Mayer's hematoxylin (Vectastain Elite ABC Kit; Vector Laboratories, Burlingame, California, USA). Subendothelial space and extracellular matrix in tunica intima and media also were positive. Distribution of immunoreaction against anti-canine apoA-I (CapoA-I) polyclonal antibody 12 appeared similar to that of CapoB-100 immunopositive signals in the aortas, coronary arteries, splenic arteries, and renal arteries. 7 , 8 CapoA-I signals were observed in the cytoplasm of endothelial cells, macrophages (Fig. 3b), and extracellular matrices (Fig. 4) in the atheromatous plaques. The swollen cytoplasm of smooth muscle cells in the tunica intima and media also reacted (Fig. 5).
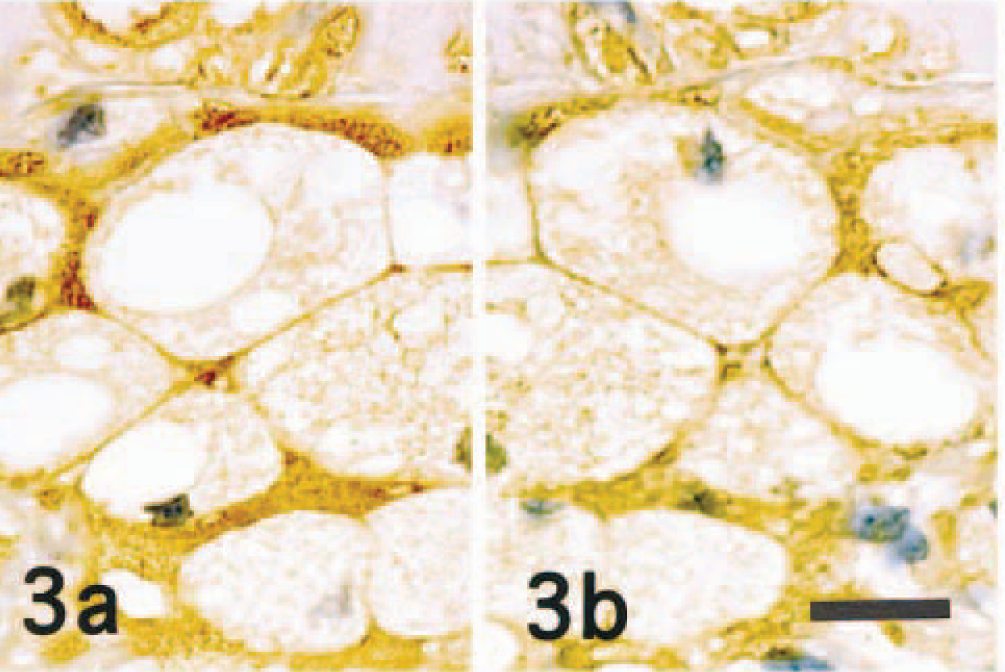
Atherosclerosis; dog. An immunohistochemical-labeling mirror image of the atheromatous lesion in the coronary artery. CapoB-100 ( Fig. 3a ) and CapoA-1 ( Fig. 3b ) immunopositive signals are seen in the foamy cytoplasm of macrophages and extracellular matrix in the lesion. ABC complex method, Mayer's hematoxylin counterstain. Bar = 20 μm.
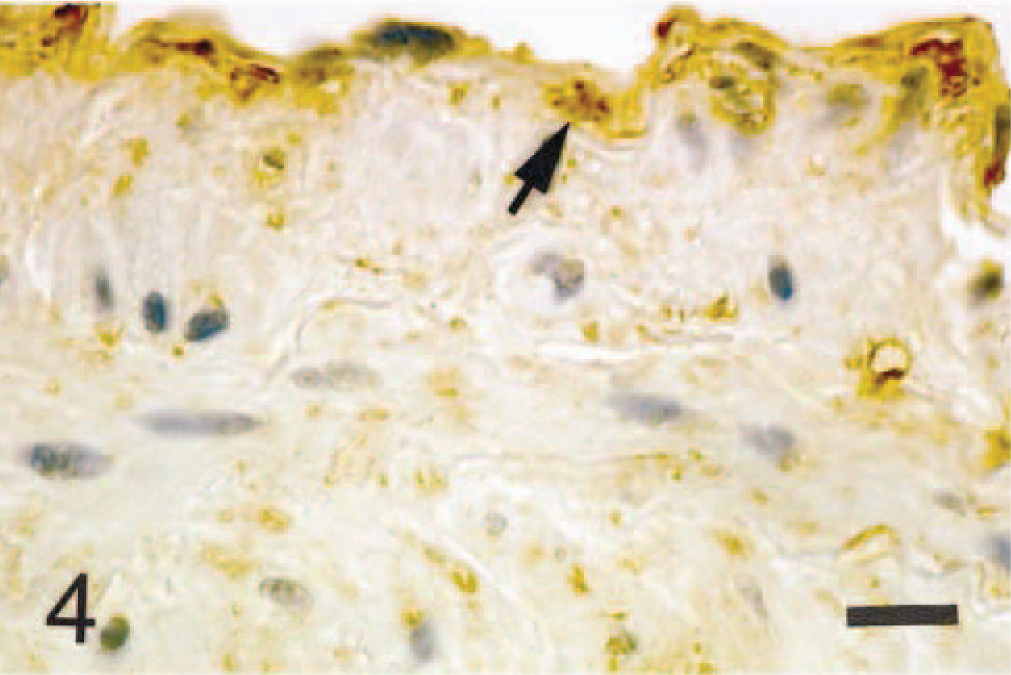
Atherosclerosis; dog. Immunohistochemical labeling of the aorta. CapoA-I immunoreactivity is seen in the endothelial cells and extracellular matrix in the intima (arrow). ABC complex method, Mayer's hematoxylin counterstain. Bar = 20 μm.
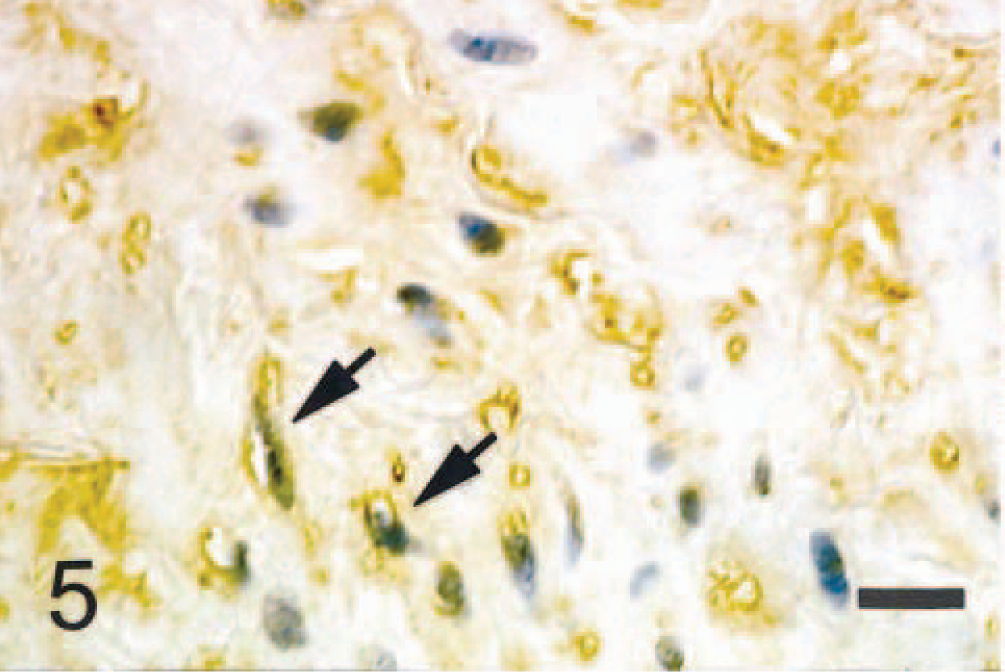
Atherosclerosis; dog. Immunohistochemical labeling of the aorta. CapoA-I immunoreactivity is seen in the cytoplasm of smooth muscle cells (arrows) in the tunica media. ABC complex method, Mayer's hematoxylin counterstain. Bar = 20 μm.
Localization of CapoB-100 and CapoA-I in the same atherosclerotic lesion was simultaneously detected by using double immunolabeling. The section immunostained with rabbit anti–CapoB-100 antibody 12 and DAB was treated with 0.005% biotin in phosphate-buffered saline (PBS) to mask avidin for 30 minutes at room temperature and then incubated with CapoA-I antibody as described above. The section was reacted with biotinylated goat anti-rabbit IgG (immunoglobulin G), followed by incubation with alkaline phosphatase–conjugated streptoavidin and alkaline phosphatase substrate Fast blue (SAB-AP Kit; Nichirei Co., Tokyo, Japan), which yielded a blue precipitate. CapoB-100 (brown signal) and CapoA-I (blue signal) were simultaneously observed in the same atheromatous lesions of the aortas, coronary arteries, splenic arteries (Fig. 6), and renal arteries. No immunopositive signals against CapoB-100 and CapoA-I antibodies were observed in walls of aortas, coronary (Fig. 7) and splenic arteries, and renal arteries and glomeruli in any samples from normal dogs.
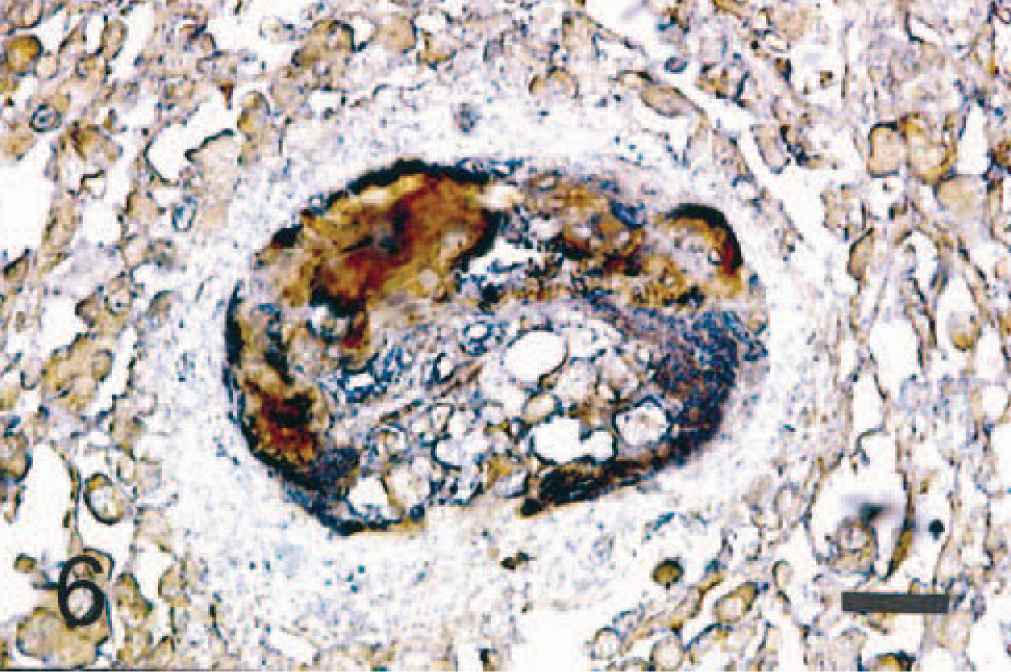
Atherosclerosis; dog. Double immunolabeling of the atheromatous lesion in the central artery of the spleen. CapoB-100 (brown signal) and CapoA-I (blue signal) are simultaneously seen in the atheromatous lesion. ABC complex method. Bar = 20 μm.
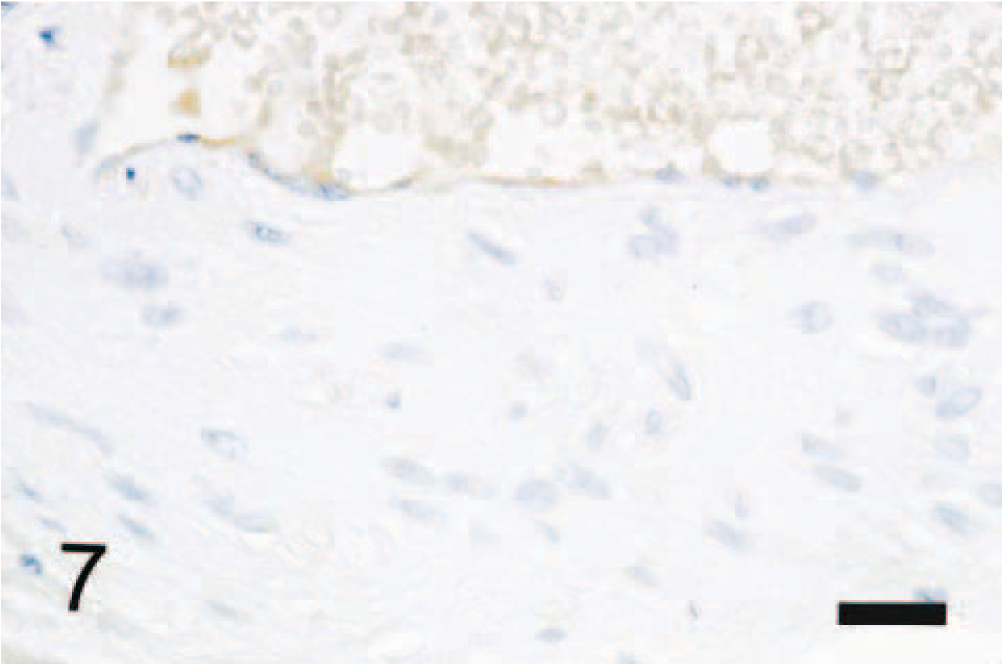
Coronary artery; dog. No immunoreactivity for CapoA-I is seen in the wall of the coronary artery. ABC complex method, Mayer's hematoxylin counterstain. Bar = 30 μm.
Immunohistochemical expression of apoB in atheromatous and atherosclerotic lesions reflects the accumulation of LDL and VLDL in the wall of arterial vessels in humans and animals. 2 , 7 , 14 Transport of LDL and VLDL can occur across intact endothelial cells either by a receptor-mediated uptake of lipoprotein or by a nonspecific uptake through micropinocytic channels. 16 Increased LDL and VLDL plasma levels constitute a major risk factor in atherosclerosis. 14 HDLs have an inverse correlation with the risk of developing atherosclerosis because of their participation in the removal of LDL from atherosclerotic lesions, their ensuing transport to the liver for excretion, their ability to inhibit the oxidation of LDL and the proatherogenic activities of oxidized LDL. Therefore, a decreased HDL plasma level also is regarded as a risk factor for the development of atherosclerosis in humans. 13 , 18 Immunohistochemical distribution of apoA in atherosclerotic lesions has been detected in a few human cases. 2 ApoA and apoB were found to be present in atherosclerotic plaques but absent in normal arterial tissue. In the plaques, both apoA and apoB were found extracellularly, primarily in the lipid core, and also in connective tissue. In addition, both apoA and apoB were found intracellularly in foam cells. 2 The implications of the localization of apoA in atherosclerotic plaques are not obvious. ApoA may merely diffuse passively into plaque along with other proteins as endothelial permeability increases or it may be actively transported to this site. 2
Recent studies in humans revealed that HDL can reduce cholesterol ester hydroperoxides to the corresponding cholesterol ester hydroxides. 5 , 15 This reducing activity of HDL is potentially antiatherogenic based on the fact that during the course of such a reduction, 15 HDL also reduces cholesterol ester hydroperoxides transferred from oxidized LDL through cholesterol ester transfer protein and thus can potentially detoxify proatherogenic oxidized LDL. 3 In addition, reduction of lipid hydroperoxides by HDL is accompanied by the formation of specifically oxidized apoA-I and apoA-II containing methionine sulfoxide as the selective oxidation product. Selective oxidation of HDL methionine residues by nucleophilic oxidants such as chloramine T inhibited subsequent lipid hydroperoxides, thus reducing activity in a dose-dependent manner. 5 , 17 Therefore, the coexistence of both apoA and apoB in human atherosclerotic lesions strongly suggests that the ability of HDL to reduce and thereby detoxify potentially proatherogenic lipid hydroperoxides is because of a reaction with specific methionine residues on apoA-I and apoA-II. However, if oxidation of methionine to methionine sulfoxide in HDL occurs extracellularly and is not reversible in biologic systems, its formation can be considered as oxidative damage, and apoA-I–A-II–containing methionine sulfoxide would be expected to accumulate at sites of oxidative stress. 5
Like oxidatively modified LDL, oxidatively modified HDL also causes intracellular cholesterol accumulation. 2 Oxidized HDL is internalized by macrophages through the same scavenger receptor that is responsible for the clearance of oxidized LDL 6 , 11 and depresses both cholesterol esterification and cholesterol biosynthesis. 2 , 5 Therefore, HDL oxidation may generate modified lipoproteins capable of modulating the cholesterol homeostasis of macrophages.
In the present study, deposition of apoB-100 and apoA-I was demonstrated in both mild and severe atherosclerotic lesions. The coexistence of apoB-100 and apoA-I was demonstrated in the extracellular matrices and cytoplasm of endothelial cells, infiltrated macrophages, and smooth muscle cells. Using double-labeling immunohistochemistry, deposits of apoB-100 and apoA-I were simultaneously demonstrated in the extracellular matrices, cytoplasm of the same endothelial cells, macrophages, and smooth muscle cells. These findings closely resemble those of the localization of apoB-100 and apoA-I in human atherosclerotic lesions. 2 Unlike human HDL, canine HDL lacks the oxidation-sensitive methionine112 and methionine148 of human apoA-I, exhibiting only slight cholesterol ester hydroperoxide–reducing activity. 4 , 5 The remaining low-reducing activity of canine HDL may be because of an interaction of lipid hydroperoxides with other redox-active amino acids, possibly including methionine86. 5 Canine HDL shows a much weaker lipid hydroperoxide–reducing activity and lower extent of formation of oxidized forms of apoA-I than does human HDL. 5 This property of canine HDL may be the reason why dogs have approximately five to six times as much high HDL as LDL. 10 However, the implication of the localization of apoA-I in canine atherosclerotic plaques remains unclear.
