Abstract
Liver tumors of unknown cause have frequently been described in polar bears. Concurrent decrease of vitamin A levels and chronic liver disease are associated with hepatic carcinogenesis in humans. More than 90% of the body's vitamin A is stored in the liver, where it is bound to an intracellular retinol-binding protein (RBP). Therefore, in this retrospective study, RBP was assessed by immunohistochemistry in liver sections of 11 polar bears. Two of these polar bears had hepatocellular carcinoma, four showed other chronic liver changes, and five had normal livers. In normal livers, the cytoplasm stained diffusely positive with intensely staining cytoplasmic granules. RBP staining was evaluated and the abundance of diffuse cytoplasmic staining and intracytoplasmic large granules was determined. All cases with pathologic liver changes had markedly decreased staining intensities for RBP compared with normal livers. The findings of this study suggest that in polar bears, as in humans, vitamin A metabolism may play a role in hepatic carcinogenesis.
Introduction
Liver tumors have been frequently described in captive bears. Tumors have been reported in the liver and bile ducts of sloth bears (Melursus ursinus), Malayan sun bears (Helarctos malayanus), and polar bears (Thalarctos maritimus). 3 , 9 , 10 , 13 Primary hepatocellular carcinoma with intrahepatic metastases was diagnosed in one 30- to 32-year-old polar bear. 22 A trabecular hepatocellular carcinoma was diagnosed in a 15-year-old polar bear and a cholangiocarcinoma in a 19-year-old bear. 16 Another report describes two 26-year-old polar bears with a malignant melanoma of the liver and numerous other tumors of the liver and kidney without further elaboration. 8 Five cases of liver tumors (one hepatoma, two biliary carcinoma, and two hepatocellular carcinoma) in polar bears are present in the archival collection at the Armed Forces Institute of Pathology (B. Williams, personal communication).
Various etiologic factors for hepatic tumors in animals and humans have been discussed. Liver cirrhosis and hepatitis B and C viral infections are well-known risk factors in humans. 4 Viral infections are also associated with hepatocellular carcinoma in birds and woodchucks. 21 , 27 Parasites such as Clonorchis or Opisthorchis flukes may cause human and animal hepatic tumors. 21 , 26 Helicobacter hepaticus causes hepatocellular tumors in laboratory mice. 25 Food contaminants such as aflatoxins, sterigmatocystine, piperonyl butoxide, or nitrosamines in the diet have been shown to be carcinogens in laboratory animals. 1 , 10 , 16 , 21 , 28 Although the cause of hepatocellular tumors in bears is unknown, a genetic component has been proposed to explain the high incidence of liver neoplasms in ursids. 20
In humans, several findings demonstrated a correlation between vitamin A metabolism and carcinogenesis. Vitamin A therapy has been administered to decrease the incidence of hepatocellular carcinomas in patients with chronic liver disease. 17 Low serum levels of retinol, the most important transport form of vitamin A, were associated with hepatocellular carcinoma in patients with chronic liver disease. Additionally, low serum retinol levels are considered as a possible risk factor for the development of hepatocellular carcinoma in humans. 18
Polar bear livers are known to have comparatively high vitamin A levels. 2 Vitamin A plays an important role for the differentiation and maintenance of specialized epithelia and also for resistance to infection. 12 Therefore, vitamin A deficiencies in the polar bear's diet will most likely pose a health problem to the animals.
Retinol represents the most important transport and storage form of vitamin A. More than 90% of the body's vitamin A reserves are stored in the liver, where it is bound to a cellular retinol-binding protein (RBP). This protein is synthesized in the liver. 19 RBP has been immunohistologically demonstrated in dogs, raccoons, silver foxes, and cats. 23 , 24 In this study, the amount of RBP in histologic liver sections of polar bears with hepatocellular carcinoma and other liver diseases was assessed by immunohistochemistry and compared with that in liver sections of normal polar bears to investigate a possible association between liver disease and positivity for RBP.
Materials and Methods
For this retrospective study, investigations were carried out on 11 polar bears (T. maritimus). Six adult and three juvenile animals (polar bear Nos. 1 and 4–11) were from the Zoologischer Garten Basel, Basel, Switzerland, and two animals (polar bear Nos. 2 and 3) from the Tiergarten Schönbrunn, Vienna, Austria (Table 1).
Case no., age, sex, diagnosis, and results of immunohistochemistry for retinol-binding-protein in 11 polar bears.∗
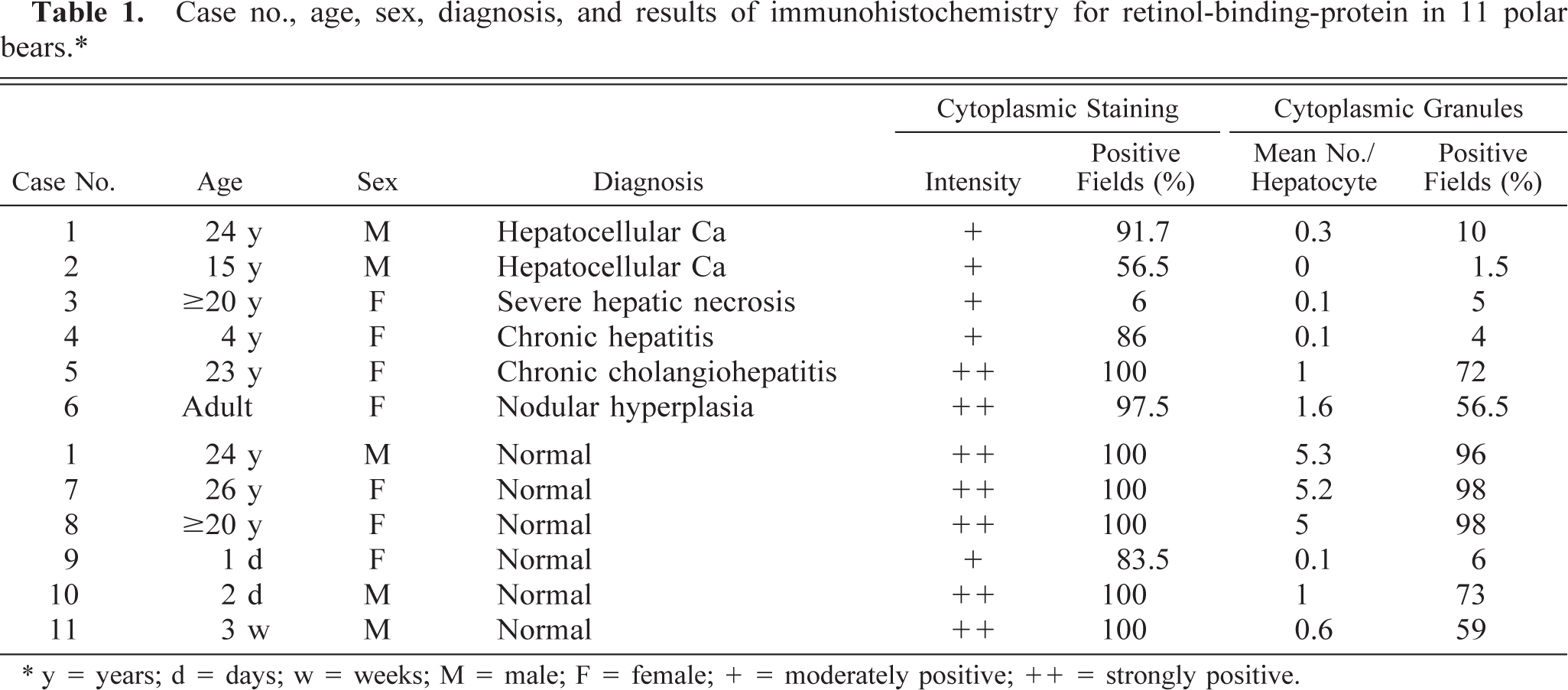
∗ y = years; d = days; w = weeks; M = male; F = female; + = moderately positive; ++ = strongly positive.
Tissues for histologic examination were fixed in 4% buffered neutral formalin, embedded in paraffin, and cut at 4 μm. Sections were stained with hematoxylin and eosin (HE). Glycogen storage was assessed by periodic acid–Schiff (PAS) staining with and without diastase digestion. Excess collagen formation was demonstrated by Van Gieson's stain. T lymphocytes, macrophages, and epithelial cells were characterized using immunohistochemistry. RBP was demonstrated in hepatocytes using a method described previously. 23 In cases diagnosed with tumors, a Von Willebrand factor staining (factor VIII–related antigen) was performed to identify endothelium. Primary antibodies, specificity, source, dilution, and pretreatment are listed in Table 2.
Antibodies, specificity, source, dilution, and pretreatment used for immunohistochemistry.

Immunohistochemical staining was performed as follows. Sections were deparaffinized and incubated for 45 minutes in 0.5% hydrogen peroxide in methanol to deactivate endogenous peroxidases. Nonspecific antibody binding was blocked by incubation for 10 minutes in Tris-buffered saline (TBS) with 20% goat serum. Sections were incubated overnight with primary antibody (diluted in 20% goat serum in TBS) in a humidified chamber at 4 C overnight. The sections were then incubated with biotinylated anti-rabbit IgG or anti-mouse IgG (DAKO, Zug, Switzerland) for 45 minutes, followed by streptavidine (DAKO) for 45 minutes, and finally with 3-amino-9-ethylcarbazole (DAKO) for 15 minutes. The slides were covered with coverslips and Aquamount (Merck, Darmstadt, Germany). All incubations except for the primary antibody were performed at room temperature.
RBP staining was assessed semiquantitatively in two steps. In the first step, the intensity of diffuse cytoplasmic staining was classified as negative, moderately positive, or strongly positive. The percentage of microscopic fields with cells having diffusely stained cytoplasm was calculated in 100 randomly chosen fields. In the same fields, presence or absence of positive intracytoplasmic granules of 2–3 μm in diameter was noted, and the percentage of fields containing hepatocytes with such granules was calculated. In the second step, 100 hepatocytes were randomly chosen, and the number of intracytoplasmic positive-staining granules (2–3 μm in diameter) per hepatocyte was determined and the mean was calculated.
Results
Cases
An overview of the examined cases is given in Table 1, indicating age, sex, and diagnosis for the histologic evaluation of the liver.
The first polar bear, a captive 24-year-old male from the Zoologischer Garten Basel, was euthanatized after several weeks of lethargy and decreasing general condition. The necropsy revealed a moderate emaciation and a severe muscular atrophy of the hind limbs. Five to six liters of serous pale brown fluid was found in the abdomen. The liver was markedly increased in size, with an irregular surface and multiple nodules varying from 5 to 15 cm in diameter. The yellow to brownish parenchyma showed extensive necrosis and coalescing areas of hemorrhage and necrosis.
The second polar bear was a 15-year-old male from the Tiergarten Schönbrunn, Vienna (polar bear No. 2). At necropsy, the moderately emaciated animal had a large mass, 25 cm in diameter, in the left hepatic lobe. Within the mass were multiple foci of necrosis and hemorrhage.
Histopathologic findings were similar for polar bear Nos. 1 and 2. The normal liver architecture was lost and replaced by large confluent, poorly demarcated lobules of tumor cells, separated by mature and immature collagen fibers, and multifocal to coalescing areas of hemorrhage and necrosis (Figs. 1, 2). Large confluent blood-filled vacuoles were limited by endothelium, as confirmed by positive Von Willebrand factor staining. Tumor cells were usually well differentiated, polyhedral, of various sizes, mostly with indistinct cell borders (Fig. 3). These had a high amount of faintly stippled eosinophilic cytoplasm, often with one or several large lipid- and glycogen-containing vacuoles. Multifocally, there were some multinucleated tumor cells (Fig. 4). The central nuclei were round, large, and pale basophilic with a large amount of coarse chromatin in the nuclear periphery and one or two large amphophilic nucleoli of 5 μm in diameter. There was mild to moderate anisocytosis and anisokaryosis. Mitotic figures were rare. Frequently, tumor cells stained slightly positive for cytokeratin. The histopathologic findings were consistent with hepatocellular carcinoma.
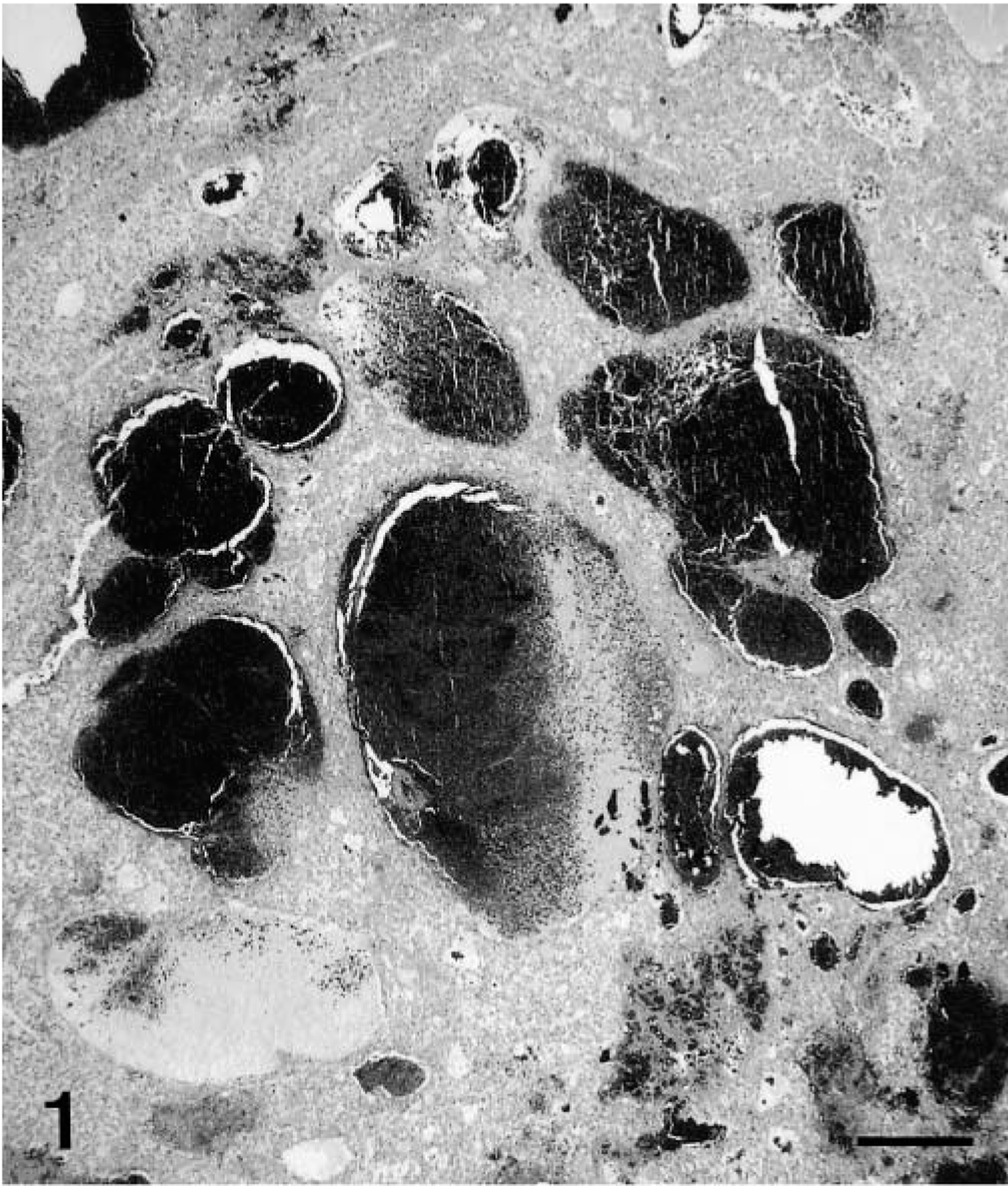
Liver; polar bear No. 2. Hepatocellular carcinoma with large blood-filled spaces. HE. Bar = 800 μm.
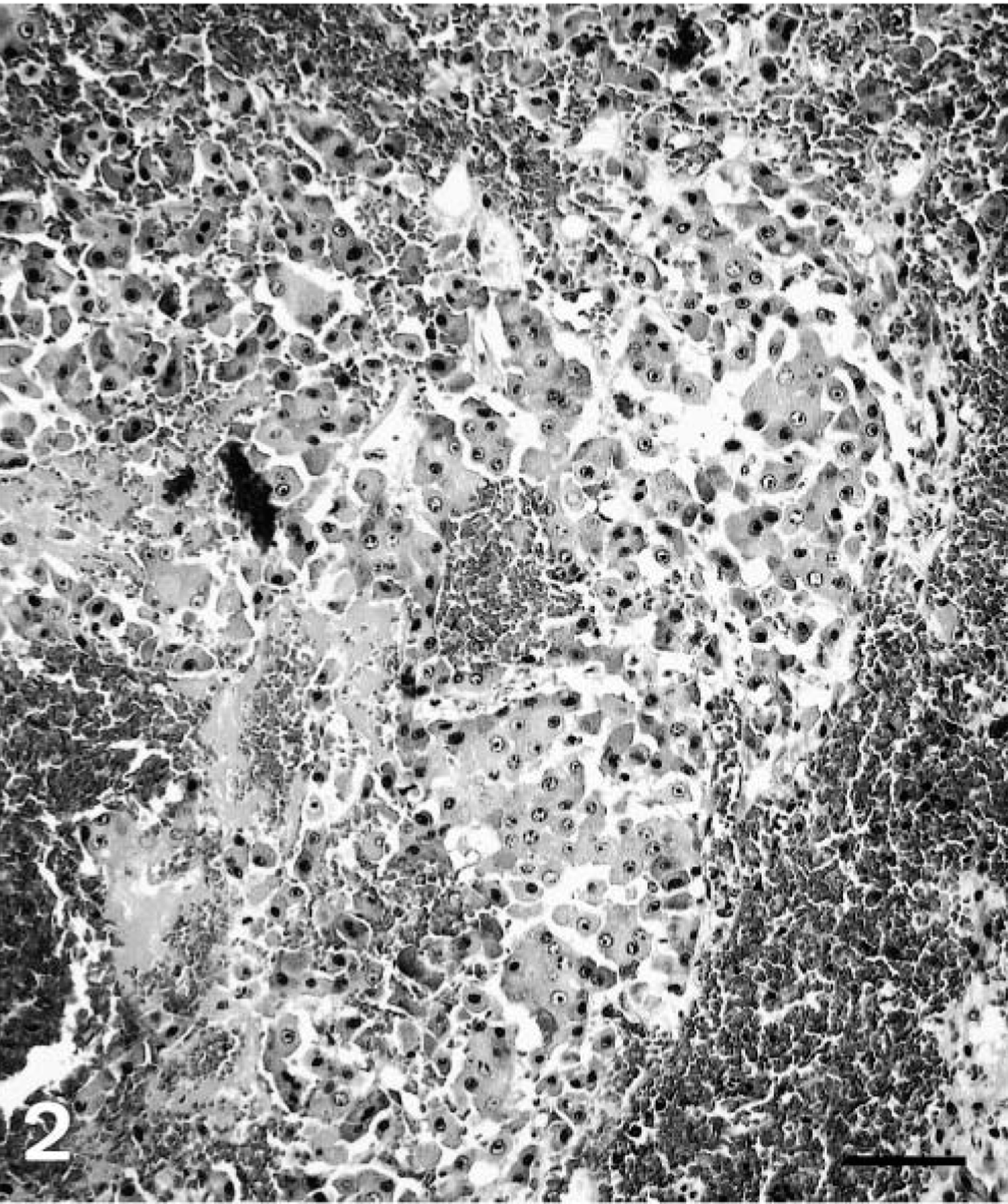
Liver; polar bear No. 1. Hepatocellular carcinoma. HE. Bar = 80 μm.
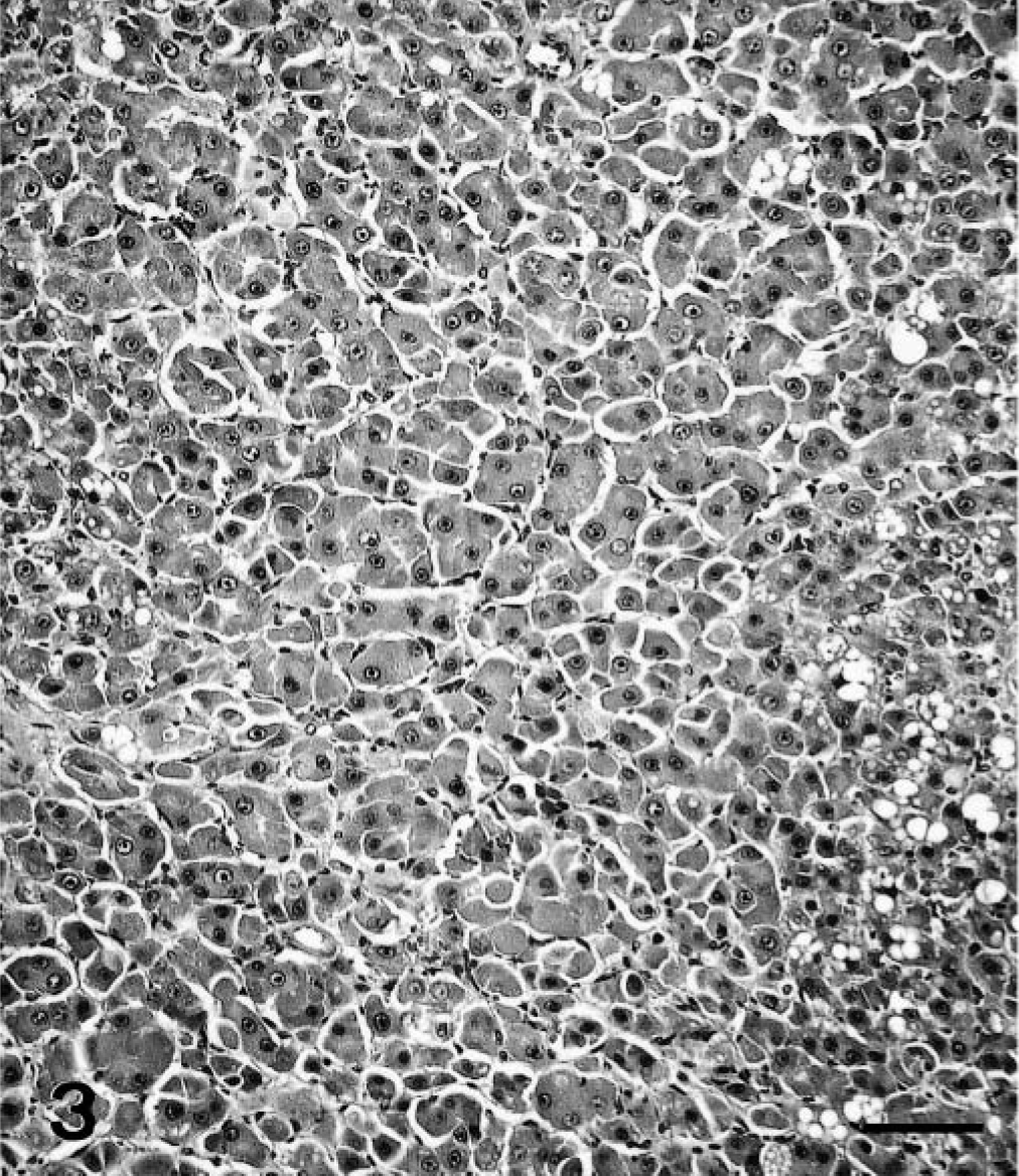
Liver; polar bear No. 1. Hepatocellular carcinoma. In multiple foci, the neoplastic liver cells were hardly distinguishable from normal hepatocytes. HE. Bar = 80 μm.
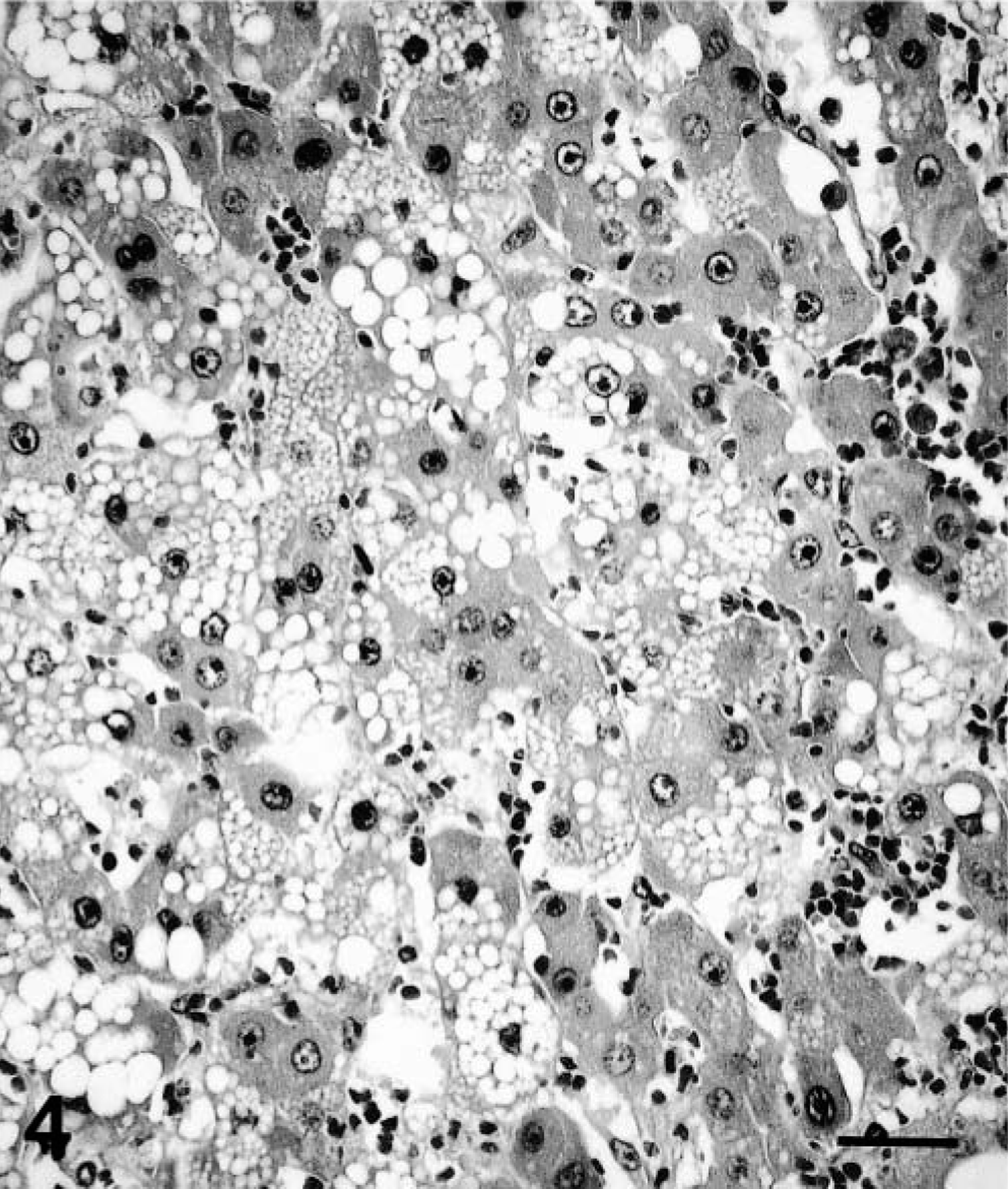
Liver; polar bear No. 2. Hepatocellular carcinoma. Note the multinucleated tumor cell. HE. Bar = 40 μm.
The third polar bear was a female adult over 20 years old. Multiple, mainly perilobular areas of necrosis and degeneration were found with dense infiltrates of predominantly lymphocytes, macrophages, and fewer neutrophils. Most of the lymphocytes were CD3 positive.
The fourth polar bear was a 4-year-old female. In the liver were multifocal to coalescing mild infiltrates with mainly lymphocytes, some macrophages, and very few neutrophils. The majority of the lymphocytes were CD3 positive. Many single hepatocytes were degenerate and necrotic. There was a diffuse, moderate to severe congestion. Bile ducts were moderately hyperplastic. The diagnosis was a moderate multifocal, chronic lymphocytic hepatitis.
The fifth polar bear was a 23-year-old female. Within the liver were multifocal to coalescing moderate to severe infiltrates of mainly lymphocytes with fewer plasma cells and macrophages. These infiltrates were generally distributed around portal triads. Additionally, there was multifocal degeneration and necrosis of individual hepatocytes. The Van Gieson's staining confirmed moderate collagen formation around the cellular infiltrates. Most of the cells in these infiltrates were CD3-positive lymphocytes. The morphologic diagnosis for this liver was moderate to severe chronic cholangiohepatitis.
The sixth polar bear was a female adult of unknown age. Apart from moderate glycogen and lipid storage, the diagnosis was multifocal nodular hyperplasia.
The histology of the liver was normal for two adult (polar bear Nos. 7 and 8) and three juvenile polar bears (Nos. 9–11).
Results of RBP staining
The results of immunohistologic examination for RBP are summarized in Table 1. Cases with hepatocellular carcinoma or other pathologic findings in the liver were compared with the RBP-staining results of the normal liver sections.
In the normal livers of adult polar bears (Nos. 7 and 8, Fig. 5), the cytoplasm of all hepatocytes showed a diffuse strongly positive staining, with equally sized, coarse, intensely red-staining granules of approximately 2–3 μm diameter. In nearly all the examined fields, hepatocytes contained an average of five of these positive granules per hepatocyte, varying from 1 to 10.
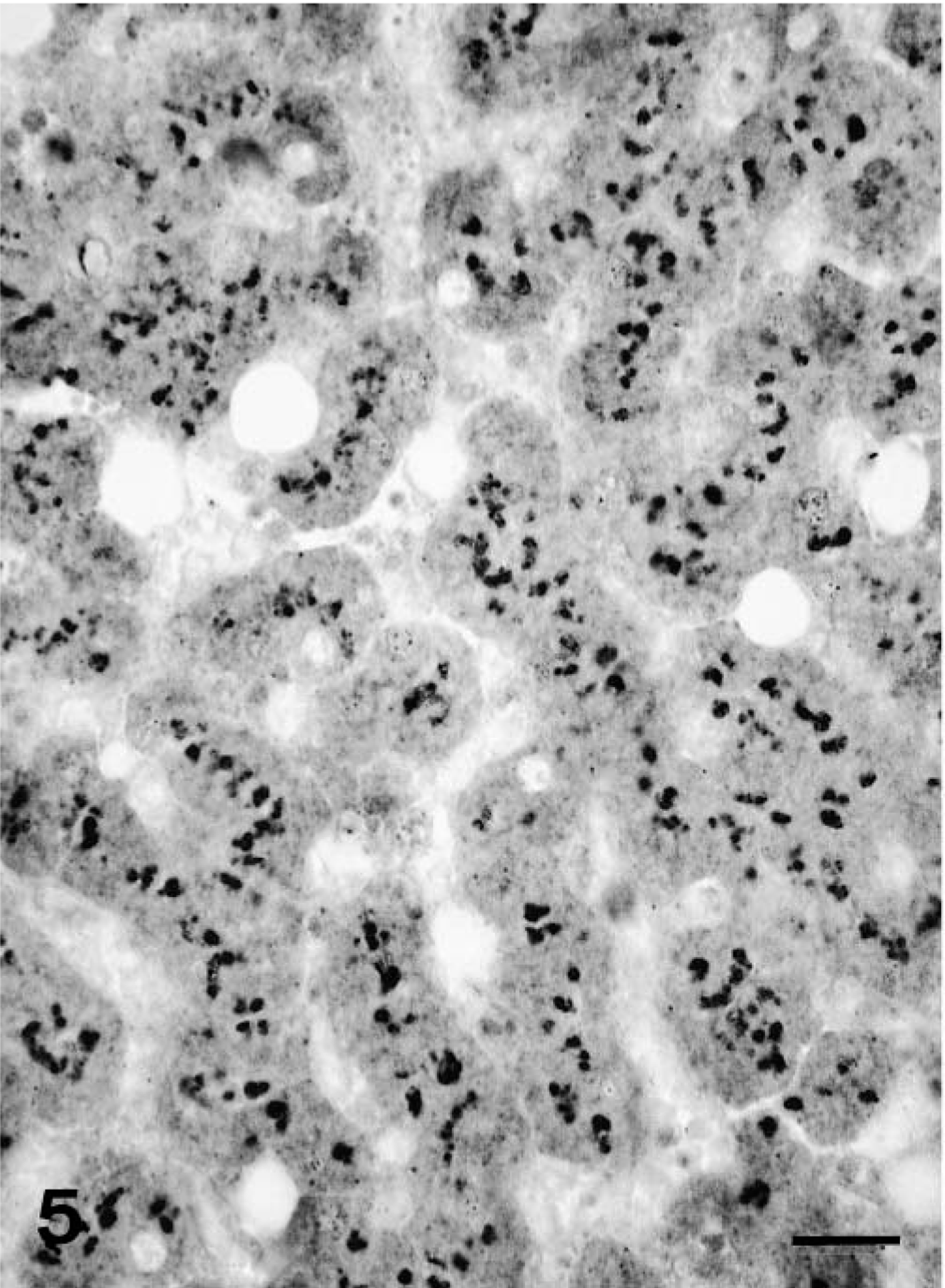
Liver; polar bear No. 7. Normal liver with intense cytoplasmic staining for RBP. Immunohistochemistry for RBP, avidin–biotin–peroxidase complex method. Bar = 25 μm.
The two hepatocellular carcinomas showed a distinct reduction of cytoplasmic staining (polar bear Nos. 1 and 2, Fig. 6). There were multiple cellular foci without any positive staining. In areas that are positive, the staining was only moderately positive. Only few fields (10% or less) had cells that contained positive granules. In polar bear No. 1, sections of normal liver were available, and in these sections, the pattern of RBP staining was identical to that observed in normal polar bear liver.
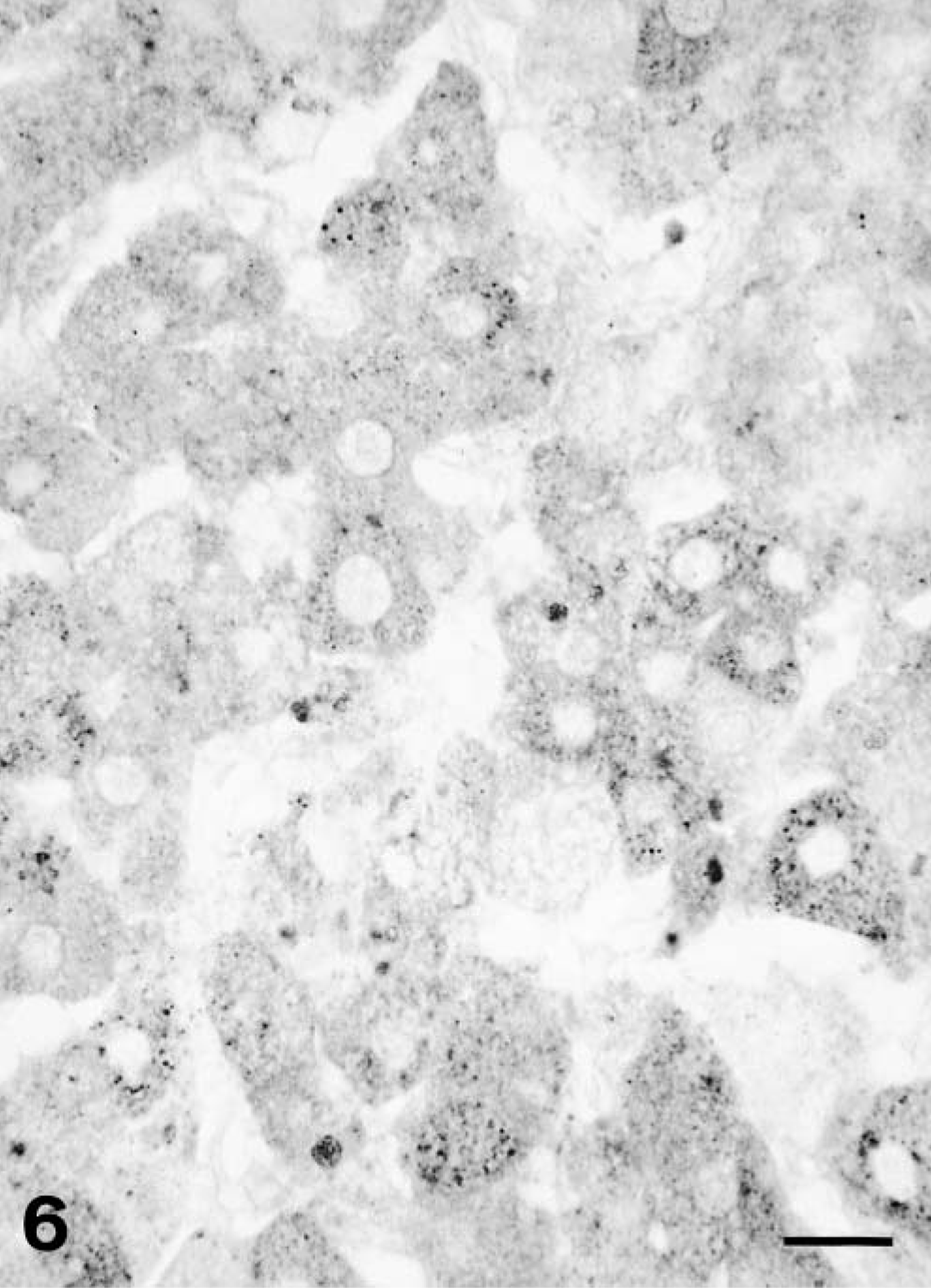
Liver; polar bear No. 1. Hepatocellular carcinoma. Only slight cytoplasmic staining for RBP. Immunohistochemistry for RBP, avidin–biotin–peroxidase complex method. Bar = 25 μm.
Cases with histopathologic liver changes (polar bear Nos. 3–6) also showed a distinct reduction of RBP staining. In polar bear No. 3, with severe liver changes, there was only one focus showing diffuse cytoplasmic positive staining. A few individual cells contained one or two positive granules. Cytoplasmic staining in polar bear No. 4 was also reduced compared with normal liver sections. Other findings were similar to polar bear No. 3. Polar bear Nos. 5 and 6 had strongly positive-staining cytoplasm. Intracytoplasmic granules could be detected in more than half of the evaluated fields, but the average number of granules per cell was clearly lower compared with the normal livers.
RBP staining was less pronounced in liver sections of the three juvenile polar bears than in normal liver from adults. It was higher in the 3-week-old polar bear compared with the two 1- or 2-day-old polar bears.
Discussion
A marked difference of staining intensities for RBP was found between normal livers, hepatocellular carcinomas, and other liver pathologies. These differences were manifested either as a decrease in diffuse cytoplasmic staining or as a reduction in positive cytoplasmic granules. In normal adult livers, strong diffuse cytoplasmic RBP staining was present. In two cases with liver changes, the cytoplasm stained was also strongly positive (polar bear Nos. 5 and 6). However, in two other cases (polar bear Nos. 3 and 4), only a moderate positive staining for RBP was present, whereas the staining intensity in tumor cases was clearly decreased. Additionally, the percentage of fields with positive cytoplasmic staining was usually lower in diseased livers. Furthermore, a mean of five intracytoplasmic granules was found in nearly every field in the normal liver sections from adult animals, whereas polar bears with liver changes had much lower numbers of these granules. These findings show that the RBP-staining intensities were clearly decreased in polar bears with chronic histopathologic liver changes. This reduction was especially striking in livers with tumors.
Immunohistochemical staining with RBP antibody has been used in other species. 23 , 24 This protein, synthesized in the liver, plays a key role in vitamin A metabolism for transport and storage. 19 Polar bears are known to have very large vitamin A reserves in the liver, with levels ranging from 3,900 to 10,380 μg/g. 2 In this retrospective study, historical data from the Zoologischer Garten Basel were available for polar bear Nos. 4–6 (vitamin levels analyzed by F. Hoffmann-La Roche Ltd., Analytical Laboratories of the Vitamin Department). Vitamin A liver content of polar bear Nos. 5 and 6 were around the inferior limit of reference values: 3,390 and 3,940 μg/g, respectively. Correspondingly, both bears had markedly reduced numbers of cytoplasmic granules compared with polar bear Nos. 7 and 8 with normal livers. In polar bear No. 4, a vitamin A liver content of 950 μg/g was distinctly lower compared with data from the literature. RBP staining was clearly reduced in this case with very rare positive granules and only moderately positive cytoplasmic staining. Compared with polar bear Nos. 5 and 6, polar bear No. 4 showed an even lower staining intensity, indicating that RBP immunostaining corresponds to vitamin A levels in polar bear livers.
These data underline the correlation between RBP-staining properties and vitamin A content of the liver. Because more than 90% of the body's vitamin A reserves are stored in the liver, the assessment of vitamin A liver content reflects the vitamin A body pool. Vitamin A is known to play an important role for differentiation and maintenance of specialized epithelial cells. The importance of vitamin A for the survival of hepatocytes during hepatic regeneration was demonstrated in rats. 7 Moreover, low serum retinol levels are associated with hepatocellular carcinoma in patients with chronic liver disease, and nutritional vitamin A therapy could decrease incidence of hepatocellular carcinomas. 17 , 18
Among captive wild mammals, hepatic neoplasms and hepatic diseases appear to be overrepresented in ursids in general and in polar bears in particular. 11 , 20 Apart from the mentioned etiologies including previous chronic liver disease, infections, or chronic exposure to carcinogens, nutritional factors like vitamin A deficiency might play a role in hepatic carcinogenesis. Vitamin A deficiency could account for the high numbers of hepatobiliary tumors in some zoological gardens, whereas other zoos, where vitamin A levels are not problematic, do not report such neoplasms. 3 , 5 , 6 , 10 , 14 , 15 Thus, marginal vitamin A levels and concurrent liver disease would represent a risk factor for the development of hepatic tumors in polar bears as described in humans. 18
These results suggest that a contributory or even causative relationship between vitamin A deficiency and chronic liver changes could be hypothesized in polar bears. Further investigations to evaluate vitamin A liver contents in healthy polar bears and those with hepatic diseases are needed to confirm this hypothesis.
