Abstract
Monocyte chemoattractant protein-1 (MCP-1) but not interleukin-8 (IL-8) was detected by in situ hybridization using a nonradioactive digoxigenin-labeled complementary DNA probe in granulomatous lesions of lymph nodes from 20 pigs with naturally occurring postweaning multisystemic wasting syndrome (PMWS). Complementary DNA probes of 375 and 266 base pairs for MCP-1 and IL-8, respectively, were generated by reverse transcription-polymerase chain reaction. The 20 pigs with PMWS had distinct positive hybridization signals for MCP-1 but not for IL-8. The hybridization signals for MCP-1 were strictly confined to the cells with granulomatous lesions, including macrophages and multinucleated giant cells. A very close cell-to-cell correlation between MCP-1 and porcine circovirus 2 was seen in serial sections of lymph nodes. Results of this study indicate that MCP-1 expression may play a role in the pathogenesis of granulomatous inflammation in pigs with PMWS.
Keywords
Inflammation is often classified as acute or chronic. Morphologically, polymorphonuclear leukocytes (PMNs) are the predominant leukocyte population at sites of acute inflammatory reactions elicited by noxious stimuli, including bacteria. 7 , 26 By contrast, infiltration by monocytes/macrophages is an important component of chronic inflammation. 7 , 21 , 26 Granulomatous inflammation is a specific type of chronic inflammation that is characterized by the accumulation of macrophages and their derived cells, epithelioid cells and multinucleated giant cells. 7 , 21
Chemotactic factors released at the site of injury or inflammation are believed to mediate the direct recruitment of monocytes into tissues. 2 , 24 , 25 Members of the chemokine supergene family have proinflammatory activity and are inducible in a number of pathophysiological processes. 24 Monocyte chemoattractant protein-1 (MCP-1), the prototypic member of the C-C subfamily, is chemoattractive for monocytes. Monocytes/macrophages are the predominant cellular sources of MCP-1. 24 MCP-1 has been demonstrated to be produced in granulomatous inflammation. 8 , 10 , 13 In contrast, interleukin-8 (IL-8), the prototypic member of the C-X-C subfamily, has chemotactic and activating properties for PMNs and lymphocytes. 2 , 7 , 24 , 25
Postweaning multisystemic wasting syndrome (PMWS) is a chronic viral infection caused by porcine circovirus 2 (PCV2). 1 , 4–6 , 17 Histologically, the disease is characterized by widespread granulomatous inflammation, multinucleated giant cells, and variable numbers of intracytoplasmic basophilic viral inclusion bodies within infiltrating histiocytes and macrophages. 4 , 6 , 17–19 Features of the histopathological lesions suggest that monocyte/macrophage infiltration may be closely related to the pathogenesis and progression of PMWS. However, there is little information regarding the factors that initiate and regulate the recruitment of monocytes to the granulomatous lesions seen in PMWS. Therefore, the objectives of this study were to determine whether MCP-1 and IL-8 are expressed in granulomatous lesions in pigs with naturally occurring PMWS.
Materials and Methods
Animals
At necropsy, samples were obtained from pigs submitted to the Department of Veterinary Pathology of Seoul National University from January 1999 to December 2000. Twenty pigs (Nos. 1–20) of approximately 65–80 days of age, from 20 different herds, were selected on the basis of clinical signs, characteristic histopathological lesions, and viral isolation. 17 All 20 pigs were positive for PCV2 but negative for porcine parvovirus, swine influenza virus, and porcine reproductive and respiratory syndrome virus by virus isolation, polymerase chain reaction (PCR), and in situ hybridization. 4 , 5 , 14 , 17 Negative control sections were prepared from 1-day-old colostrum-deprived pigs that had not been exposed to any viral and bacterial pathogens. Conventional 60- and 80-day-old pigs that were negative for bacterial isolation also were used as negative controls. Lymph node section from a 4-year-old dog naturally infected with Mycobacterium avium was also used as negative control. Lung section from a 65-day-old pig naturally infected with Actinobacillus pleuropneumoniae was used as positive control for IL-8. This lymph node had granulomatous lesions.
Tissue processing
Samples of lung, liver, kidney, tonsil, lymph node, and large and small intestines from infected and noninfected animals were fixed in 10% (wt/vol) neutral buffered formalin for 24–48 hours, embedded in paraffin, sectioned at 4 μm, floated on a water bath containing diethylpyrocarbonate-treated water, and mounted on positively charged slides (Superfrost/Plus slide, Erie Scientific Co., Portsmouth, NH).
RNA extraction
Lymph nodes from 20 pigs with PMWS were used for extraction of MCP-1 and IL-8. RNA was extracted from lymph nodes with Trizol LS reagent (GIBCO-BRL, Grand Island, NY), according to the manufacturer's instructions. RNA extracts were treated with DNase I (GIBCO-BRL) to eliminate genomic DNA contamination.
Primer
A 375–base pair (bp) complementary DNA (cDNA) fragment was used as the probe. The forward and reverse primers were 5′-TCACCAGCAGCAAGTGT-3′ (nucleotides 206–223) and 5′-CTGAGATTCACAGAGGA-3′ (nucleotides 605–621), respectively. 11 The primers for IL-8 were used as described previously. 9 The forward and reverse primers were 5′-TTTCTGCAGCTCTCTGTGAGG-3′ (nucleotides 127–147) and 5′-CTGCTGTTGTTGTTGCTTCTC-3′ (nucleotides 372–392), respectively. The primers amplified a 266-bp cDNA fragment.
Reverse transcription–polymerase chain reaction
For the first-strand cDNA synthesis, 1 μl of the RNA (5 ng/μl) was supplemented in a total reaction volume of 20 μl with 1× reverse transcription buffer (50 mM Tris-HCl, 8 mM MgCl2, 30 mM KCl, 1 mM dithiothreitol, pH 8.3), 0.5 mM (each) deoxynucleotide triphosphates (dNTPs), 2.5 μM random hexanucleotide mixture, 20 U of RNase inhibitor, and 50 U of Moloney murine leukemia virus reverse transcriptase. After incubation for 15 minutes at 42 C, the mixture was incubated for 5 minutes at 99 C to denature the products. The mixture was then chilled on ice. Positive controls for MCP-1 and IL-8 were prepared as described previously. 9 , 11
The composition of the PCR mixture (150 μl) was 30 μl of cDNA (5 ng/μl), 2 μl of each primer (250 nM), 15 μl of 10× PCR buffer (10 mM Tris-HCl, 40 mM KCl, 1.5 mM MgCl2, pH 8.3), 1.2 μl of each dNTP (0.2 mM), 2.5 unit of Taq polymerase (Bioneer Corp., Cheungwon, Republic of Korea) in a volume of 29 μl, and 67.2 μl of distilled water. The PCR reaction of MCP-1 was done under the following conditions in a thermal cycler (Perkin-Elmer-Cetus, Norwalk, CT): 35 cycles of denaturation at 94 C for 1 minute, annealing at 55 C for 1 minute, and elongation at 72 C for 2 minutes. The PCR reaction of IL-8 was done under the following conditions in a thermal cycler (Perkin-Elmer-Cetus): 35 cycles of denaturation at 94 C for 45 seconds, annealing at 62 C for 45 seconds, and elongation at 72 C for 45 seconds.
Preparation of labeled probe
Reverse transcription–polymerase chain reaction (RT-PCR) products of MCP-1 and IL-8 were purified using a 30-kd cutoff membrane ultrafiltration filter. The nucleotide sequences of the purified RT-PCR products were determined by use of BigDye chemistry with the ABI Prism Sequencer (Applied Biosystems, Foster City, CA). Sequencing was performed of the purified RT-PCR products before RT-PCR products were labeled by random priming with digoxigenin-dUTP (Boehringer Mannheim, Indianapolis, IN), according to the manufacturer's instructions. The probe for PCV2 was prepared as described previously. 16
In situ hybridization
Sections were deparaffinized in xylene and rehydrated in phosphate-buffered saline (PBS) (pH 7.4, 0.01 M) for 5 minutes. Deproteinization was carried out in 0.2 N HCl for 20 minutes at room temperature. Tissues were then digested at 37 C for 20 minutes in 100 μg/ml proteinase K (GIBCO-BRL) in PBS. Serial sections of each tissue section examined were treated with RNase A (Boehringer Mannheim) at 100 μg/ml in 10 mM Tris-HCl (pH 7.4) for 30 minutes at 37 C to remove target RNA as a specificity control. After digestion, tissues were fixed in 4% paraformaldehyde in PBS for 10 minutes. After rinsing twice with PBS, the slides were acetylated in 300 ml of 0.1 mM triethanolamine-HCl buffer (pH 8.0) to which 0.75 ml of acetic anhydride (0.25%) had been added. After 5 minutes, an additional 0.75 ml of acetic anhydride was added, and 5 minutes later the slides were rinsed in 2× saline sodium citrate (SSC) (1× SSC contains 50 mM NaCl and 15 mM sodium citrate, pH 7.0). The slides were allowed to equilibrate for 60 minutes in a standard hybridization buffer that consisted of 5× SSC with 50% deionized formamide, 10× 2% buffered blocking solution (Boehringer Mannheim), 0.1% N-lauroylsarcosine, and 0.02% sodium dodecyl sulfate.
Hybridization was done overnight at 45 C for PCV2, MCP-1, and IL-8. The digoxigenin-labeled probe (0.1 ng/μl) was diluted in 300 μl of the standard hybridization buffer, heated for 10 minutes at 95 C on a heating block, and quenched on ice before being applied to the tissue sections. Approximately, 50 ng of the digoxigenin-labeled probe was added to the standard hybridization buffer (50 μl), which was then layered over the section. Fluid was held in place by a coverslip, and the edges were sealed with rubber cement. After overnight hybridization, sections were thoroughly washed twice in 4× SSC for 5 minutes at room temperature, twice in 2× SSC for 10 minutes at 37 C, twice in 0.2× SSC for 5 minutes at room temperature, and once in maleic acid buffer (100 mM maleic acid and 150 mM NaCl, pH 7.5) for 5 minutes at room temperature.
For detection of hybridization, sections were incubated with anti-digoxigenin conjugated with alkaline phosphatase (Boehringer Mannheim) diluted 1:250 in 0.1 M Tris-HCl (pH 7.4) and 0.15 M NaCl with 1% blocking reagent (Boehringer Mannheim). After three washes in buffer, substrate consisting of nitroblue tetrazolium and 5-bromocresyl-3-indolylphosphate was layered over the sections. Color was allowed to develop for 5–8 hours in the dark, and the reaction was stopped by dipping slides briefly in Tri–ethylenediaminetetraacetic acid buffer (10 mM Tris-HCl and 1 mM ethylenediaminetetraacetic acid, pH 8.0). Sections were counterstained with 0.5% methyl green, and the slides were then washed with distilled water for 1 minute, allowed to dry completely, dipped into the absolute xylene, and coverslipped with Canada balsam mounting medium (Hayashi Pure Chemical Industries Ltd., Osaka, Japan).
Statistical analysis
Hybridization signals of PCV2, MCP-1, and IL-8 were given a ranked score of 0–3 by simple subjective system: 0 = no hybridization signal, 1 = minimal hybridization signal, and 2 = intense hybridization signal. The in situ hybridization scores were subjected to assess relationships by the Pearson correlation analysis. P < 0.05 was considered to be statistically significant.
Results
Reverse transcription–polymerase chain reaction
Amplification of template cDNA with primers of MCP-1 or IL-8 resulted in amplified products corresponding to those of the predicted size, namely, 375 bp (MCP-1) and 266 bp (IL-8). PCR products were sequenced and their identity confirmed as MCP-1 and IL-8. To determine expression of MCP-1 and IL-8, RT-PCR analyses were performed using RNA extracted from lymph node of all pigs with PMWS. MCP-1 was detected in the lymph nodes (Fig. 1), but IL-8 was not detected in these lymph nodes. MCP-1 and IL-8 were not detected in negative control pigs.
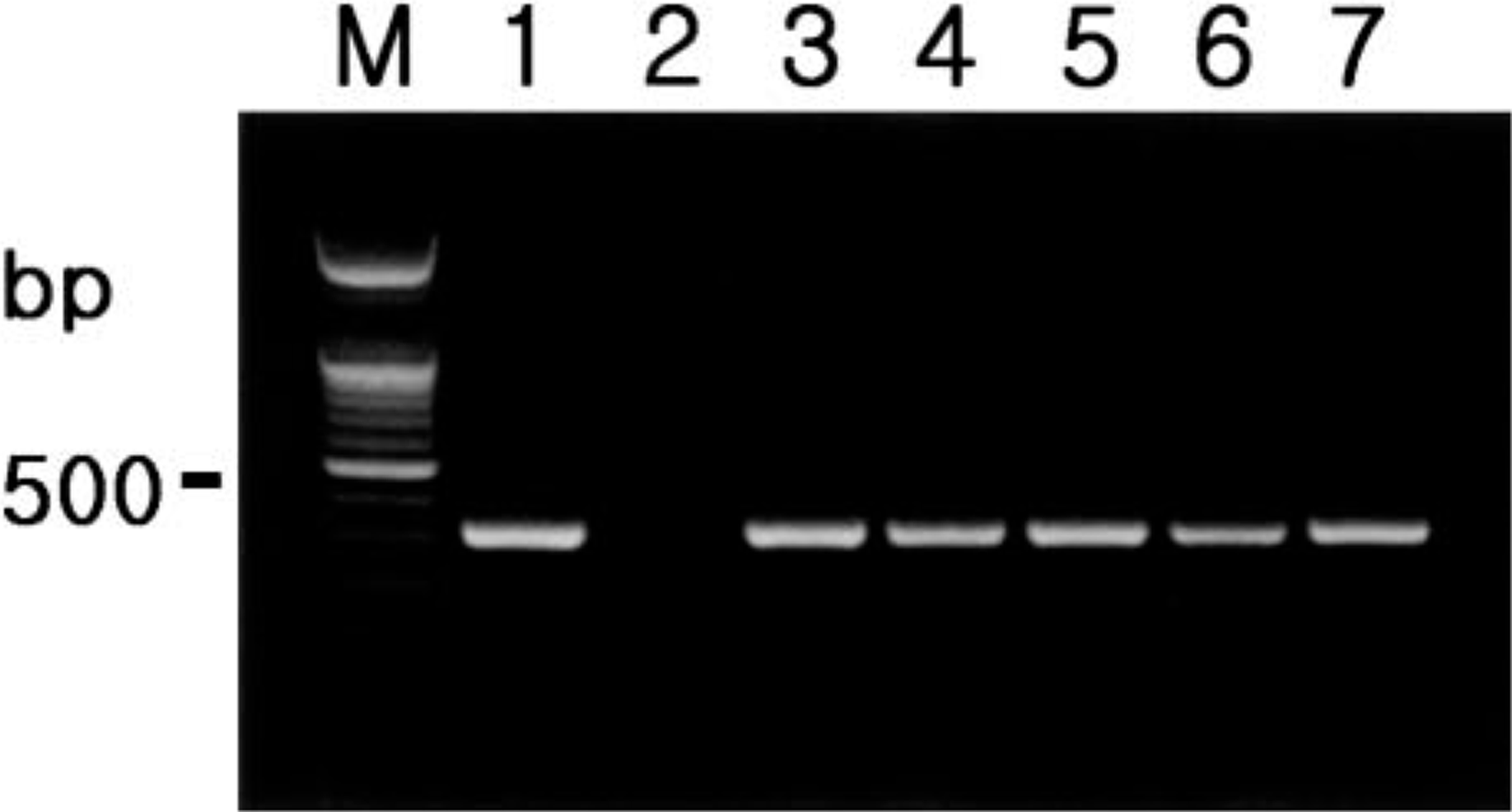
Agarose gel electrophoresis of PCR-amplified MCP-1 cDNA products. From left to right: M = 100-bp DNA ladder; lane 1 = positive control; lane 2 = negative control; lane 3 = positive MCP-1 cDNA from pig No. 1; lane 4 = positive MCP-1 cDNA from pig No. 6; lane 5 = positive MCP-1 cDNA from pig No. 9; lane 6 = positive MCP-1 cDNA from pig No. 16; lane 7 = positive NOS2 cDNA from pig No. 20.
Histopathology
Microscopic lesions in lymph nodes were similar for all pigs with PMWS. The most unique lesion was multifocal granulomatous inflammation, characterized by infiltrates of epithelioid macrophages and multinucleated giant cells (Fig. 2a). There were intracytoplasmic and intranuclear amphophilic inclusion bodies in epithelioid macrophages and multinucleated giant cells. Lymph nodes also were depleted of lymphocytes, and this was characterized by basophilic pyknotic nucleic and karyorrhectic debris.
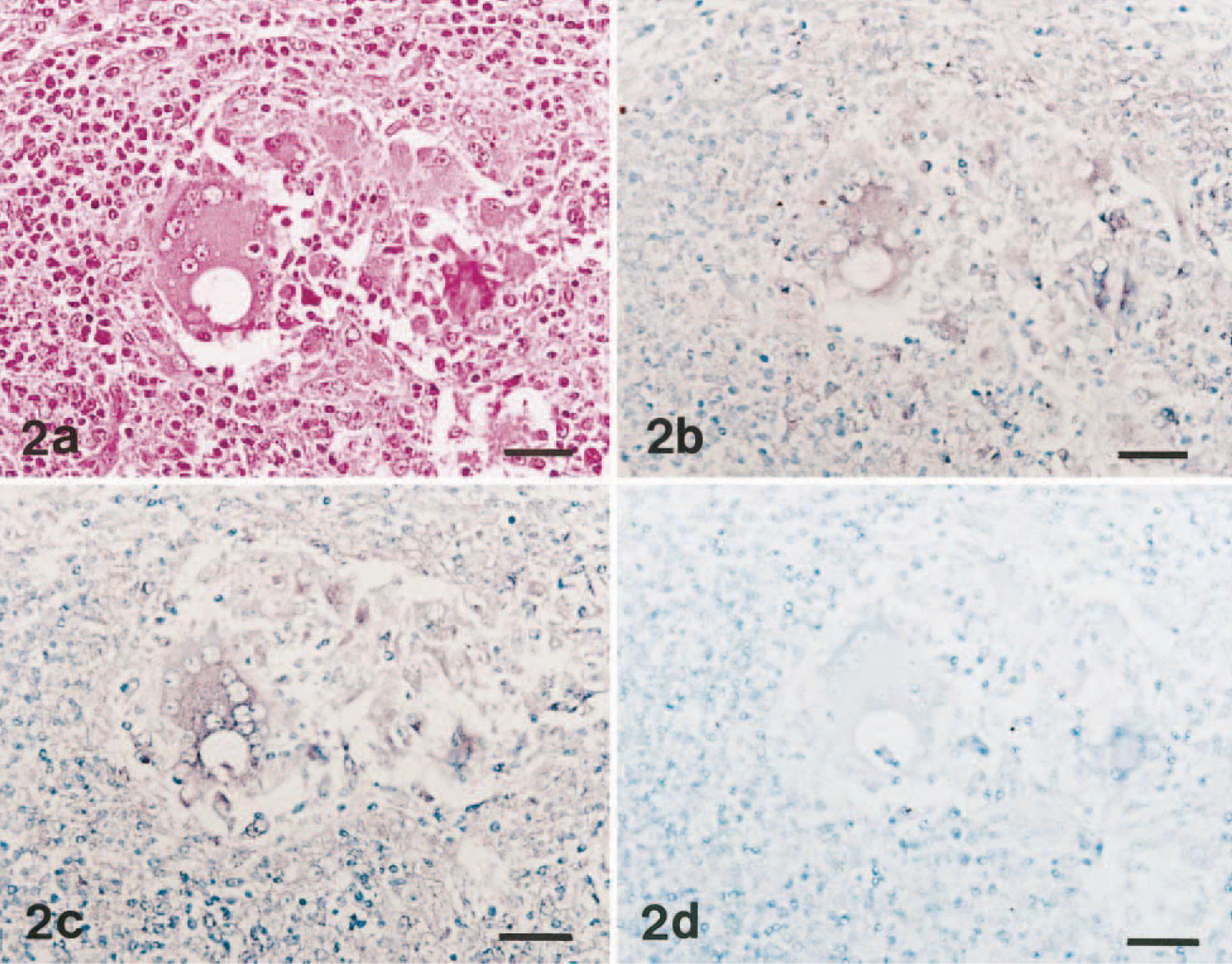
Lymph node; pig No. 6 with naturally occurring PMWS. Fig. 2a. Granulomatous foci characterized by infiltration with macrophages and multinucleated giant cells. HE. Bar = 55 μm. Fig. 2b. Serial section of the cells in Fig. 2a showing the same cells positive for PCV2 (brown reaction). In situ hybridization: DNA probe, nitroblue tetrazolium/5-bromocresyl-3-indolylphosphate, methyl green counterstain. Bar = 55 μm. Fig. 2c. Serial section of the cells in Fig. 2a showing the same cells positive for MCP-1 (brown reaction). In situ hybridization: cDNA probe, nitroblue tetrazolium/5-bromocresyl-3-indolylphosphate, methyl green counterstain. Bar = 55 μm. Fig. 2d. Serial section of the cells in Fig. 2a showing the same cells negative for IL-8. In situ hybridization: cDNA probe, nitroblue tetrazolium/5-bromocresyl-3-indolylphosphate, methyl green counterstain. Bar = 55 μm.
In situ hybridization
All 20 pigs with PMWS had distinct and positive hybridization signals for MCP-1 but not for IL-8. The morphology of host cells was preserved despite the relatively high temperature required during the incubation procedure. Signal intensity varied within and between anatomical structures in any one section and between pigs. Positive cells were identified as dark brown or black reaction products in the cytoplasm without background staining. Strong hybridization signals for PCV2 were detected in the cytoplasm of epithelioid macrophages and multinucleated giant cells in lymph nodes from pigs with PMWS (Fig. 2b). The cytoplasm of scattered macrophages exhibited a less intense, dispersed hybridization signal. The distribution of PCV2-positive cells in the lymph node was multifocal.
A strong hybridization signal for MCP-1 was seen in epithelioid macrophages and multinucleated giant cells of lymph nodes (Fig. 2c). In addition, the hybridization signals were confined to cells in areas of granulomatous inflammation. Hybridization signals for IL-8 were not seen in epithelioid macrophages and multinucleated giant cells (Fig. 2d). A very close cell-to-cell correlation between MCP-1 and PCV2 in serial sections from lymph nodes was confirmed by in situ hybridization (Fig. 2b, c).
Pretreatment with RNase A eliminated hybridization signals for both MCP-1 and IL-8 from 20 pigs with naturally occurring PMWS and positive control pigs. Sections from negative control pigs showed no hybridization signal for MCP-1 and IL-8. Hybridization signal of IL-8 was detected in lung sections from a pig with A. pleuropneumoniae infection. No hybridization signal was detected in lymph node sections from a dog with M. avium infection.
Statistical analysis
The mean score of in situ hybridization is 2.4 ± 0.75 for PCV2 and 2.6 ± 0.51 for MCP-1. The score of in situ hybridization in PCV2 was correlated with the score of in situ hybridization in MCP-1 (r s = 0.50, P < 0.05).
Discussion
In this study, in situ hybridization with nonradioactive digoxigenin-labeled probes was successfully applied to PCV2-infected lymph node to detect MCP-1 and to identify the sites at which they were expressed. Intense and consistent expression of MCP-1 was demonstrated in lesions of granulomatous inflammation caused by PCV2. In contrast, expression of MCP-1 was minimal in noninflammed lymph nodes of infected pigs and in normal lung from control pigs. In situ hybridization of serial sections of the lymph node indicates that the majority of areas containing PCV2-positive cells also have numerous MCP-1–positive cells. Although the exact manner in which PCV2 and MCP-1 interact is not completely understood, PCV2 induces MCP-1 expression by macrophages and other inflammatory cells in infected lymph nodes. The results strongly suggest that MCP-1 may be involved in lesion development or progression during PCV2 infection.
The results of this study indicate that macrophages, epithelioid cells, and multinucleated giant cells may participate in the recruitment of mononuclear cells to a site of granulomatous inflammation by producing MCP-1. MCP-1 has been demonstrated to be produced during granulomatous inflammation and to participate in it. 8 , 10 , 13 Although in situ hybridization and RT-PCR detected macrophages and multinucleated giant cells as the primary sources of MCP-1, the detection of MCP-1 nucleic acid does not necessarily equate with functional activity of MCP-1 production in vivo.
Chemotactic properties of MCP-1 for monocytes correlate with the presence of monocytes/macrophages in the granulomatous lesions. Factors that initiate and regulate the recruitment of monocytes are likely to be important mediators in activating the host response for granulomatous inflammation. 7 Thus, secretion of MCP-1 provides a potential mechanism to account for the recruitment of monocytes observed in the granulomatous inflammatory process. 8 , 10 , 13 MCP-1 has been shown to be a product of primarily macrophages. 24 The expression of MCP-1 by macrophages may be important for the initial stimulation of monocyte chemotaxis. Once monocytes are recruited to sites of granulomatous inflammation, the expression of MCP-1 by monocytes/macrophages may serve to further amplify monocyte recruitment.
The inflammatory response is a tightly ordered sequence of events controlled, in part, by the regulated expression of cell surface and soluble molecules. 2 , 7 , 22 At the site of PCV2 infection, elicited blood monocytes and surrounding macrophages are activated and synthesize MCP-1 with autocrine and paracrine effector activities. The role of MCP-1 in granulomatous inflammation is believed to be related to its capacity to induce chemotaxis and to regulate trafficking of mononuclear cells at the molecular levels. 8 The mechanism by which MCP-1 recruits mononuclear cells is still unclear, although chemokines of the C-C superfamily act through signal transduction events such as G-protein–coupled receptors, leukotriene formation, and rapid changes in Ca2+. 3
Multinucleated giant cells are highly stimulated cells of macrophage lineage, and they are a terminal stage of differentiation. 20 Multinucleated giant cells were found to contain considerable amount of hybridizable MCP-1 nucleic acid; they should not be regarded as nonfunctioning cells 23 because they are likely to be actively synthesizing many products. In this study, multinucleated giant cells have been shown to contain MCP-1 nucleic acid; they may also be important sources of MCP-1. Because multinucleated giant cells are a common cellular component of granulomatous inflammation in PWMS, 4 , 6 , 15 , 17–19 it is reasonable to propose that multinucleated giant cells contribute to the initiation and maintenance of the granulomatous inflammatory process.
The exact mechanisms by which granulomatous inflammation is induced by PCV2 remain unknown. A correlation between the presence of MCP-1 expression and PCV2 in serial sections of the lymph node suggests that the regulated expression of MCP-1 by mononuclear cells in response to PCV2 and thus PCV2 plays an important role in granulomatous inflammation. The ability of MCP-1 to induce integrin expression and respiratory burst activity 12 indicates that there are different pathways in which MCP-1 may influence monocyte/macrophage function in PMWS. Future investigations that focus on the role of MCP-1 in the upregulation of granulomatous inflammation may provide new understanding of the pathophysiology of PCV2 infection and PMWS.
Footnotes
Acknowledgements
This research was supported by Ministry of Agriculture, Forestry and Fisheries-Special Grants Research Program (MAFF-SGRP) and Brain Korea 21 Project, Republic of Korea.
