Abstract
To determine the transmissibility of scrapie to Rocky Mountain elk (
Keywords
Scrapie is a naturally occurring transmissible spongiform encephalopathy (TSE) of sheep and goats. It has also been proposed as the possible origin of chronic wasting disease (CWD) in cervids. 18 In scrapie-infected sheep there is an accumulation of an abnormal form of prion protein (PrPres) in tissues of the central nervous system (CNS) and lymphatic system. Detection of PrPres in these tissues and characteristic histopathologic changes in the brain are the basis of currently available diagnostic methods for TSEs. 7
This article documents preliminary findings of an as yet incomplete investigation of Rocky Mountain elk (
Eight 3–4-month-old male castrated elk calves that were born in captivity were obtained from two Midwestern states (Iowa and Kansas). They were assigned to inoculated (
The inoculum (No. 13-7) was prepared from a pool of 13 scrapie-affected sheep brainstems (all sheep were clinical, and the brains were PrPres-positive by immunohistochemistry) from seven source flocks. The brain tissue was ground in a mechanical grinder, gentamicin was added at 100 μg/ml, and the final concentration of 10% (w/v) was made with physiologic buffered saline.
The elk calves were inoculated intracerebrally with 1 ml of the scrapie brain inoculum as described previously. 3 In brief, the animals were sedated with xylazine, a midline incision was made in the skin at the junction of the parietal and frontal bones, and a 2-mm hole was drilled through the calvarium. The inoculum was injected into the midbrain via a 22-gauge, 9-cm-long needle while withdrawing the needle from the brain. The skin incision was closed with a single suture. Two elk calves (controls) were not inoculated.
A complete necropsy was conducted within 1 hour after death or euthanasia of the animals. Representative samples of major organs, including one-half of sagitally cut brain, and representative areas of the spinal cord (cervical, thoracic, and lumbosacral) were immersion-fixed in 10% neutral buffered formalin. The remainder of the brain was frozen for immunodetection of PrPres using a commercial (Prionics-Check) western blot method
15
and for detection of scrapie-associated fibrils (SAFs) using negative stain electron microscopy.
16
The brain was retained in fixative for not less than 3 weeks. It was cut into 2–4 mm wide coronal sections, and various anatomic sites (
Preliminary findings in five elk experimentally inoculated with sheep scrapie agent (No. 13-7).

∗ IHC = immunohistochemistry for PrPres; SAFs = scrapie-associated fibrils; ND = not done; WB = western blot (Prionics-Check); +/– = lesions or antigen present/absent.
Six weeks postinoculation (PI), one inoculated calf (No. 1; Table 1) developed progressive clinical signs of encephalitis. It was euthanatized at 7 weeks PI, and at necropsy a large abscess was present bilaterally in the cerebrum. Two other inoculated animals were either euthanatized (No. 3, Table 1) or died (No. 2) at 6 and 15 months PI because of self-induced accidental traumas. No other gross or microscopic lesions were present in these two animals.
At 25 months PI, elk No. 4 (Table 1) died after a brief neurologic episode (falling and paddling with its legs while in lateral recumbency) that lasted for approximately 1 minute. Prior to this terminal episode, the elk had developed a slightly hostile attitude toward the animal caretakers. However, at that time, it was presumed that the behavior was within the normal range of a wild animal in confinement. Ten months later (35 months PI; Table 1), elk No. 5 exhibited similar terminal neurologic signs and died within 20 minutes of falling. Prior to the neurologic signs, both elk had shown slightly reduced appetite for approximately 4 weeks and as a result had lost some body weight. At necropsy no gross lesions were observed. Microscopically, lesions of spongiform encephalopathy (vacuolation of neuronal perikarya and neuropil) were seen in the brains (Figs. 1–3) and spinal cords (Fig. 4) of both animals, and the lesions were morphologically similar in severity. At the affected sites there was also mild multifocal increase in glial cells, but neuronal degeneration was not a prominent feature in any of the sections.
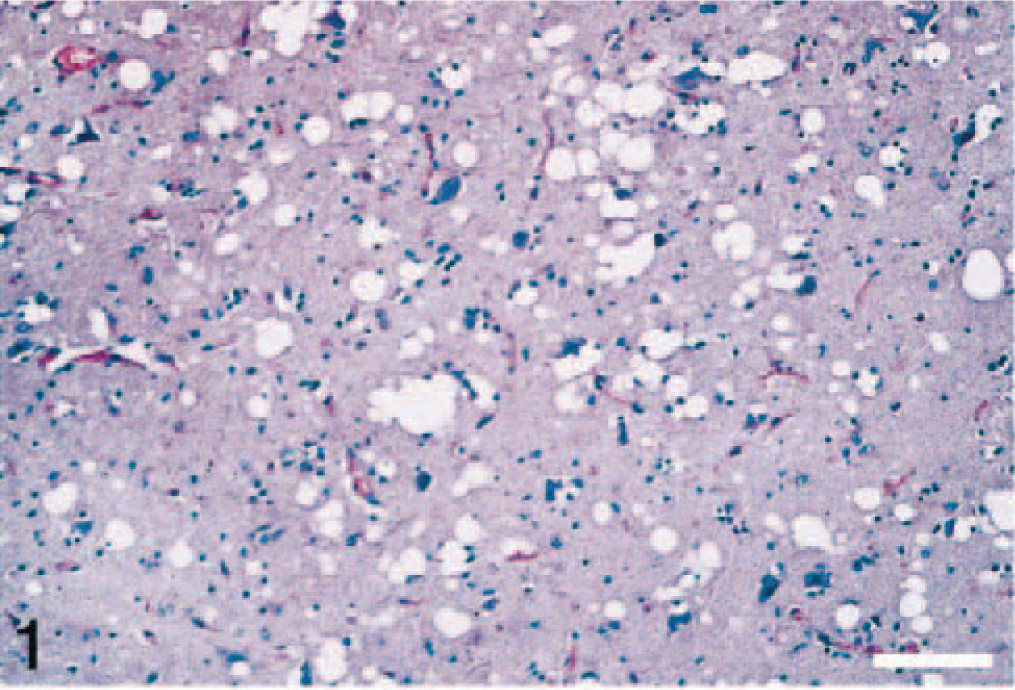
Brain; thalamus of elk No. 4. There is extensive vacuolation in the neuropil. HE. Bar = 70 μm.
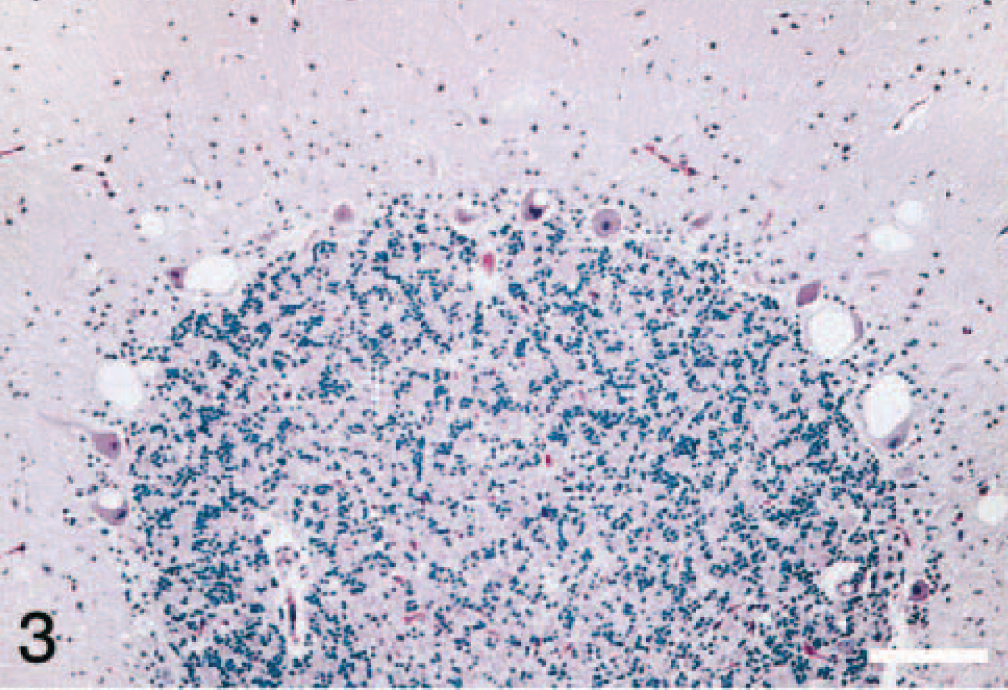
Brain; cerebellum of elk No. 4. Single or multiple cytoplasmic vacuoles are present in the Purkinje cells. HE. Bar = 70 μm.
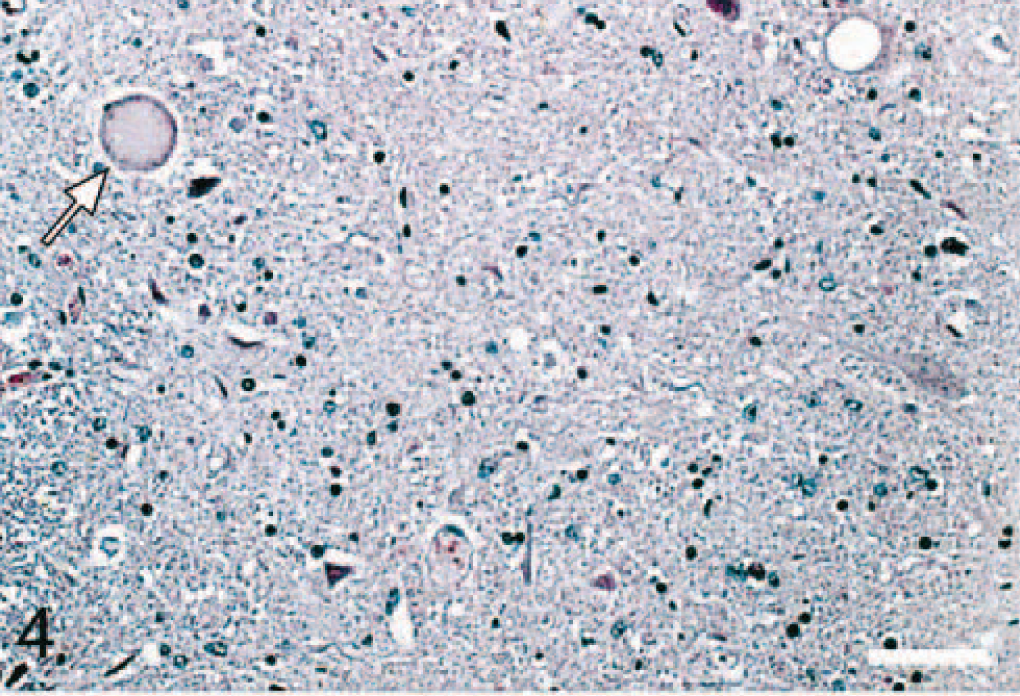
Brain; thoracic spinal cord of elk No. 4. There is presence of one degenerate neuron (arrow head) and a single vacuole in neuronal perikaryon (arrow). HE. Bar = 70 μm.
In the brain, the lesions were much more severe and extensive in the thalamus and cerebellum. In the thalamus there was widespread vacuolation in the neuropil (Fig. 1), but vacuolation of neuronal cytoplasm was not a predominant feature. However, in the cerebellum, besides the presence of variable-sized vacuoles in the neuropil of the molecular layer (Fig. 2), there were prominent single or multiple cytoplasmic vacuoles in Purkinje cells (Fig. 3). Examination of hemisections through the brainstem showed characteristic spongiform lesions in various nuclei, including the dorsal nucleus of the vagus nerve. However, spongiform change was much more severe and extensive in the pontine nucleus than in the nucleus of the dorsal vagus.
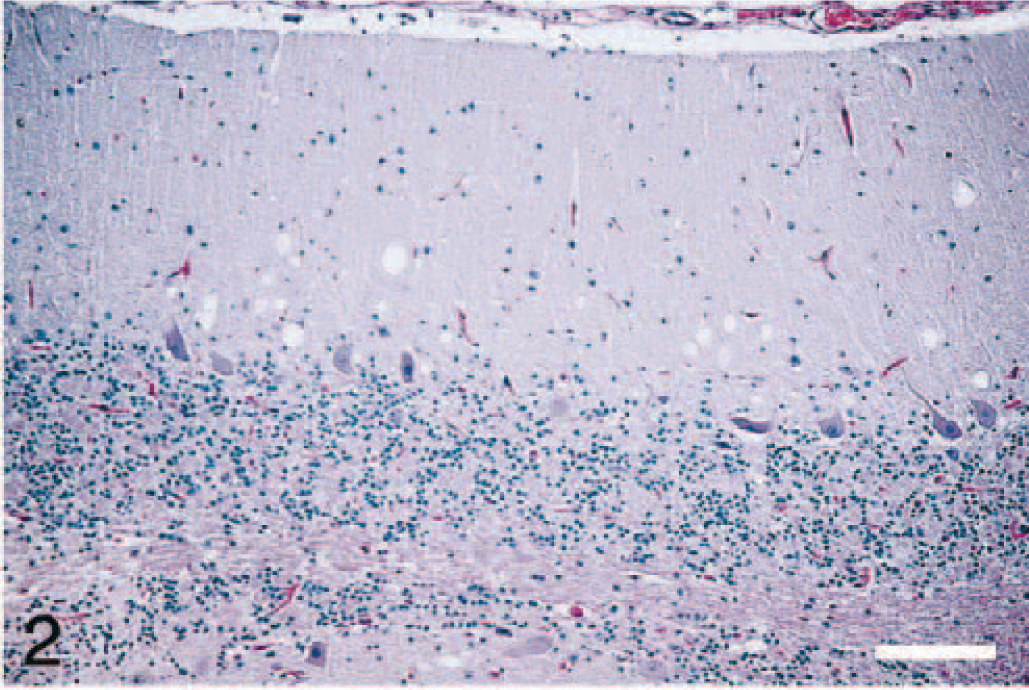
Brain; cerebellum of elk No. 4. Variable-sized clear vacuoles are present in the neuropil of the molecular layer. HE. Bar = 120 μm.
IHC revealed PrPres in the brains (cerebrum, cerebellum, and brainstem) and in the spinal cords (only cervical cord was available from elk No. 5). The immunostaining was diffusely distributed throughout gray matter neuropil in all parts of the CNS, including both granular and molecular layers of cerebellum (Figs. 5, 6). The staining pattern was primarily punctate and granular, with some small aggregates and only a few accumulations large enough to be described as plaques. Some neurons contained large, stained granules in the perikaryon, but more commonly the location of PrPres was perineuronal, forming a ring around the cell surface.
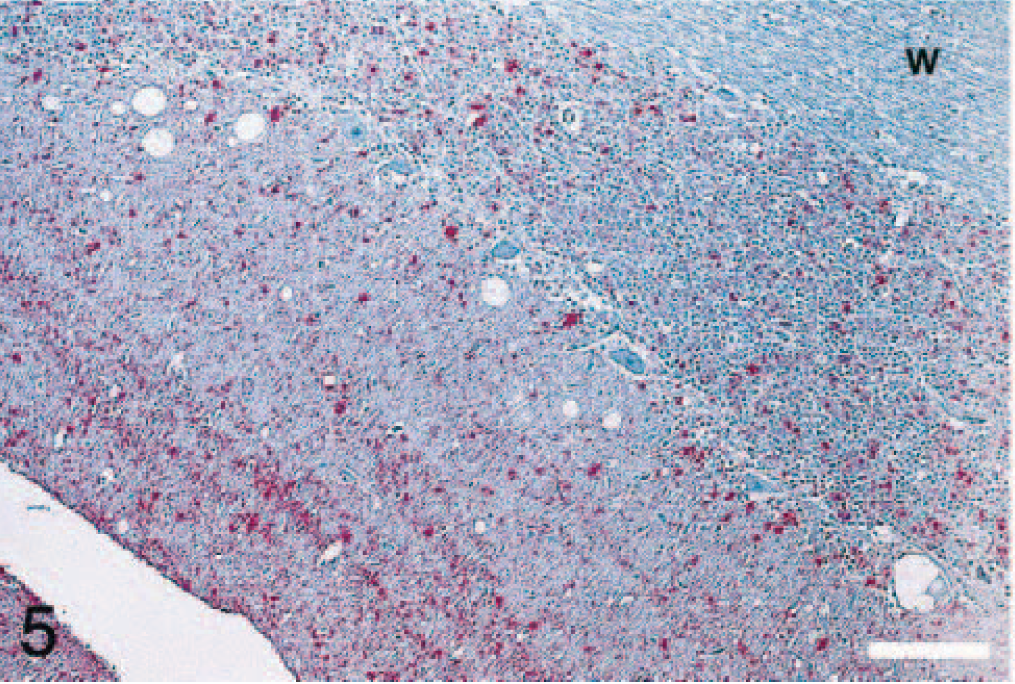
Brain; cerebellum of elk No. 4. There is presence of PrPres in molecular and granular layers of the cerebellum. PrPres staining is not present in the cerebellar white matter (w). Stained for PrPres by IHC. Bar = 100 μm.
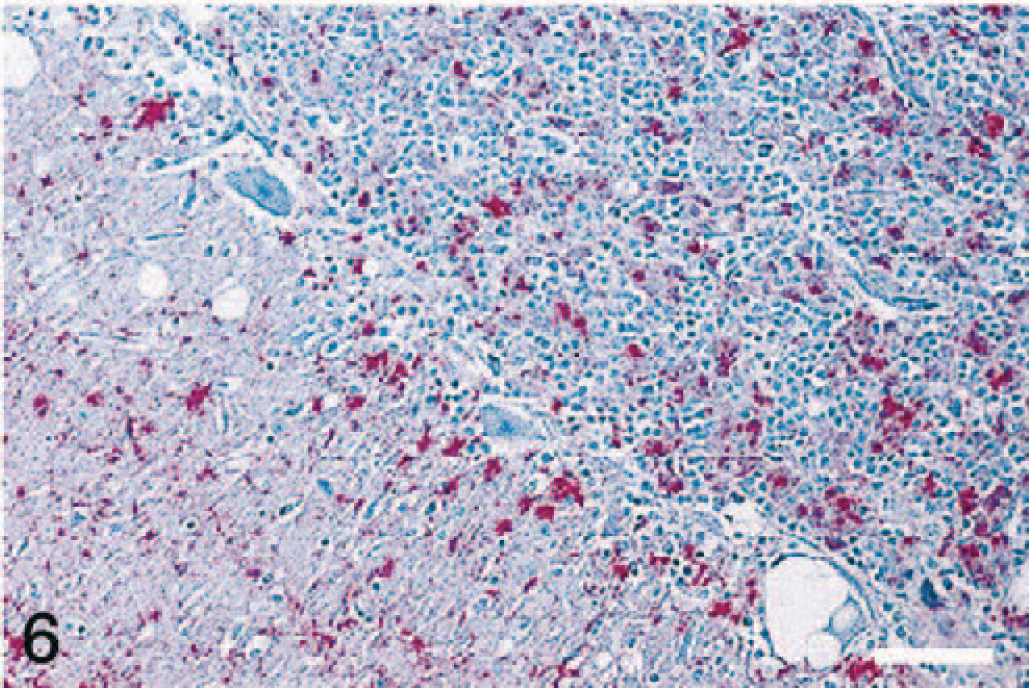
Higher magnification showing PrPres in molecular and granular layers of the cerebellum. Stained for PrPres by IHC. Bar = 75 μm.
The brains of both elk (Nos. 4 and 5) were positive for PrPres by western blot (Fig. 7) and for SAFs by negative stain electron microscopy (Fig. 8). Neither PrPres nor SAFs were detected in the CNS of the other three elk that were euthanatized prior to the death of elk No. 4 (Table 1).
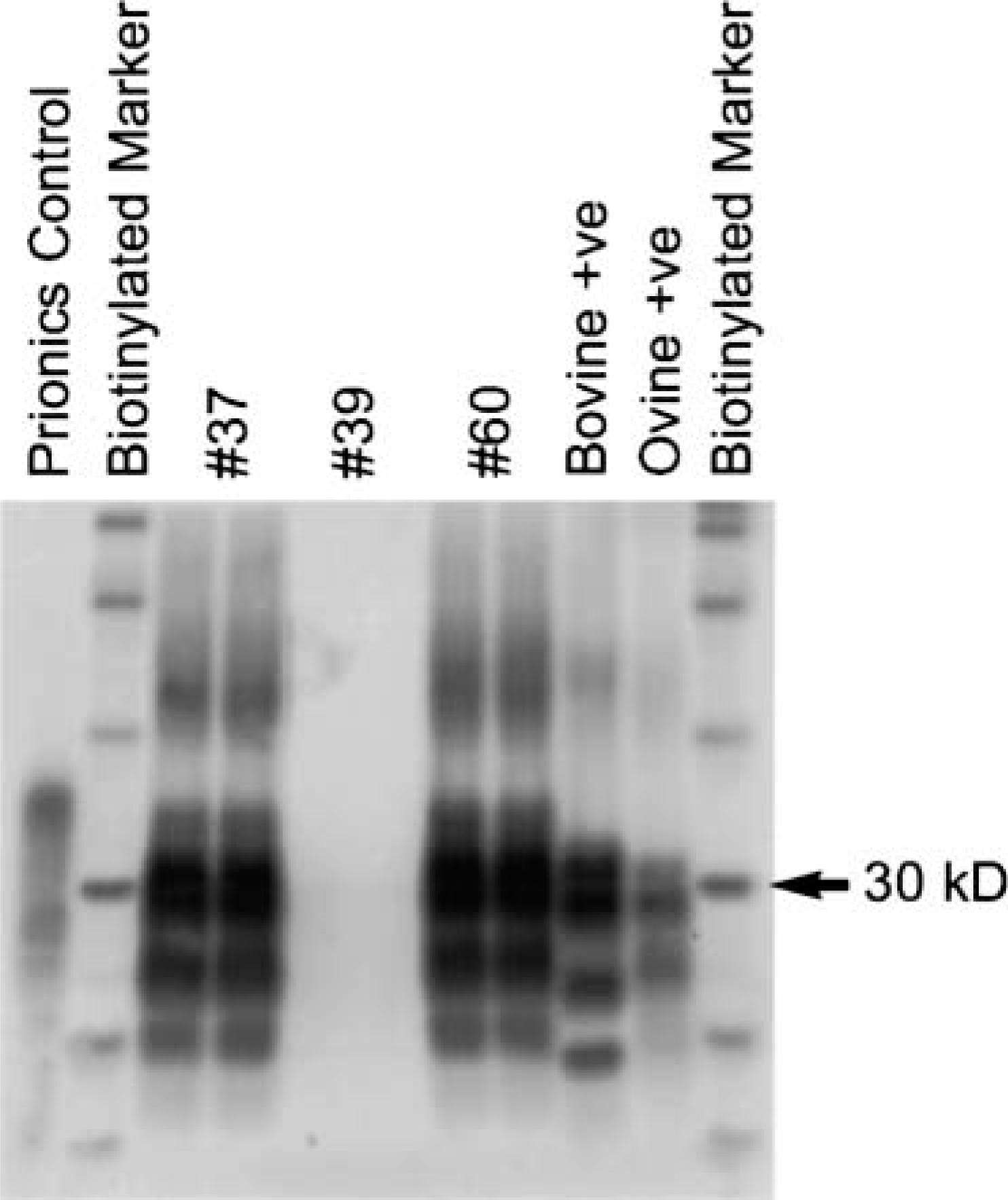
Western immunoblot showing distinct profile of PrPres (molecular weight 27–30 kD) in elk Nos. 37 and 60. No signal is seen in elk No. 39, classified as negative. A normal PrPc control (molecular weight 33–35 kD) is in the first lane (Prionics control). Molecular weight marker positions are indicated on the right-hand side of the image. Bovine +ve and ovine +ve refer to samples positive for bovine spongiform encephalopathy and scrapie, respectively.
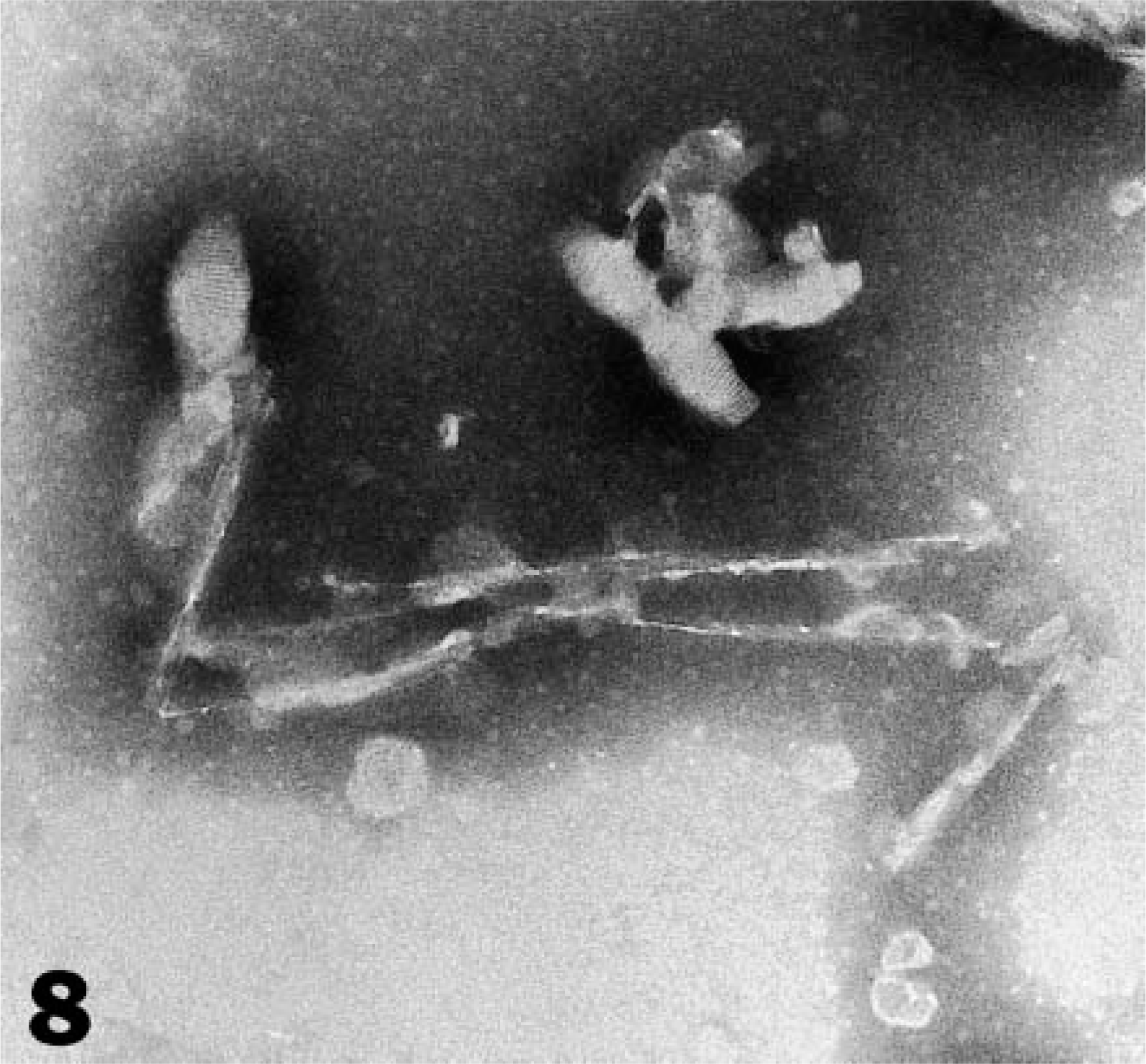
Negatively stained SAF observed in tissue extract (caudal medulla) from elk No. 60.
Scrapie, like all other TSEs, is characterized by a long incubation period, which in sheep and goats is of 2 to 4 years duration. 4 Similarly, the incubation period of CWD in elk is seldom less than 18 months. 19 Therefore, based on the incubation time of scrapie in elk Nos. 4 and 5, it is not possible to distinguish scrapie and CWD infections in elk.
Regarding clinical signs of CWD in elk, the disease is characterized by severe emaciation, changes in behavior, and excessive salivation. 20 In our study, we did notice some change in behavior in one animal (hostile to animal handlers) and terminal signs of CNS abnormality. However, excessive salivation was not noticed, and although moderate weight loss was observed at necropsy, frank emaciation was not a feature of this experimental infection.
Although the transmission route used in the present investigation does not mimic the natural route of infection (which in the wild would most likely be via the ingestion of scrapie-infected material), the study does demonstrate that elk are susceptible to sheep scrapie. The per os route of infection would undoubtedly require a higher infectious dose, and even so, successful transmission may be much more difficult or impossible to accomplish.
In this study, lesions of spongiform encephalopathy were found throughout the CNS of elk with terminal neurologic signs, following infection with the scrapie agent. Although the spongiform change was more severe in certain anatomic locations of the brain (cerebellum, thalamus, pontine nucleus), in general these findings are indistinguishable from those reported in elk with CWD infection. 20
In previous successful intracerebral transmission experiments of scrapie to cattle, there were no diagnostically significant neurohistopathologic lesions in recipient animals. 1–3 The present findings of extensive lesions of spongiform encephalopathy in scrapie-inoculated elk could be due to biological differences of the host species or due to strain differences in the inocula sources used. It could be speculated from these findings that both the agents of scrapie and CWD have a common lineage (i.e., CWD originated as a cross-species transmission of scrapie). Strain typing of the agents involved might provide some insight in resolving this question.
The pattern of PrPres immunostaining in elk inoculated with scrapie was similar to that described previously in elk with naturally acquired CWD, 5,14 although large plaques were uncommon, and accumulations around blood vessels were not observed. Also absent were the “florid plaques” considered characteristic of CWD in captive mule deer. 5,9 These plaques, which consist of PrPres accumulations surrounded by vacuoles, are also considered pathognomonic for variant Creutzfeldt-Jakob disease in humans. 8,17
We conclude that sheep scrapie agent can be transmitted to elk by intracerebral inoculation and the infection results in severe, widely distributed lesions and accumulations of PrPres in the CNS of affected animals. Because only two of the inoculated elk developed TSE, it could be assumed that the affected elk were genetically the most susceptible to the infection. Such genetic predisposition has been proposed for CWD infection in elk. 13
Forty-three months after the scrapie challenge, the one remaining inoculated elk and two uninoculated (control) elk are alive and apparently healthy. As with previous interspecies TSE transmissions experiments at NADC, 3,6 the remaining elk will be kept under observation for at least 3 more years (a total of 7 years PI) before the experiment is terminated.
Footnotes
Acknowledgements
We thank Dr. Lawayne Nusz for assistance with the clinical and necropsy examinations. Expert technical assistance was provided by Martha Church, Jean Donald, and Jennifer Slovak at NADC, Ames, Iowa, USA, and Linda Davis and Donna Barnicle at VLA, Weybridge, UK.
Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
